Abstract
Background. Locomotor training of rats with thoracic contusion spinal cord injuries can induce task-specific changes in stepping but rarely results in improved overground locomotion, possibly due to a ceiling effect. Thus, the authors hypothesize that incompletely injured rats maximally retrain themselves while moving about in their cages over the first few weeks postinjury. Objective. To test the hypothesis using hindlimb immobilization after mild thoracic contusion spinal cord injury in adult female rats. A passive stretch protocol was included as an independent treatment. Methods. Wheelchairs were used to hold the hindlimbs stationary in an extended position leaving the forelimbs free. The wheelchairs were used for 15 to 18 hours per day, 5 days per week for 8 weeks, beginning at 4 days postinjury. A 20-minute passive hindlimb stretch therapy was applied to half of the animals. Results. Hindlimb locomotor function of the wheelchair group was not different from controls at 1 week postinjury but declined significantly over the next 4 weeks. Passive stretch had no influence on wheelchair animals but limited functional recovery of normally housed animals, preventing them from regaining forelimb–hindlimb coordination. Following 8 weeks of wheelchair immobilization and stretch therapy, only the wheelchair group displayed an improvement in function when returned to normal housing but retained significant deficits in stepping and coordination out to 16 weeks. Conclusion. Hindlimb immobilization and passive stretch may hinder or conceal the normal course of functional recovery of spinal cord injured rats. These observations have implications for the management of acute clinical spinal cord injuries.
Introduction
A potential impediment to the translation of activity-based rehabilitation following spinal cord injury (SCI) from animal models to patients is that the conditions of recovery in animal models do not parallel those of patients. Most patients are immobile over the first few weeks following injury, except for muscle stretch and passive joint range-of-motion (ROM) therapies.1,2 In contrast, animals used in experimental studies of SCI begin to move about in their cages within a few days or a week of injury. With a few exceptions,3-5 the majority of animal studies do not mention the use of stretch or ROM therapies, yet these are widely accepted in clinical practice worldwide,6,7 and the Consortium for Spinal Cord Medicine prescribes that physical therapy begin within the first week postinjury and continue throughout the acute phase. A recent review of clinical trials of SCI rehabilitation found these therapies to be ineffective when compared with no intervention or conventional care.8,9 The effects of immobilization and stretching/ROM maneuvers in animal models of SCI remains largely unstudied.
Step training with weight support is a promising form of activity-based rehabilitation for SCI patients that is often used in conjunction with stretch/ROM therapies. The step-training approach is deeply rooted in the elegant studies by Grillner, Rossignol, Edgerton, and others showing that adult cats with complete spinal transections can be retrained to perform quality hindlimb stepping via repeated exposure to the activity on a treadmill. Afferent input associated with phasic limb movements, loading, and paw or foot contact10-12 is thought to retrain spinal circuitry below the injury. However, improvements in overground ambulation for patients participating in step training are variable and far less than expected based on the preclinical feline studies.13-15 Similar to clinical studies, step training in rodent models of incomplete SCI has shown only modest, task-specific improvements in treadmill stepping that rarely lead to improvements in overground stepping.16-20 In contrast to the clinical situation, however, adult rats with all but the most severe of incomplete injuries show remarkable spontaneous recovery leading some authors, including ourselves, to suggest that early in-cage activity provides sufficient amounts of appropriate afferent input to bring about substantial, even maximal, retraining that is difficult to improve on using a variety of approaches including step training on the treadmill, stepping in shallow water, or swimming.17,19,20-24
The current study was undertaken to test the hypothesis that early in-cage activity is responsible for the dramatic functional improvements seen in adult rats with incomplete SCI. We limited hindlimb activity and altered the pattern of activity-dependent afferent input by placing the animals in wheelchairs, 15 to 18 hours per day for 5 days a week starting at 4 days postinjury. In addition, we explored the influence of a daily stretch protocol on the profile of recovery in hindlimb-immobilized animals and normally housed animals as controls. We found that periodic hindlimb immobilization in a wheelchair had a negative and lasting effect on functional recovery in rats with mild SCIs and that a daily passive stretch therapy had a lasting negative impact on recovery for normally housed SCI animals, but was not a factor for those experiencing hindlimb immobilization.
Methods
Spinal Cord Injury and Experimental Design
Twenty-one female adult Sprague-Dawley rats (190-215 g) were used for this study. All procedures involving experimental animals were performed according to the guidelines of the University of Louisville Institutional Animal Care and Use Committee. Animals were assigned to 3 experimental groups: Wheelchair/Stretch (WC/SR), wheelchair/non-stretch (WC/non-SR), non-wheelchair/stretch (non-WC/SR), n=5 each or non-wheelchair/non-stretch (non-WC/non-SR) controls, n=6. Animals received contusion injuries at the T10 spinal cord level as described previously.21,24,25
Figure 1 shows a timeline of the study and all assessments. On postinjury day 4, animals in the 2 wheelchair (WC) groups spent 15 to 18 hours per day (from approximately 4

Wheelchair immobilization and stretch protocol was for an 8-week period (dashed line) followed by 8 weeks of normal (2 per cage) housing. The schedules of injury and treatments are shown below the timeline, and the schedules of assessments are shown above the timeline.
