Abstract
Background. Leukoaraiosis describes ischemic white matter lesions, a leading cause of gait disturbance in the elderly. Objective. Our aim was to improve gait and balance in patients with leukoaraiosis by combining a single session of transcranial direct current stimulation (tDCS) and physical training (PT). Methods. We delivered anodal tDCS over midline motor and premotor areas in 9 patients with leukoaraiosis. Patients underwent gait and balance training during tDCS stimulation (real/sham). This was repeated 1 week later with the stimulation crossed-over (sham/real) in a double-blind design. Assessments included gait velocity, stride length, stride length variability (primary gait outcomes), and a quantitative retropulsion test (primary balance outcome). Results. Combining tDCS and PT improved gait velocity, stride length, stride length variability, and balance (all at P ≤ .05). Overall, training without tDCS showed no significant effects. Conclusions. Combined anodal tDCS and PT improves gait and balance in this patient group, suggesting that tDCS could be an effective adjunct to PT in patients with leukoaraiosis, for whom no treatment is currently available.
Keywords
Introduction
Leukoaraiosis (white matter lesions or small vessel disease) are hyperintensities in the cerebral subcortical white matter and are associated with gait and balance dysfunction and falls in the elderly. 1 There are no evidence-based treatments for the gait disorder and balance dysfunction in these patients.
Physical therapy is an established treatment for patients with neurological gait disorders and has been used in conjunction with transcranial direct current stimulation (tDCS) to augment the effects of gait training 2 and lower limb rehabilitation 3 in patients with hemiparesis following established cortical strokes. To date, the combination of noninvasive stimulation with simultaneous gait training has not been studied in patients with primary gait disorders such as leukoaraiosis.
Primary motor and premotor cortices are relevant to the control of gait in relation to leukoaraiosis. 4 We attempted to facilitate neural activity in this network by combining physical training with tDCS of the primary motor leg area and premotor cortex bilaterally. 5
The primary gait outcomes of this exploratory pilot study were gait velocity and stride length, which correlate with disease severity. 6 Our primary balance outcome was recovery of stance in the retropulsion test, postural instability being a feature of leukoaraiosis, and a common cause of falls in these patients. 7 Secondary outcomes were test duration (a surrogate measure of gait velocity), and angular trunk velocity and amplitude.
Methods and Materials
Patients
Local ethics approval and participant informed consent was obtained. Patients were prospectively recruited over a 12-month period. Inclusion criteria consisted of a gait and balance disturbance with onset >1 year, in patients who responded affirmatively to the question: “Have you noticed changes in your normal walking or balance?” and had a radiological diagnosis of leukoaraiosis (Fazekas score 2 to 4, 8 Figure 1A for representative scan). Ten patients entered the study; 1 patient was unable to attend the second session and was excluded from the analysis. Brain scans were reviewed by 2 independent neuroradiologists. Patients with severe freezing, daily falls, dementia, or other conditions that could affect gait/balance were excluded. Eighteen healthy age-matched individuals took part as controls and performed only baseline tests (without tDCS). Whereas the main comparison was between affected individuals undergoing tDCS or sham, this control group was included to assess whether any gait or balance improvements were clinically meaningful. Patients’ mean age (79.4 years, range = 71-89 years) was not statistically significant from that of controls (mean age = 76.1 years, range = 66-89 years; P = .13). See Table 1 for demographic and clinical data. Based on published neurophysiological data, 5 for an analysis of variance (ANOVA) design a sample size calculation revealed that 8 patients or more would be sufficient to observe a proof-of-principle effect (α = .05 and power = 80%).
