Abstract
Background. Autonomic dysfunction after spinal cord injury (SCI) is an under-researched area when compared with motor and sensory dysfunction. Cardiovascular autonomic dysfunction is a particular concern, leading to impaired control of blood pressure and heart rate. Objectives. (1) To determine the prevalence of hypotension in individuals with SCI during and after rehabilitation; (2) To investigate changes in cardiovascular variables during and after rehabilitation; (3) To evaluate the influence of personal and lesion characteristics on cardiovascular variables. Methods. Cardiovascular variables (resting systolic [SAP] and diastolic [DAP] arterial pressures and resting [HRrest] and peak heart rates [HRpeak]) were measured on 5 test occasions: start of inpatient rehabilitation, 3 months later, at discharge, and at 1 and 5 years after discharge. The time course and effects of personal and lesion characteristics on cardiovascular variables were studied using multilevel regression analyses. Results. The prevalence of hypotension was unchanged during rehabilitation and for 5 years after discharge. Odds for hypotension were highest in those with cervical and high thoracic lesions, younger individuals, and men. DAP increased during the 5 years after discharge. HRrest decreased during and after rehabilitation. SAP, DAP, HRrest, and HRpeak were lowest in those with cervical and high thoracic lesions. SAP and DAP increased with age; HRpeak decreased with age. Conclusions. These longitudinal data provide normative values for blood pressure and heart rate changes with time after injury according to lesion and personal characteristics. These results can be used to guide clinical practice and place changes in cardiovascular function caused by interventions in perspective.
Introduction
Autonomic dysfunction is common after spinal cord injury (SCI), but this has traditionally been an under-researched area compared with motor and sensory dysfunction. Indeed, these issues have only recently gained significant attention in the research arena. 1 This is particularly pertinent given that 2 surveys have shown that individuals with SCI consider that their quality of life would improve significantly with regaining autonomic functions such as bowel, bladder, and sexual function2,3 and the resolution of cardiovascular dysfunction such as autonomic dysreflexia (AD; sudden and profound hypertension in response to sensory stimuli from below the lesion). 2
Cardiovascular autonomic dysfunction is a particular concern after SCI. 4 With high level lesions (at or above the fifth thoracic level, T5), disruption of descending spinal sympathetic pathways can result in basal sympathetic hypoactivity with unopposed parasympathetic control to the heart. 5 This leads to impaired blood pressure control, manifested by low resting blood pressure, orthostatic hypotension (OH; further profound falls in blood pressure when upright), and bouts of extremely high blood pressure during AD. 6 There may also be an increased risk for cardiac arrhythmia as a result of autonomic imbalance after SCI.7,8 Although AD and arrhythmia can have serious consequences, the repeated episodes of severe hypotension that occur throughout the day can also be troublesome in terms of quality of life, leading to feelings of general fatigue,9,10 difficulty participating in activities of daily living and rehabilitation, 11 and impaired cognitive function.12,13
Impairment of cardiovascular autonomic pathways is not quantified by the standard assessment of severity of SCI, the American Spinal Injury Association Impairment Scale (AIS). 14 Resting blood pressure and heart rate (HRrest) can, however, be used as relatively simple measures indicating severity of impairment to cardiovascular sympathetic pathways. A negative correlation between resting blood pressure and the level of the SCI has been shown previously.1,6,15 The peak heart rate (HRpeak) that can be achieved during exercise is decreased if sympathetic control of the heart is abolished after SCI (usually only in those with lesions at or above T5), with exercise-induced increases in heart rate presumably achieved by vagal withdrawal. Indeed, the HRpeak achieved after autonomically complete SCI above T5 (approximately 105 BPM) 16 is comparable to that of able-bodied controls during complete vagal blockade (approximately 100 BPM). 17
To the best of our knowledge, it is unknown how the prevalence of hypotension and resting blood pressure and HRrest change with time after acute SCI. One recent cross-sectional study did examine cardiovascular parameters in a large cohort of individuals with SCI 18 and found that individuals with high level lesions or who had been injured for longer periods of time had lower blood pressures. Men with SCI were reported to have higher blood pressures and lower heart rates than women. However, in this study, only individuals with incomplete lesions to motor and sensory pathways were examined, and any changes in cardiovascular control with time after injury or during inpatient rehabilitation were not examined. Therefore, the objectives of our study were: (1) to determine the prevalence of hypotension in individuals with SCI during and after inpatient rehabilitation; (2) to investigate the time course of resting blood pressure, HRrest, and HRpeak changes with exercise, during inpatient rehabilitation and for up to 5 years after discharge; (3) to evaluate the influence of personal (age and gender) and lesion (level and completeness) characteristics on hypotension, resting blood pressure, HRrest, and HRpeak.
Methods
Participants
This study was part of the Dutch prospective cohort study “Physical strain, work capacity and mechanisms of restoration of mobility in the rehabilitation of persons with SCI.” 19 The study comprised 5 measurement sessions. Ethics approval was received from the medical ethics committee of SRL/iRv Hoensbroeck for the first 4 measurements, and the medical ethics committee of the University Medical Center Utrecht approved the addition of the fifth measurement. This study also received ethical approval from the Simon Fraser University Department of Research Ethics for all measurements. All investigations were performed in association with the Declaration of Helsinki of the World Medical Association. All participants gave written informed consent.
We recruited participants from 8 rehabilitation centers that are specialized in SCI rehabilitation in the Netherlands. Individuals were eligible to participate if they had an acute traumatic or nontraumatic SCI; were between the ages of 18 and 65 years; were classified as A, B, C, or D on the AIS Scale 20 ; were expected to remain wheelchair dependent; did not have a progressive disease or psychiatric problem; and had sufficient understanding of the Dutch language to understand the purpose of the study and the testing methods. In this study, we excluded participants with known preexisting cardiovascular disease, diabetes mellitus, or cardiac diseases from our analyses. We included participants on whom we obtained data for blood pressure and HRrest and HRpeak on at least 2 of the 5 occasions.
Design
We collected data on 5 different occasions: at the start of active rehabilitation, when participants could sit for 3 to 4 hours (start of rehabilitation); 3 months later (3 months); at discharge from inpatient rehabilitation (discharge); 1 year after discharge (1 year); and 5 years after discharge (5 years). Trained research assistants with paramedical backgrounds collected the data using standardized procedures at the different sites.
Cardiovascular Parameters
A physician recorded resting systolic (SAP) and diastolic (DAP) arterial pressures using a manual sphygmomanometer while participants were seated in their wheelchair. The measurements were recorded once, at the same time of the day during every visit. Participants were asked to eat a light meal only, to refrain from smoking and drinking caffeine or alcohol for 2 hours before testing, and to void their bladders. Participants continued taking their regular medications. We determined prevalence of hypotension using World Health Organization (WHO) criteria 21 : for men, SAP <110 mm Hg; for women, SAP<100 mm Hg. We recorded HRrest and HRpeak using a heart rate monitor (Polar Sport tester, Polar Electro Inc, Kempele, Finland) before and during a graded maximal aerobic capacity test. We determined the prevalence of resting bradycardia (using 2 cutoffs: <60 BPM and <50 BPM) as well as the prevalence of elevated HRrest (>80 BPM) and tachycardia (>100 BPM). Prevalence data are reported for the SCI group as a whole and for 3 subgroups defined according to lesion level.
