Abstract
Background
White matter hyperintensities (WMH) are considered to contribute to diminished brain reserve, negatively impacting on stroke recovery. While WMH identified in the chronic phase after stroke have been associated with post-stroke aphasia, the contribution of premorbid WMH to the early recovery of language across production and comprehension has not been investigated.
Objective
To investigate the relationship between premorbid WMH severity and longitudinal comprehension and production outcomes in aphasia, after controlling for stroke lesion variables.
Methods
Longitudinal behavioral data from individuals with a left-hemisphere stroke were included at the early subacute (n = 37) and chronic (n = 28) stage. Spoken language comprehension and production abilities were assessed at both timepoints using word and sentence-level tasks. Magnetic resonance imaging (MRI) was performed at the early subacute stage to derive stroke lesion variables (volume and proportion damage to critical regions) and WMH severity rating.
Results
The presence of severe WMH explained an additional 18% and 25% variance in early subacute (t = −3.00, p = .004) and chronic (t = −3.60, P = .001) language comprehension abilities respectively, after controlling for stroke lesion variables. WMH did not predict additional variance of language production scores.
Conclusions
Subacute clinical MRI can be used to improve prognoses of recovery of aphasia after stroke. We demonstrate that severe early subacute WMH add to the prediction of impaired longitudinal language recovery in comprehension, but not production. This emphasizes the need to consider different domains of language when investigating novel neurobiological predictors of aphasia recovery.
Keywords
Introduction
Poststroke aphasia affects approximately 25 million individuals worldwide, 1 with multiple neurological, demographic, and health-related factors contributing to recovery.2,3 While some studies have succeeded in predicting almost half of the variability in post-stroke aphasia outcomes by considering the contribution of different lesion characteriscs, 4 recovery among individuals with aphasia (IWA) remains highly variable and unpredictable.5,6 Clinicians need reliable predictors 7 of residual aphasia, beside lesion characteristic, to allow them to deliver meaningful prognoses to patients and caregivers. 3 In some stroke survivors, the quantifiable premorbid integrity of the brain, “brain reserve” in the presence of neural pathology, may increase susceptibility to more severe impairments, or to diminished recovery potential. 8 Such measures of “brain reserve” may therefore improve prediction of functional outcomes after stroke.3,9
Small vessel disease (SVD) is a common cerebrovascular pathology associated with aging and cerebrovascular risk factors 10 that is typically detected in midlife and found in the majority of people over 60 years. 11 After stroke, premorbid SVD burden has been shown to be associated with poorer outcomes12,13 and therefore purported as a possible index of diminished “brain reserve.”14,15 White matter hyperintensities (WMH), the most commonly assessed radiological marker of SVD burden, 10 is most frequently assessed using the Fazekas scale 16 ; which differentiates between WMH severity (mild, moderate, severe) within both periventricular white matter hyperintensities (PVWMH) and deep white matter hyperintensities (DWMH). WMH are considered to be a radiological manifestation of progressive white matter integrity loss17,18 and have been associated with poor cognitive skills,19,20 including language.21,22 Given that WMH burden likely reflects the disintegration of white matter pathways, assumed to contribute to effectiveness of neuroplastic recovery processes reliant on these pathways, markers of WMH severity could be considered a surrogate measure of premorbid “brain reserve.” 9 Therefore, measures of early WMH severity may explain some of the variability observed across many cognitive domains and skills after stroke, including language.
Whilst a detrimental role of WMH burden on language has been suggested in both elderly individuals21,22 and people with aphasia,23 -25 this evidence is either limited to WMH assessed within the chronic stage of recovery,3,24 -26 or limited by temporal inconsistencies in WMH and behavioral assessments. 23 WMH represent a dynamic pathology that progresses over time, 27 usually leading to an increase in WMH severity 28 but has been shown to reduce in severity in a smaller portion of patients. 29 As a result of the evolving nature of WMH lesions, the timing of WMH assessment must be taken into consideration when used as an explanatory variable in stroke. To this end, premorbid WMH burden is likely to be most accurately assessed on neuroimaging acquired within the early stages after stroke, reflecting the individual premorbid “brain reserve” available for optimal recovery. 30
To date, 4 studies have identified an association between language (overall aphasia severity, naming and fluency) and chronic WMH burden.3,24 -26 Whilst Wilmskoetter et al 25 did not find a direct relationship between overall severity of aphasia and chronic WMH burden, they did show that PVWMH severity (using Fazekas scale) mediated a relationship between the number of long-ranging white matter fibers and overall aphasia severity. Basilakos et al 24 also found a relationship between chronic WMH severity (again using the Fazekas scale) and the rate of decline in language abilities in chronic aphasia. Most recently, Johnson et al 3 demonstrated, in a large sample of patients, that chronic WMH severity (using the Fazekas scale) contributed to language outcomes, together with many other predictors. When considering naming skills alone, Varkanitsa et al 26 found no association between naming scores and WMH in the chronic post-stroke stage, however they did find a negative association between participants’ response to naming therapy and both total chronic Fazekas WMH, and DWMH scores. Finally, Wright et al 23 demonstrated that both subacute WMH, as assessed by the Cardiovascular Health Study scale, 31 and brain atrophy, calculated as a ratio of brain volume to cerebrospinal fluid volume, were associated with chronic naming and fluency deficits. Whilst Wright et al 23 is the only study to date to have investigated the contributions of subacute WMH to aphasia outcomes, an important limitation of this study was the heterogenous timing of the neuroimaging (early subacute stage) and behavioral (range 3-157 months post onset) data acquisition. These inconsistencies in the timing of behavioral assessment have prevented researchers from disentangling the effects of premorbid WMH on early compared to chronic recovery processes.