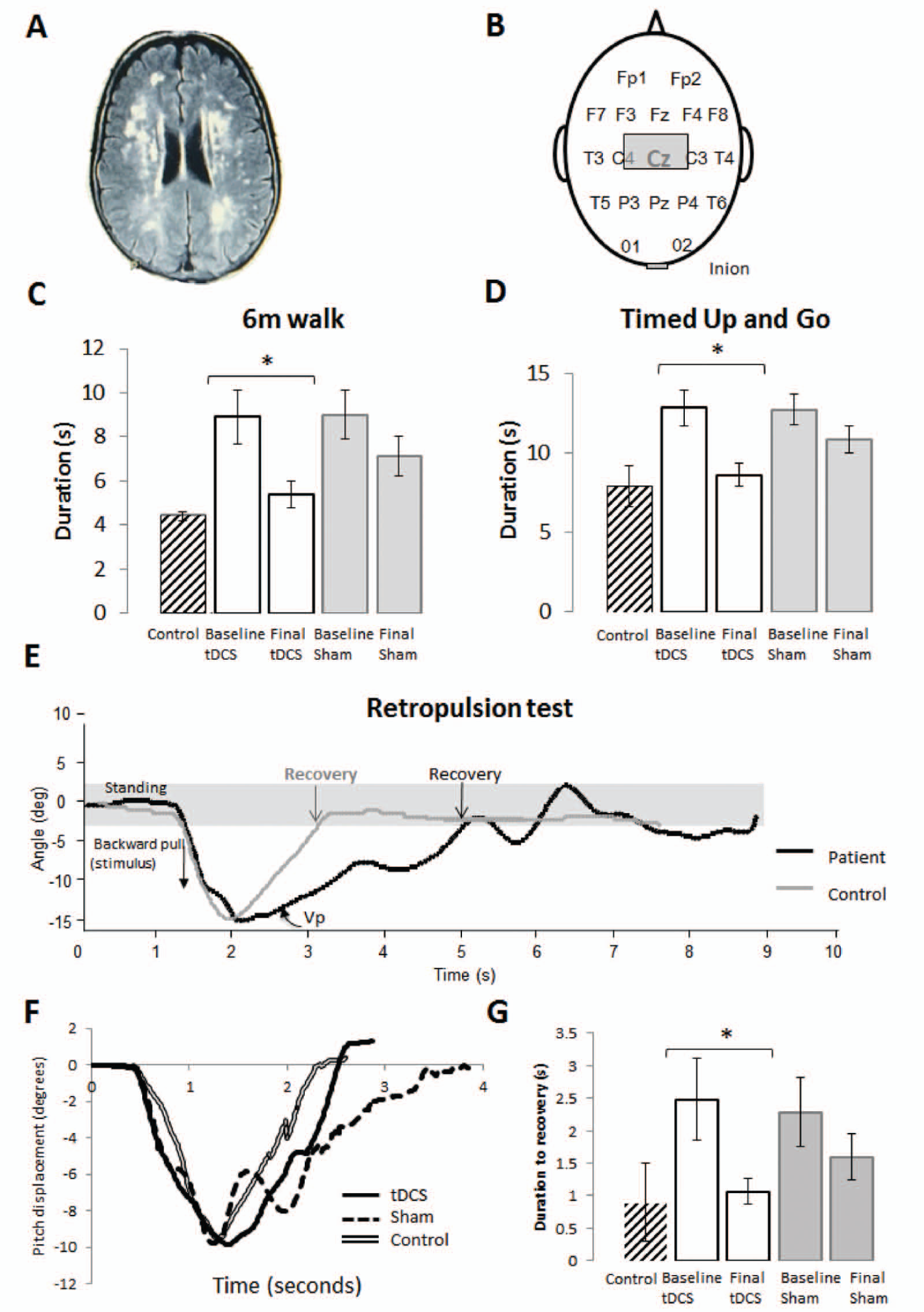
(A) Representative axial cut fluid attenuated inverted (FLAIR) sequence magnetic resonance image (1.5 T) of a patient that took part in this study showing multiple areas of white matter hyperintensity (Fazekas score 3). (B) Transcranial direct current stimulation (tDCS) electrode placement with the anode at Cz and cathode at the inion. (C) Group averages of duration for the 6 m walk and (D) Timed Up and Go (TUG) test for patients receiving either tDCS (n = 9) or sham (n = 9), before or after the crossover. Note that patients receiving tDCS in the first session were crossed over to receive sham 1 week later, and vice versa for those receiving sham in the first session. Vertical bars represent standard errors. *Denotes an effect of combining training + tDCS; P < .05. (E) Retropulsion test. Subjects received a forceful pull backward from the shoulders (stimulus). The time taken to recover pitch trunk displacement to ± 2 cm of baseline was recorded offline using the SwayStar, which is easy to measure objectively (recovery). We also recorded the peak pull velocity and displacement (to ascertain stimulus consistency) and peak recovery phase velocity within the first second following maximum pitch backward displacement. The peak velocity (Vp) within the first second following maximum peak pitch displacement was measured for the recovery phase in all subjects as a function of displacement over 100 ms. (F) Time taken to regain a pitch angular displacement of ±2 cm of baseline following a backward pull on the shoulders. *P < .01. (G) Group averages of retropulsion test responses. Vertical bars represent standard errors of the mean. *P < .01.
