Abstract
Background. The majority of studies investigating the neural mechanisms underlying treatment-induced recovery in aphasia have focused on the cortical regions associated with language processing. However, the integrity of the white matter connecting these regions may also be crucial to understanding treatment mechanisms. Objective. This study investigated the integrity of the arcuate fasciculus (AF) and uncinate fasciculus (UF) before and after treatment for anomia in people with aphasia. Method. Eight people with aphasia received 12 treatment sessions to improve naming; alternating between phonologically-based and semantic-based tasks, with high angular resolution diffusion imaging conducted pre and post treatment. The mean generalized fractional anisotropy (GFA), a measure of fiber integrity, and number of fibers in the AF and UF were compared pre and post treatment, as well as with a group of 14 healthy older controls. Results. Pre treatment, participants with aphasia had significantly fewer fibers and lower mean GFA in the left AF compared with controls. Post treatment, mean GFA increased in the left AF to be statistically equivalent to controls. Additionally, mean GFA in the left AF pre and post treatment positively correlated with maintenance of the phonologically based treatment. No differences were found in the right AF, or the UF in either hemisphere, between participants with aphasia and controls, and no changes were observed in these tracts following treatment. Conclusions. Anomia treatments may improve the integrity of the white matter connecting cortical language regions. These preliminary results add to the understanding of the mechanisms underlying treatment outcomes in people with aphasia post stroke.
Introduction
Impaired naming is a common difficulty in people with aphasia post stroke and may arise because of deficits in lexical-semantic processing, access to the word form, or a combination of the two. 1 The rehabilitation of naming impairments typically aims to identify the locus of breakdown in word retrieval and target the impaired process using specific linguistic tasks. 2 However, an individual’s response to a particular treatment approach cannot always be predicted by the locus of impairment. 3 Thus, investigation of the neural mechanisms underlying different treatment approaches may aid in predicting responses to treatments targeting different processing components. Previous studies have examined how patterns of brain activity or the integrity of specific cortical regions may relate to improvements in naming following specific treatments.4,5 However, white matter pathways may also be damaged post stroke, disrupting the connections between these cortical regions. Investigating the relationship between white matter connectivity and response to different treatment approaches may provide additional information to predict treatment outcomes. 6
The idea that cortical regions essential for language can become disconnected dates back to Wernicke’s 7 network model of language, which proposed language impairments can be caused by a disruption of the white matter pathway connecting the frontal center for “motor imagery” with the temporal centre of “acoustic images.” This disconnection account of language impairments was extended by Lichtheim, 8 and more recently by Geschwind. 9 However, these models have relied on lesion-deficit correlations of post mortem dissections. With advances in neuroimaging techniques, such as diffusion imaging, the anatomical basis of language networks can be explored in the living brain.
Using diffusion imaging–based tractography, the probable orientation and integrity of white matter tracts can be mapped, allowing for inferences to be made regarding how cortical areas responsible for language may be networked. This technique allows for investigation into how particular language impairments post stroke may reflect damage to this network, how this damage may relate to an individual’s response to treatment, as well as how white matter structure may change as a result of treatment. A major fiber tract that has been examined in studies of language processing is the arcuate fasciculus (AF), considered to be a component of the superior longitudinal fasciculus, connecting the classical Broca’s and Wernicke’s language areas in the frontal and temporal cortices, respectively. 10 The function of the AF has been associated with phonological processing, where electrical stimulation of this tract has elicited phonemic paraphasias.11,12
However, different theories concerning the structure and function of this white matter connection have also been proposed. Catani et al 13 found the AF to project to 3 different cortical regions: a long segment connecting the frontal and temporal cortices, a posterior lateral segment connecting temporal and parietal cortices, and an anterior lateral segment connecting frontal and parietal cortices. It was suggested that the long segment creates a direct phonological pathway between the 2 regions, while the posterior and anterior segments create an indirect pathway for lexical-semantic processing via the parietal lobe. In contrast, Glasser and Rilling 14 described 2 distinct segments of the AF: a segment linking the inferior frontal cortex to the posterior superior temporal gyrus associated with phonological processing, similar to the long segment described by Catani et al 13 and a segment linking the inferior frontal cortex to middle and inferior temporal regions associated with lexical-semantic processing.
In addition to the AF, other more ventrally located pathways have also been suggested to contribute to language circuitry. For example, the uncinate fasciculus (UF) has been suggested to connect frontal and temporal language regions related to semantic processing.10,15,16 Saur et al 17 proposed 2 distinct pathways connecting frontal and temporal regions. In addition to the dorsal pathway along the AF, a more ventrally located pathway along extreme capsule was found to connect middle and inferior temporal regions with the ventrolateral prefrontal cortex. The functional interpretation of these pathways, based on functional magnetic resonance imaging activation during repetition and auditory comprehension tasks, was that the dorsal pathway was responsible for mapping sound to articulation, and the ventral pathway supported mapping sound to meaning.
The functions of different language pathways have also been examined by investigating damage to white matter poststroke. For example, Zhang et al 18 examined a group of 10 participants with conduction aphasia, characterized by poor repetition, and found reduced fractional anisotropy (FA) values in the left hemisphere AF, as well as increased FA values in the right hemisphere, compared with controls. Similarly, Hosomi et al 19 found a loss of leftward asymmetry in the number of AF fibers to predict the presence of aphasia at discharge in a group of 13 acute stroke patients. Breier et al 20 found greater damage to the left AF to be associated with impairments in repetition in 20 participants with aphasia, independent of damage to the cortical regions that may be involved in phonological processing. However, damage to the UF was not found to be associated with impairments in confrontation naming. Marchina et al 21 also found lesion load in the AF, but not the extreme capsule or UF, to predict performance on spontaneous speech and naming measures in a group of 30 participants with aphasia.
