Abstract
Background
White matter hyperintensities (WMH) are a radiological marker of brain health that has been associated with language status in poststroke aphasia; however, its association with language treatment outcomes remains unknown.
Objective
To determine whether WMH in the right hemisphere (RH) predict response to language therapy independently from demographics and stroke lesion–related factors in poststroke aphasia.
Methods
We used the Fazekas scale to rate WMH in the RH in 30 patients with poststroke aphasia who received language treatment. We developed ordinal regression models to examine language treatment effects as a function of WMH severity after controlling for aphasia severity, stroke lesion volume, time post onset, age, and education level. We also evaluated associations between WMH severity and both pre-treatment naming ability and executive function.
Results
The severity of WMH in the RH predicted treatment response independently from demographic and stroke-related factors such that patients with less severe WMH exhibited better treatment outcome. WMH scores were not significantly correlated with pretreatment language scores, but they were significantly correlated with pretreatment scores of executive function.
Conclusion
We suggest that the severity of WMH in the RH is a clinically relevant predictor of treatment response in this population.
Keywords
Introduction
Aphasia, the loss or impairment of language function due to brain damage, affects one-third of stroke survivors 1 leading to poor functional communication and quality of life. 2 Although aphasia recovery occurs even in the chronic phase 3 and despite the effectiveness of language therapy, 4 not all people with poststroke aphasia (PWA) improve after therapy. 5
The prediction of aphasia recovery has largely focused on left hemisphere lesion-related factors, including lesion size and location 6 and pretreatment cognitive skills. 7 However, the integrity of the brain tissue spared by the stroke may contribute to neural plasticity and influence treatment response independently of left hemisphere damage. 8 White matter hyperintensities (WMH) of presumed vascular origin is a relevant radiological marker of brain health. WMH severity has been associated with aging and cognitive impairment9,10 and language status in poststroke aphasia, although relevant evidence in this group is limited.11-13
Here, we sought to determine whether WMH in the right hemisphere (RH) predicts response to language therapy in poststroke aphasia independently from demographics and stroke-related factors. We hypothesized that WMH severity in the RH would predict treatment gains, after controlling for age and education, aphasia severity, left hemisphere stroke lesion volume (ie, lesion volume hereafter), and time post onset. We also hypothesized that WMH severity in the RH would not be associated with stroke-related measures (ie, lesion volume, time post onset and aphasia severity) and pretreatment naming performance. The rationale is that WMH are a magnetic resonance imaging (MRI) marker of small vessel disease, 14 which can be indicative of the brain state prior to stroke, whereas stroke-related measures and pretreatment naming performance reflect poststroke effects in the affected hemisphere. Finally, several population-based studies have provided clear evidence that WMH are related to cognitive impairment, particularly to reductions in information processing speed and executive dysfunction.10,15-17 This is probably because WMH may cause secondary loss of communication between critical cortical regions leading to subsequent loss in function.15,18 WMH have also been associated with worse cognitive performance and recovery after stroke.19,20 Based on this literature, we expected WMH in the RH to be associated with pretreatment nonverbal executive function in PWA.
Methods
Participants
The study included a retrospective sample of 30 participants (10 female; mean age = 61 ± 10.73 years; mean education level = 15 ± 2.11 years) with persistent aphasia following a single left hemisphere stroke (time poststroke onset = 52 ± 50.12 months). No participant reported major psychiatric disorders, neurological disease other than stroke, or any active medical condition that precluded participation. They all primarily spoke English and had normal or corrected-to-normal vision and adequate hearing. All participants provided informed consent to undergo study procedures approved by the Boston University and Massachusetts General Hospital Institutional Review Boards.
Language Assessments and Treatment
Prior and after treatment, participants completed standardized language assessments, including the Western Aphasia Battery–Revised (WAB-R) to determine aphasia type and overall language severity (aphasia quotient, AQ), 21 the Boston Naming Test (BNT), 22 and the Pyramids and Palm Trees Test (PAPT) 23 to assess picture naming and semantic processing respectively. The subtests symbol trails, mazes, and design generation of the Cognitive-Linguistic Quick Test (CLQT) 24 were also administered to generate a composite score of nonverbal executive function.
Participants received semantic feature analysis treatment 25 for up to 12 weeks. They attended up to 24 two-hour treatment sessions either 2 or 3 times per week or until they reached ≥90% accuracy on 2 consecutive weekly probes tracking change in naming throughout treatment. Treatment effects were determined by calculating individual proportion of potential maximal gain (PMG) with the following formula: [(mean posttreatment score – mean pretreatment score)/(total number of trained items – mean pretreatment score)]. PMG has been utilized in previous research 26 including prior reports on the same patient cohort.27,28 Further assessment and treatment protocol details are available elsewhere.27,28
Participants’ demographics as well as stroke and WMH lesion data and behavioral data are reported in Table 1.