Demographic and Clinical Data for Patients and Control Subjects.
Subjects were randomized to receive either real tDCS or sham stimulation in the first session. All subjects were “crossed over” to receive either real or sham stimulation in the second session 1 week later. Both the examiner and patients were blinded to the tDCS protocol.
Outcome Measures
Subjects performed a 6-m walk, Timed Up and Go (TUG), and retropulsion test (Figure 1E), twice in each session: “baseline” and “final” assessments. Following “baseline” assessments, patients underwent physical training (PT) consisting of a 15-minute balance and gait training session (Figure 2) concurrently with stimulation (real or sham). Subjects then immediately repeated the gait and balance tests (“final” assessment). Assessments took on average approximately 15 minutes to complete.

Exercise protocol for patients in the physical training group.
Semiquantitative assessment consisted of the modified Tinetti test, which is a reliable measure of gait function in both healthy 9 and patient populations. 10 All tests were recorded on video camera (Veho Kuzo HD camcorder, Southampton, UK) to assess gait velocity, stride length, and stride length variability. Observational assessments of gait have been previously validated in the clinical setting. 11
Subjects were instructed to “walk as quickly, but safely, as possible.” The duration of walking during the middle 4 m of the 6-m walk (Figure 3B) was used to calculate mean gait velocity (m/s) in order to avoid variability from acceleration/deceleration effects. Stride length (distance from the initial contact of one foot with the ground to the following contact of the same foot) was measured against floor markers 15 cm apart, using frame-by-frame video analysis. Stride length variability was calculated using the variability index (CoVar = SD/mean × 100). Mean gait velocity for the 6-m walk obtained from the SwayStar (see below) was cross-referenced with the video data acquisition for validation (Supplementary Figure S1A, available online at nnr.sagepub.com/supplemental).
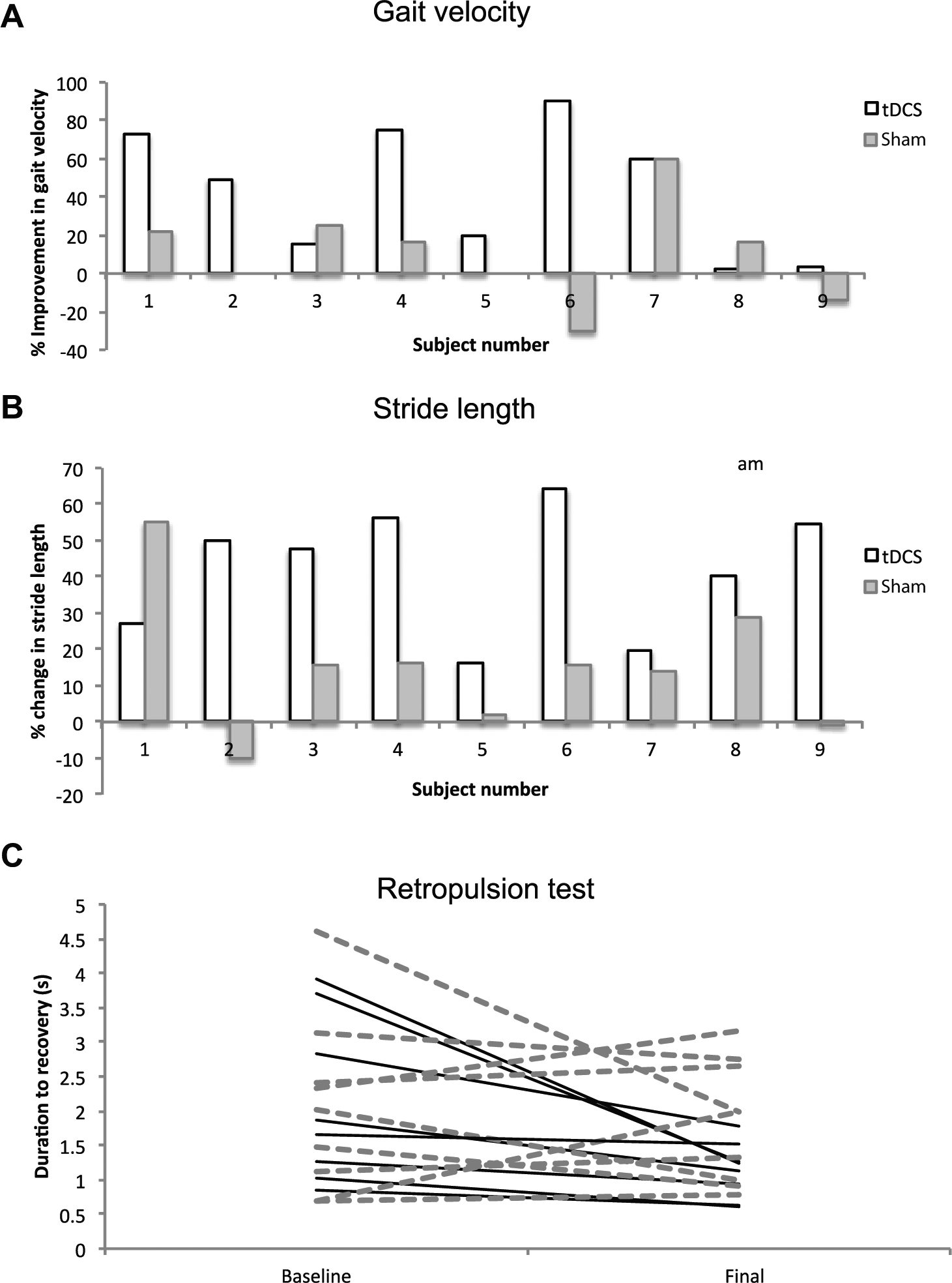
Individual patient data showing percentage change in gait velocity (A) and stride length variability (B), and individual responses to the retropulsion test at baseline and final assessments (C).