Measures of maximal aerobic capacity were obtained during a standardized peak wheelchair exercise test on a treadmill. 22 The test was preceded by 5 minutes of rest, seated in the testing wheelchair. As before, participants had a light meal before testing, abstained from caffeine and alcohol for at least 2 hours before the test, and emptied their bladder before the test. HRrest was determined as the mean over the last 30 s of this 5-minute rest period (this rest period was not immediately followed by the onset of exercise to avoid an anticipatory heart rate response to upcoming peak physical activity). During the determination of the maximal capacity, the speed of the treadmill stayed constant and was set at 2 km/h for individuals with tetraplegia and at 4 km/h for individuals with paraplegia; 3 km/h was used in cases where the other options were too slow or too fast for the individual. The treadmill incline was increased by 0.36° every minute until the test was terminated because of exhaustion or inability to keep pace with the speed. The complete testing protocol and equipment used for the aerobic capacity test were described previously by Kilkens et al. 22 HRpeak was determined as the highest 5-s average HR measured during the entire test.
Personal and Lesion Characteristics
We collected age, gender, and lesion characteristics at the start of rehabilitation and at discharge. A physiatrist determined lesion characteristics (level and motor/sensory completeness) using AIS criteria. 20 Cardiac sympathetic innervation arises from spinal segments T1 to T5. 5 Therefore, participants were divided into 3 subgroups according to lesion level at discharge from rehabilitation: individuals with cervical lesions (cervical); lesions at the levels T1 to T5 (high thoracic), and lesions at T6 or below (low). In the cervical lesion group, supraspinal control to sympathetic pathways to both the heart and vascular system can be impaired; the high thoracic group can have damage to some sympathetic pathways to the heart and the main components of the vascular system in terms of blood pressure control; the low injury level group will have spared control of the sympathetic pathways to the heart and major vascular beds controlling blood pressure.4,5 We defined a lesion as complete when participants were graded as AIS A and incomplete when they were graded as AIS B, C, or D at discharge from rehabilitation.
Statistical Analyses
We determined descriptive statistics (means and standard deviations [SD]) of SAP, DAP, HRrest, and HRpeak for all test occasions. We studied changes in SAP, DAP, HRrest, and HRpeak using random coefficient analysis (MLwiN),23,24 with 3 levels (center, participant, and test occasion). The dependent variables of this regression analysis were the blood pressure or heart rate variables. First, we modeled SAP, DAP, HRrest. or HRpeak over time using time periods as categorical variables (dummy) with discharge as the reference to be able to distinguish between changes during and after rehabilitation. The regression coefficient for a time dummy describes the predicted change in blood pressure or heart rate over that time period.
Second, we investigated the longitudinal relationships between blood pressure or heart rate with lesion and personal characteristics using univariate analyses. Lesion level (2 dummies with the low lesion group as reference and then in a subsequent analysis with the cervical lesion group as reference to be able to compare all 3 groups), motor completeness of the lesion (incomplete = 0; complete = 1), age (continuous variable), and gender (women = 0; men = 1) were used as independent variables in this second phase of analysis. Independent variables that showed a P value <0.1 were added to the multivariate model. We then used a backward elimination technique until only significant determinants remained (P < 0.05). This final multivariate model, however, always included the 2 lesion dummy variables using the low lesion group as reference.
To study the interaction effects of personal or lesion characteristics with time, the time dummies, the personal or lesion characteristics, and their interactions were entered in models with SAP, DAP, HRrest, and HRpeak as dependent variables.
We performed the same analyses with a binomial random coefficient analysis to investigate the change in the percentage of participants that were hypotensive (no hypotension = 0; hypotension = 1) during and after rehabilitation. These analyses were also performed for bradycardia (<60 BPM and <50 BPM), elevated heart rate (>80 BPM), and tachycardia (>100 BPM).
Results
Participants
A total of 197 individuals participated in the study on 2 or more test occasions. Participant characteristics are described in Table 1. The total number of participants per test/occasion (blood pressure and maximal aerobic capacity test) varied between 59 and 194 (Table 2). Of all participants at discharge, 40% had a cervical lesion, 19% a high thoracic lesion, and 41% a low level lesion. At discharge, 47% of the participants had a complete lesion (AIS A), and 53% had incomplete lesions (AIS B, C, or D). The average age at discharge was 40.0 years (SD = 14.1) and the average time since injury was 322.4 days (SD = 159.7). In our sample, 74% were male.
Participant Characteristics.
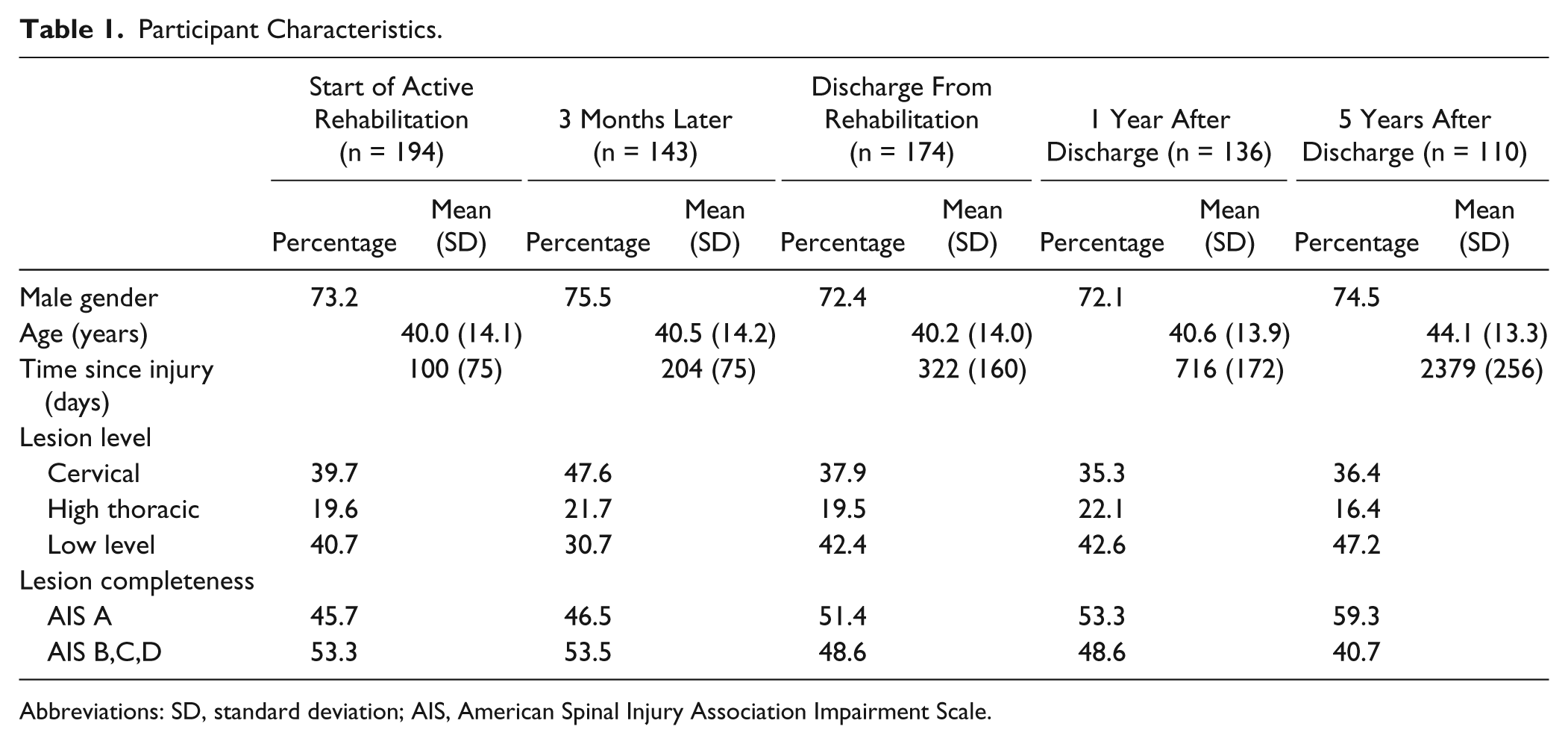
Abbreviations: SD, standard deviation; AIS, American Spinal Injury Association Impairment Scale.