Demographic, Behavioral and Stroke and WMH Lesion Data for All PWA.
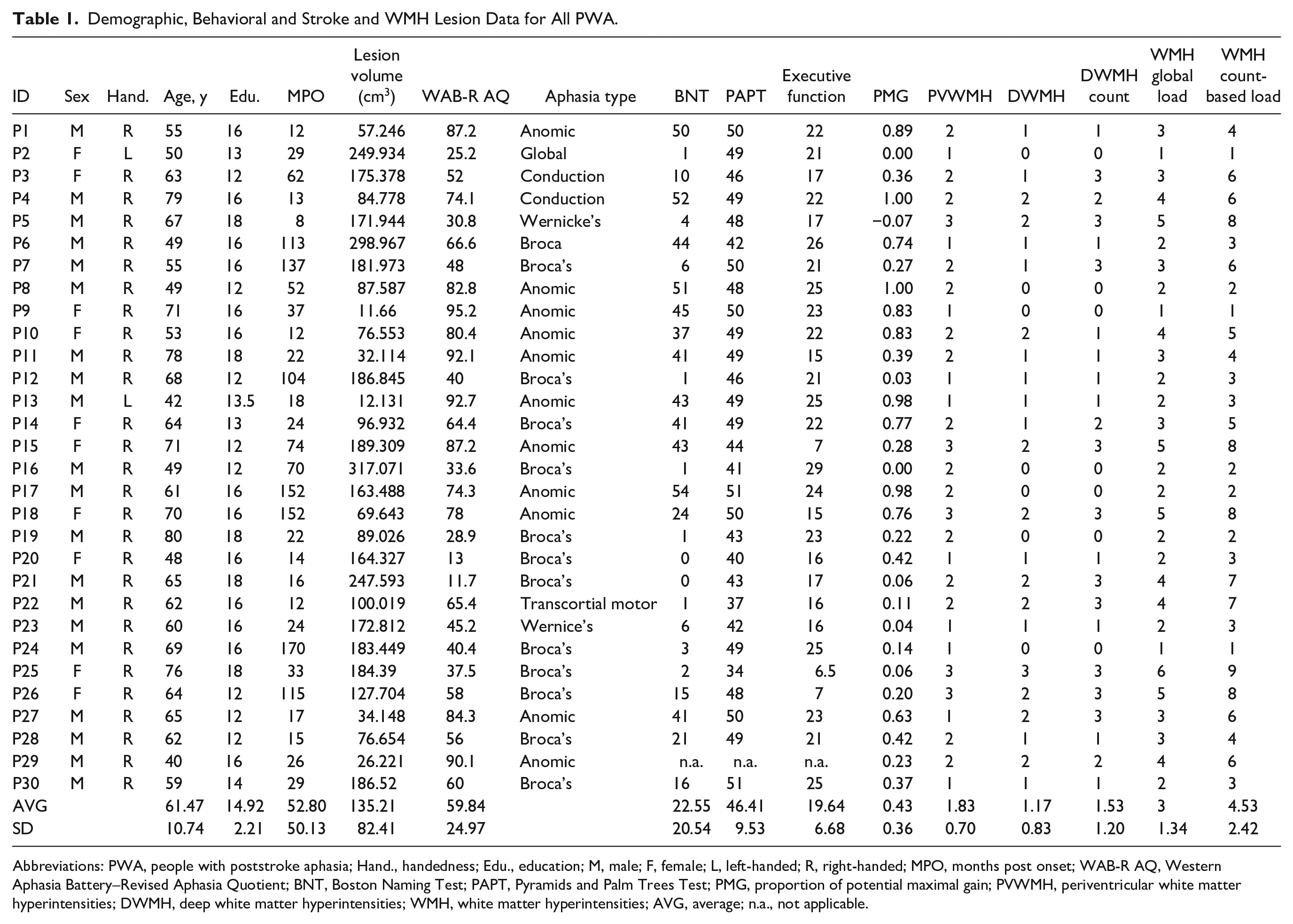
Abbreviations: PWA, people with poststroke aphasia; Hand., handedness; Edu., education; M, male; F, female; L, left-handed; R, right-handed; MPO, months post onset; WAB-R AQ, Western Aphasia Battery–Revised Aphasia Quotient; BNT, Boston Naming Test; PAPT, Pyramids and Palm Trees Test; PMG, proportion of potential maximal gain; PVWMH, periventricular white matter hyperintensities; DWMH, deep white matter hyperintensities; WMH, white matter hyperintensities; AVG, average; n.a., not applicable.