Studies investigating the relationship between white matter integrity and response to treatment, or changes in white matter following treatment, are scarce. Schlaug et al 22 employed diffusion imaging to investigate the plasticity of the right AF following melodic intonation therapy, a treatment thought to engage right hemisphere mechanisms. Significant increases were found in the number and volume of AF fibers in the right hemisphere following treatment, and it was suggested that these changes may reflect increased myelinization and possibly the formation of axon collaterals to improve frontotemporal connections in the right hemisphere. Breier et al 23 examined changes in the AF with a single case study employing constraint-induced language therapy, where an increase in FA, a measure of white matter integrity, of the left AF was found post treatment.
Although the above studies provide some evidence of a relationship between changes in white matter integrity of the AF and treatment outcome in aphasia, the therapies focused on new treatments not routinely used in clinical practice and did not target any specific linguistic processes. The most common treatment in aphasia is for word retrieval, 2 yet the effect of this specific form of treatment on white matter connectivity has not been investigated. Additionally, other white matter tracts that contribute to the language network may also be important for treatment outcome. The current study addressed these issues by investigating changes in the integrity of both the AF and UF following phonologically and semantically based treatment for anomia and examining the relationship between white matter integrity (pre and post treatment) and treatment outcome.
Methods
Participants
8 participants with aphasia (5 female) and 14 healthy older controls (8 female) completed the study. Participants with aphasia ranged from 41 to 69 years (mean = 56.38 years, standard deviation [SD] = 9.15) and time post single left cerebrovascular accident ranged from 17 to 170 months (mean = 52.25 months, SD = 49.84). Control participants ranged in age from 49 to 81 years (mean = 61.71 years, SD = 10.07). No significant difference in age was found between participants with aphasia and controls (P = .20). All participants had English as a first or primary language prior to stroke, were right handed according to the Edinburgh Handedness Inventory, 24 with normal or corrected-to-normal vision and hearing. All participants were screened for cognitive impairments and depression using the Mini Mental State Examination 25 and the Glasgow Depression Scale, 26 respectively. None of the participants had a history of any other neurological disease or disorder, mental illness, head trauma, alcoholism, cerebral tumor or abscess, severe dysarthria or apraxia of speech, or any contraindications for magnetic resonance imaging. All participants gave their informed consent prior to participation in the study.
All participants were assessed using the Boston Naming Test, 27 the Pyramids and Palm Trees, 28 Western Aphasia Battery, 29 and the Comprehensive Aphasia Test. 30 A naming battery of 476 items from the International Picture Naming Project Database 31 was administered twice within the same week for participants with aphasia, and on one occasion for control participants. See Table 1 for demographic information and language assessment results for participants with aphasia.
Demographic Information and Language Assessment Results for Participants With Aphasia.
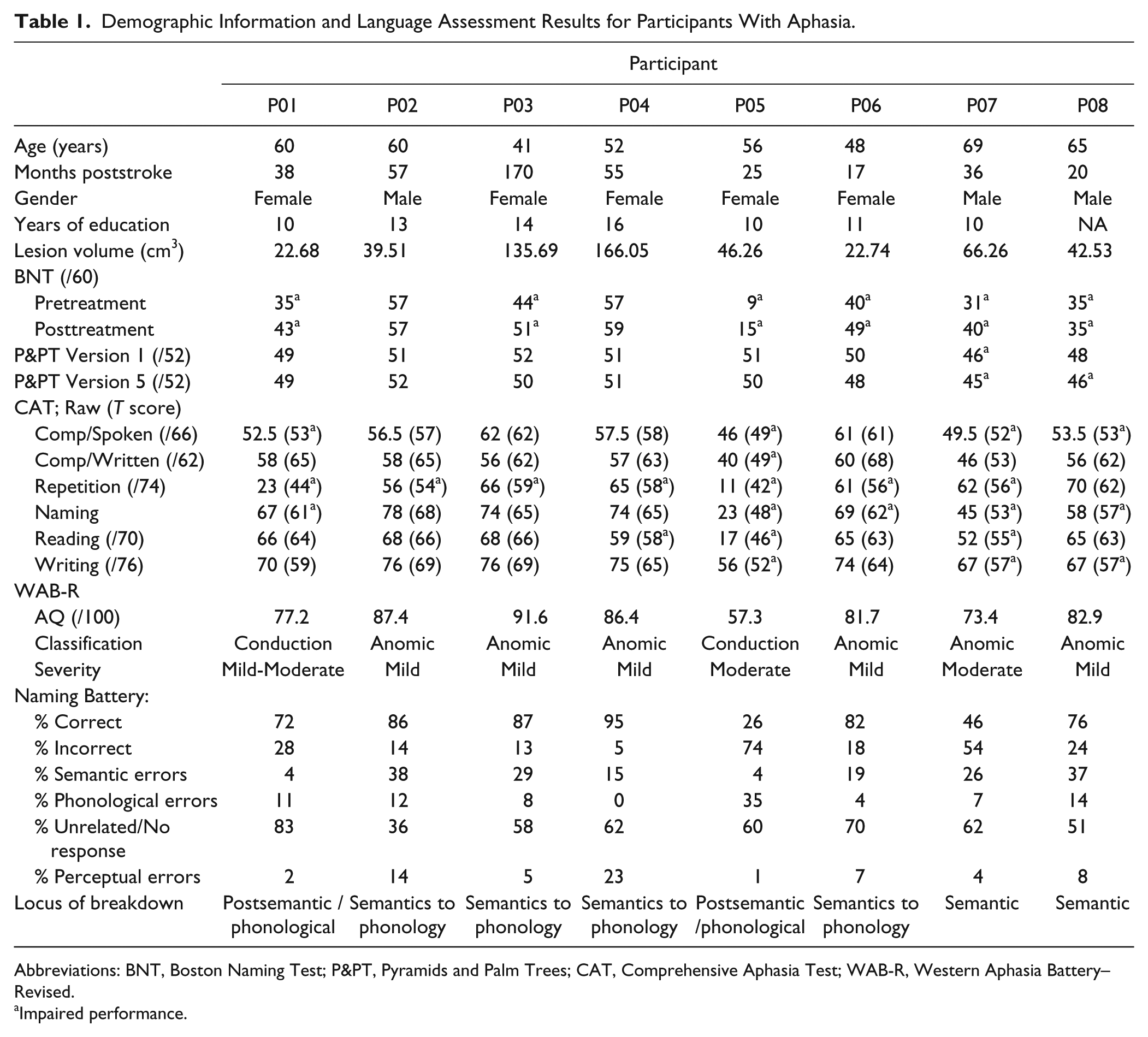
Abbreviations: BNT, Boston Naming Test; P&PT, Pyramids and Palm Trees; CAT, Comprehensive Aphasia Test; WAB-R, Western Aphasia Battery–Revised.