Cardiovascular Variables a at All Test Occasions According to Lesion Level.
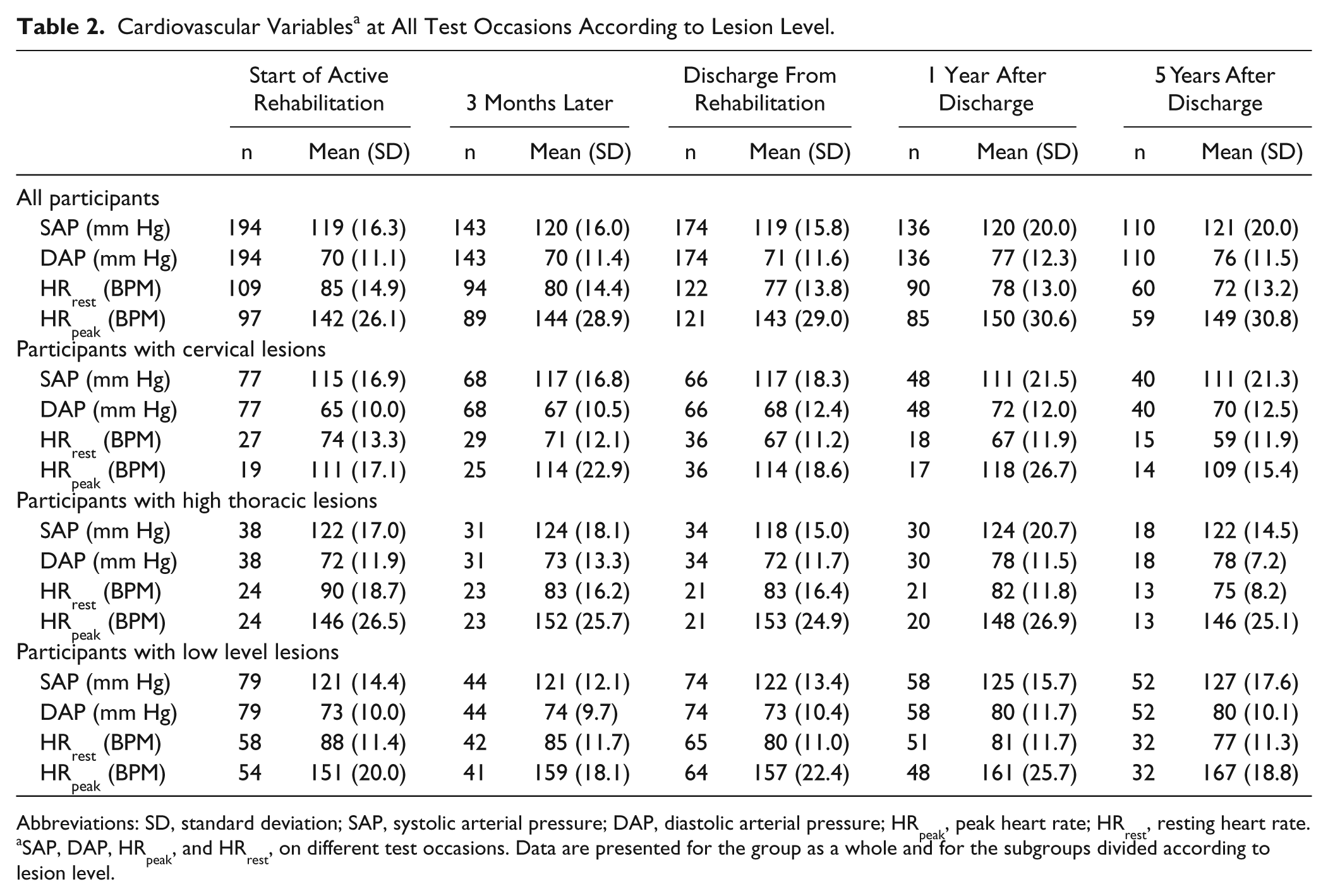
Abbreviations: SD, standard deviation; SAP, systolic arterial pressure; DAP, diastolic arterial pressure; HRpeak, peak heart rate; HRrest, resting heart rate.
SAP, DAP, HRpeak, and HRrest, on different test occasions. Data are presented for the group as a whole and for the subgroups divided according to lesion level.
Time Course and Determinants of Resting SAP and DAP
The time course of changes in SAP and DAP can be seen in Table 2. We found no significant change over time in SAP (Figure 1 and Table 3). DAP did not change during the period of rehabilitation but increased in the first 5 years after discharge. Of the personal and lesion characteristics, age and lesion level were related to both SAP and DAP. For every 10-year increase in age, the SAP increased by 3.6 mm Hg, and the DAP increased by 1.7 mm Hg (Table 3). Both SAP and DAP were significantly lower in those with cervical lesions when compared with those with high thoracic and low level lesions. Also, the DAP in the high thoracic group was significantly lower compared with that in the low level group (Figures 1A and 1B).
