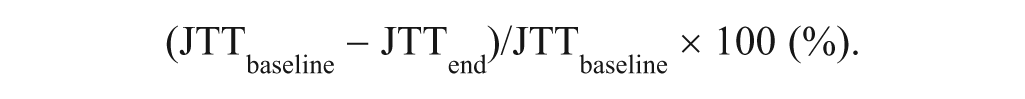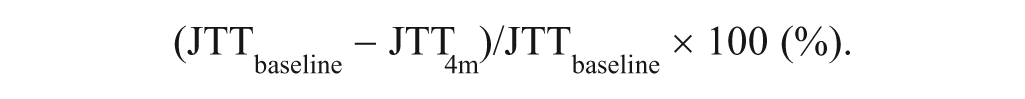Abstract
Background. Somatosensory stimulation in the form of repetitive peripheral nerve stimulation (RPSS) is a promising strategy to improve motor function of the upper limb in chronic stroke. Home-based RPSS may be an alternative to hospital-based RPSS. Objectives. To investigate the feasibility and safety of an innovative program of home-based RPSS combined with motor training and to collect preliminary data on the efficacy of this program to enhance hand motor function in patients in the chronic phase after stroke. Methods. Twenty patients were randomized to either active or sham RPSS associated with daily motor training performed at home over 4 consecutive weeks. All the patients were able to perform tasks of the Jebsen–Taylor Test (JTT). The primary outcome measures were feasibility, evaluated by self-reported compliance with the intervention, and safety (adverse events). Secondary outcomes comprised improvements in hand function in the JTT after end of treatment and after a 4-month follow-up period. Results. There were no relevant adverse events. Compliance with RPSS and motor training was significantly greater in the active group than in the sham group. Upper extremity performance improved significantly more in the active group compared with the sham group at the end of treatment. This difference remained significant 4 months later, even when differences in compliance with motor training were considered. Conclusions. Home-based active RPSS associated with motor training was feasible, was safe, and led to long-lasting enhancement of paretic arm performance in the chronic phase after stroke for those who can perform the JTT. These results point to the need for an efficacy trial.
Keywords
Introduction
Stroke incidence is greater in low- and middle-income countries than in high-income countries and the burden of stroke disability is expected to increase over the next decades. 1 One of the main differences between stroke care in developed and developing countries is the availability of inpatient rehabilitation facilities. In developing countries, rehabilitation often relies strongly on outpatient care. 2 Compliance with outpatient rehabilitation is limited by costs and a range of social, psychological, and practical barriers such as the paucity of adapted transportation for persons with restricted mobility. In this context, home-based rehabilitation emerges as an attractive alternative concept for patient care. 3
Cultural, social, and educational barriers challenge the feasibility of home-based rehabilitation in low- and middle-income countries, and further research is needed to guide the adaptation of potentially effective home-based interventions to local conditions in these countries. In this context, we have developed a novel home-based rehabilitation program to improve functional ability of the paretic arm. The protocol focused on motor rehabilitation of the paretic arm because upper limb weakness is common and significantly contributes to disability from stroke. 4
The program combined motor training with neurostimulation in the form of repetitive peripheral nerve sensory stimulation (RPSS). RPSS predominantly targets afferent proprioceptive fibers with little or no stimulation of motor or cutaneous fibers. Thereby, RPSS increases motor cortical excitability5,6 and induces task-related activation of the primary motor, primary sensory, and premotor cortices.7,8 It is therefore an excellent tool to enhance activity in the sensorimotor system in a way that capitalizes on activity-dependent brain plasticity when combined with motor training.
In proof-of-principle studies, beneficial effects of RPSS on motor performance or motor training were documented in patients in the chronic or subacute phase after stroke.9-14 In all these studies, RPSS was administered in outpatient medical facilities or laboratories. To apply RPSS at home, we opted to use a novel strategy, by administering RPSS through a portable device that could be easily managed by patients. The device is similar to a wristwatch and delivers biphasic square-wave electrical pulses at 31 Hz.
Our goal was to perform a proof-of-principle study in order to (a) investigate the feasibility and safety of an innovative program of home-based RPSS combined with motor training in a middle-income country and (b) collect preliminary data on the efficacy of this innovative program to enhance hand motor function in patients in the chronic phase after stroke.
Methods
In this pilot, randomized, double-blinded clinical trial, we compared the feasibility, safety, and efficacy of motor training combined with either active or sham RPSS in patients in the chronic phase after stroke with mild to moderate hand paresis. All patients were able to perform activities of the Jebsen–Taylor Test 15 (JTT) that assesses hand function. Performance is measured as the time (in seconds) required to perform tasks such as picking up small objects, stacking checkers, turning cards, picking up beans with a spoon, and moving light and heavy cans.