The neural networks underpinning the language skills required for effective communication are likely more extensive than the networks required to support single-word production tasks, potentially making language more susceptible to the anatomically diffuse effects of WMH than identified in studies investigating single word level production only.23,26 Moreover, comprehension skills may be particularly vulnerable to the adverse effects of WMH burden given the frequently identified association of language comprehension after stroke with both a range of cognitive abilities,32,33 and the upregulation of domain-general cognitive neural networks. 34 Given that WMH can compromise multiple domains of cognitive processing,20,35 language comprehension skills may be more vulnerable than other aspects of language, to the adverse effects of WMH burden. A global measure of aphasia severity that incorporates several aspects and domains of language, as used in two previous studies,3,24 may be insufficient to identify any differential contributions across language domains. To this end more research is required to investigate the potential role of premorbid WMH across both production and comprehension, using a range of levels of language processing including single word, sentence and discourse level tasks.
To this end, we aimed to examine the contribution of WMH assessed at an early subacute stage (subsequently referred to as “early subacute WMH”), a surrogate measure of premorbid brain reserve, to post-stroke aphasia outcomes. Specifically, we examined the extent to which language comprehension and production skills are differentially impacted by early subacute WMH burden at two different phases of recovery (early subacute and chronic). Given that stroke lesion volume and anatomy have been shown to be a reasonably reliable predictor of aphasia,4,36,37 we investigated if WMH burden could explain any additional variance, over and above stroke lesion volume and distribution, within the language network.
Methods
Participants
This study retrospectively analyzed data from two post- stroke aphasia studies. Inclusion criteria were (a) a single left hemisphere stroke, diagnosed using clinical structural neuroimaging, (b) the presence of aphasia, diagnosed using the WAB, 38 (c) English as primary language, (d) availability for an initial assessment at two to six weeks poststroke onset, (e) able to provide informed consent. Exclusion criteria were (a) history of neurological disorder, mental illness, head trauma, alcoholism, or cerebral tumor, (b) contraindications to magnetic resonance imaging (MRI), (c) severity of deficits precluding informed consent, (d) severe dysarthria or apraxia of speech (determined by a speech pathologist), (e) severe hearing impairment. Apraxia of speech was assessed on the Apraxia Battery for adults (ABA-2). 39 In one of the aphasia studies, half of the IWA were randomized into language therapy treatment. Participants enrolled at early subacute timepoint were invited for a chronic follow-up assessment at six months poststroke. In total, 37 IWA were included at early subacute stage (19% haemorrhagic stroke) and 28 IWA returned for follow-up assessment (18% haemorrhagic stroke). Education was scored into eight levels according to the UNESCO International Standard Classification of Education (ISCED). 40 The study received approval from the University of Queensland Medical Research Ethics Committee and the Queensland Health Human Research Ethics Committee.
Language Assessment
Participants underwent language assessment at both timepoints: initial early subacute assessment (range: 17-47 days, mean 27 days) and chronic follow-up assessment (range 183-237; mean 205 days). For each participant, spoken language comprehension (SpoComp) and spoken language production (SpoProd) performance was measured at both early subacute and chronic timepoints. The SpoComp score was derived from a combined score of the Auditory Word, Sentence and Paragraph comprehension subtests from the Comprehensive Aphasia Test (CAT). 41 The SpoProd score was derived by combining the Fluency and Naming (nouns and verbs) CAT subtests and a Picture description task 38 (see Supplemental material Figure 1 for details of picture description task and analysis). Picture description task was not administered to three patients at early subacute stage and these patients were excluded from Subacute SpoProd analysis. Subtests of the CAT were double scored by experienced speech pathologists blinded to neurological and demographic data, and to the timepoint. Interrater reliability, available for speech production scores (fluency, naming) for one of the included datasets, was 70%.