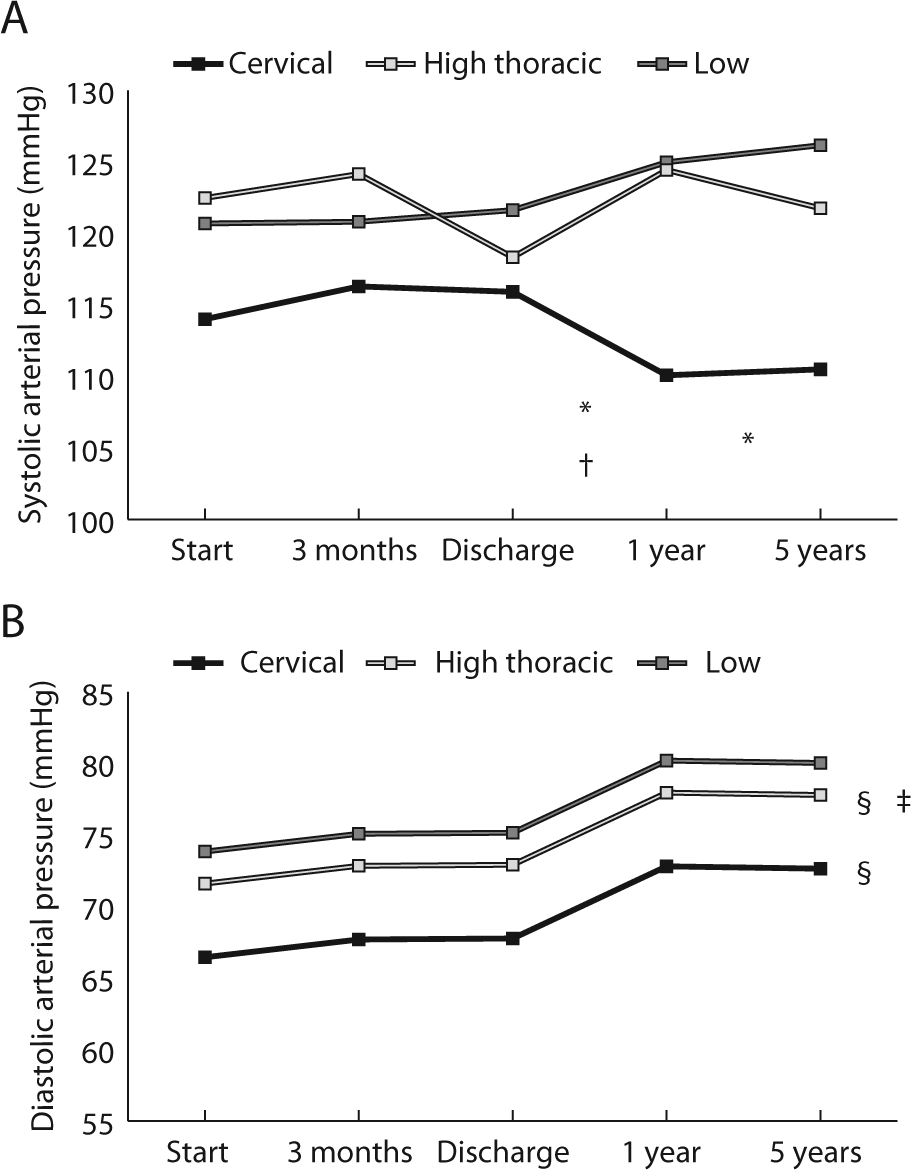
Regression model for systolic and diastolic arterial pressures (SAP and DAP): A. Modeled interaction between time course and lesion level for SAP. The change over time from discharge to 1 year is significantly different between the cervical and low lesion groups (*) and between cervical and high thoracic groups (†). The change over time from discharge to 5 years is significantly different between cervical and low lesion groups (*). In the cervical group, SAP decreases after discharge, whereas in the high thoracic and low level lesion group, it increases. B. Modeled time course for DAP during and after rehabilitation. We did not find a significant interaction effect between time and lesion level (indicated by the parallel lines). DAP is significantly lower in both cervical and high thoracic groups compared with the low lesion group (§) and significantly higher in the high thoracic lesion group compared with the cervical group (‡).
Blood Pressure and Heart Rate Regression Analyses. a
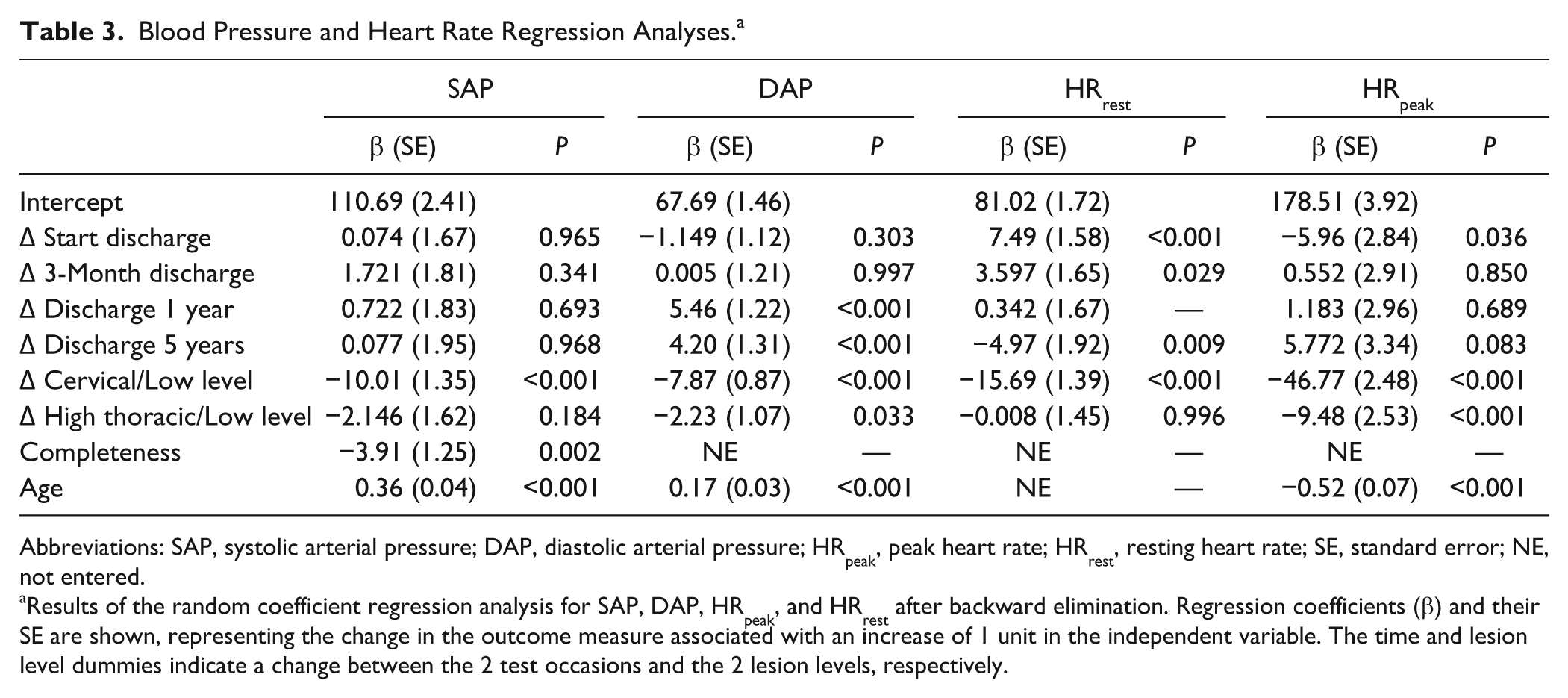
Abbreviations: SAP, systolic arterial pressure; DAP, diastolic arterial pressure; HRpeak, peak heart rate; HRrest, resting heart rate; SE, standard error; NE, not entered.