Participants
Recruitment was performed by searching medical files from patients followed by the Stroke Group at Hospital das Clínicas/São Paulo University Medical School. Potentially eligible patients were invited to participate. In addition, the protocol was announced in the local media, and patients not followed at our institution who were in the protocol were also screened. The screening process involved a telephone interview to check inclusion and exclusion criteria. Patients still considered eligible after this step were invited for evaluation in the Neurostimulation Laboratory.
Inclusion criteria comprised (a) age between 18 and 80 years; (b) single ischemic or hemorrhagic stroke in a cerebral hemisphere, documented by computed tomography (CT) or magnetic resonance imaging (MRI); (c) stroke onset at least 6 months earlier; and (d) hand paresis with preserved ability to perform all tasks of the JTT. Exclusion criteria comprised (a) other morbid neurological conditions, (b) hand anesthesia, (c) shoulder or hand pain, (d) debilitating chronic diseases, (e) inability to provide informed consent due to severe aphasia or cognitive impairment, and (f) left-handedness before the stroke.
The protocol was approved by the Ethics Committee of Hospital das Clínicas/São Paulo University, and all participants gave written informed consent prior to participation. This study was registered in clinicaltrials.gov (NCT1309-06).
Baseline Measures
The following characteristics were evaluated at baseline: age, sex, chronicity, type of stroke (ischemic/hemorrhagic), affected hemisphere, JTT scores, Fugl-Meyer Assessment of sensorimotor recovery after stroke (motor performance, upper limb), 16 Functional Independence Measure, 17 National Institutes of Health Stroke Scale, and Modified Rankin Scale. 18
Experimental Design
Randomization, allocation concealment, and blinding
Patients were randomly assigned in blocks by the principal investigator with a basic random number computerized generator in a 1:1 ratio to receive either active or sham RPSS associated with daily motor training performed at home, during 4 weeks.
Performance in the JTT was videotaped during testing and rated by a researcher blinded to group assignment at Hospital das Clínicas/São Paulo University. Patients were blinded to the experimental hypothesis and were not allowed to interact with each other or to comment on the interventions with the researcher who evaluated outcomes. They were informed that they would be randomized to either a high or a low intensity of RPSS and that they would not know or choose which type of RPSS they would receive during the protocol. After the last session of treatment, patients were asked whether they believed RPSS had or not been administered.
Interventions
Peripheral stimulation
Patients were instructed to use a “ReliefBand” device (Woodside Biomedical, Carlsbad, California) for 2 hours, daily, immediately before motor training. This device is similar to a wrist watch, is powered by a 6-V battery, and delivers biphasic square-wave electrical nerve stimulation through surface electrodes at a frequency of 31 Hz. There are 5 different levels of stimulation intensity, ranging from 1 (minimum) to 5 (maximum).
In the active group, the device was positioned so that the electrodes faced the anterior surface of the wrist, stimulating the median nerve. Before the first session of treatment, the investigator demonstrated how the device should be positioned. Patients were instructed to wash and dry the paretic wrist and hand well. They had to position the device, apply gel to the skin, and turn the device on by pressing the POWER button until a green light blinked. The intensity of stimulation was set to elicit maximum paresthesias on the hand palm and/or digits without causing pain, discomfort, or visible muscle contraction, a procedure used in previous RPSS studies. 13
In the sham group, similar instructions were provided, except that a thick polyester wristband was interposed between the wrist and the device, and electrodes were positioned to face the dorsal surface of the wrist so that they faced away from the median nerve. Therefore, no effective electrical stimulation was administered to the nerve even though the set up was literally identical.
Motor training
Patients were familiarized with the JTT tasks in the laboratory 1 day before home-based treatment started. Patients were instructed to perform 5 of the 7 tasks of the original JTT: writing, turning cards, picking small objects, picking beans with a spoon, and stacking checkers. Scores were calculated as the sum of times required to perform each of the tasks, in seconds. Standardized instructions were given to patients and all measurements were videotaped. The JTT is a valid, reliable tool that measures the time required to perform seven tasks with the upper limb.15,19 Higher scores indicate worse performance.
During familiarization, the JTT was repeated with the paretic upper limb until a stable performance level was reached (difference <20% between 3 consecutive scores). After familiarization, patients were instructed to perform the 5 tasks at home immediately after each session of RPSS, in 2 blocks of training, daily over 4 consecutive weeks. In each block, each task was performed once. All tasks had to be performed with the paretic hand, without aid from the nonparetic hand. Patients were instructed to rest between the blocks if they felt fatigued but were allowed to perform the tasks without rest. To practice the tasks at home, each patient received a kit with the materials of the 5 JTT items used.