Neuroimaging
Neuroimaging Protocol
Early subacute neuroimaging data were acquired 2 to 6 weeks poststroke. Data from the first study (n = 13) 42 were collected using Siemens 3 Tesla Trio scanner (Siemens, Erlangen) with a 12-channel head coil. During the same scanning session, a high-resolution 3D T1-weighted anatomical image (MP-RAGE; TR 1900 milliseconds (ms); TE 2.4 ms; TI 900 ms; (0.9 mm)3 resolution) and 2D T2-weighted FLAIR image (TE 87 ms, TR 9000 ms, TI 2500 ms, 36 3 mm slices, 0.9 × 0.9 mm in-plane resolution) were acquired for each subject. Data from the second study (n = 24) was collected using a Siemens 3 Tesla MAGNETOM Prisma scanner (Siemens, Erlangen) using a 20-channel head coil. A high-resolution 3D T1-weighted anatomical image (MP2RAGE; Marques et al., 2010; TR 4000 ms; TE 2.91 ms; TI1 700 ms; TI2 2220 ms; FA1 6 degrees; FA2 7 deg; (1 mm)3 resolution) and 3D T2-weighted FLAIR image (TE 386 ms, TR 5000 ms, TI 1800 ms, (1 mm)3 resolution) were acquired for each subject.
Lesion Delineation and Image Processing
Stroke lesions were manually delineated on high-resolution T1-weighted sequences in patient space using MRIcron (https://www.nitrc.org/projects/mricron) by two authors (KG and VV) and verified by two senior authors (KM and SB), blinded to behavioral and demographic data. T2-weighted FLAIR scans were consulted to improve the delineation of stroke lesion where unclear, that is, in cases of haemorrhagic stroke. The stroke lesion volume was calculated in native space for subsequent analyses.
For region of interest (ROI) analyses, T1-weighted sequences and lesion masks were normalized to MNI space using Clinical Toolbox (2012, https://www.nitrc.org/projects/clinicaltbx/) within SPM (version 12, https://www.fil.ion.ucl.ac.uk/spm/) in Matlab (version 2017, https://www.mathworks.com/). Normalized lesion masks were manually revised (KG, VV) where necessary and verified by two authors (KM and SB). The stroke lesion volume was calculated in native space for subsequent analyses. Disagreements during the manual drawing process were resolved through group discussion (KG, VV, SB, KM).
Lesion Volume Within Language ROIs
ROI masks in MNI space were created in DSI studio software (https://dsi-studio.labsolver.org), using the Automated anatomical atlas 3 (AAL3). 43 Given the limited number of participants, the number of ROIs was restricted to the four most frequently identified cortical regions associated with aphasia; Broca’s area (pars triangularis + pars opercularis), insula, superior temporal gyrus (STG) and a combined region of both angular gyrus (AG) and supramarginal gyrus (SMG). Finally, the lesion “load” or proportion damaged of each ROI was calculated by inclusively masking each language ROI with the normalized lesion, and dividing by the total volume of the ROI.
White Matter Hyperintensities Assessment
Using T2-weighted FLAIR sequences, the severity of WMH was assessed according to the Fazekas scale 44 in (a) the periventricular white matter (PVWMH; scores ranging from 0 to 3), (b) deep white matter territories (DWMH; scores 0-3), and (c) a combined WMH (a + b) severity score (ranging from 0 to 6). Using the combined WMH severity score, severity was then classified as none present (0), mild (1-2), moderate (3-4), severe (5-6). WMH were assessed using the whole brain, disregarding the territory of stroke lesion. In cases where the stroke lesion and/or the gliosis extended toward the left ventricular region and prevented the assessment of PVWMH in the left hemisphere, only PVWMH in the right hemisphere was assessed, and was assumed to have been bilateral pre-stroke for the purposes of scoring. The assessment was performed by consensus of three authors, including a certified radiologist for each dataset (VV, SB, LZ), blinded to behavioral and demographic details. In cases of a disagreement, the radiologist made the final decision.
Statistical Analysis
Relationships between the demographic and imaging characteristics were explored using Pearson correlations. Univariate linear regression was used to investigate the relationship between language therapy treatment and chronic SpoComp and chronic SpoProd scores. Stepwise linear regression models were used to investigate the relationship between early subacute stroke lesion characteristics, WMH characteristics and longitudinal language outcomes. All analyses were conducted in IBM SPSS Statistics for Windows, Version 22.0.