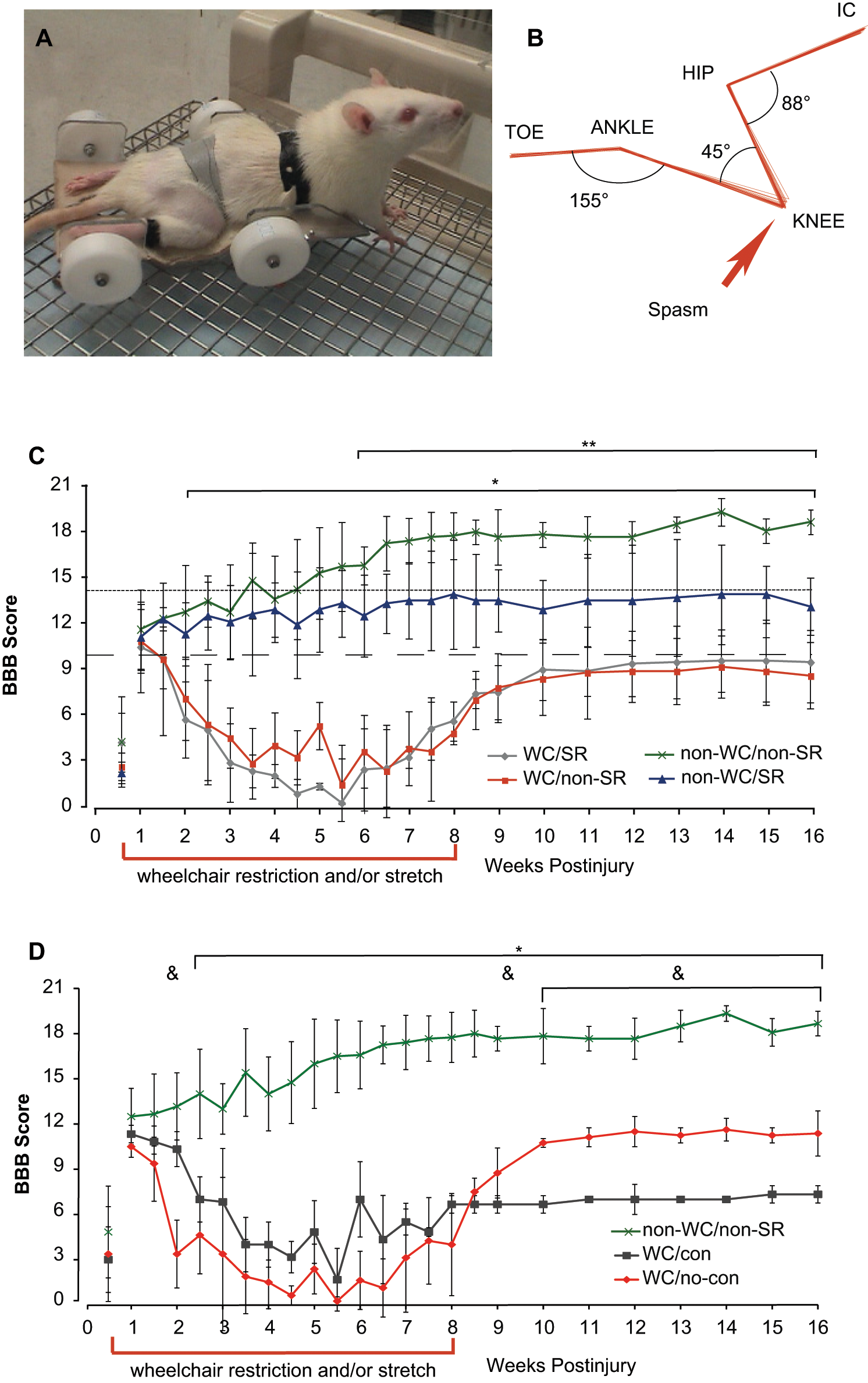
(A) An injured animal in a wheelchair. Velcro straps held the animal and hindlimbs in place. (B) A 3-dimensional kinematic analysis of the right hindlimb during wheelchair immobilization. The anatomic landmarks represented are the iliac crest (IC), hip, knee, ankle, and toe. Some movement of the knee or ankle was sometimes seen during spasms, as shown here for the knee. For (C) and (D) all data are shown as means ± standard deviation. (C) The BBB scores over time for all 4 experimental groups (WC, wheelchair; SR, stretch). All animals scored 21 preinjury. Significant differences were found for both WC groups (n = 5) compared with non-WC/non-SR controls (*, n = 6, P < .05). Animals receiving the SR intervention (non-WC/SR, n = 5) had BBB scores that were significantly different from non-WC/non-SR controls (**, n = 6, p < .05, (D) The BBB scores over time for groups based on presence (con) or absence (no-con) of ankle contractures. WC/con (n = 3) and WC/no-con (n = 4) groups have significantly lower BBB scores compared with controls for weeks 2 to 16 (*, n = 6, P < .05). Scores for the WC/con group are significantly different from the WC/no-con group at weeks 2, 6, and 10 to 16 (&, P < .05).