Each subject was contacted weekly via telephone to ask whether they were complying with the instructions given, to reinforce the importance of complying with the interventions, and to answer subjects’ questions and concerns.
Outcome Measures
Primary outcome measures
The primary outcome measures concerned the feasibility, evaluated by compliance with the interventions, and safety.
On the seventh day after onset of treatment, patients were required to demonstrate to one of the investigators (RLSF) how they wore the RPSS device at home, as well as how they trained the motor tasks. Compliance with RPSS and with motor training was assessed through a daily written log and quantified as follows:
Number of logged days/Number of planned days (device use) × 100 (%) Number of logged days/Number of planned days (motor training) × 100 (%)
After end of treatment, patients were asked the following questions to investigate their satisfaction with the interventions: “Did you like the treatment?,” “Was the treatment important for your recovery,” “Was the peripheral nerve stimulated?,” and “Would you do this treatment again?”
Patients were encouraged to note adverse events on their daily logs and were instructed to report these effects to the investigators whenever they happened. In addition, the weekly catch-up sessions proactively addressed potential adverse events.
Secondary outcome measures
Secondary outcome measures were derived from the comparison of JTT scores at baseline, at end of treatment (JTTend), and 4 months after end of treatment (JTT4m; Figure 1, overall treatment schedule). Previous research indicates that baseline performance influences the effects of the combined RPSS–motor training intervention with greater improvements being observed in more severely affected patients than in less affected patients. 13 To correct for possible differences in responsiveness to therapy associated with different baseline performances, we chose to evaluate relative improvements in relation to baseline, rather than raw JTT scores. Baseline-corrected treatment effects have been commonly evaluated in rehabilitation studies.9,20-24 Therefore, improvement at end of treatment was calculated as follows:
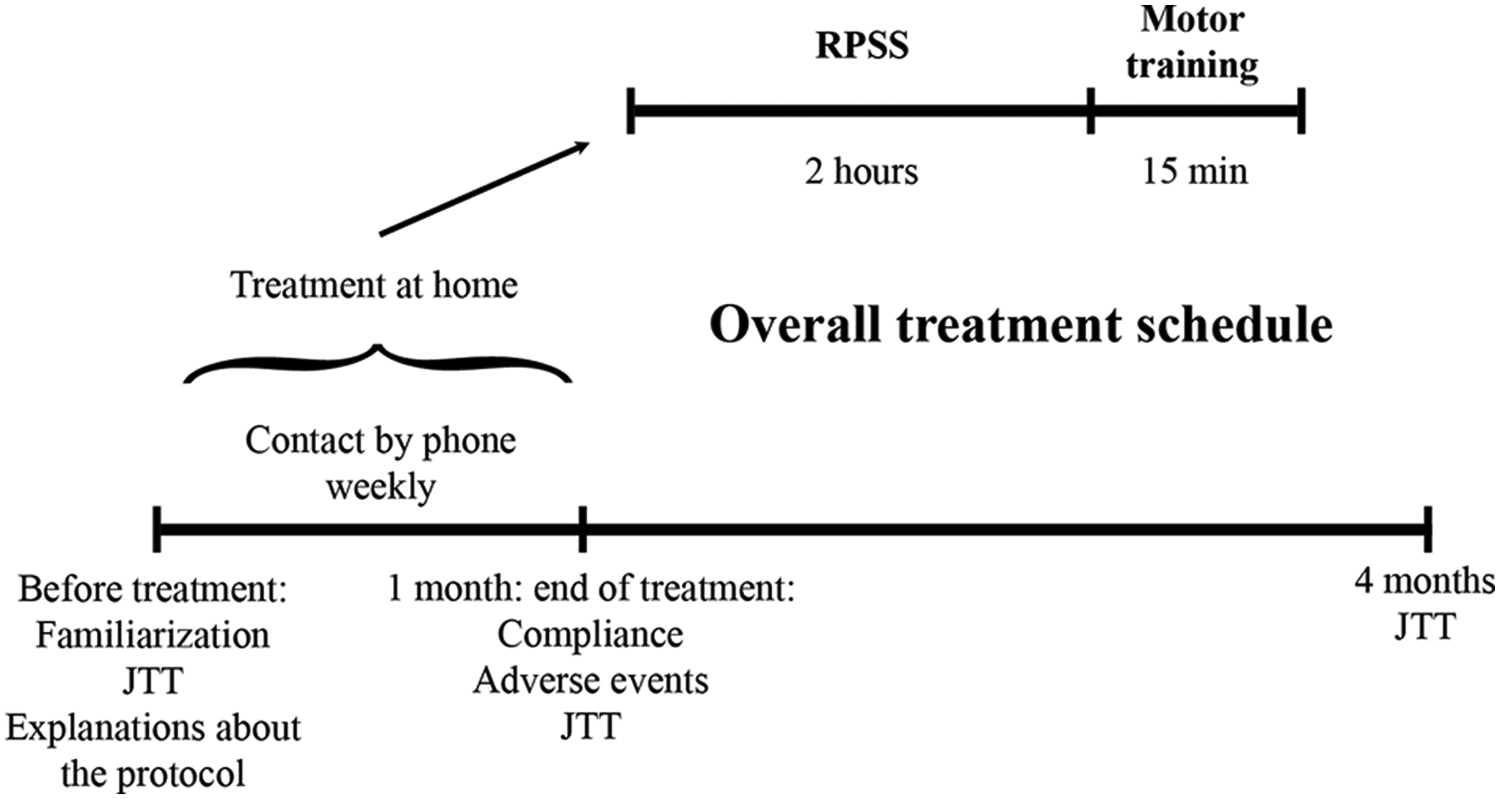
Experimental design and timeline. Abbreviations: RPSS, repetitive peripheral nerve sensory stimulation; JTT, Jebsen–Taylor Test.