In Model 1, we examined if early subacute WMH burden predicted additional variance in longitudinal SpoComp and SpoProd scores, after factoring out any significant effect of stroke lesion volume. In Model 1A, a stepwise linear regression was used to test if stroke lesion volume alone predicted language outcomes. In Model 2A, a stepwise linear regression was used to test if early subacute WMH explained variance in outcomes after controlling for stroke lesion volume, if significant.
In Model 2, we examined if early subacute WMH predicted additional variance in longitudinal SpoComp and SpoProd scores, after factoring out proportion stroke damage to critical language ROIs. In Model 2A, a stepwise linear regression was used to test if proportion damage to the insula, Broca’s area, STG, and AG+SMG ROIs predicted language outcomes. In Model 2B, a stepwise linear regression was used to test if early subacute WMH explained variance in outcomes after controlling for proportion damage to critical ROIs, if significant (see Figure 1 for an illustration of the different models and model sequence). Several individual outcome measures from the Fazekas scale 44 (PVWMH, DWMH, combined WMH, and a WMH severity rating) were included in the models in a stepwise fashion.
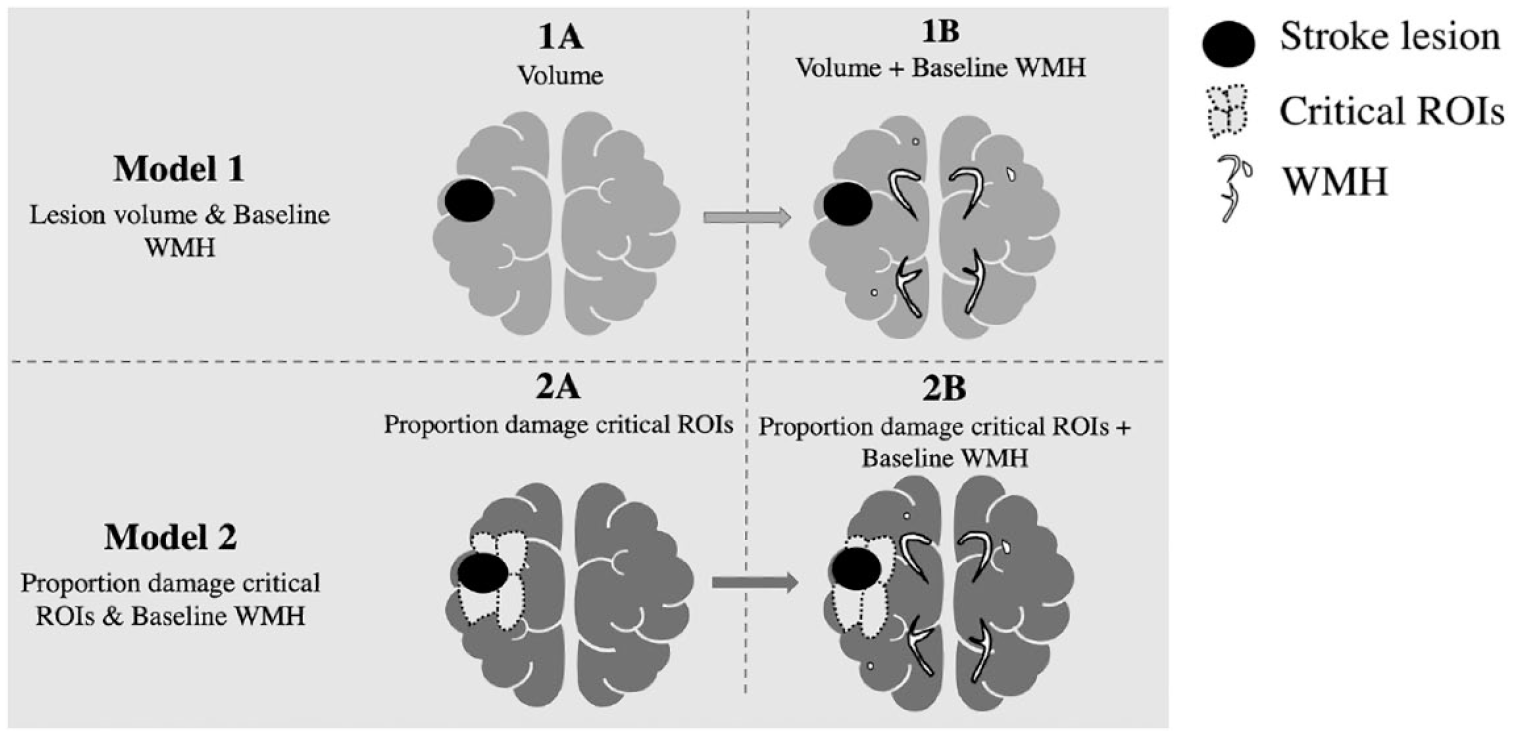
An illustration of the anatomical components included in two step-wise linear regression models (Model 1, Model 2) employed in this study.