Impaired performance.
Procedure
For participants with aphasia, language assessments were completed over 3 sessions prior to the first scan, and results used to determine the primary locus of breakdown in word retrieval and selection of individual treatment items. Following the scan, each participant with aphasia received 4 weeks of therapy (3 sessions per week), where the type of treatment task (semantic or phonological) was alternated each session and order counterbalanced among participants. Within 7 to 12 days after the final therapy session, participants with aphasia underwent a second scan.
The semantic therapy task employed was Semantic Feature Analysis (SFA), 32 and the phonological therapy task employed was Phonological Components Analysis (PCA). 33 A total of 120 individualized items were selected for each participant, including 30 “able to name items” (named correctly on both attempts of the naming battery), and 90 “unable to name items” (named incorrectly on at least one attempt of the naming battery). The 90 “unable to name items” were divided into 3 sets of 30 items; one set was treated with PCA, one set was treated with SFA, and one set was untreated. The SFA and PCA tasks were administered in accordance with previous studies employing these tasks.32-34 For full details, see van Hees et al, 35 where the behavioral aspects of this treatment have been described in detail. Two baseline naming measures (B1 and B2) were collected during the initial assessment sessions, and the third baseline (B3) was collected prior to the first treatment session. Post therapy measures were collected immediately after treatment (P1) and at follow-up assessment 2 to 3 weeks after the final session (P2). For the control group, language assessment was completed over 2 sessions, followed by one scan.
Image Acquisition and Processing
Images were acquired using a 4-Tesla Bruker MedSpec magnetic resonance imaging system (Bruker BioSpin GmbH, Rheinstetten, Germany) using a transverse electromagnetic head coil. Diffusion imaging was performed using a diffusion-weighted dual-echo echo-planar imaging sequence (repetition time [TR] = 8250 ms; echo time [TE] = 92.3 ms; 6 b = 0; 50 b = 1177; 230 mm field of view [FOV]; 55 × 2 mm slices). Pre processing consisted of eddy current correction and brain extraction using FSL software v 4.1.6. 36 The q-ball fit and deterministic fiber tracking was performed using Diffusion Toolkit software, 37 using the standard FACT method. 38 Orientation distribution functions were calculated using the spherical harmonic basis method. 39 The final tracts were smoothed with a B-spline filter.
A 2 regions of interest approach was used to reconstruct the AF and UF in native space. Using the color-coded directional white matter map, seed and target regions were manually selected in the axial planes (see Figure 1). For the AF, the seed mask was drawn in the posterior parietal portion of the superior longitudinal fasciculus, and the target mask was drawn in the descending portion of the superior longitudinal fasciculus in order to extract the AF connecting frontal and temporal cortices. 40 For the UF, the seed mask was drawn in white matter of the inferior frontal lobe, and the target mask drawn inferiorly to the seed in order to dissect the UF from the occipito-frontal fasciculus. 41 A minimum length filter of 10 mm was applied before the total number of fibers and mean generalized fractional anisotropy (GFA), a measure of fiber tract integrity that takes into account crossing fibers, were extracted for each tract.