Improvement at 4 months later was calculated as follows:
This was a “hypothesis-generating” project aimed at generating data that would allow a power analysis to determine sample size for a more advanced investigation.
Analysis
Baseline characteristics in the active and sham groups were compared with χ2 tests, Fisher exact tests, or Mann–Whitney tests when appropriate.
Compliance between the active and sham groups, in regard to RPSS and motor training, was evaluated with Mann–Whitney tests.
Baseline-corrected treatment effects were evaluated with repeated-measures analysis of variance (ANOVARM) with factors GROUP (“active” and “sham”), TIME (“end of treatment” and “4 months’ follow-up”); COMPLIANCE WITH MOTOR TRAINING was included in the model as a covariate. Tukey tests were used for post hoc analysis. This way, we compared short-term (immediately after end of treatment) and long-term (4 months later) differences in effects of the active and sham interventions.
Intent-to treat analysis was performed, that is, all patients were included in the analysis. A statistically significant result was considered at a p value of <.05.
Effect size (Cohen d) was calculated for improvement in JTT performance, as follows:
where n = number of subjects.
Results
After screening 1731 patients, 20 were included between February 2007 and September 2009 (Figure 2). Ten patients were included in the active group and 10 in the sham group. There were no significant differences in baseline characteristics between patients in the 2 groups (Table 1).
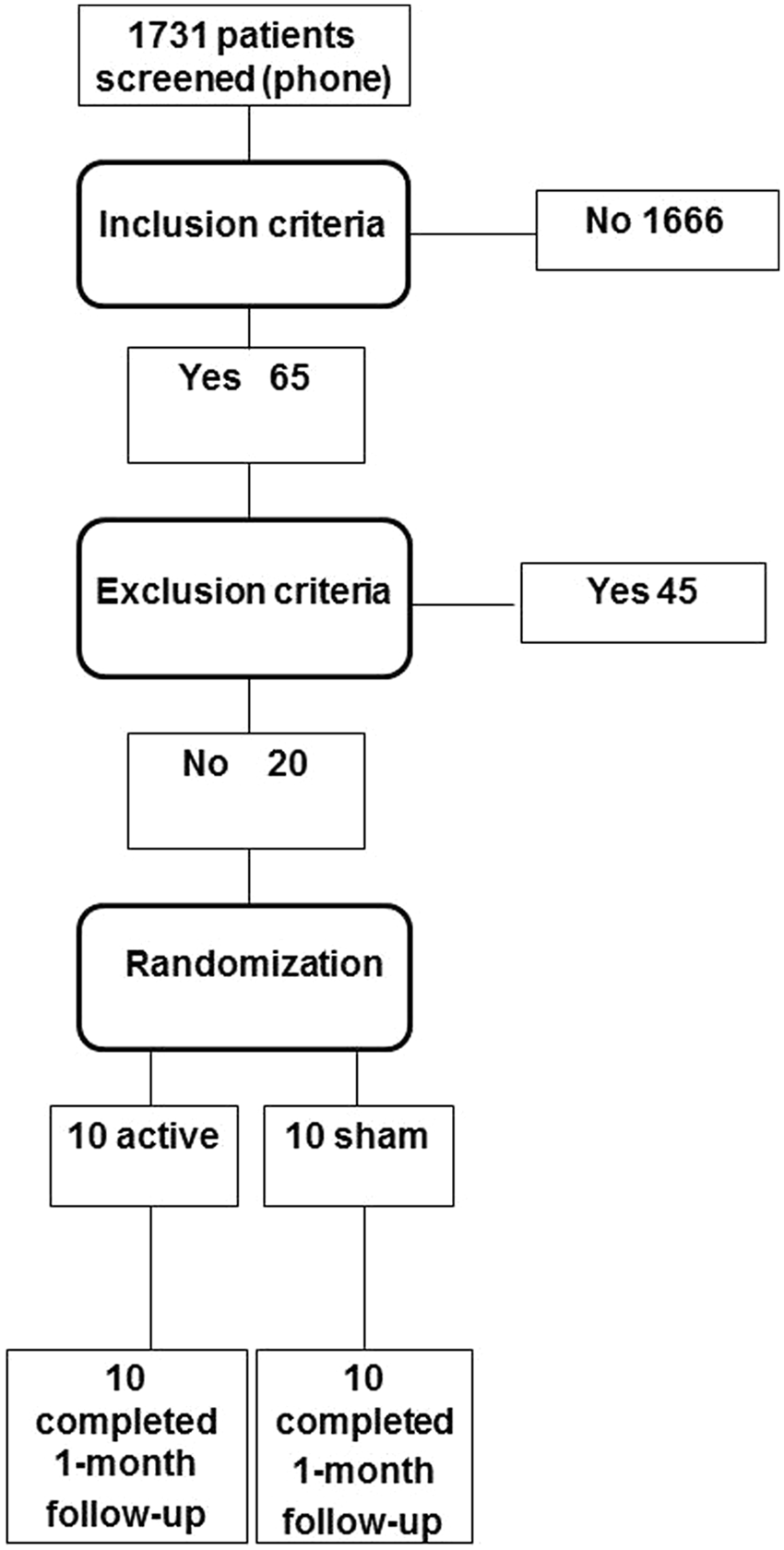
Flow diagram of patients through the trial.
Baseline Characteristics.
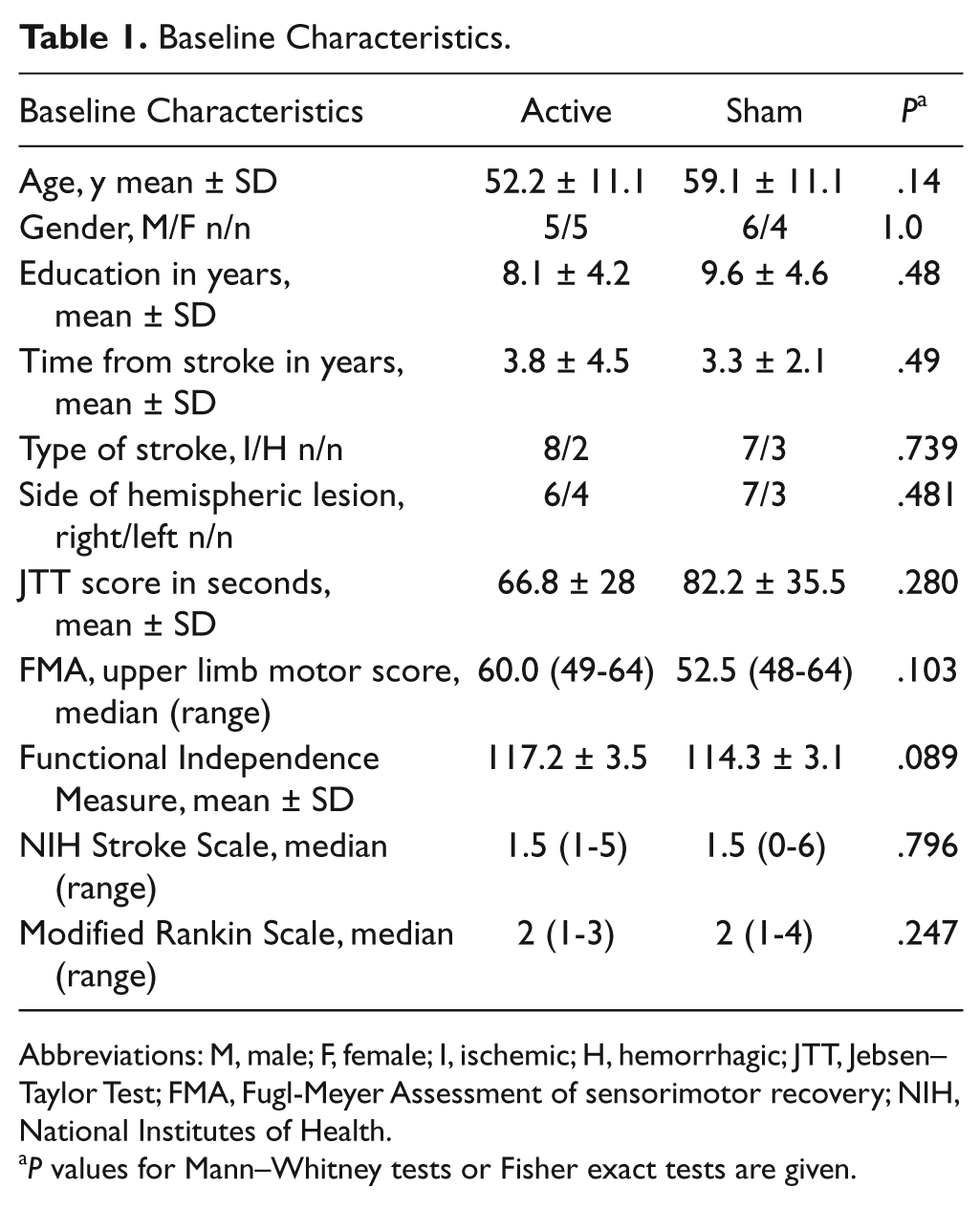
Abbreviations: M, male; F, female; I, ischemic; H, hemorrhagic; JTT, Jebsen–Taylor Test; FMA, Fugl-Meyer Assessment of sensorimotor recovery; NIH, National Institutes of Health.