Rat Wheelchair Design
The wheelchairs used held the hindlimbs in a position that resembled the early hindlimb dragging phase of recovery following SCI (Figure 2A). Figure 2B shows a representative sample of kinematic analysis of hindlimb position, at rest, during WC immobilization. Hindlimbs were statically positioned with joint angles of approximately 88° (hip), 45° (knee), and 155° (ankle) and were held in position using Velcro straps.
Daily Care and Stretch Protocol
In addition to standard postoperative care, all animals received daily care 5 mornings per week for the first 8 weeks of study. Daily care consisted of washing with warm water and towel dry and massage of the hindlimbs in a distal to proximal direction for pressure relief and circulation. The left side hindlimb tibialis anterior (TA) and medial and lateral gastrocnemii (MG and LG, respectively) were harvested on termination to assess muscle atrophy. MG and LG were weighed together.
Following daily care, animals in the SR groups received a 20-minute hindlimb stretch protocol, consisting of 2 sets of bilateral hindlimb manipulations that held each joint in maximally flexed and extended positions for 30 seconds each. Handlers documented each stretch session and noted the side and joint order and observations about joint stiffness and any loss of ROM.
Behavioral and Kinematic Assessments
Each animal was assessed using the BBB Open Field Locomotor Scale and the Louisville Swim Scale (LSS) throughout the study.16,28 In addition, animals were assessed periodically for sensory changes using Hargreave device (thermal hyperalgesia) and von Frey filaments (somatosensory hypersensitivity).
To quantitatively assess hindlimb movement during walking and swimming, we performed kinematic analysis at various time points throughout the study as previously described.23,24 Briefly, the movements of the hindlimbs during overground stepping and swimming were quantified using markers placed over the iliac crest (I), hip (H), ankle (A), and toe (T), and digital video was analyzed using MaxTraq3D (Innovision Systems, Columbiaville, Michigan).
Electrophysiological Assessment
Some WC animals developed ankle pathologies that resulted in a severe loss of ROM. To assess the tone of, and estimate spasticity in an ankle extensor, we performed simultaneous bilateral electromyographic (EMG) recordings of lateral gastrocnemius muscles (LG) at rest and during tail and paw pinches for all animals. Tail and paw pinch involved closing a rubberized paper clamp 1-in. from the base of the tail and onto the plantar and dorsal aspects of the paw for approximately 1 second, repeated 5 times over a period of 3 to 5 minutes. Recordings were filtered and rectified for analysis in Axograph 4.0 on a Macintosh G4 computer to determine the timing of burst onset and offset. The percent bilateral co-contraction of LG muscles during tail and paw pinch was calculated as a surrogate indicator of spasticity.30,31 The duration of co-activity was divided by the total duration of activity (either muscle) per set of bursts and converted to a percentage.
Histological Outcomes
All animals were euthanized at the end of week 16 and perfused transcardially with 4% paraformaldehyde and calcium-free tyrodes solution. The entire length of spinal cord was removed for histological analysis, and sciatic nerves from each left hindlimb were harvested for analysis of cross-sectional area. Adjacent sets of spinal cord sections were stained for white (eriochrome cyanine) or gray matter (cresyl echt violet for Nissl substance) and processed for spared white matter at the epicenter, damaged gray matter, and cavity volume as previously described.29,32
In Vivo Scanning of Bone Architecture
Micro computed tomography (micro-CT) scanning was performed of the distal femurs at 6 and 13 weeks postinjury as described previously. 33 Briefly, each rat that did not receive the SR protocol was positioned with the left hindlimb flexed and the right hindlimb extended in the lower column of a custom-built animal holder. Scans were performed with a custom scanner using a 225-kV X-ray source and a 1024 × 1024 digital camera. Each scan covered a 3-mm distance below the knee joint as 214 slices. Bone volume fraction was calculated as bone volume over total volume and images were reconstructed using VGStudio Max (Volume Graphics, Heidelberg, Germany). Data are presented in Figure 2 as means ± standard deviation (SD) based on contracture groupings within the WC/non-SR group: no-contracture, n = 2; contracture, n = 3; and non-WC/non-SR controls, n = 6.
Statistical Analysis
All data are presented as group means with standard deviations (±SD). Outcome measures were analyzed using repeated-measures analysis of variance (ANOVA) with groups as a factor or 1-way ANOVA and were followed by Tukey HSD or Bonferroni post hoc t tests where appropriate. Differences between groups were considered statistically significant for P values of at least ≤.05. The binomial proportions test was used to determine the validity of regrouping animals based on presence or absence of ankle contracture (contracture and no-contracture, respectively) and to determine differences in the number of animals per group with ankle contractures. Proportions were checked to ensure the sample size was not too small nor the proportions too extreme for comparison. 35 No corrections to the proportions were required. All data were sorted and assessed for significant differences as follows: (a) based on the original experimental design WC/SR, WC/non-SR, non-WC/SR, non-WC/non-SR controls, and in cases where the data indicate a bimodal distribution and (b) based on contracture or no-contracture.