Results of the random coefficient regression analysis for SAP, DAP, HRpeak, and HRrest after backward elimination. Regression coefficients (β) and their SE are shown, representing the change in the outcome measure associated with an increase of 1 unit in the independent variable. The time and lesion level dummies indicate a change between the 2 test occasions and the 2 lesion levels, respectively.
We found an interaction effect between time and lesion level for SAP but not for DAP. In those with cervical lesions, SAP decreased after discharge, whereas in those with low lesions it increased. Also, the time course of the blood pressure change between discharge and 1 year was different in those with cervical and high thoracic lesions; SAP in those with high thoracic lesions increased from discharge to 1 year (Figure 1A), whereas in those with cervical lesions, it did not. The time course from discharge to 5 years after did not differ between those with cervical and high thoracic lesions. No other interaction effects between time and any personal or lesion characteristics were found.
Time Course and Determinants of HRrest and HRpeak
HRrest and HRpeak over time can be seen in Table 2. We did not find a significant change over time in HRpeak (Figure 2 and Table 3). HRrest decreased significantly during inpatient rehabilitation and decreased further from time of discharge to 5 years after discharge. Of the personal and lesion characteristics, only lesion level was related to both HRpeak and HRrest after the backward elimination technique. Both HRpeak and HRrest were lower in those with cervical lesions when compared with those with low level and high thoracic lesions (Figures 2A and 2B). Also, HRpeak was lower in those with high thoracic lesions when compared with those with low lesions (Figure 2B). Age was related to HRpeak only; HRpeak decreased by 5.2 BPM for every 10-year increase in age. We found no significant interaction effects between time and any personal or lesion characteristics.
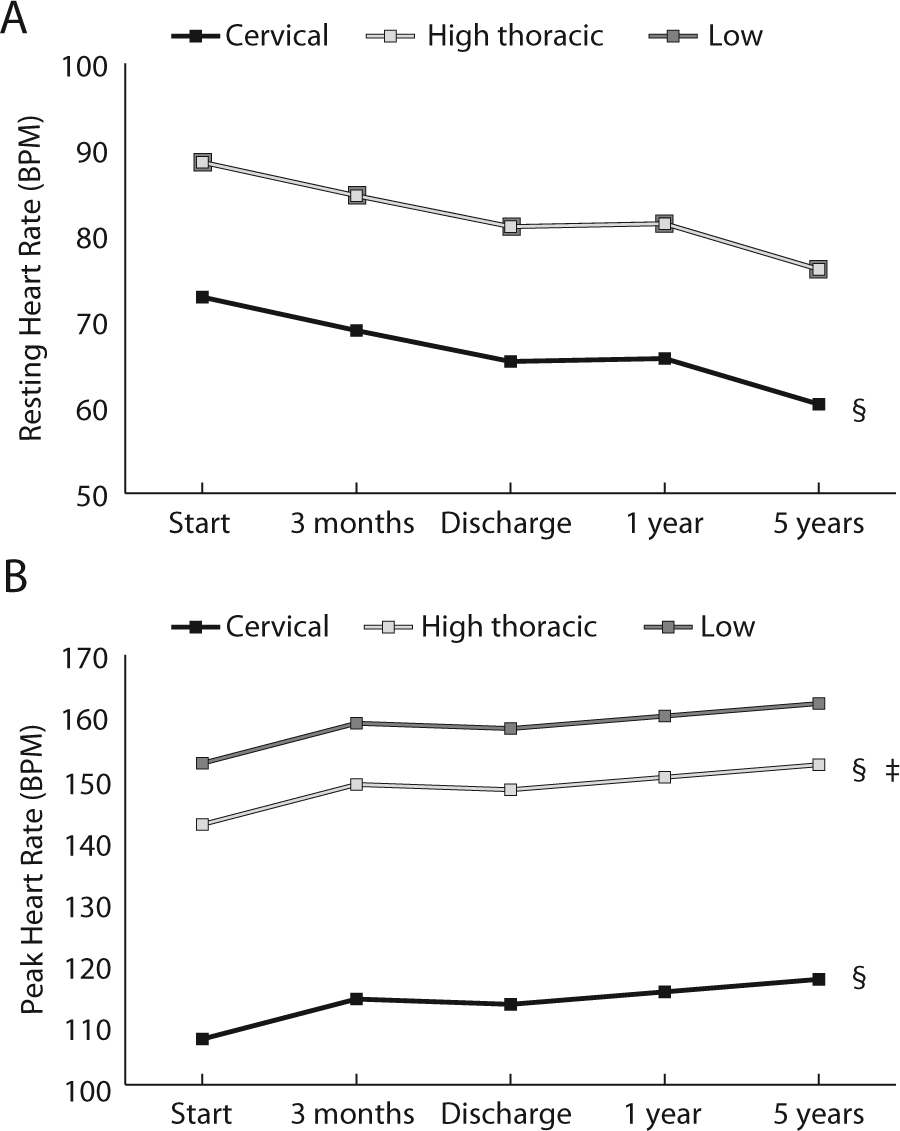
Regression model for resting and peak heart rate (HRrest and HRpeak): A. Modeled time course of HRrest during and after rehabilitation. There is a minimal difference between low level and high thoracic groups (the lines are overlapping). We did not find a significant interaction effect between time and lesion level (indicated by the parallel lines). The cervical group has significantly lower HRrest than the low lesion group (§). B. Modeled time course of HRpeak during and after rehabilitation. We did not find a significant interaction effect between time and lesion level (indicated by the parallel lines). The cervical and high thoracic groups have significantly lower HRpeak compared with the low lesion group (§). The high thoracic group has significantly higher HRpeak compared with the cervical lesion group (‡).
Prevalence of Hypotension
Overall, 33% of the participants had hypotension at the start of rehabilitation according to WHO criteria. 21 Table 4 shows the prevalence of hypotension on the different test occasions and for different lesion groups.