P values for Mann–Whitney tests or Fisher exact tests are given.
Primary Outcome Measures: Feasibility and Safety
All patients were able to demonstrate the correct use of the RPSS device and the performance of the JTT-based practice tasks as instructed on the seventh day after onset of treatment.
Compliance with RPSS was very good in both groups with a mean (± standard deviation) of 98.2% ± 4.5% in the active group and 82.8% ± 20.1% in the sham group but was significantly greater in the active group (P = .003). Compliance with motor training was also significantly greater in the active (80.0% ± 41.2%) than in the sham group (40.7% ± 43.8%), P = .007. Compliance with RPSS was significantly higher than compliance with motor training in both active and sham groups (P = .002).
No relevant adverse events related to active treatment were reported. Only 1 of the 10 patients in the sham group (patient 3) had a nocebo effect and noted that she did not like the treatment. This patient, who suffered from expression aphasia, noted pain and paresthesias in the entire paretic upper limb after starting the sham treatment. She only reported the symptoms when she used the paretic arm to perform activities that she did not enjoy (house chores), but not while using the paretic arm for leisure activities. The symptoms were reported during the last phone call from the therapist, which took place before the posttreatment evaluation session scheduled for the end of the treatment. During this evaluation, the patient’s caregiver informed the research team that treatment had been stopped after the 16th session but this had not been reported to the therapist during the first 3 weekly phone calls. The neurologic examination was unchanged compared with the baseline examination, before inclusion in the study. She was treated with drugs for 8 days and the symptoms improved, but she continued to indicate that she had mild pain in the back of the paretic hand, in a distribution that was not consistent with a neurologic etiology. The patient was anxious, and an anxyolitic benzodiazepine drug was administered for 5 days. The symptoms resolved completely. Given the complexity of the symptoms and the pattern of their occurrence, it is possible that this patient had a somatization disorder associated with anxiety and possible secondary gains (eg, stopping performing activities that she disliked to do at home, according to her sister).
Four patients, 3 out of 10 in the sham group and 1 One out of 10 in the active group, felt that the treatment was not important for their recovery. 1 out of 10 patients in the sham group noted that they would not undergo the treatment again. In all, 2 out of 10 patients in the sham group and 1 out of 10 in the active group reported that they did not think nerve stimulation was administered in the protocol.
Secondary Outcome Measures: Baseline-Corrected Treatment Effects
All patients attended the evaluation session scheduled immediately after the end of the intervention. However, only 7 out of 10 patients in the active group and 6 out of 10 patients in the sham group attended the 4-month follow-up session.
Individual scores of the JTT are shown in Table 2. With regard to improvement in JTT performance compared with baseline, ANOVARM revealed a significant effect of GROUP (F = 5.02; P = .038), with no effect of TIME (F = 0.35; P = .57) or interaction GROUP × TIME (F = .01; P = .94; Figure 3). Upper extremity performance improved significantly more in the active group compared with the sham group at the end of treatment (14.3%, confidence interval = 1.06% to 25.6%). Therefore, improvements were greater in the active group not only immediately after the end of treatment but were also sustained 4 months later. A power analysis (α-error = .05, β-error = .20) indicated that 23 subjects per group would be needed to detect a difference of 15% in improvement in JTT performance between the groups at 1 month after treatment.
Individual Improvement in JTT (%) in the Active Group and in the Sham Group, at the End of the Treatment (JTTend) and 4 Months after the End of the Treatment (JTT4m), Compared With Baseline.