Results
Behavioral Assessments
BBB assessments were performed twice weekly for the first 8 weeks during which wheelchair immobilization (WC) and/or hindlimb stretch (SR) were interventions (Figure 2C and D, dashed bar along time axis) and then weekly for the remainder of the 16-week study. Animals were taken out of the wheelchairs, underwent daily care with or without SR, and were placed into standard housing, 2 per cage, for at least 4 hours prior to any assessments. All BBB assessments for both WC groups are compared with control animals (Figure 2C, WC/SR n = 5, WC/non-SR n = 5 vs non-WC/non-SR n = 6), and non-WC animals that received SR are compared with controls separately (Figure 2C, non-WC/SR n = 5 vs non-WC/non-SR n = 6). The group averages were similar for all 4 groups in 2 aspects: (a) at postinjury day 4 all animals, as expected, were dragging their hindlimbs with no weight support and had varying degrees of hindlimb joint movement; (b) at postinjury day 7, most animals had some degree of weight support and plantar stepping. Figure 2C shows a profile of recovery that was significantly different for both WC groups as compared with controls beginning at week 2 through the remainder of the 16-week study (±SD, n = 5/6, P ≤ .05). Loss of function for WC groups became evident at week 2, and the decline in BBB scores continued until weeks 4 to 5. At week 4, WC animals exhibited slight to extensive movement of hindlimb joints and were consistently dragging their hindlimbs (WC/SR group average BBB = 1.9 ± 1.9, WC/non-SR = 3.9 ± 2.3); in contrast, the control group had recovered consistent weight-supported plantar stepping with consistent coordination (non-WC/non-SR, BBB = 14.0 ± 2.4).
The wheelchair immobilization and stretch protocol ended at week 8, and statistically significant deficits in BBB scores remained for both WC groups (WC/SR 5.5 ± 2.1, WC/non-SR 4.7 ± 3.3), when compared with control animals whose scores had reached a plateau (non-WC/non-SR 17.8 ± 1.7). WC groups regained some function between weeks 6 and 10; however, group averages indicate that the inability to achieve weight support with consistent plantar stepping persisted for the duration of the study (BBB < 10 denoted by the dashed line in Figure 2C). These data represent a substantial effect of intermittent WC immobilization in animals that could move around in their standard cages for approximately 8 hours per weekday and continuously (approximately 56 hours) each weekend.
Any effect of the daily SR protocol on WC animals was minimal and/or was rendered undetectable by the negative influence of wheelchair immobilization. We anticipated the need for circulatory and muscle maintenance for WC animals, and the SR protocol was included as an effort to alleviate any pathologies associated with hindlimb immobilization. Surprisingly, the BBB scores of the non-wheelchair/SR (n = 5) suggest they were detrimentally affected when compared with controls (Figure 2C). Beginning at week 6, the group mean BBB scores were significantly lower (non-WC/SR 12.4 ± 2.2) than the non-SR control group (±SD, n = 5/6, P ≤ .05). The dotted horizontal line, BBB = 14, indicates consistent forelimb–hindlimb coordination; although the standard deviation for this group is high, ~3 points, these data show that receiving a 20-minute protocol of hindlimb stretch therapy daily produced a group average BBB score indicating an inability to achieve consistent coordination.
Prevalence of Ankle Contractures
Beginning at postinjury weeks 4 to 5, some WC animals experienced pathological changes to their ankle joints resulting in negligible ROM.36-38 Details of these findings are shown in the supplementary material. Briefly, 9 out of the 20 total ankles developed contractures independent of inclusion in the SR protocol, showing that the stretch protocol did not influence the probability of developing ankle contractures.
Figure 2D represents the same data as shown in Figure 2C, but is regrouped based on presence (WC/contracture, n = 3) or absence (WC/no-contracture, n = 4) of ankle contractures in the WC animals. The profile of recovery during weeks 2 to 4, indicating a loss of function for WC animals, remains similar to Figure 2C. WC animals, with and without ankle contractures, have significantly lower scores when compared with controls beginning at week 2.5 and persisting to the end of the study. Weeks 10 to 16 reveal significantly different degrees of functional recovery between animals that had no contractures versus those that did develop chronic ankle pathology. During weeks 10 to 16, WC animals with no discernable ankle pathology recovered frequent weight-supported stepping lacking coordination (WC/no-contracture highest mean BBB score 11.6 ± 0.75, week 14), whereas animals with ankle contracture could not achieve extensive movement of all 3 hindlimb joints nor plantar placement (WC/contracture highest mean score 7.3 ± 0.58, week 15).
Kinematic Assessment of Stepping
For stepping, group 3D kinematic excursion data were quantified for the hip, knee, and ankle (Figure 3A). Group averages were compared at weeks 6 and 11. At week 6, WC animals showed significantly reduced mean excursions for all 3 joints when compared with non-WC animals (Figure 3A). Hip and knee positions were relatively flexed (70° and 40°, respectively), whereas the ankles were extended (140° to 160°; Figure 3B and D). By week 11, both WC groups regained hip excursions that were not different from controls and also regained some knee excursion, which remained significantly different from non-WC animals by having a lower trough value (flexion; Figure 3C and E). Mean ankle excursions at week 11 were significantly different for the 2 WC groups compared with both non-WC groups, and also between WC groups (WC groups contracture/no contracture; Figure 3A). By week 11, WC animals without contractures had recovered sufficient ROM about the ankle to achieve weight-supported, plantar stepping (Figure 3A and C), whereas animals with ankle contractures had very low excursion ranges reflecting their very limited ankle ROM. There were no differences between non-WC groups with or without SR at any time points, indicating that deficits observed using the BBB scale were in interlimb coordination and not in joint excursions during stepping.