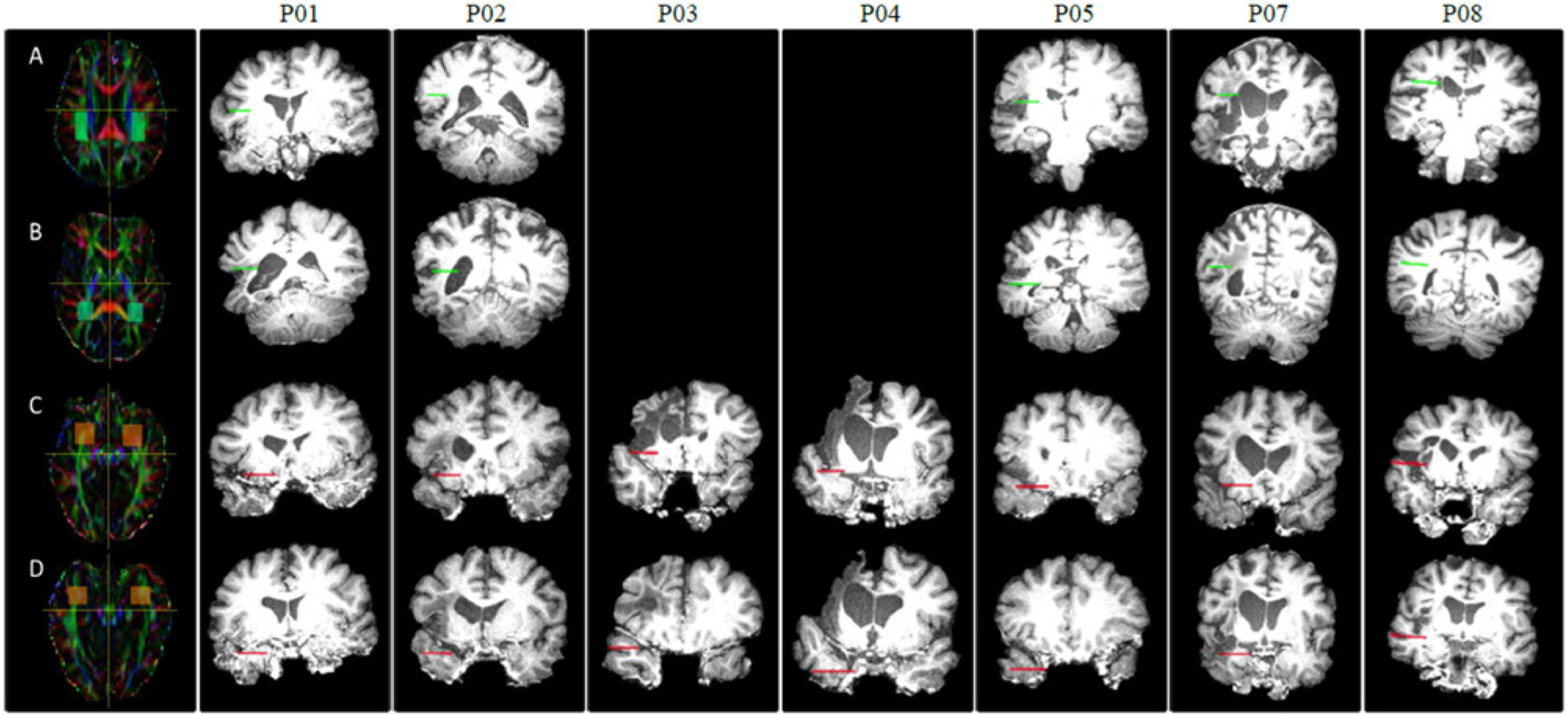
Two regions of interest (ROIs) approach using directionally encoded tensor maps in native space. Each row displays the left hemisphere ROIs overlayed on each participant’s T1 structural image to show the impact of lesions on the position of the white matter tracts. (A) Seed and (B) target ROIs for construction of the arcuate fasciculus (AF) pathway. (C) Seed and (D) target ROIs for construction of the uncinate fasciculus (UF) pathway. Because of large lesions, the left AF was not able to be extracted for P03 and P04.
Results
Control Group
Independent-samples t tests were conducted to identify any differences between hemispheres for the control group. A significantly greater number of fibers were found in the left AF compared with the right AF (P = .01), but no significant difference was found between mean GFA in the left AF compared with the right AF (P = .15). No significant difference was found between number of fibers in the left UF compared with the right UF (P = .09), or between mean GFA in the left UF compared with the right UF (P = .45). See Table 2 for a summary of results.
Summary of Tractography Results: Mean Number of Fibers and Mean GFA for Each tract in Controls and Participants With Aphasia Pre and Posttreatment.
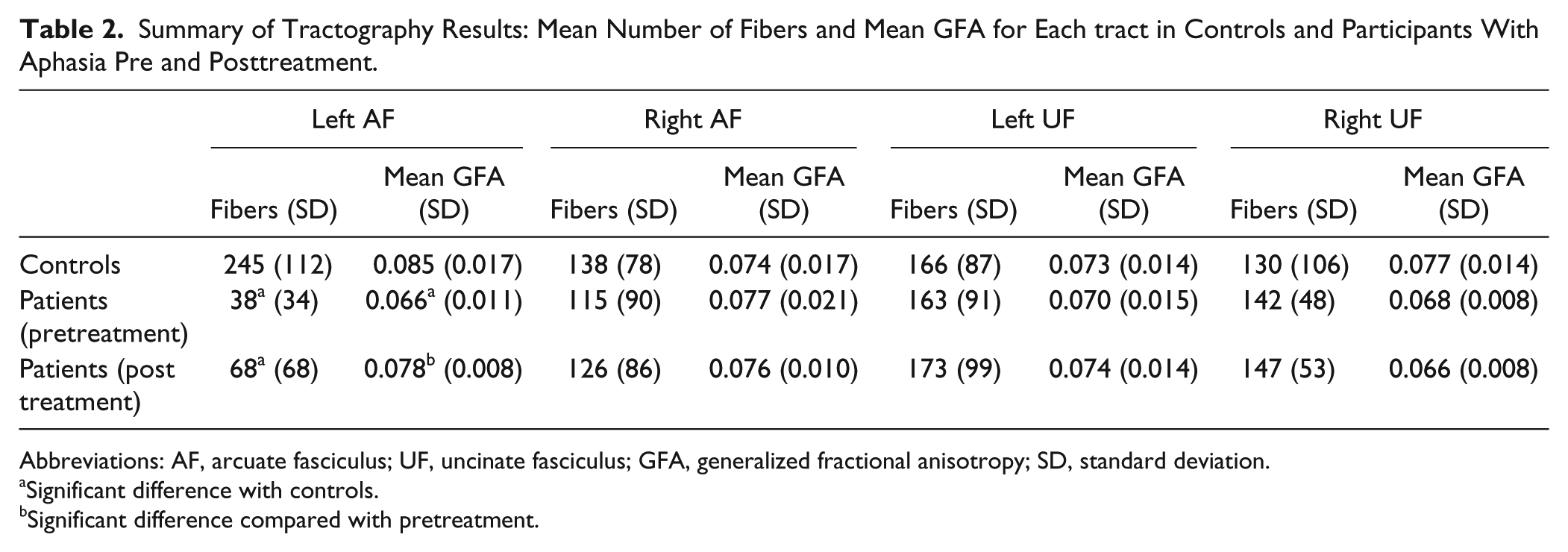
Abbreviations: AF, arcuate fasciculus; UF, uncinate fasciculus; GFA, generalized fractional anisotropy; SD, standard deviation.