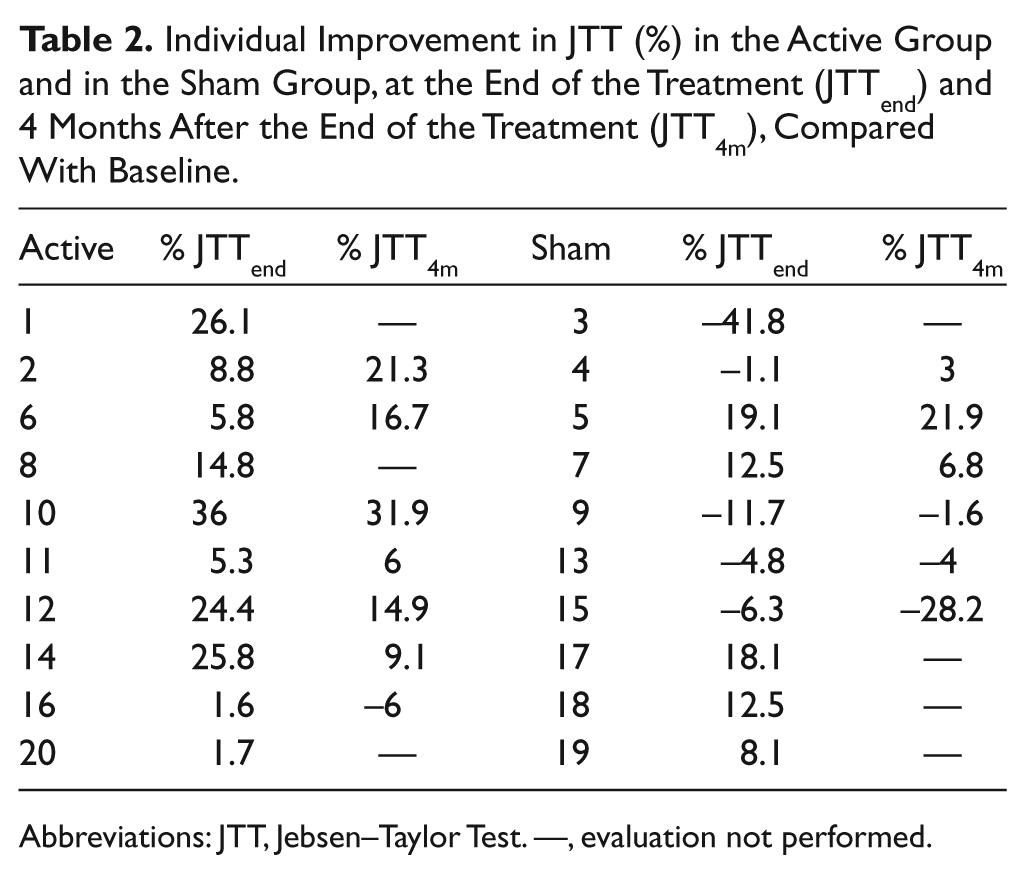
Abbreviations: JTT, Jebsen–Taylor Test. —, evaluation not performed.
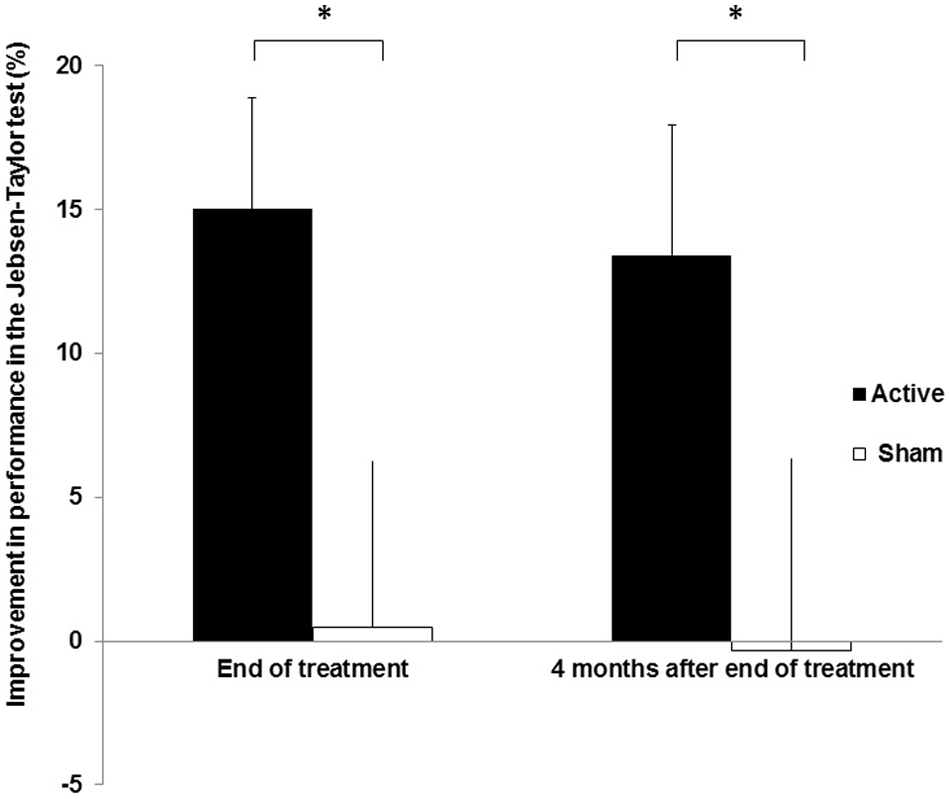
Changes in Jebsen–Taylor Test scores at end of treatment and at 4 months after end of treatment, compared with baseline, in the active and sham groups (mean ± standard error, %).