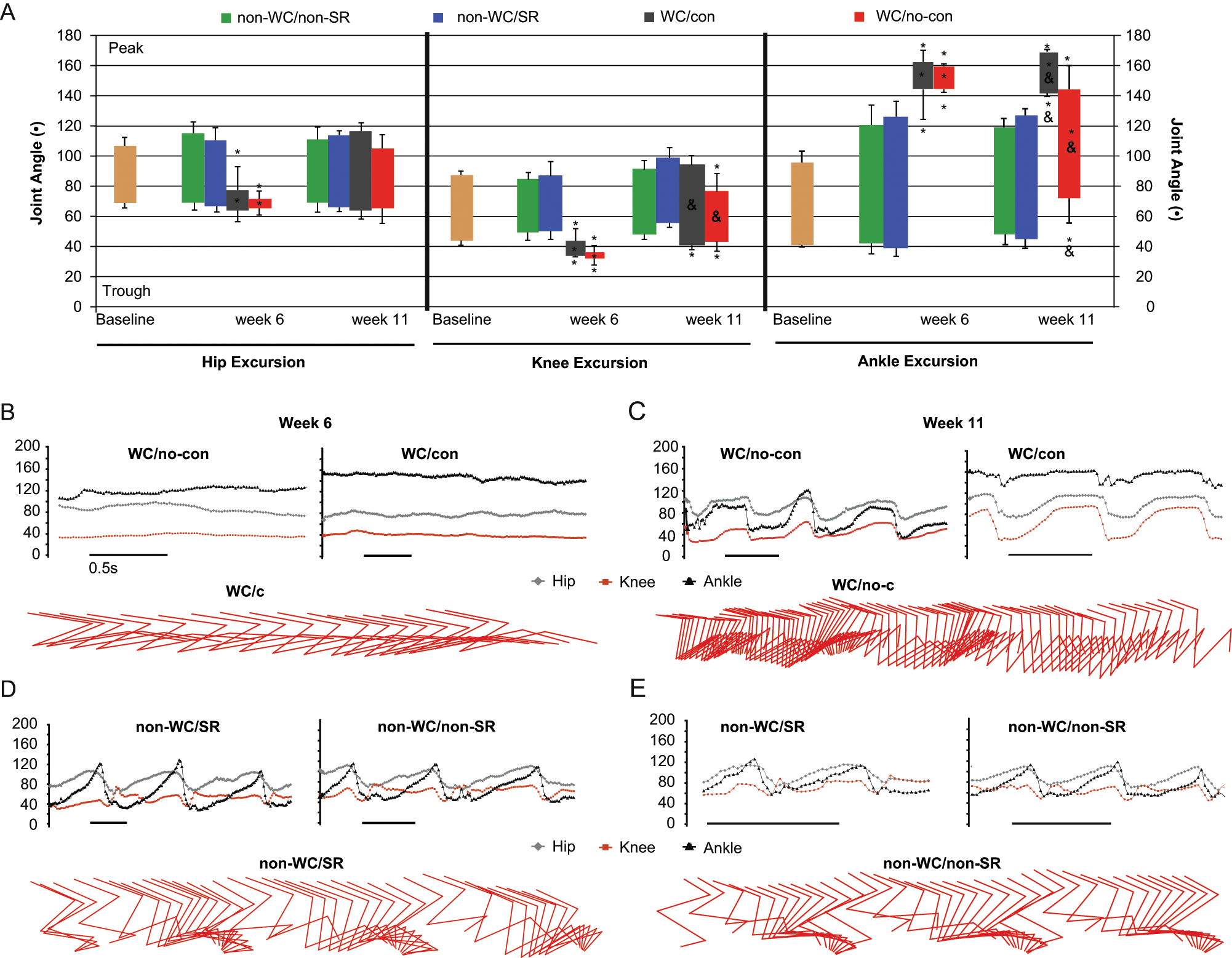
Three-dimensional kinematic analysis of hindlimb joint excursions during overground stepping. All data are shown as group means ± standard deviation. (A) Joint excursion is presented as the range of angles through which the joint moves and is calculated as maximum peak angle (extension) minus the trough angle (flexion). Baseline excursion data represent the preinjury average for all animals. Symbols within each bar represent significant differences in average excursion; symbols above and below represent significant differences in average peak or trough angles; (*) indicates significant differences for both WC groups (WC/con, n = 3, and WC/no-con, n = 4) when compared with both non-WC groups (non-WC/SR, n = 5, and non-WC/non-SR controls, n = 6), P < .05. (&) represents significant differences between the 2 WC groups, P < .05. (B) through (D) show 2 examples of excursion plots (joint angle changes over time) and examples of stick figures. (B) and (C) show excursion plots for WC rats with and without contractures and stick figures showing extended-limb dragging (B) and flexed limb dragging (C) in WC animals with and without contractures, respectively. WC/con animals displayed cyclic hip and knee movement despite the reduced ankle ROM. (D) and (E) show excursion plots and stick figures for non-WC animals that represent near-normal stepping of the hindlimbs that was not different from baseline at either time point.