Results
Population Characteristics and Variable Correlations
All demographic, stroke lesion and WMH characteristics at early subacute stage are presented in Table 1 and for those who returned for chronic assessment is presented in Table 2. A lesion overlay of all lesions can be found in Figure 2. The combined WMH score did not correlate with total stroke lesion volume (P = .86) or stroke lesion load within any language ROI (insula: P = 0.31, Broca’s area: P = .49, STG: P = .38, AG+SMG: P = .76). Combined WMH score did not correlate with educational level (P = .34) but showed high degree of multicollinearity with age (r = .75, P < .01) (See Table 1). Univariate simple linear regression indicated that there was no relationship between language therapy participation and chronic SpoComp (P = .58) and chronic SpoProd (P = .46) scores.
Details of Socio-Demographic Information, Early Subacute Stroke Lesion Variables, Early Subacute WMH Indices of IWA Included at Early Subacute Stage.

Abbreviations: %, percentage; STG, superior temporal gyrus; AG, angular gyrus; SMG, supramarginal gyrus; PVWMH, periventricular white matter hyperintensities; DWMH, deep white matter hyperintensities.
Educational levels scored according to the UNESCO ISCED classification system 40 (0: early childhood, 1: primary education, 2: lower secondary education, 3: upper secondary education, 4: post-secondary non-tertiary education, 5: short-cycle tertiary education, 6: bachelor’s or equivalent, 7: master’s or equivalent, 8: doctoral or equivalent).
Details of Socio-Demographic Information, Early Subacute Stroke Lesion Variables, Early Subacute WMH Indices of IWA Who Returned For Chronic Assessment.

Abbreviations: %, percentage; STG, superior temporal gyrus; AG, angular gyrus; SMG, supramarginal gyrus; PVWMH, periventricular white matter hyperintensities; DWMH, deep white matter hyperintensities.
Educational levels scored according to the UNESCO ISCED classification system 40 (0: early childhood, 1: primary education, 2: lower secondary education, 3: upper secondary education, 4: post-secondary non-tertiary education, 5: short-cycle tertiary education, 6: bachelor’s or equivalent, 7: master’s or equivalent, 8: doctoral or equivalent).
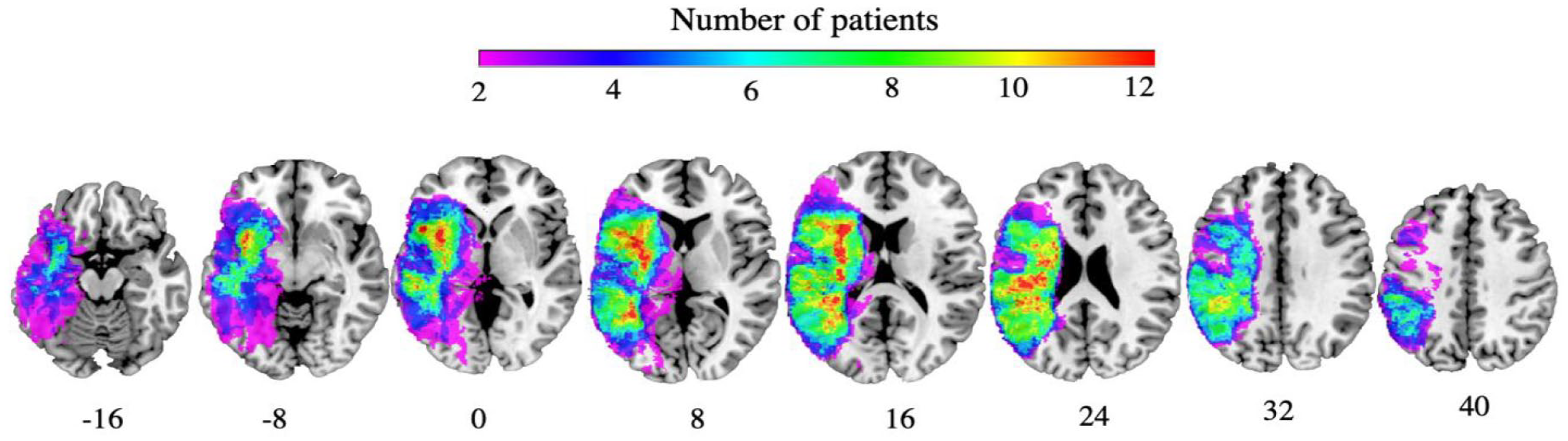
Lesion overlay early subacute stage (n = 37). Warmer colors indicate greater areas of lesion overlap. Coordinates are reported in MNI space.