Significant difference with controls.
Significant difference compared with pretreatment.
Participants With Aphasia
Tractography Results
Because of technical problems, diffusion data could not be collected from one participant (P06). Additionally, because of large lesions, the left AF was not able to be extracted in 2 participants (P03 and P04). Thus, tractography analyses were conducted on 5 participants for the left AF (P01, P02, P05, P07, and P08) and on 7 participants for the right AF as well as the UF in both hemispheres (P01, P02, P03, P04, P05, P07, and P08).
Independent-samples t tests were conducted to identify any differences between controls and participants with aphasia both pre and post treatment. Paired-samples t tests were also conducted to examine changes from pre to post treatment. Compared with controls, participants with aphasia had significantly fewer fibers, t(17) = −4.01, P = .001, and lower mean GFA, t(17) = −2.19, P = .04, in the left AF pre treatment. Post treatment, there was still a significant difference between the number of fibers, t(17) = −3.29, P = .004, compared with controls, but no longer a significant difference between groups in terms of mean GFA, t(17) = −0.72, P = .48. For the right AF, no significant differences were found between number of fibers or mean GFA pre or post treatment (P > .05) compared with controls. No significant differences were found between number of fibers or mean GFA pre or post treatment for the UF in either hemisphere (P > .05), between participants with aphasia and controls (see Table 2 for a summary of results).
For pre versus posttreatment comparisons, a significant increase in mean GFA was found from pre to post treatment in the left AF, t(4) = 3.21, P = .03 (see Figure 2 for individual results). No significant difference in mean GFA was found from pre to posttreatment in the right AF, t(6) = −0.28, P = .79. No significant difference was found between the number of fibers from pre to post treatment in the left AF, t(4) = 1.60, P = .19, or in the right AF, t(6) = 1.70, P = .14.
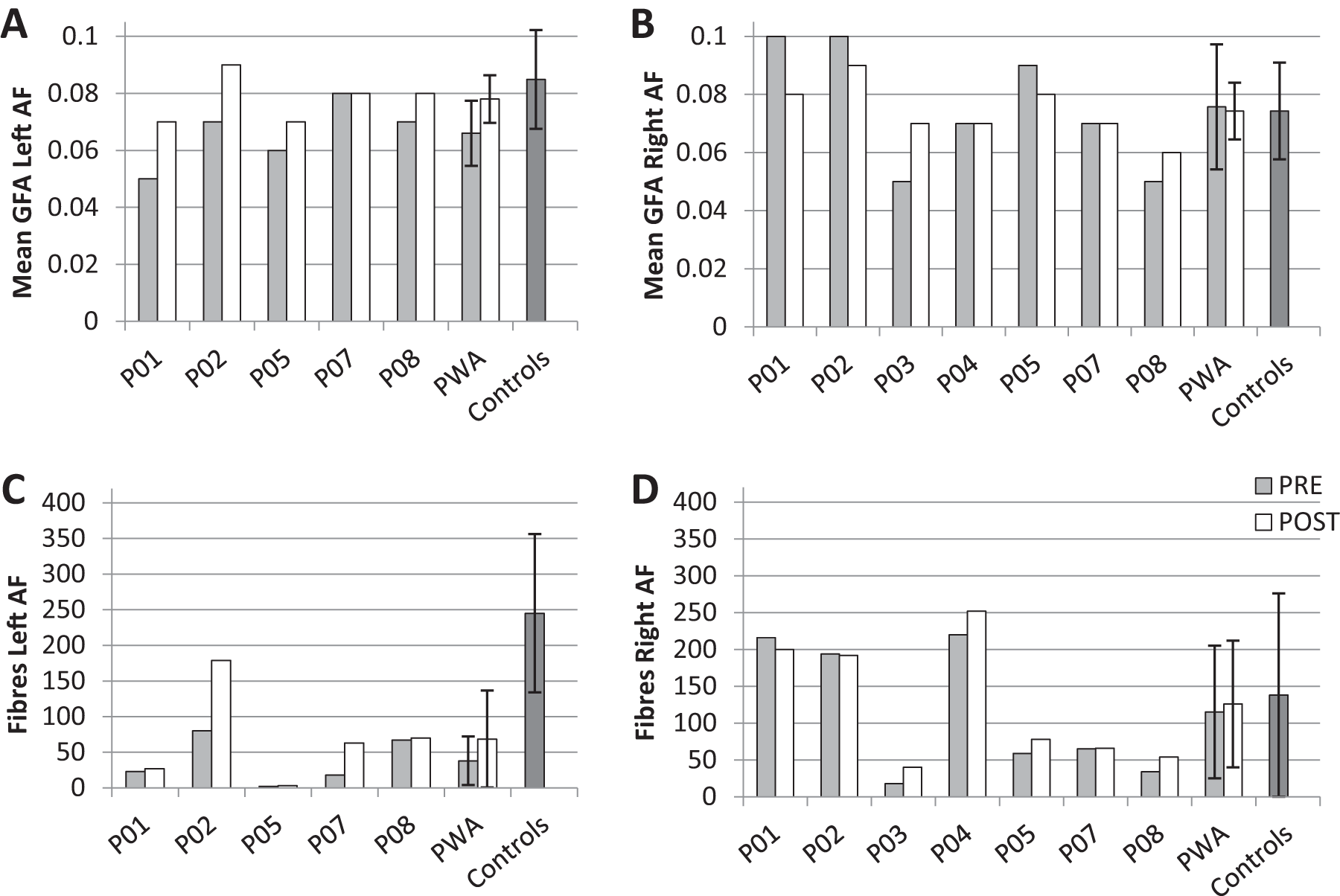
Individual and group tractography results: Mean GFA of the (A) left AF and (B) right AF and number of fibers of the (C) left AF, and (D) right AF. GFA, generalized fractional anisotropy; AF, arcuate fasciculus; PWA, participants with aphasia. Error bars indicate standard deviation.