Prevalence of Hypotension, Bradycardia, Elevated Heart Rate, and Tachycardia on All Test Occasions According to Lesion Level. a
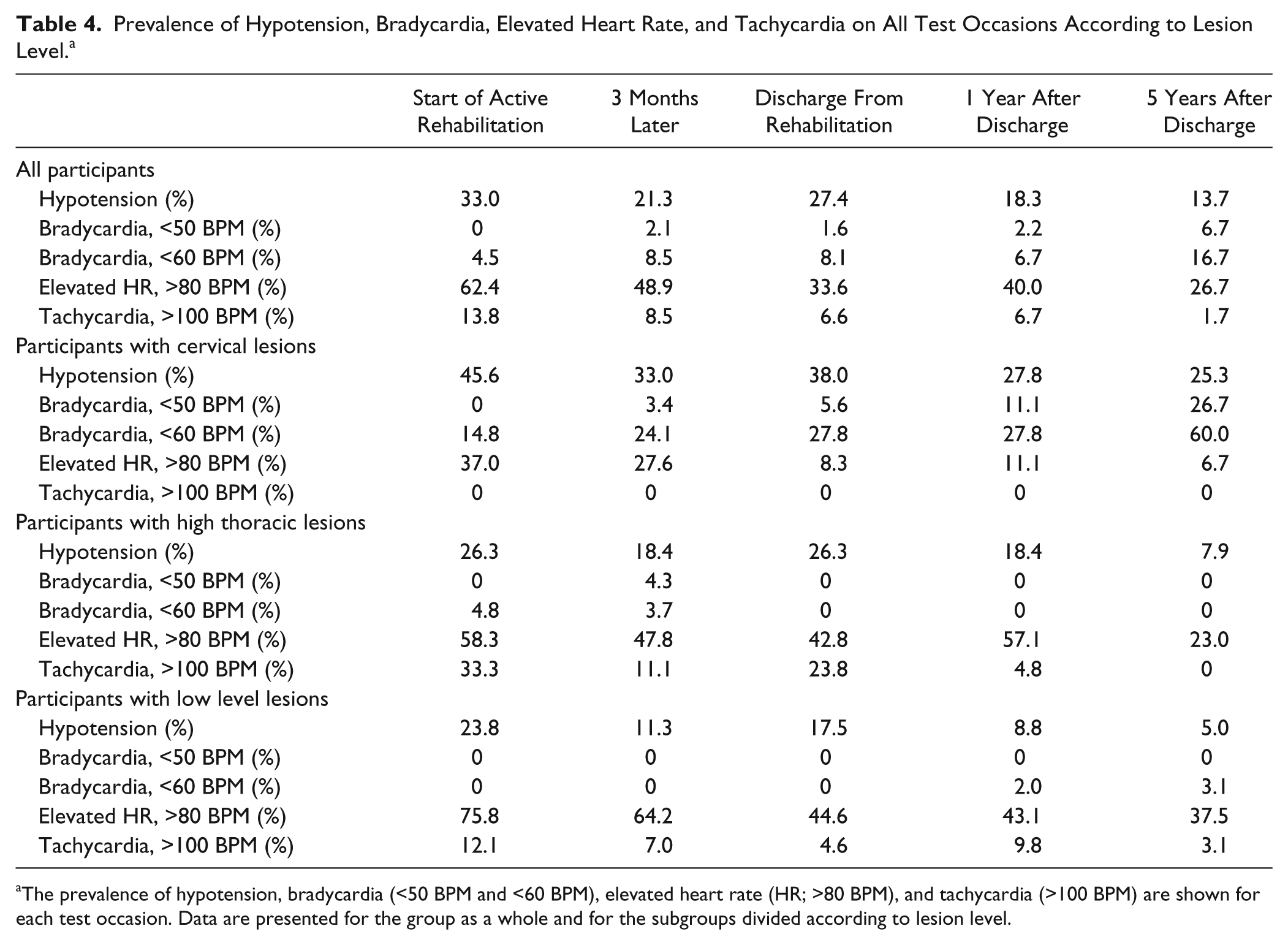
The prevalence of hypotension, bradycardia (<50 BPM and <60 BPM), elevated heart rate (HR; >80 BPM), and tachycardia (>100 BPM) are shown for each test occasion. Data are presented for the group as a whole and for the subgroups divided according to lesion level.
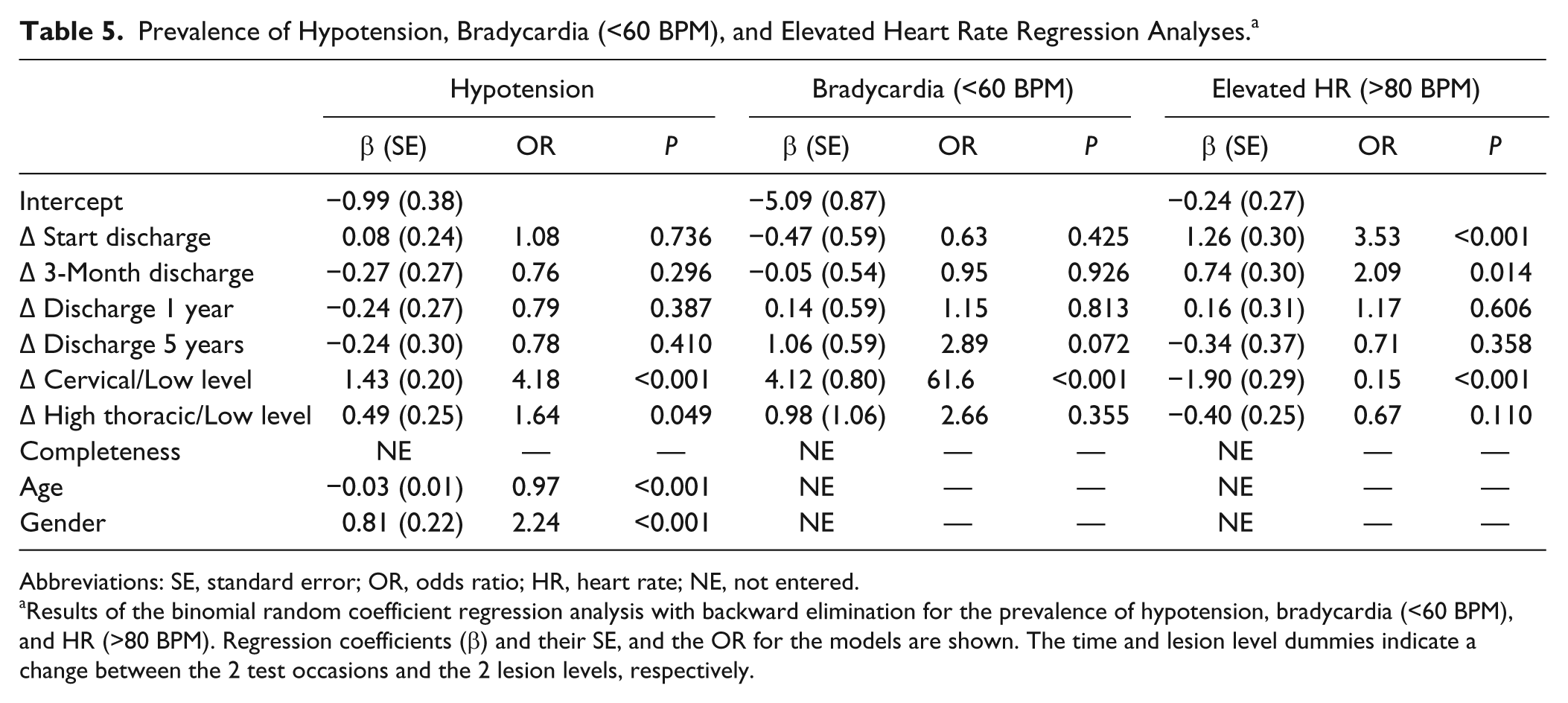
We did not find a significant change in the prevalence of hypotension over time. Table 5 shows the final regression model. The odds of having hypotension were 4.18 times greater in those with cervical lesions than in those with low lesions and 1.64 times greater in individuals with high thoracic lesions than in those with low lesions. The odds were 2.70 times greater in individuals with cervical lesions than in those with high thoracic lesions. Also, the prevalence of hypotension decreased with age; the odds were 0.75 times greater for every 10-year increase in age. Men had 2.24 times the odds of having hypotension compared with women. Completeness of injury as determined by AIS classification did not influence the odds of having hypotension.