Swimming Assessments
Swimming was used as an assessment of locomotion that does not require the ability to support body weight. Details of the swimming results can be found in the supplementary material. Briefly, both wheelchair groups exhibited significantly lower scores for swimming than the non-WC groups during weeks 3, 5, 7, and 11 (Supplementary Figure 1). No differences in LSS scores or LSS subscores were found for non-WC/non-SR and non-WC/SR groups despite the differences previously identified in overground stepping (Figure 2). However, BBB subscores were significantly better for the non-WC animals that did not receive the SR protocol (non-WC/non-SR; Supplementary Figure 1). These observations show that the SR protocol appears to negatively influence the recovery of stepping more strongly than the recovery of swimming.
Bone and Muscle Loss During Hindlimb Restriction
Results for the micro-CT scanning of hindlimb tibiae, body and muscle weight, and hindlimb EMG recording can be found in the supplemental material. Briefly, no significant differences were found for bone volume ratio and body or hindlimb muscle weights. Analysis of EMG recordings showed that WC animals had significantly greater co-contraction than non-WC animals in response to tail pinch, but no differences were seen in response to paw pinch (Supplementary Figure 2).
Histological Assessments
Using a 1-way ANOVA, no significant differences were found in the percentage of spared white matter (cross-sectional area), gray matter loss, or cavity volume (mm3) at the epicenter among any groups (Table 1). The amount of spared white matter at the epicenter (30%) is consistent with the functional recovery achieved by the control group as measured by the BBB scale (~18). 29 BBB scores of ~10 at week 1 and terminal scores of ~18 for injured controls are higher than expected with a 200 kD contusion injury using the Infinite Horizons system. We attribute the relatively mild injuries to the custom-made system we used to stabilize the vertebrae, which was novel for us at the time. Nonetheless, all the animals were injured in a consistent manner.
Mean Spared White Matter Cross-Sectional Area (CSA) at the Injury Epicenter (mm2), Gray Matter Volume Loss (mm3), and Cavity Volume (mm3) a
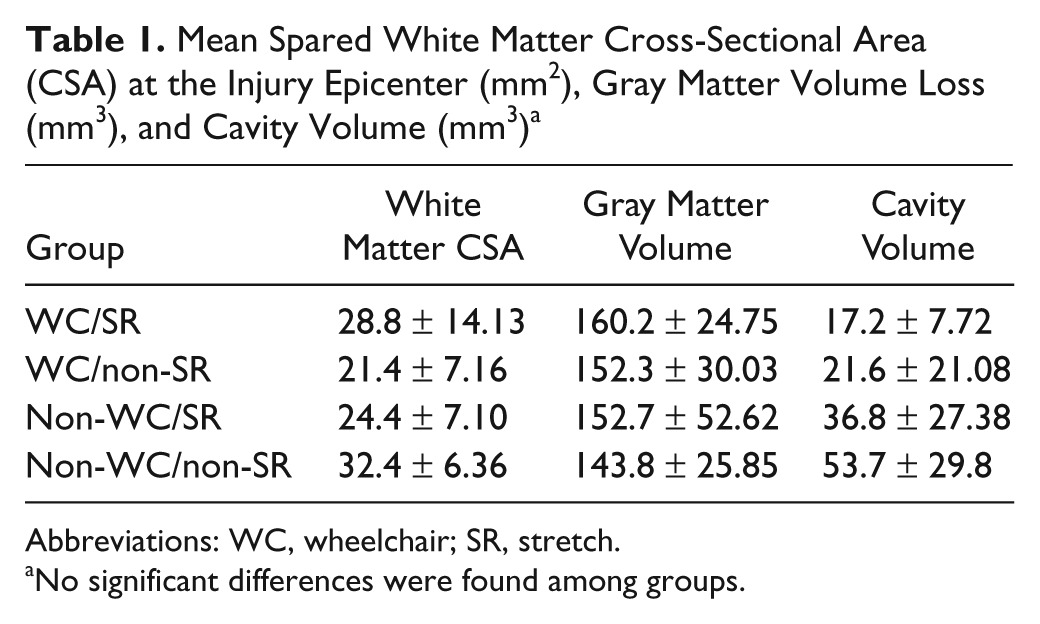
Abbreviations: WC, wheelchair; SR, stretch.
No significant differences were found among groups.