MRI Data Acquisition and Analysis
Imaging data were collected just before the beginning of treatment at the Athinoula A. Martinos Center in Charlestown, MA, USA on a 3T Siemens Trio Tim MRI scanner using a 20-channel head and neck coil. The MRI protocol included a high-resolution T1-weighted 3D sagittal volume (parameters: repetition time/echo time [TR/TE] = 2300/2.91 ms, T1 = 900 ms, flip angle = 9°, matrix = 256 × 256 mm, slice thickness = 1 mm3, 176 sagittal slices) and a T2-FLAIR (T2–fluid attenuated inversion recovery) 2D axial volume sequence (parameters: TR/TE/TI = 9000/90/2500 ms, matrix = 220 mm, field of view [FOV] = 220 mm, slice thickness = 5 mm with 0 mm gap, 35 slices, with a parallel imaging acceleration factor of 2).
Lesion maps were manually drawn slice-by-slice on T1 structural images by trained research assistants blinded to patient’s behavioral scores using MRIcron (www.mccauslandcenter.sc.edu/crnl/mricron/) (see Figure 1 for a lesion overlay map for all participants). Lesion maps and T1-weighted images were normalized to MNI space using SPM12. Normalized lesion maps were filtered at 50% and lesion volumes were calculated using in-house Matlab scripts. 28
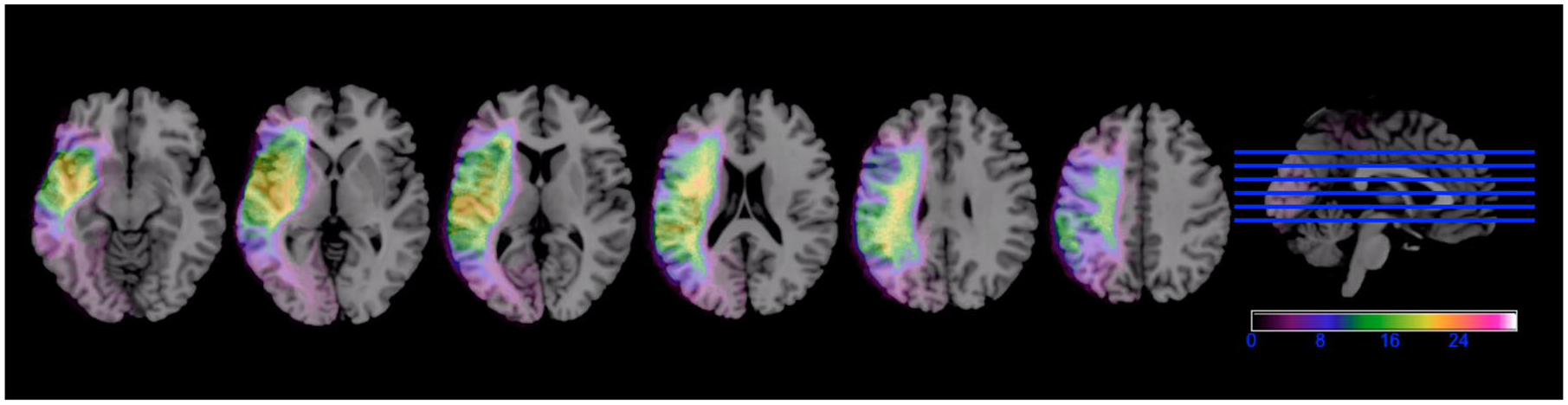
Left hemisphere lesion overlay map for all 30 people with poststroke aphasia (PWA). The heat colors represent higher number of PWA with overlapping lesions. Left hemisphere is presented on the left.