Prevalence of Hypotension, Bradycardia (<60 BPM), and Elevated Heart Rate Regression Analyses. a
Abbreviations: SE, standard error; OR, odds ratio; HR, heart rate; NE, not entered.
Results of the binomial random coefficient regression analysis with backward elimination for the prevalence of hypotension, bradycardia (<60 BPM), and HR (>80 BPM). Regression coefficients (β) and their SE, and the OR for the models are shown. The time and lesion level dummies indicate a change between the 2 test occasions and the 2 lesion levels, respectively.
Prevalence of Bradycardia, Elevated Heart Rate, and Tachycardia
Because of the low number of positive outcomes for bradycardia (<50 BPM) and tachycardia (>100 BPM), it was not possible to run the binomial random coefficient analysis on these variables. The descriptive values for these prevalence data are shown in Table 4. We did not find a significant change in the prevalence of bradycardia (<60 BPM) over time (Table 5). The odds of having bradycardia was 61.50 times greater in those with cervical lesions than in those with low lesions, and 23.08 times greater in those with high thoracic lesions than in those with low lesions. Age, gender, and completeness of injury by AIS classification did not influence the odds for bradycardia. The prevalence of an elevated HR decreased during and after rehabilitation (Table 4). The odds of having an elevated HR was 0.15 times lower in those with cervical lesions than in those with low lesions. Age, gender, and completeness of injury by AIS classification did not influence the odds for an elevated HR. Table 5 shows the final regression model.
Discussion
This is the first study to investigate the time course of blood pressure, HRrest, and HRpeak over the first 5 years after SCI using a longitudinal approach and to provide normative data in a relatively large cohort that is subdivided according to time after injury over 5 years and by lesion level. We are also the first to not only identify associations between lesion or personal characteristics and cardiovascular dysfunction, but also to quantify the magnitude of increased risk associated with these traits. We found that the prevalence of hypotension did not change during and after rehabilitation: however, it was related to lesion level (positively), age (negatively), and gender. We found no change in SAP over time, but DAP increased during the first 5 years after discharge. SAP and DAP were related to age (positively) and lesion level (negatively). The time course for SAP was different between the different lesion levels. HRpeak did not change over time, whereas HRrest decreased during and after rehabilitation. Lesion level was negatively associated with both HRpeak and HRrest, and age was negatively associated with HRpeak. The prevalence of bradycardia remained the same over time, whereas the prevalence of elevated HR improved (decreased) during and after rehabilitation. The prevalence of bradycardia (<60 BPM) was positively correlated with lesion level, and the prevalence of elevated HR (>80 BPM) was negatively correlated with lesion level.
We found a greater prevalence of hypotension in those with cervical and high-thoracic lesions than in those with injury levels below T5. The prevalence was greatest in those with cervical lesions. This is not surprising because individuals with cervical lesions can lose control over spinal sympathetic pathways to the heart and vasculature, and therefore, a consequent reduction in resting blood pressure and a greater susceptibility to hypotension might be expected. Individuals with high thoracic lesions can retain sympathetic control over the heart but control of the splanchnic vasculature may be impaired. This can explain the effective titration of resting blood pressure across lesion levels after SCI. Age and gender influenced the odds of hypotension after SCI, whereby older individuals had lower odds compared with younger individuals, and men had higher odds compared with women. In able-bodied individuals, increasing age has been shown to be related to higher blood pressures as a result of arterial stiffening25,26 and a higher prevalence of hypertension, 27 and this is in agreement with our finding of a lower prevalence of hypotension with aging after SCI. We do not know why men had higher odds of hypotension compared with women. We considered the possibility that the men in our study had more severe injuries than the women, but this was not the case. The completeness of injury based on AIS score had no effect on the prevalence of hypotension, indicating that AIS completeness is not a strong predictor for impairment to cardiovascular autonomic pathways, as previously reported by our laboratory.6,28,29
The increase in SAP found in those with low level and high thoracic lesions could be related to adopting a more sedentary lifestyle after injury, as shown in a subset of this study population. 30 The failure of SAP to increase with time in those with cervical SCI can be explained by the lack of sympathetic control to the heart and vasculature. In this case, an increase in SAP as a result of a more sedentary lifestyle is unlikely because this increase has been related to elevated sympathetic tone and coordinated control of sympathetic tone after cervical SCI is often lost. 31 This finding is in agreement with several cross-sectional studies of individuals with chronic SCI,16,32-35 which reported that resting BP and HR continued to be lower in those with high lesions compared with those with lower lesions in the chronic phase after injury. Some studies6,35 report lower SAP, DAP, and HRrest compared to our findings, which can be explained by the fact that their studies included a greater proportion of individuals with complete injuries compared with our mixed population. Other studies with lesion characteristics more similar to that in our study in terms of completeness of injuries found similar SAP, DAP, and HRrest.18,36,37 However, a major limitation of these studies is that they do not show changes in cardiovascular parameters over time but rather a snapshot in the chronic phase of injury.
The HRpeak in individuals with cervical lesions was low (111 ± 17.1 BPM at the start of rehabilitation and 109 ± 15.4 BPM 5 years after discharge). This diminished heart rate response to exercise is most likely a result of impaired supraspinal control of cardiac sympathetic outflow in this group. In the absence of coordinated sympathetic control of heart rate, the remaining heart rate response to exercise in these individuals is presumably largely mediated by vagal withdrawal. Indeed, the HRpeak in this group is comparable to that of able-bodied controls during complete vagal blockade (approximately 100 BPM). 17
The variability in HRpeak in this group is likely to be a result of the fact that not all these participants had a complete injury, and therefore, some of them would have preserved cardiovascular autonomic function and higher HRpeak.
The greater prevalence of bradycardia in those with cervical lesions underscores the vagal predominance in this group, in whom there is a greater potential for injury to descending spinal cardiac sympathetic pathways. Similarly, those with low lesions had higher HRrest. It is interesting to note that the prevalence of elevated heart rates in the low lesion group decreased with time after injury, and there are numerous factors (including the influence of rehabilitation, recovery of cardiovascular deconditioning, improved blood volume status, and medication adjustment) that could underlie this change. The prevalence of bradycardia in those with cervical injuries was a consistent feature, compatible with lack of improvement in cardiac sympathetic function over time, and this is also reflected in the failure of the impaired HRpeak to improve with time in this group.