For the retropulsion test, 12 subjects stood in a comfortable stance position with feet at shoulder width, and eyes open. They were warned that they would receive a sudden and firm shoulder pull, delivered by the examiner standing directly behind the subject. The examiner then delivered a single pull using sufficient strength to force subjects to take at least one corrective step backward. The test was repeated a total of 3 times. The same examiner (DK) performed the retropulsion test on every subject to reduce the pull strength variability. The shoulder pull forces were not quantitatively controlled but recordings showed that subjects’ backward trunk velocity was approximately equal (mean backward velocity = 32.5 °/s, SD = 5.8). Trunk sway recordings were started just prior to the pull and terminated 5 seconds later, or until loss of balance occurred. Subjects were prevented from falling by the examiner and an assistant standing next to the subject.
Trunk Sway Measurement System
A device encompassing 2 digitally based angular velocity transducers arranged to measure angular trunk displacement and velocity in the roll (coronal) and pitch (sagittal) planes (SwayStar System, Balance International Innovations GmbH, Iseltwald, Switzerland) was used in all subjects. The transducers were encased in a lightweight Bluetooth cordless device attached to an elasticized motorcycle belt, which was easily fitted on the subjects back at the L2-3 level. Angular deviations were calculated using online-trapezoid integration of the angular velocities. 13 The device has been extensively used in patients with neurological diseases to assess gait and balance.14,15
Transcranial Direct Current Stimulation
Given that the cortical representation of gait is bihemispheric, we aimed to deliver anodal tDCS to the motor cortex of both hemispheres simultaneously. This is a novel montage that increases cortical excitability in lower limb areas in healthy subjects without significant effects over the cerebellum. 5 A DC stimulating rectangular saline-soaked sponge electrode (10 cm × 4 cm; surface area 40 cm2) was placed 10% to 20% anterior to Cz as measured from the midline of the stimulating electrode (Figure 1B). The reference electrode (4 cm × 4 cm) was positioned at the inion. A 2-mA current (current density 0.05 mA/cm2) was delivered by a Magstim Eldith DC stimulator (NeuroConn, Ilmenau, Germany) during the exercise session for 15 minutes. Sham stimulation was identical to real stimulation except that the current was delivered for only 30 seconds. 16 As part of a proof-of-principle study, each patient received a single session of active (and sham) tDCS rather than repeated treatment sessions.