WMH in the RH were defined following the recent STandards for Reporting Vascular changes on nEuroimaging (STRIVE) 14 as signal abnormality of variable size in the white matter that shows hyperintensity on T2-FLAIR weighted images without cavitation. T2-FLAIR images were rated for WMH severity in the right hemisphere only, using the Fazekas scale. 29 Periventricular WMH (PWMH) were graded as 0 (absent), 1 (caps or pencil-thin periventricular lining), 2 (smooth haloing or thick lining), and 3 (irregular WMH extending into the deep white matter). Deep WMH (DWMH) were graded as 0 (absent), 1 (small punctate or nodular lesions), 2 (beginning confluent lesions), and 3 (confluent lesions). The number of DWMH lesions (DWMH count) was also computed for a more fine-grained quantitative approach. Following previous literature, 2 composite scores were calculated to capture overall WMH burden: (a) WMH global load (PVWMH score + DWMH score) and (b) WMH count-based load score (PVWMH score + DWMH score + DWMH count score). (Both DWMH metrics were included to account for cases with mild DWMH and several lesions [ie, severe DWMH count] and cases with severe DWMH and fewer lesions [ie, mild DWMH count].) These composite scores allow assessing the total severity and extent of WMH in the hemisphere of interest (see Table 1 for individual patients’ rating scores). Scans were rated by the first and second author achieving high interrater reliability (Cohen’s κ: PVWMH = .79, DWMH = .90, DWMH count = .95). Rating discrepancies were resolved by the third author.
Statistical Analyses
PMG was the dependent treatment outcome variable. Because PMG scores were not normally distributed (see Supplemental Figure 1), they were divided into quartiles as done in previous research12,30,31 resulting in meaningful groups (ie, low to high gradient of treatment response) for statistical analyses. WMH scores were dichotomized into absent-mild and moderate-to-severe as follows: PVWMH: absent-mild = 0-1, moderate-to-severe = 2-3; DWMH: absent-mild = 0-1, moderate-to-severe = 2-3; DWMH count: absent-mild = 0-1, moderate-to-severe = 2-3; WMH global load: absent-mild = 1-2, moderate-to-severe = 3-6; WMH count-based load: absent-mild = 1-4, moderate-to-severe = 5-9.
Five multivariable ordinal regression models evaluated the effects of WMH in the RH on language treatment outcomes (ie, one model for each WMH score). In all models, age, education, time post onset, total lesion volume and aphasia severity were included as covariates to assess whether WMH predict treatment outcomes independently from demographics and stroke-related factors. Spearman correlations between WMH and both BNT and CLQT scores assessed the relationship between WMH severity and pretreatment naming and nonverbal executive function. Raw scores were used in all correlation analyses.
Results
The participants’ demographics, time post onset, aphasia severity, lesion volume, PMG scores, and WMH ratings in the RH are reported according to PMG quartiles in Table 2. Multivariate analyses were conducted after excluding collinearity (see Supplemental Table 1 for univariate analyses showing nonsignificant correlations between WMH scores and covariates: age, education, time post onset, total lesion volume, aphasia severity). Importantly, models 2 and 3 (Table 3) revealed that both deep WMH scores (ie, DWMH and DWMH count, respectively) were significant predictors of PMG scores. Specifically, PWA with more severe DWMH and more lesions, presented with lower odds of belonging to a higher PMG quartile (ie, better response to treatment) compared to PWA with absent-to-mild DWMH and fewer lesions, after controlling for stroke-related and demographic factors. When considering total WMH burden, both composite scores (ie, WMH global load and WMH count-based load) were also significant predictors of PMG (models 4 and 5, respectively) with higher composite scores being associated with lower odds of belonging to a higher PMG quartile compared with lower scores (see Figure 2A-C for example cases). Also, stroke-related factors but not demographic factors were associated with PMG scores. Aphasia severity was a significant predictor of PMG in all models such that higher WAB-AQ scores (ie, lower aphasia severity) were associated with higher odds of belonging to a higher PMG quartile relative to lower WAB-AQ scores. Lesion volume was a significant predictor in model 4 only, indicating that PWA with larger lesions had lower odds of being in a higher PMG quartile compared with PWA with smaller lesions.
Median and Range of Demographics, Aphasia Severity, PMG, and WMH Ratings per PMG Quartile.