For the UF, no significant difference in mean GFA was found from pre to post treatment in the left UF, t(6) = 1.162, P = .29, or in the right UF, t(6) = −0.26, P = .81. Similarly, no significant difference was found between the number of fibers from pre to post treatment in the left UF, t(6) = 1.707, P = .14, or in the right UF, t(6) = 0.58, P = .58.
Treatment Results
Treatment results were analyzed using accuracy scores from the 5 participants from whom the left AF was able to be extracted (P01, P02, P05, P07, and P08), and from the 7 participants from whom the right AF and UF bilaterally were able to be extracted (P01, P02, P03, P04, P05, P07, and P08). Repeated-measures analyses of variance were performed to identify significant differences in naming accuracy from pre to post treatment for each item set (PCA, SFA, and untreated). For items sets that showed a significant change in naming accuracy, post hoc pairwise comparisons were used to examine significant differences in naming accuracy both immediately post treatment and at follow-up assessment, compared to the final baseline measure (corrected for multiple comparisons using a Bonferonni correction).
For items treated with PCA, both subgroups showed a significant change in naming accuracy: n = 5, F(2, 3) = 50.21, P = .005, = 0.97; n = 7, F(2, 5) = 89.80, P < .001, = 0.97. Post hoc pairwise comparisons revealed significant improvements in naming accuracy both immediately post treatment (n = 5, P = .008; n = 7, P < .001) and at follow-up assessment (n = 5, P < .001; n = 7, P < .001) compared with the final baseline. For items treated with SFA, a significant change in naming accuracy was only found for the subgroup of 7 participants: n = 5, F(2, 3) = 7.97, P = .06; n = 7, F(2, 5) = 16.04, P = .007, = 0.87). Post hoc pairwise comparisons for the 7 participants identified with a significant change in naming accuracy revealed significant improvements both immediately post treatment (P = .005) and at follow-up assessment (P = .003), compared with the final baseline. No significant change was found for the untreated set in either subgroup: n = 5, F(2, 3) = 0.212, P = .82; n = 7, F(2, 5) = 1.295, P = .35. See Figure 3 for individual results.
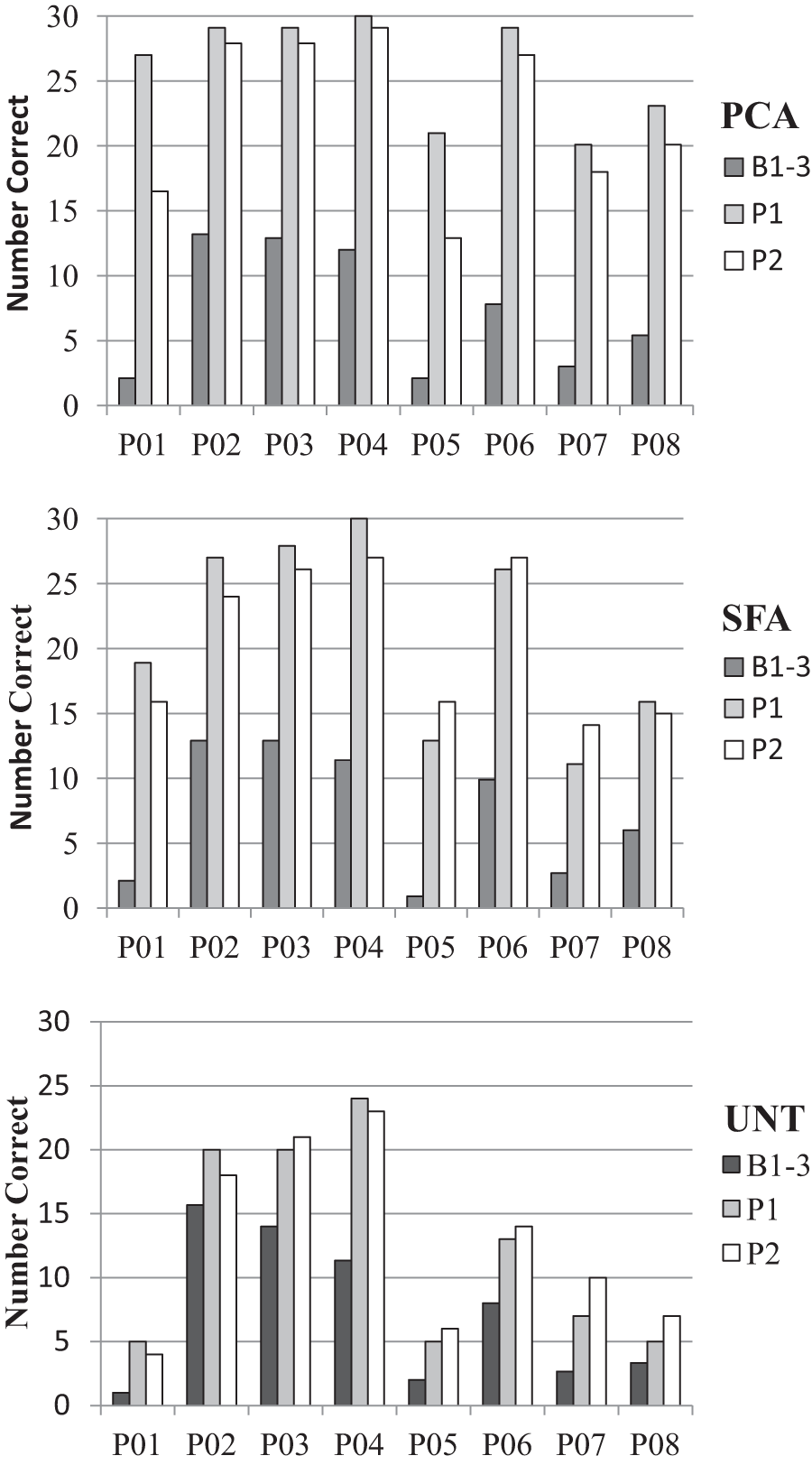
Individual treatment results. B1-3, average of baseline 1-3 (pre treatment); P1, immediately post treatment; P2, follow-up 2 to 3 weeks posttreatment; PCA, items treated with Phonological Components Analysis; SFA, items treated with Semantic Feature Analysis; UNT, untreated control items.