Discussion
It has been more than 3 decades since Sten Grillner and his talented trainees began referring to “the central generation of locomotion” while studying the low spinal cat model.39,40 Since then, this model of acute transection has been used to characterize the capabilities of the lumbar spinal cord and the principles underlying locomotor retraining on the treadmill. The work of many laboratories including those of Edgerton, Rossignol, and Barbeau have identified several issues including phasic limb loading, cutaneous paw contact, and load progression as critical to retraining.12,41-46 In addition, training is thought to be task specific in that improvements are largely limited to the trained task.11,21,47,48
Translating these key principles to the rodent and human models, however, has not been straightforward. Inherent to these 3 models—human, cat, and rat—are a number of critical differences, as reviewed by Majczynski and Slawinska, 49 which may hinder translation and complicate the interpretation of experimental results. For example, most rodent studies use midthoracic transections/contusions or incomplete lacerations and acute or subacute retraining. In contrast, most clinical studies involve chronic incomplete or dyscomplete cervical or high-thoracic contusions. These 3 models also have very different locomotor characteristics: bipedal versus quadrupedal, high versus low versus very low center of gravity. The characteristics of balance, trunk stability, and posture and the consequences of errors in stepping vary considerably and dramatically influence the capabilities of each species during the critical acute and subacute postinjury time period. Rodents are very active within days of injury whereas patients are largely immobile for several weeks or months, experiencing little more than transfer, stretching, and passive ROM therapies during this period. Apart from the substantial studies conducted by Dobkin and colleagues, rarely will patients enter an activity-based rehabilitation setting before 3 to 6 months postinjury.2,14
Importantly, step training of spinal cord injured rodents consistently demonstrates task-specific changes in hindlimb function but rarely achieves frank improvements in overground stepping. Fouad and colleagues 17 used daily treadmill training in an over-hemisection rat model and were unable to uncover improvements in overground stepping using the BBB scale and kinematics as outcomes. As also alluded to earlier by de Leon et al, 50 Fouad et al 17 concluded that spontaneous recovery resulting from self-training was substantial and that treadmill training provided no added benefit. Subsequently, Multon et al 18 used manually assisted treadmill training of rats with T7 microballoon compression injuries to demonstrate that small improvements in BBB scores could be achieved. Notably, these animals were housed individually, without enrichment. 27 Invariably, studies using rats with all but the most severe incomplete injuries report substantial improvements in overground locomotion over the first few weeks postinjury.17-19,29,51,52 Similar reports for cats following thoracic contusion or incomplete laceration injuries are limited, 53 with the majority of studies being focused on task-specific improvements of limb function on the treadmill.
Taken together, these observations suggest that the in-cage activity exhibited by rats with acute incomplete SCIs should be viewed as cumulative step training. Therefore, we hypothesized that incompletely injured rats maximally retrain themselves within the first few weeks after injury. To test this hypothesis, we designed a 4-wheeled cart or wheelchair that dramatically restricts hindlimb movement by holding the limbs in an extended-drag position and alters the normal pattern of afferent input while still permitting good in-cage mobility. Anticipating that hindlimb immobilization might induce pathologies of muscles and joints, we also developed a stretching protocol based on standard-of-care clinical physical therapy. 9 We found that neither intermittent hindlimb immobilization (WC) nor stretch (SR) initiated at 4 days postinjury, prior to the resolution of spinal shock, had any influence on the dramatic early increase in hindlimb function. Both SR and non-SR and WC and non-WC groups had similar BBB scores at 7 days postinjury (10.6 ± 1.43 and 11.3 ± 1.85, respectively), suggesting that this phase of recovery is likely not dependent on activity or patterns of afferent input. Thereafter, WC (SR and non-SR) animals experienced a dramatic loss of hindlimb function leaving them unable to generate weight-supporting plantar steps by 3 weeks postinjury. Poor hindlimb function, assessed using the BBB scale and kinematically, persisted for the 8-week duration of WC immobilization. When returned to normal housing at 8 weeks postinjury, function rebounded somewhat during weeks 8 to 10 but remained significantly below the non-WC/non-SR control group out to week 16, suggesting that the spinal cord circuitry was not as responsive to in-cage retraining beyond 8 weeks postinjury. Both WC/SR and WC/non-SR groups also developed ankle pathologies at a 40% to 50% rate that dramatically limited ROM. Only modest decreases in muscle weights were observed for the LG and MG only (not the TA), and the WC/SR group experienced loss similar to the WC/non-SR group. The SR protocol did appear to ameliorate a decrease in bone volume ratio seen in the WC/non-SR group; however, our group sizes were too small to achieve significance with this measure. Importantly, when the animals with ankle pathologies were excluded, a significant difference in the BBB scores and kinematic measures persisted, showing that WC immobilization had a dramatic and lasting negative influence on locomotor function.
Conceptually, there are interesting parallels between the current work, the literature discussed above, and the work of Maier et al, 54 using a rat model of unilateral corticospinal tract injury. Maier and colleagues showed that constraining the use of one limb, thus forcing the use of the impaired limb, led to dramatic improvements in function that were correlated to increased contralateral collateralization of corticospinal fibers in the cervical gray matter. When not forced to use the forelimb on the injured side, it remains immobile and the long term functional (and anatomical) consequences of dis-use are dramatic.