Data Analysis
Video and SwayStar data were acquired simultaneously for all tests. For the video data, the first 3 and the last 2 steps of each gait cycle were eliminated from the data set to remove acceleration and deceleration effects.
We performed a 2-way repeated-measures ANOVA [INTERVENTIONtDCS,sham and PHASEbaseline,final] to evaluate overall effects of physical training with and without stimulation. Statistical analysis was carried out using SPSS 18.0 with significance set at P ≤ .05 (2-tailed).
Results
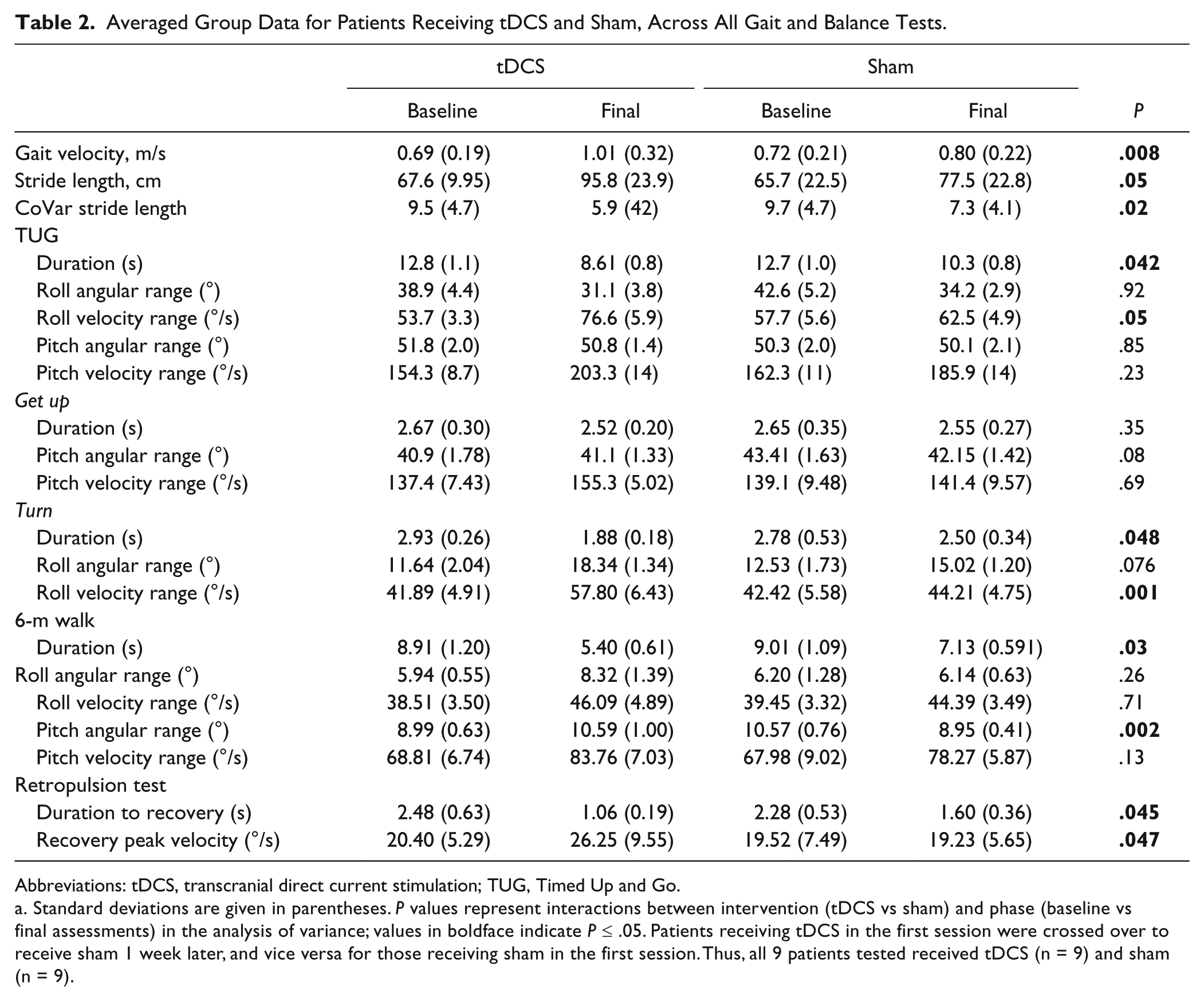
Patients performed significantly worse than controls in all tests based on the first assessment results. We observed no order effects for any of the variables measured (ie, no significant difference between baseline trials in the first and second sessions). Table 2 shows the absolute values, for baseline and final assessments, for patients receiving tDCS and sham across all outcome measures.