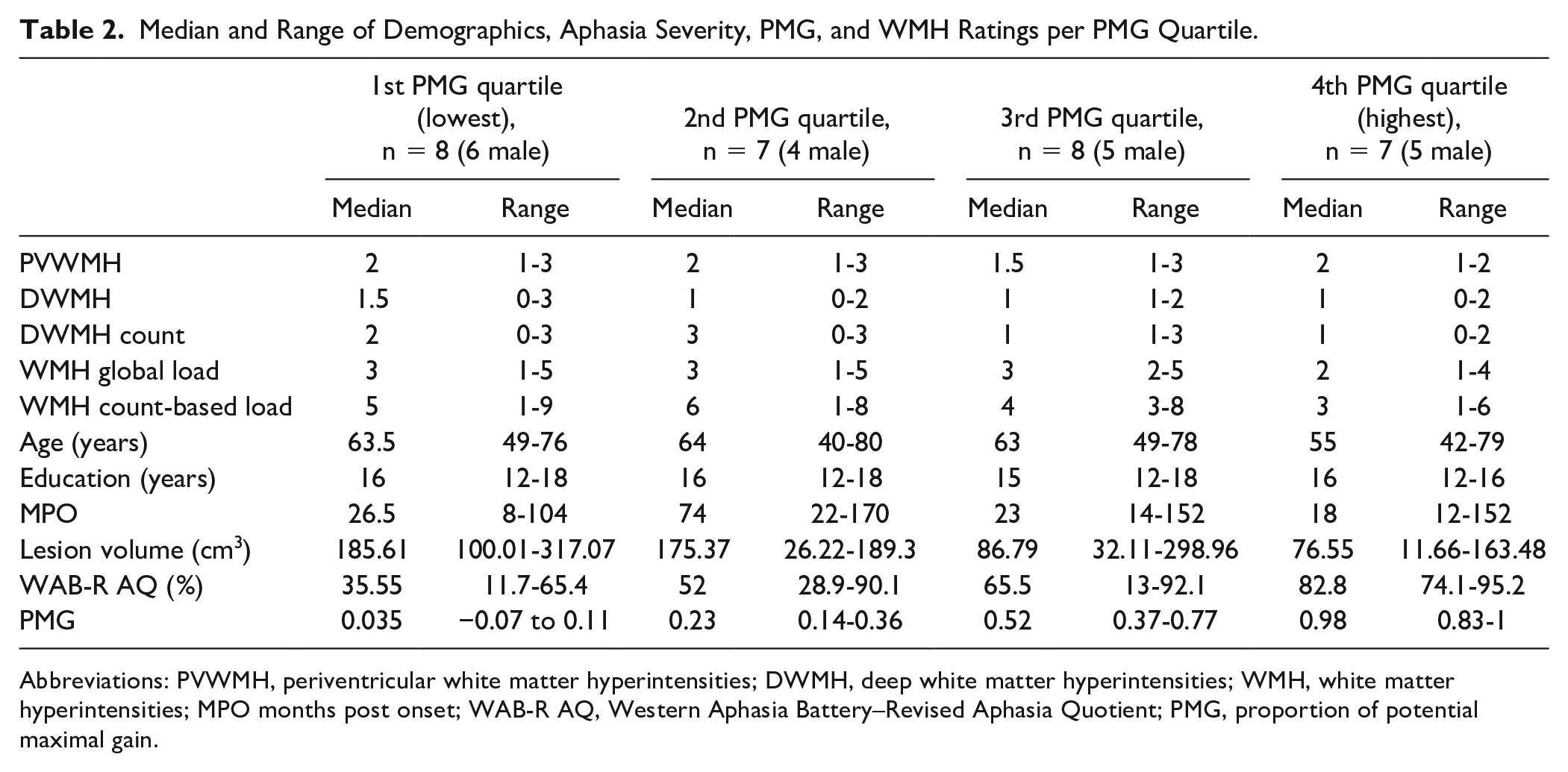
Abbreviations: PVWMH, periventricular white matter hyperintensities; DWMH, deep white matter hyperintensities; WMH, white matter hyperintensities; MPO months post onset; WAB-R AQ, Western Aphasia Battery–Revised Aphasia Quotient; PMG, proportion of potential maximal gain.
Results of Ordinal Regressions.