In contrast to the anticipated loss of function brought about by WC hindlimb immobilization, we also observed a significant and surprising difference between the SR and non-SR groups of non-WC animals, indicating that our stretch protocol was having a detrimental effect on hindlimb function. The differences between groups developed somewhat gradually between weeks 2 and 5, suggesting that the SR protocol was bringing about a functional plateau earlier than observed in the non-SR control animals. Despite stretching/ROM being a standard-of-care practiced clinically, systematic literature reviews have suggested that these approaches have mixed benefits or the data appear inconclusive at best. 9 While many dubious explanations may be put forward for our observations, the most feasible is that our stretching protocol was activating barrages of afferent activity that disrupted lumbosacral circuitry and plasticity. The Grau laboratory, using an instrumental learning paradigm, has demonstrated that randomly applied (uncontrollable) noxious inputs can disrupt spinal cord plasticity associated with both the short-term establishment phase and long-term maintenance phase of instrumental learning in the fully transected rat preparation.55,56 Importantly, they also uncovered evidence that only 6 minutes (cumulative) of uncontrollable noxious input (tail shock) can negatively alter the normal profile of locomotor recovery in a contusion model of SCI. 57 Thus, we posit that our SR protocol, acting as an uncontrollable input, had a negative influence on the process of plasticity that leads to or allows in-cage activity-dependent functional recovery. The non-WC/SR animals in our study were high functioning; they exhibited consistent weight-supported stepping as measured by the BBB and were not different from controls when assessed kinematically while moving freely overground and swimming, indicating the stretch protocol was not debilitating. The clinical implications of these observations are significant, and future work should address the utility of stretch-based therapies for avoidance of peripheral pathologies versus the potential for negative long-term consequences on spinal learning and locomotor retraining.
WC-immobilized animals also exhibited higher levels of antagonist muscle co-contraction in response to tail pinch, as a surrogate measure of spasticity, compared with non-WC animals. In addition, we routinely observed bouts of bilateral hindlimb spasms during wheelchair immobilization, which were not quantified. The combination of ankle contractures and spasticity suggests that the immobilization was not just preventing movement-related afferent input but was possibly resulting in uncontrollable, noxious afferent input. Thus, while immobilization or inactivity may prevent optimal in-cage retraining, it remains to be determined how an absence or lack of activity-dependent afferent input compares with an abundance of activity-independent (uncontrollable), possibly noxious, input to influence functional recovery. 57
Recently, we discovered that providing 50% to 60% body weight support with 5 cm of water allows young adult female SD rats with moderately severe (25 g-cm) T9 contusion injuries to generate high-quality plantar hindlimb stepping with near-normal kinematics as early as 1 week postinjury in the absence of an applied retraining strategy. 23 In an elegant piece of work, Courtine and colleagues 58 showed in fully transected rats that a combination of serotonergic agonists, epidural stimulation, and weight support allowed near-normal stepping (albeit bipedal) to be expressed at 1 week postinjury. In the current study, nightly WC immobilization on days 4, 5, and 6 postinjury, as spinal shock was resolving, had no influence on the day 7 BBB scores. Taken together, these findings suggest that a robust ability to generate a stepping pattern is present immediately postinjury and that this capacity is not dependent on an acute retraining phenomenon. Importantly, however, in our shallow-water stepping model, we found that the acute stepping capacity was not maintained in the absence of continued exposure to the activity (“retraining”). This is in good agreement with the work of Courtine et al, 58 described above, who found that excellent patterns induced at 1 week (with the “full combination” treatment) were not present at 9 weeks in animals that did not receive treadmill training in the intervening period. Thus, it follows that hindlimb immobilization of animals with mild to moderate spinal cord injuries sufficiently disrupts the patterns of afferent input from the hindlimbs to prevent the maintenance of the acute capacity leading to a dramatic drop in locomotor function over the first few weeks postinjury. With the caveat that intense acute activity may be detrimental to traumatically injured spinal cord, 22 our results support suggestions that appropriate activity, applied acutely, may enhance or optimize overall functional outcomes following a contusive SCI.
The current findings suggest that limiting early in-cage activity can dramatically alter functional recovery of locomotion in a rodent model of mild to moderate SCI. Surprisingly, we also show that a hindlimb stretch protocol can have a negative effect on recovery in normally housed animals, preventing the development of forelimb–hindlimb coordination, while having no apparent influence on recovery in hindlimb immobilized animals or on the development of ankle contractures. These findings imply that the immobility experienced by the majority of spinal cord–injured patients, and some aspects of standard-of-care therapies, may significantly hinder functional recovery acutely and also potentially limit the responsiveness of the spinal cord circuitry to retraining efforts initiated at later time points.
Footnotes
Acknowledgements
The authors wish to acknowledge the excellent technical assistance provided by the KSCIRC Core staff: Christine Nunn, Kim Fentress, and Johnny Morehouse. Thanks also to Ashley Whelan for technical assistance and Seid Waddell for assistance with the micro-CT scanning and data analysis. We would also like to thank the staff of the Research Resources Center and Drs Vicky Kyle and Mary Proctor for veterinary assistance. We recognize and thank W. Lee Smith, PT, at the Frazier Rehab Institute, Louisville, Kentucky, for technical advice regarding hindlimb stretch, and the laboratory of Susan J. Harkema, PhD, for research opportunities in rehabilitation after spinal cord injury in a clinical setting.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants to DSKM from the Kentucky Spinal Cord and Head Injury Research Trust (KSCHIRT) and the NIH (NINDS R01 NS052292 & NCRR 1P20-RR15576).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