Model 1: Contribution of Total Stroke Lesion Volume and Early Subacute WMH Burden
SpoComp scores
Simple linear regression in Model 1A showed a significant negative relationship between stroke lesion volume and subacute SpoComp scores (R2 = .16, F[1, 35) = 6.91, P = .013), with scores decreasing by −0.17 points for each cm3 increase in stroke lesion volume. In Model 1B, that included WMH burden after factoring out stroke lesion volume, a significant relationship was found between severe WMH and SpoComp (t = −3.00, P = .004). The addition of severe WMH burden into Model 1B explained an extra 18% of variance compared to Model 1A (R2 = .34, F[2, 34] = 8.76, p < .001). Scores were on average 2.87 lower in IWA with severe WMH burden than those without.
With chronic SpoComp scores, there was no relationship with lesion volume in Model 1A. Model 1B, which included WMH burden only, indicated that there was a significant effect of severe WMH (R2 = .24, F[1,26] = 8.49, P = .007) on chronic SpoComp, such that IWA with severe WMH burden scored 2.63 lower than IWA without severe WMH, irrespective of stroke lesion volume.
SpoProd scores
Simple stepwise linear regression in Models 1A yielded a negative relationship between stroke lesion volume and both subacute (R2 = .17, F[1, 32] = 6.49, P = .016) and chronic SpoProd (R2 = .22, F(1, 26) = 7.54, P = .011) scores, with SpoProd scores decreasing by −0.36 and −0.43, for each cm3 increase in stroke lesion volume for subacute and chronic timepoints respectively. In Models 1B, that included WMH burden after factoring out stroke lesion volume, WMH did not contribute to explaining additional variance in either early subacute or chronic SpoProd scores.
Model 2: Contribution of Proportion Damage to Critical ROI & Early Subacute WMH Burden
SpoComp scores
Model 2A showed that there was no relationship between proportion damage to critical ROIs and subacute SpoComp measures. In Model 2B, that included WMH only, results of linear regression indicated that there was a significant effect of severe WMH on early subacute SpoComp (R2 = .17, F[1,35] = 7.36, P = .01), that is IWA with severe WMH scored on average -2.86 lower on SpoComp measures than those without severe WMH (t = −2.71, P = .01), irrespective of stroke lesion distribution.
Model 2A indicated that there was a combined significant relationship between proportion damage to the insula (β = −.30, t = 3.19, P = .004) and Broca’s area (β = 0.22, t = 2.17, P = .039) ROIs and chronic SpoComp scores (R2 = .29, F[2, 25] = 6.93, P = .013). In the subsequent Model 2B, stepwise multiple linear regression indicated that there was a significant effect of the severe WMH burden on chronic SpoComp scores (R2 = .54, F[3, 24] = 9.48, P < .001). IWA with severe WMH scored on average 2.65 points lower on chronic SpoComp, after controlling for lesion load within the insula and Broca’s area ROIs, than those without severe WMH (t = −3.60, P = .001). The inclusion of severe WMH burden to Model 2B explained an extra 25% of variance in chronic SpoComp scores.
SpoProd scores
Stepwise multiple linear regression in Model 2A demonstrated a combined significant effect of proportion damage to the insula (β = −.32, t = −2.36, P = .024) and SMG+AG (β = −.57, t = −2.55, P = .016) ROIs on subacute SpoProd scores (R2 = .28, F[2, 31] = 6.01, P = .006). In Model 2B, WMH measures did not explain variance in subacute SpoProd language outcomes, over and above the proportion damage to critical ROIs.
Model 2A revealed a significant effect of lesion load within STG ROI on chronic SpoProd (R2 = .17, F[1, 26] = 5.40, P = .028). In Model 2B, WMH measures did not explain variance in chronic SpoProd language outcomes, over and above the proportion damage to critical ROIs.
To ensure that our results were not impacted by the inclusion of haemorrhagic stroke, we re-ran all the models with ischemic stroke only and our results regarding the WMH burden predictor were not impacted by this change (see Supplementary material for details of subanalyses).