Furthermore, for white matter tracts that showed a significant change from pre to post treatment in either number of fibers or mean GFA, Pearson’s correlations were conducted to examine the relationship between imaging measures and naming improvement for item sets that showed significant improvement following treatment. Thus, correlations were performed between mean GFA in the left AF and naming improvement following PCA (n = 5). No significant correlations were found between mean GFA and immediate improvement; however, a significant positive correlation was found between maintenance scores and pre treatment mean GFA (r = 0.90, P = .04) as well as post treatment mean GFA (r = 0.93, P = .02). See Figure 4 for scatterplots of the correlation results.
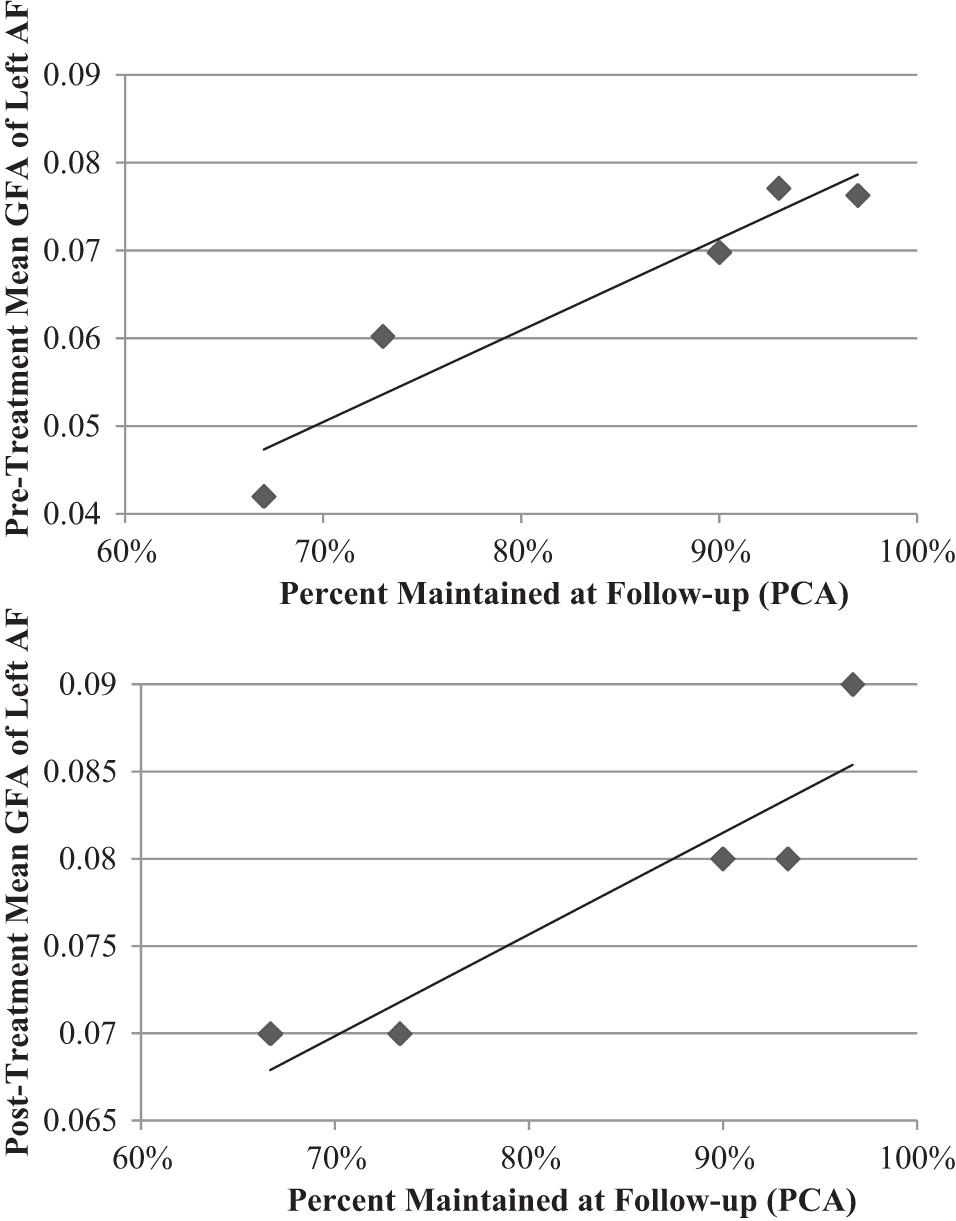
Scatterplots showing correlation between pre and posttreatment mean GFA in the left AF and maintenance of treatment outcome for items treated with PCA. GFA, generalized fractional anisotropy; AF, arcuate fasciculus; PCA, Phonological Components Analysis.