Averaged Group Data for Patients Receiving tDCS and Sham, Across All Gait and Balance Tests.
Abbreviations: tDCS, transcranial direct current stimulation; TUG, Timed Up and Go.
Standard deviations are given in parentheses. P values represent interactions between intervention (tDCS vs sham) and phase (baseline vs final assessments) in the analysis of variance; values in boldface indicate P ≤ .05. Patients receiving tDCS in the first session were crossed over to receive sham 1 week later, and vice versa for those receiving sham in the first session. Thus, all 9 patients tested received tDCS (n = 9) and sham (n = 9).
Gait Velocity, Stride Length, Stride Length Variability
As expected, gait velocity correlated well with stride length and stride length variability (Supplementary Figure S1, available online at nnr.sagepub.com/supplemental.
Mean “fast-walking” gait velocity in healthy controls was 1.69 m/s (SD = 0.30) and 0.70 m/s (SD = 0.24) in the leukoaraiosis group (paired t test, P < .01). We observed an interaction between tDCS and phase (ANOVA intervention phase; F = 11.7, P = .008) suggesting a combined benefit of tDCS with physical training (PT). A post hoc analysis revealed no effect of PT alone (baseline vs final assessment in the sham group; paired t test, P = .44). Percentage improvement in gait velocity in tDCS and sham groups for individual patients is shown in Figure 3A (paired t test between tDCS and sham, P = .02).
Patients had reduced stride length at first assessment compared with controls (paired t test, P = .047). We observed an interaction between tDCS and phase (F = 5.0, P = .05) suggesting a combined benefit of tDCS with PT. There was no isolated effect of PT alone (paired t test, P = .29). A histogram showing individual percentage improvements in stride length for both tDCS and sham is displayed in Figure 3B (paired t test between tDCS and sham, P = .006).
Stride length variability was greater in patients for the first assessments compared with controls (paired t test, P = .001). We observed an interaction between tDCS and phase (F = 8.2, P = .02). Post hoc analysis revealed no benefit of PT alone (paired t test, P = .52). The percentage improvement in stride length variability was significant for tDCS compared with sham (paired t test, P = .019).
6-Meter Walk
We observed an interaction between tDCS and phase (F = 17.0, P = .003; Figure 1C) but no isolated effect of PT on post hoc analysis (P = .09). There was a significant percentage improvement in 6-m walk duration with tDCS compared with sham (paired t test, P = .048).
Timed Up and Go
There was a significant interaction between tDCS and phase (F = 4.88, P = .042; Figure 1D). Training alone also reduced TUG duration (paired t test, P = .05). Percentage improvement in TUG duration was significantly greater with tDCS than sham (paired t test, P = .03).
Retropulsion Test
The baseline measurement correlated well with both the Fazekas score (r = 0.76, P = .003) and number of falls in the preceding year (r = 0.79, P < .001). The retropulsive stimulus was reproducible as the average peak backward displacement and peak backward trunk velocity did not differ across groups (paired t test real vs sham groups, P = .80). The duration to recovery of trunk posture was reduced in patients receiving tDCS + PT (F = 5.6, P = .045; Figure 1F and G). There was no effect of training alone (paired t test, P > .1). Individual responses at baseline and final assessments are shown in Figure 3C.