Discussion
This study attempted to tease apart the concomitant effects of stroke pathology and WMH pathology, assessed on early subacute scans, on the inter-individual variation observed in the recovery of post-stroke aphasia. Whilst studies to date have been unable to disentangle the effects of premorbid WMH from the chronic progression of WMH, we mitigated these temporal inconsistencies, between scan acquisition and language assessment, by restricting the assessment of WMH to the first six weeks after stroke. In doing so, we have been able to demonstrate that early subacute WMH burden, a proxy measure of premorbid brain reserve, impacts on longitudinal language outcomes after stroke.
To extend our current understating of the relationship between WMH pathology and aphasia outcomes, measures of both complex language production and comprehension skills were interrogated separately. We therefore provide novel evidence that early subacute WMH burden impacts these skills differentially, with the presence of severe WMH negatively impacting language comprehension, but not production, skills across both the subacute and chronic phases of recovery. As such, this work highlights the need for the different domains of propositional language to be investigated separately, when investigating possible neurobiological predictors of language recovery after stroke. It also demonstrates the utility of clinically—derived measures of early subacute WMH burden in contributing to providing more reliable and specific clinical predictors of recovery of language after stroke.
Early Subacute WMH and Language Outcomes in Spoken Comprehension and Production
In contrast to the study by Wright et al, 23 who identified an independent association between chronic fluency and naming impairments and WMH severity, after controlling for cardiovascular risk factors, diabetes, depression rates and gray and white matter damage, this study, along with Varkanitsa et al, 26 found no relationship between WMH burden and language production impairments in IWA. Whilst these discordant findings are likely driven by the diverse assessment of language production skills and common methodological limitations (eg, small sample sizes, temporal inconsistencies between WMH and behavioral assessment data acquisition, and the different co-variates used across studies), it is clear that further research is needed to disentangle the impact of premorbid WMH burden on both discrete and complex language production skills in aphasia.
Notably, the association of language comprehension deficits in aphasia and WMH burden has not been previously investigated. Whilst it is beyond our data to determine why language comprehension skills appeared to be more susceptible to severe WMH burden than language production skills, evidence suggesting both distinct neural contributions to these language skills,45,46 along with a partial overlap of neural networks supporting both cognitive processes and language comprehension32,47 -49 may help to explain this differential impact.
Behaviorally, the contribution of executive processing (control, inhibition, maintenance, manipulation) to normal speech comprehension has been demonstrated.32,48,49 Given the evidence base demonstrating that executive functioning is often compromised by WMH, any functional neurologically mediated relationship between language comprehension and executive processing, is likely to be more susceptible to the cumulative effects of premorbid WMH burden.
From an anatomical perspective, a neural region that has been shown to be involved in both language comprehension45,49 and executive processing50 -52 is the bilateral prefrontal cortex. White matter projections from the prefrontal cortex are particularly susceptible to integrity loss53,54 and WMH damage within these regions have been consistently linked to cognitive deficits.20,35 It is therefore possible that premorbid white matter disruption within these bilateral networks negatively impacts linguistic (ie, comprehension) and/or non-linguistic (ie, executive processing) neuroplastic mechanisms required for successful recovery of comprehension.
To this end, future studies should include rigorous and comprehensive assessment of language domains thought to be impacted by the different patterns of neurological damage caused by a stroke. This behavioral evidence could then systematically be combined with structural and functional neuroimaging data to evaluate the differential impact of WMH burden on effective communication and recovery of language.
Early Subacute WMH burden and Mechanisms of Aphasia Recovery
In aphasia, stroke pathology most commonly occurs within the primary left-lateralized language network, and successful recovery requires optimization and strengthening of spared neural resources by either upregulating the remaining language network and/or by upregulating other networks to support language function, for a detailed review see Stefaniak et al. 55 Premorbid WMH burden may diminish individuals’ brain reserve capacity and so impede either network upregulation or adaption during the crucial early recovery period. We found that the presence of severe WMH burden predicted language performance after stroke. If brain reserve is defined as a set of structural properties of the brain that threshold the expression of functional impairment, 8 severe WMH burden may place individuals below that threshold and predispose them to not only more pronounced post-stroke language impairment, but also diminished language recovery potential.
The neurobiological mechanisms by which premorbid WMH contribute to reduced brain reserve in stroke remains to be determined. Severe WMH are indicative of globally fragmented white matter structural connectivity. 17 The importance of global white matter integrity is becoming increasingly recognized as a potential biomarker of language recovery in aphasia. Bonilha et al 56 provided seminal evidence that global white matter integrity was associated with effectiveness of anomia treatment. While this study did not take WMH burden into consideration, a further study by Wilmskoetter et al 25 used a mediation analysis to show that severe periventricular WMH was associated with fewer long-range white matter fibers, and in turn fewer long-range fibers were associated with overall aphasia severity. Whether WMH lesions observed on structural scans are indicative of globally fragmented white matter integrity poststroke, which in turn diminish recovery potential in aphasia, requires further investigation.