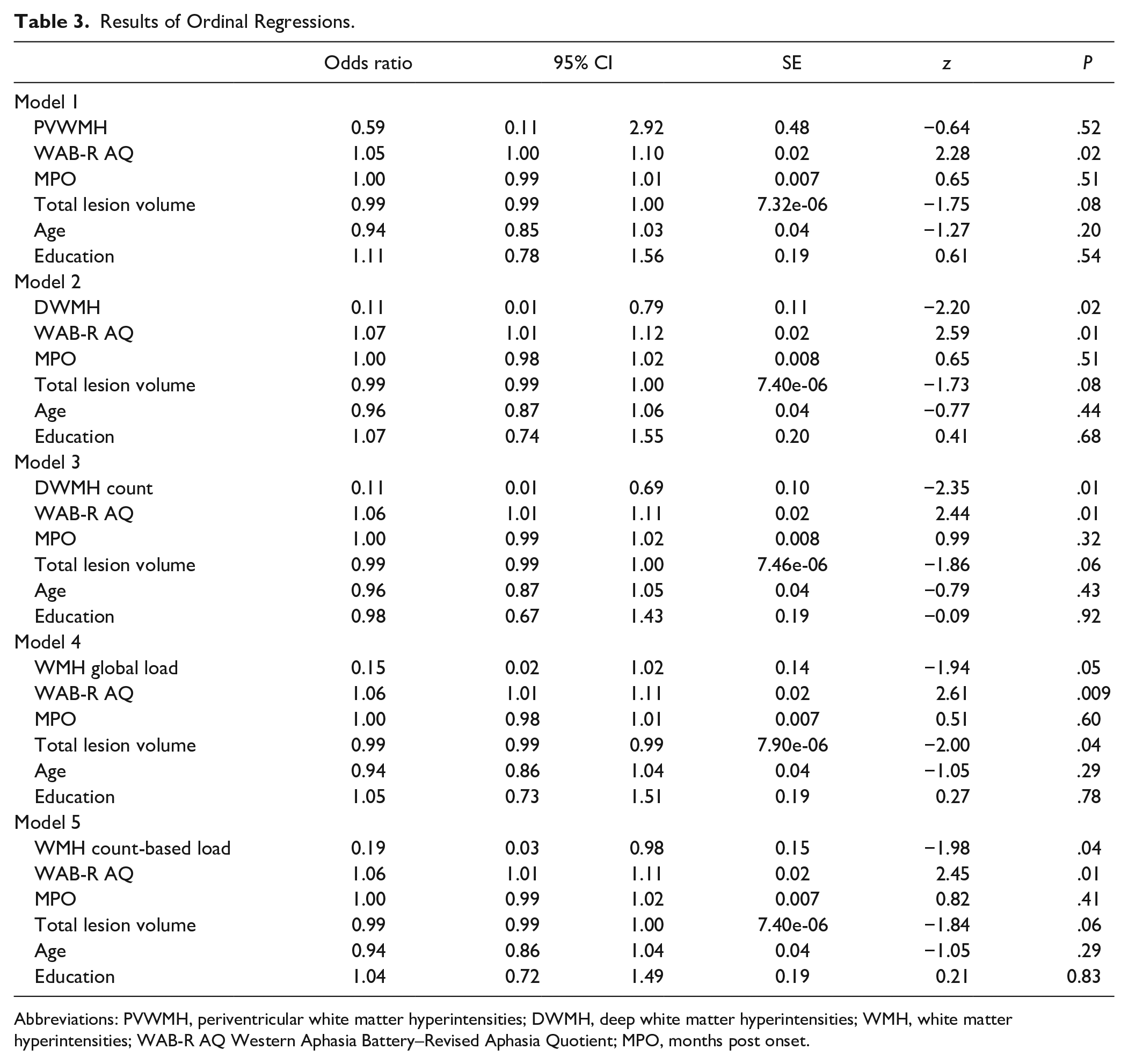
Abbreviations: PVWMH, periventricular white matter hyperintensities; DWMH, deep white matter hyperintensities; WMH, white matter hyperintensities; WAB-R AQ Western Aphasia Battery–Revised Aphasia Quotient; MPO, months post onset.
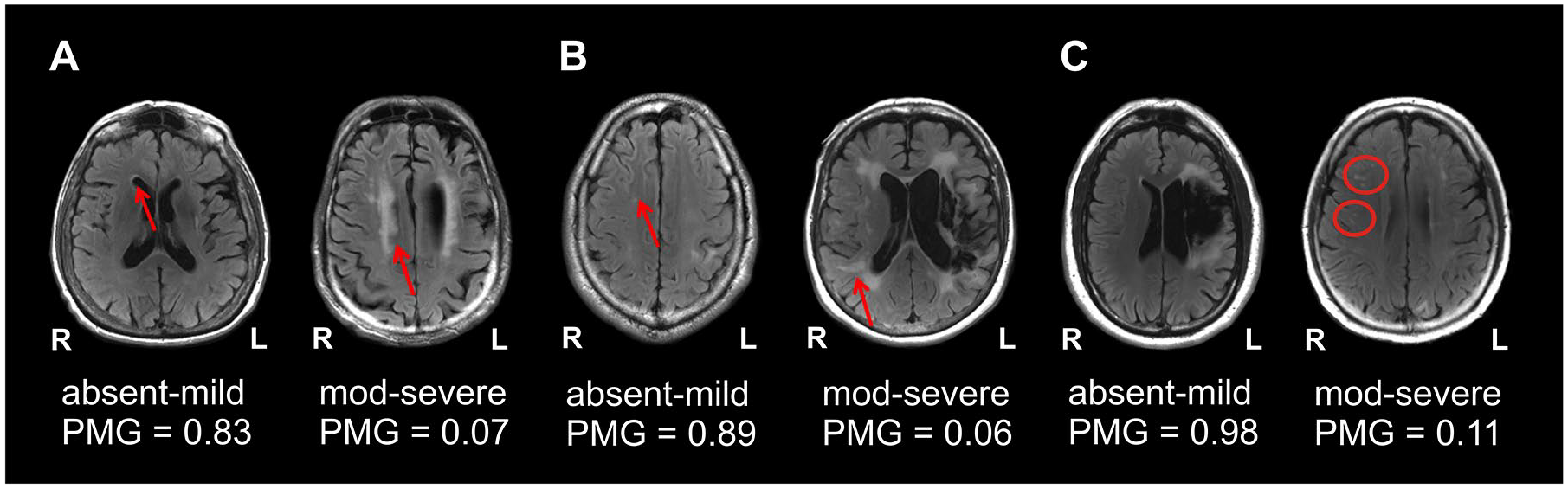
Examples of the association between WMH and treatment outcomes. Panels A-C present the MRI scans (radiological convention) of 6 PWA showing from left to right, that absent-mild WMH are associated with higher PMG scores whereas moderate-severe WMH are associated with worse PMG scores across all three classifications of WMH lesions: periventricular WMH (A), deep WMH (B) and deep WMH lesion count (ie, based on the number of lesions) (C).