Discussion
The principal result is that the combination of tDCS and locomotor training improves clinically relevant locomotor performance as measured by stride length, stride length variability, and gait velocity in patients with leukoaraiosis. We also observed improvements in various measures of trunk movement (Table 2). Our results support the finding that tDCS applied during the motor practice (“voluntary drive” 17 ), improves performance.3,18 Indeed, we cannot comment on whether this effect relates to motor practice or motor learning, but previous work has shown improvements in motor adaptive learning using an identical tDCS montage. 5
This result suggests that targeting neocortical regions involved with gait using noninvasive stimulation may be one therapeutic strategy to overcome the cortico–subcortical disconnection caused by white matter lesions in leukoaraiosis. Motor improvements with brain stimulation protocols are far less common for gait than upper and lower limb function.3,19 Cortical locomotor centers can, however, be modulated bilaterally with our tDCS montage. 5
Neither the use of long-term gait training with robotic trainers and treadmills 20 nor the combination of tDCS and robotic training 2 have managed to produce effects such as those observed in this study, although we emphasize that those studies have focused on patients with consolidated strokes, rather than patients with a primary gait disorder. One explanation is that physical training using voluntary drive (vs robotic assistance) induces changes in excitability in relevant motor cortex 21 that may strengthen corticospinal and intracortical networks, whereas tDCS lowers the threshold for these changes to occur. Indeed, PT alone (sham group) was insufficient to improve gait, perhaps because the threshold for the cortical excitability changes that training can induce was not reached without tDCS. Although the magnitude of the increase in gait velocity observed was somewhat unexpected, motor improvements of this magnitude have been previously observed within single sessions combining noninvasive brain stimulation techniques with peripheral nerve stimulation. 22 Further studies in a larger cohort of patients will help determine the everyday clinical applicability of this technique.
The effect of tDCS on balance has not to our knowledge been previously investigated. Although the response to the retropulsion test undoubtedly involves a coordinated neuronal network, including cortical (M1) and subcortical (basal ganglia) structures, vestibular and spinal reflexes, one unanswered question was whether tDCS could impact on this network, either through direct actions on the motor cortex or as a result of the more widespread effects of tDCS that are increasingly recognized. 23 Nevertheless, given that the retropulsion test is abnormal in patients with frontobasal ganglia disorders, the observed effects of tDCS most likely reflect changes to corticosubcortical networks. The combination of PT and off-line tDCS (applied during learning rather than execution of the task) improves balance in patients with leukoaraiosis that may be related to preferential effects on reflex activity, rather than voluntary motor performance. 24 Alternatively, neurostimulatory effects over phasic execution of movement such as a postural response may result from an increase in spontaneous neuronal firing rate. 25 Further studies dissecting the underlying mechanisms involved are warranted.
Study Limitations and Future Implications
As with most neurostimulation studies, one should not falsely attribute behavioral effects to the structures directly beneath the electrodes or coil. As such, our effects may not be restricted to the primary motor or prefrontal cortices. Although a smaller electrode surface area and reduced current density results in more focal cortical stimulation, a larger electrode size can induce a more distributed, if less intense, effect beneath the electrode 26 that can be helpful for bihemispheric primary and premotor cortex stimulation, as in this study. As with many exploratory studies, multiple outcome measures were assessed to investigate the effect of tDCS and PT on various aspects of gait, which is a limitation of this study.
In summary, combining locomotor training with tDCS may offer a treatment option for patients with gait disorder due to leukoaraiosis. Given that gait variability is a functional indicator of gait dysfunction in leukoaraiosis, anodal tDCS as used here may entrain a more symmetrical locomotor pattern that could prove clinically relevant. To provide evidence of the effectiveness of these treatments further randomized controlled trials with larger samples of patients are warranted.
Footnotes
Acknowledgements
We are grateful to all the staff at the Hospital Sirio Libanes in Buenos Aires. We thank the participants for their contribution of time and information to the study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JHA works as a consultant for Balance International Innovations GmbH that manufacture some of the equipment used in this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AMB is supported by a grant from the Medical Research Council (United Kingdom).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