Limitations and Future Directions
Whilst we present a well-controlled longitudinal investigation of the relationship between premorbid WMH and post-stroke aphasia, our findings are limited by the relatively small number of IWA at the early subacute stage, with further attrition at the chronic timepoint. This may have limited our ability to identify additional associations between early subacute WMH and language measures. Given the high multicollinearity between WMH and age, we were also unable to separate the independent contributions of these characteristics to language outcomes.
Secondly, to ensure appropriate relationship between statistical observations and variables in the regression analyses, the sample size of the current study prevented us from examining a larger number of cortical and subcortical stroke ROIs. Stroke lesion distribution within the language network has been shown to be a robust predictor of differential aphasic impairments,4,57,58 therefore larger studies would be better equipped to examine whether more nuanced assessment of primary stroke lesion (ie, assessment of a higher number of cortical and subcortical language network ROIs) can refine the impact of premorbid WMH.
The ubiquity of WMH in older age suggests that WMH are not exclusively pathological and may also accompany healthy aging.17,18 To understand their role in aphasia recovery, future research needs to focus on identifying the conditions under which WMH are indicative of pathological aging, such as their severity, anatomical distribution and underlying microstructural damage.
To this end, future studies should strive to include larger cohorts with more balanced WMH severity distribution. To separate the effects of premorbid WMH and WMH progression requires a multi-timepoint study design, with an early scan and additional scans acquired at controlled chronic timepoints, ideally years apart. Considering the network-based architecture of cognition, 59 focus may need to shift from WMH severity within isolated regions (ie, periventricular white matter, deep white matter) toward the interaction of regions within and across both the lesioned language network in stroke, and spared cognitive networks frequently structurally compromised by WMH. In addition, structural and functional data needs to be paired with comprehensive behavioral evaluation of verbal and non-verbal cognition as to further probe the underlying mechanisms by which WMH hamper language recovery in stroke.
Conclusion
Existing biomarkers do not account for the variable recovery observed among IWA, highlighting an urgent need to identify novel predictors of recovery and/or variables that help explain some of this variability.2,3,5,6 Acute stroke pathology and premorbid white matter hyperintensities have distinct aetiologies and chronologies, and research into their combined contribution to functional stroke outcomes is in the early stages. This is the first study to demonstrate that premorbid WMH impact post-stroke language performance. Further, it is the first study to consider how this differentially impacts on the different modalities of language, and the different phases of post-stroke recovery. Our findings, in combination with evidence that WMH primarily affect non-language white matter tracts (for a review see Biesbroek et al 60 ), highlights the need for more targeted research into the contribution of spared neural structures and networks, beyond known language networks, in the mechanics of aphasia recovery. 34
WMH burden, and brain reserve in general, are best conceptualized as variables that refine and/or modulate existing biomarkers, such as stroke lesion variables. Before the stroke event, IWA from this study were not experiencing language deficits, even when harboring the most severe WMH burden. This highlights the compounding, rather than causal, effect in which WMH lead to more pronounced impairments and/or attenuate successful recovery from aphasia. The inclusion of WMH along with stroke lesion characteristics predicting aphasia recovery may contribute to improving predictions, and therefore provide more meaningful prognoses. Severe WMH burden can be readily assessed on clinical T2-weighted FLAIR images or computed tomography (CT) images and requires minimal specialist training. This renders its inclusion into prognostication processes not only clinically feasible but, given our findings, also clinically meaningful.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683231168384 – Supplemental material for Early Subacute White Matter Hyperintensities and Recovery of Language After Stroke
Supplemental material, sj-docx-1-nnr-10.1177_15459683231168384 for Early Subacute White Matter Hyperintensities and Recovery of Language After Stroke by Veronika Vadinova, Aleksi J. Sihvonen, Kimberley L. Garden, Laura Ziraldo, Tracy Roxbury, Kate O’Brien, David A. Copland, Katie L. McMahon and Sonia L. E. Brownsett in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We would like to thank all the participants in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: VV and KG are supported by the Research Training Program, the Commonwealth Government of Australia. VV, DC and KG were supported by the NHMRC-funded Predicting and Promoting Aphasia Recovery (1104194). SB was supported by the NHMRC-funded Centre of Research Excellence in Aphasia Recovery and Rehabilitation (APP1153236). AJS was supported by the Finnish Cultural Foundation (191230), Orion Research Foundation sr, Maire Taponen Foundation, and Signe and Ane Gyllenberg Foundation.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