Discussion
The aim of this study was to investigate changes in the integrity of language-related white matter tracts following treatment of anomia poststroke. The number of fibers and mean GFA values of the AF and the UF in both hemispheres were investigated pre and post treatment and compared with a healthy older control group. The results for the AF in the control group are consistent with previous studies that have found greater numbers of fibers, fiber density, and FA values in the dominant left hemisphere.42,43 Compared with the control group, participants with aphasia were found to have significantly fewer fibers, as well as lower mean GFA, in the left AF pretreatment. However, with increases in mean GFA post treatment, there was no longer a significant difference between participants with aphasia and controls for mean GFA in the left AF.
An increase in mean GFA following treatment in the absence of a significant increase in the fiber count, suggests that treatment was improving the integrity of the existing fibers in the left AF. That is, there is no change to the principal direction, but to the ability to diffuse in this direction compared with orthogonal directions. Training-induced increases in fiber integrity were also found in a study conducted by Scholz et al 44 and were suggested to represent activity-dependent modulation of myelination in white matter tracts. Increased integrity of the left AF was also found following constraint induced aphasia therapy in a single case study conducted by Breier et al. 23 Thus, the current study provides further evidence of increased integrity of the left AF following successful treatment of anomia poststroke. Additionally, changes in mean GFA following therapy were specific to the left hemisphere, with no change found in the right hemisphere AF. These results are in contrast to a study conducted by Schlaug et al, 22 who found increased integrity and number of fibers in the right AF following a melodic intonation treatment assumed to target right hemisphere mechanisms. This discrepancy may suggest that changes in the left or right AF may depend on the type of treatment task employed. However, the participants in study by Schlaug et al also had more severe language impairments, and thus may be more reliant on preserved right hemisphere mechanisms.
Previous studies have found damage to the left AF to be associated with deficits in phonological processing, particularly repetition.18,20 As the 5 participants from whom the left AF was able to be extracted only showed a significant improvement in naming items treated with PCA, the integrity of the left AF may relate to the outcomes of phonologically based therapy. However, mean GFA in the left AF only correlated with how well therapy improvements were maintained for items treated with PCA, it did not relate to immediate improvement. As the ultimate goal is for patients to maintain improvements beyond the therapy period, this result is important for clinical practice. Although participants with lower mean GFA may have made significant behavioral gains immediately post treatment, these gains were not able to be maintained as well as for participants with higher mean GFA in the left AF.
In contrast to the AF, no significant differences were found between the left and right UF in the control group, as well as between participants with aphasia and controls in either hemisphere. Additionally, no changes in the UF were found from pre to post treatment. Although the UF has been suggested to be involved in language processing as it connects the anterior temporal lobe to the inferior frontal lobe, its functional role is unclear. Additionally, the UF is more difficult to measure successfully given the sensitivity of the diffusion sequence to artifacts around the sinuses.
Previous studies have found damage to the UF following surgical resection of the anterior temporal cortex to be associated with naming impairments 45 and reduced FA values in the UF have been associated with the semantic variant of primary progressive aphasia,46,47 suggesting a role in lexical-semantic processing. However, UF lesion load was not associated with measures of spontaneous speech and naming in a group of 30 participants with aphasia. 21 One explanation for the discrepancies between the results of the AF and UF is that the AF is more left lateralized, whereas the UF is more bilateral. Thus, the language functions of the UF may be more easily compensated for following left hemisphere damage.48,49 However, it was also suggested in the study by Marchina et al that the AF is more likely to be damaged post stroke and therefore may be more likely to show changes following treatment. As no difference was found in number of fibers or mean GFA of the UF in participants with aphasia compared with controls, this may explain why no change was found following treatment. Alternatively, although previous studies have linked the UF to semantic processing, the UF may play a subsidiary or connecting role with the inferior fronto-occipital fasciculus,16,50 which may explain the absence of results for the UF following treatment employing a semantic-based task.
One limitation of this study was that the left AF was not able to be extracted in the 2 participants with large lesions (P03 and P04). Despite extensive damage to the left AF, these 2 participants both showed significant improvements following treatment. These results may reflect a technical limitation of the tractography technique in the presence of a large lesion, or may suggest that other compensatory mechanisms are involved. Examination of the individual results shows that P04 had the highest number of fibers in the right AF, both pre and post treatment, which may represent reorganization that has already occurred. In contrast, P03 had fewer numbers of fibers, but showed an increase in mean GFA of the right AF following treatment. Such results suggest that these participants may have engaged preserved right hemisphere mechanisms.
The current study has provided supporting evidence that anomia treatments can effect changes in the integrity of the left AF, with pre and post treatment GFA found to correlate with maintenance of improvement following therapy targeting phonological processing. However, as there were only a small number of participants, these results should be considered as preliminary findings. The positive results warrant further research with a larger cohort of participants to improve their generalizability. Determining predictors for the outcome of specific treatments may aid in providing more effective and targeted treatment for people with aphasia.
Footnotes
Acknowledgements
The authors would like to acknowledge the University of Queensland’s Aphasia Registry for the recruitment of participants with aphasia, and the University of Queensland’s Ageing Mind Initiative for recruitment of control participants. We also thank Aiman Al Najjar and Charlene Pearson for their contribution to data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Wesley Research Institute grant, awarded to Copland, McMahon, and Read (Grant No. WRI1206COLE). Copland was supported by a National Health and Medical Research Council Career Development Award and Australian Research Council Future Fellowship (Grant No. FT100100976), and de Zubicaray was supported by an Australian Research Council Future Fellowship (Grant No. FT0991634).