There are no prior longitudinal studies that have examined changes in these cardiovascular parameters with time after injury. However, there are cross-sectional studies that predominantly focus on chronic SCI (>1 year after injury). Prevalence of resting hypotension is not frequently reported in these studies. One study showed a prevalence of 33% in their overall sample (482 individuals with SCI) and 51% in a subset of those with AIS complete tetraplegia, but this was based on self-reported hypotension (by a questionnaire). 38 The prevalence of OH is more commonly reported. OH is a variable related to resting blood pressure measured in a seated position (as it incorporates a gravitational effect) and is defined as a drop in SAP of 20 mm Hg or a drop in DAP of 10 mm Hg or more when moved from supine to seated/upright. We have previously reported a prevalence of OH of 50% in individuals with cervical lesions 6 and 47% in individuals with lesions above T5. 37 Recently, Sisto et al 18 reported a lower prevalence of OH of 21% in individuals with mostly cervical injuries. This lower prevalence can be explained by including only individuals with highly preserved function (AIS C and D). In these types of injuries, spinal sympathetic pathways may be more likely to remain intact, and individuals might thus be protected from the cardiovascular abnormalities that often accompany high level injuries. Even though they did not report the prevalence of resting hypotension, this group found lower resting blood pressure in those with cervical lesions than in those with high and low thoracic injuries, suggesting a higher risk for resting hypotension in this subgroup.
Several studies have investigated cardiovascular adaptations after SCI.39,40 They found an increased leg vascular resistance in individuals with SCI, even though sympathetic control over the heart and vasculature was impaired, as a result of vascular 40 and hormonal adaptations after injury. 39 These adaptations were proposed to counteract the deleterious effects of the loss of normal sympathetic regulation of blood pressure. The findings of the present study indicate that these adaptations might not be sufficient to prevent hypotension; it is tempting to speculate that without these adaptations, we might predict even more severe and prevalent hypotension.
Strengths and Limitations
The main strength of the present study is the large sample size, incorporating all levels and degrees of completeness of injury, and its longitudinal design. All participants were prone to cardiovascular deconditioning because of the use of a wheelchair, making level of injury and autonomic dysfunction the primary factors underlying any differences in blood pressure and heart rate. No previous studies have investigated the time course of changes in these cardiovascular variables after SCI. An additional strength of the study is the identification not only of associations between lesion or personal characteristics and cardiovascular dysfunction but the quantification of the magnitude of increased risk associated with these traits.
One limitation of this study is that not all participants completed the test protocols at all time points. Although, of course, this is not the ideal scenario, it is the clinical reality of these types of studies. We chose to analyze the full data set with a statistical approach that is validated for use with incomplete data sets.23,24 This approach has 2 advantages: the statistical power is greater, and it avoids the selection bias encountered by analyzing only those who performed all measurements (often healthier persons or those who are unemployed, for example). This selection bias would also not be desirable when it comes to the generalizability of the results to clinical practice. However, we also completed repeated-measures analysis of variance on only those participants who completed all testing sessions (data not shown) and found no meaningful differences between the outcomes of the 2 approaches, further supporting the validity of our findings. Similarly, another limitation is the higher drop-out rates for the maximal aerobic capacity test, leading to a lower sample size for this variable. 19 The heart rate data may, therefore, be biased toward those who were willing and/or able to perform the maximal aerobic capacity test (without cardiovascular or musculoskeletal complaints and likely excluding those using power wheelchairs), potentially overestimating HRpeak and HRrest.
Our statistical analyses of the prevalence data for bradycardia and elevated heart rate/tachycardia were limited by the low number of positive responses. This was particularly so for the subgroup analyses and for the prevalence of bradycardia (<50 BPM) and tachycardia (>100 BPM). The analysis of the prevalence of bradycardia (<60 BPM) was also somewhat constrained by this issue and should be interpreted with caution.
Another limitation is that some of the resting blood pressure measurements might have been obscured by episodes of AD. Because it is challenging to accurately determine the occurrence of these episodes retrospectively, it is conceivable that some resting blood pressure values might be artificially elevated by episodes of AD. This could lead to an overestimation of the mean SAP and DAP in individuals with cervical and high thoracic lesions, and consequently an underestimation of the prevalence of hypotension in these groups. To estimate how often this occurred, in those at risk for AD (lesions at or above T5), we qualified AD as a SAP value of 30 mm Hg above the average SAP over the test occasions for that individual. Using this approach, we estimated that episodes of AD occurred in a total of 23 tests out of 757 tests analyzed. Since we could not be sure that these represent true episodes of AD because of the retrospective nature of this study, we included all SAP measurements in our analyses. However, it should be kept in mind that this could lead to a modest underestimation of the prevalence of hypotension in our sample.
Implications
At the start of active rehabilitation, 33% of the participants had resting hypotension, and this did not significantly improve over time, at least until 5 years after rehabilitation. Individuals with cervical lesions are most prone to resting hypotension and bradycardia when compared with those with high thoracic and low level injuries. In previous studies, it has been shown that OH is even more prevalent (~50%) in this population of individuals with cervical or high thoracic SCI.5,31 This combination of resting hypotension and OH can lead to intolerably low blood pressures, with subsequent reductions in cerebral blood flow, presyncope, syncope, cognitive impairment, and fatigue. 37 We also showed that the impaired HR responses to exercise in those with cervical lesions do not improve over time, with a prolonged negative impact on exercise tolerance and ability to participate in physical activity. These complications have a significant negative impact on quality of life in this population. 11 The assumption that these are issues related primarily to deconditioning in the acute stage after injury and that they recover with time is incorrect and should be challenged. These findings suggest that management of (orthostatic) hypotension and cardiac dysfunction should remain a priority for those with injuries above T5 into the chronic phase of injury.
We are the first to provide data on the time course of cardiovascular changes after injury. These data can be used to guide clinical practice and to place changes in cardiovascular function as a result of interventions in perspective, relative to that expected with standard treatment alone, as has been shown for motor and sensory function.41-43 We also identified those individuals at greatest risk for cardiovascular dysfunction considering age, gender, and level and completeness of injury. These specific odds of cardiovascular dysfunction may be useful to guide targeted patient therapy.
Footnotes
Acknowledgements
We would like to thank the participants, the 8 participating rehabilitation centers, and especially the research assistants for collecting the data: Sacha van Langeveld (De Hoogstraat, Utrecht); Annelieke Niezen and Peter Luthart (Rehabilitation Center Amsterdam); Marijke Schuitemaker (Het Roessingh, Enschede); Karin Postma, Rogier Broeksteeg, and Jan Kamberg (Rijndam Revalidatiecentrum, Rotterdam); Jos Bloemen (Hoensbroeck Revalidatiecentrum, Hoensbroek); Hennie Rijken (Sint Maartenskliniek, Nijmegen); Ferry Woldring (Beatrixoord, Haren); and Linda Valent (Heliomare, Wijk aan Zee).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by a Heart and Stroke Foundation of Canada New Investigator Award (VEC) and by the Dutch Health Research and Development Council, ZonMw Rehabilitation program, grant no. 1435.0003 and 1435.0025.