WMH scores were not significantly correlated with pretreatment naming scores although they were significantly correlated with pretreatment scores of executive function. Post-hoc correlations showed that executive function scores were not significantly associated with total lesion volume, aphasia severity and performance on the BNT but they were significantly correlated with both PMG raw scores and PMG quartiles (Table 4).
Results of Correlation Analyses B: Spearman’s ρ Values. a

Abbreviations: WAB-R AQ, Western Aphasia Battery–Revised Aphasia Quotient; BNT, Boston Naming Test; PMG, proportion of potential maximal gain.
P values are provided in parenthesis when statistically significant.
Discussion
Our main findings revealed that the severity of WMH in the RH, particularly deep WMH, predicted language treatment outcome in PWA independently from demographic factors and stroke-related factors. Furthermore, WMH in the RH were not associated with stroke-related measures, including time post onset and lesion volume, which is not unexpected given that WMH reflect a chronic degenerative process unrelated to stroke insult. These results are consistent with prior research showing that WMH in the noninfarcted hemisphere can predict poststroke naming outcomes 12 and longitudinal changes in aphasia severity. 13 While these studies highlight the critical role of WMH in language recovery, no previously published research has examined their impact on treatment-induced recovery in poststroke aphasia. Our study provides initial evidence that WMH also play a critical role in modulating treatment-dependent recovery in poststroke aphasia beyond the effects of stroke on language brain regions in the left hemisphere.
A number of potential mechanisms might explain the association between WMH and treatment response in our cohort. WMH are a well-established neuroimaging biomarker of progressive small vessel disease related to cumulative brain damage and associated cognitive impairment.14,32 The presumed underlying pathophysiology is reduced microscopic white matter connectivity leading to a loss of communication between different cortical regions via long white matter tract damage18,32 and hence loss of function. 15 Therefore, their presence and severity could index the overall status of brain health signaling the integrity of brain tissue beyond the stroke lesion. 12 Crucially, language reorganization and neural plasticity in poststroke aphasia rely on the integrity of bilateral white matter tracts 8 and treatment success has been associated with increased reliance on perilesional or spared brain regions in one or both hemispheres.18,33 Therefore, increased WMH burden may negatively affect treatment outcomes by reducing global network connectivity and efficiency 34 and disrupting white matter connections between key regions that sustain language-specific and domain-general processes shown to support language recovery. 33
In line with this interpretation, our findings revealed that WMH in the RH (both DWMH and PVWMH) were associated with nonverbal executive function at pretreatment, which was further associated with PMG scores reflecting direct treatment outcomes. These results align with previous evidence for the detrimental effects of WMH on executive function and other aspects of domain-general cognition in stroke-fee individuals10,15,35,36 and individuals with other neurological conditions, 37 as well as the association between pretreatment executive function scores and anomia therapy outcomes in PWA. 26 As in prior research, 13 we did not find a relationship between WMH in the RH and pretreatment aphasia severity or pretreatment language scores. Although more recent research has provided evidence for such association, 38 the contrast in findings across studies might be related to differences in sample size and WMH severity between patient cohorts and require further investigation. Altogether, our findings suggest that the cumulative effects of WMH on otherwise stroke-spared brain tissue in poststroke aphasia may lead to poor therapy outcomes by decreasing neural and cognitive resources that are essential for recovery, thus reducing their functionality and availability to support improvement during language treatment.
Of note, although PWMH were accounted for in the composite scores reflecting overall WMH burden, they were not a significant independent predictor of treatment response. This is in agreement with the suggestion that distinct WMH patterns have differing pathogenesis, risk factors, effects on the brain and hence clinical consequences. 39 Indeed, postmortem MRI-histopathological studies suggest that each type of WMH reflects distinct neuropathological changes and severity. In brief, higher DWMH load is associated with increasing severity of demyelination and axonal damage, hence more severe disconnection and microstructural damage, compared to PWMH. 39
Studying the role of WMH on treatment-induced recovery in aphasia has important clinical implications. 40 Our findings contribute to our current understanding of relevant anatomical factors that modulate treatment response in aphasia, that have been otherwise traditionally focused on just patient-related and stroke-related factors. 6 Determining relevant predictors of therapy outcome can inform individual prognosis, treatment planning, and prompt allocation of additional care and support resources. Importantly, the detection of high WMH severity in poor responders to treatment can help identify PWA at risk for cognitive decline in order to track their long-term language and cognitive status and consider additional neuropsychological stimulation in their rehabilitation programs.
Despite the cross-sectional nature of our study, it is worth considering that the accumulation of WMH over time can lead to long-term negative cognitive effects. Both population- and hospital-based cohort studies indicate that the progression of WMH is related to a parallel decline in cognitive function and a considerable risk of developing dementia.10,40-42 Given the high prevalence of WMH in the older adult population and their relationship with cognitive function and recovery in PWA, we argue that WMH should be identified and assessed for prognostic purposes. Longitudinal studies capturing the progression of WMH in PWA after stroke may provide further insights into the relationship between WMH and responsiveness to language therapy.
A few limitations in the present study should be considered for future research. We evaluated WMH in the RH using the Fazekas scale, a validated and widely used metric in the small vessel disease field of research. 43 While visual rating scales provide limited measurements of WMH, they have excellent intra- and interobserver agreement, they can be readily used in clinical settings, and scores on this scale closely correlate with WMH volumetric assessments. 43 In addition our predefined cut-off used for the distinction between absent-mild versus moderate-severe WMH in different locations (ie, PVWMH and DWMH) was sensitive enough to capture the topographic differences and independent associations consistently across different statistical models. Advanced volumetric methods adapted to measure WMH in patients with large stroke lesions could be used to obtain exact WMH volumes and localization. These metrics may allow to identify more subtle associations between WMH and treatment outcomes, and to examine regional variability in WMH location by assessing the relationship between the specific topography of WMH and cognitive impairment as done in prior research with other populations.44,45 Our findings require further replication with larger samples to rule out the possibility of selection bias and with patient cohorts receiving different types of treatment to ensure generalization of our findings. Also, future research should examine other neuroanatomical markers of small vessel disease to evaluate its overall effects on therapy response in PWA as a whole-brain disorder. 18 Finally, due to the retrospective nature of our study, we could not control for the time post onset at which treatment was initiated across patients. Although all PWA were in the chronic phase of stroke and time post onset was included in our models as a confound variable, the range of time post onset at which therapy was initiated is broad in our cohort, which may affect the magnitude of treatment responsiveness, especially in more chronic patients. Future research should examine the relationship between WMH severity and response to language therapy in more homogeneous cohorts in this regard and at time points closer to the stroke event.
Conclusions
Our findings indicate that WMH in the RH can predict treatment-induced recovery in post-stroke aphasia independently from well-established individual-dependent and stroke-related factors and suggest that WMH burden may affect the availability of neural and cognitive resources that support language improvement. We support the use of WMH of presumed vascular origin on MRI, as a proxy of neural tissue integrity and overall brain health that should be further studied as a neuroanatomical marker of language recovery in intervention trials and general aphasia research.
Supplemental Material
supplamental_materials_NNR – Supplemental material for White Matter Hyperintensities Predict Response to Language Treatment in Poststroke Aphasia
Supplemental material, supplamental_materials_NNR for White Matter Hyperintensities Predict Response to Language Treatment in Poststroke Aphasia by Maria Varkanitsa, Claudia Peñaloza, Andreas Charidimou, David Caplan and Swathi Kiran in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to thank all participants for their time and commitment to this study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Swathi Kiran serves as a consultant for The Learning Corporation with no scientific overlap with the present study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been supported by the Dudley Allen Sargent Research Fund of Boston University, post-doctoral grant awarded to Maria Varkanitsa and Claudia Peñaloza and by NIH/NIDCD grant P50DC012283 (Center for the Neurobiology of Language Recovery).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