Because compliance with motor training was significantly greater in the active group than in the sham group, the significant effect of GROUP might not reflect a difference between effects of the active and sham interventions but may simply be related to more intensive motor training in the active group. Therefore, we performed an additional post hoc ANOVARM, adding compliance with motor training as a covariate in the model. We found a significant effect of GROUP (F = 6.77; P = .0019) with no effect of TIME (F = 0.29; P = .60) or interaction GROUP × TIME (F = 0.01; P = .94). These results indicate that the beneficial effect of active RPSS could not be attributed to the greater extent of motor training in this group compared with the sham group.
Discussion
The present study was designed to obtain initial evidence for the feasibility, safety, and efficacy of a combined RPSS–motor training intervention implemented in the home environment. Thereby the experiment was designed to accommodate potential constraints in health care access and delivery, as well as cultural and societal barriers to treatment compliance. Data obtained from 20 patients suggest that (a) home-based RPSS combined with motor training was feasible in a middle-income country and was not associated with relevant adverse events; (b) active RPSS was superior to sham RPSS on enhancing effects of motor training; and (c) the beneficial effect of active compared with sham RPSS was sustained, even 4 months after end of treatment.
The compliance data revealed that patients generally conformed well to the treatment regime. However, compliance was higher for RPSS (wearing the stimulation device) than the motor training. One might argue that this discrepancy in compliance is because of the passive nature of RPSS, compared with active motor training. However, it is also possible that the written information from the compliance logs may not be reliable in our sample. Patients reported that they were not familiar with written logs and that they often forgot to register motor training and use of the stimulation device in the logs, despite having trained and used the device as instructed. Future research might therefore incorporate a treatment contract, a standard tool used in psychology to enhance treatment compliance. Alternatively, compliance might be monitored more directly through the use of an electrical device via a physical sensor or a timer.25,26
The present study used a sham protocol to investigate the added benefit of peripheral stimulation. Choosing an appropriate sham is a challenge in this context. Patients may notice that no intervention is being applied, because the sham procedure induces no sensation and may hence become overall less motivated. Systematic differences in motivation, associated with the RPSS protocol, could therefore explain the compliance differences between the active and the sham group. However, this is an unlikely explanation for the 40.7% compliance rate in the sham group, because 80% of the patients in this group believed that they received active RPSS during the study. Still, 30% of the patients in the sham group and only 10% in the active group reported that RPSS had not been important for their recovery. We therefore suggest that the enhanced compliance with motor training was driven by a subjective impression of improvement in motor function that occurred to a greater extent in the active group than the sham group.
Data from the JTT suggest better performance in the active RPSS than the sham group for both, the testing after the treatment and the 4-month follow-up interval. These improvements were independent of greater motor training. It is therefore reasonable to conclude that either compliance in the sham group was underestimated because of poor registration of information in this group or that there was a ceiling effect of motor training, that is, additional training of the tasks did not lead to greater benefit. Therefore, the most plausible explanation to our results is that active home-based RPSS enhanced effects of motor training to a greater extent than sham RPSS. However, a number of caveats in the data must be considered. First, the difference observed between the active and sham groups might have been influenced by the deterioration in JTT performance in 5 subjects in the sham group. In one of these subjects (patient 3), the nocebo effect probably had a substantial impact on performance. In the other 4 patients, no adverse events were reported. They showed slight worsening after the end of treatment that could reflect fluctuation in performance related to motor control, despite familiarization with the tasks that were implemented at baseline until a stable performance was reached in all patients. Multiple baseline measures after familiarization may decrease fluctuation in performance in future efficacy trials. Second, dropouts limited the power to detect significant differences in performance between end of treatment and 4 months later. Therefore, it is possible that improvement in performance might not be sustained if a greater number of patients had been examined. Still, this is a speculation and larger studies are necessary to elucidate this point.
We screened many potential subjects and included relatively few patients with stroke, as often happens in rehabilitation trials. Exclusion of patients with recurrent strokes and of patients not able to perform the JTT allowed us to evaluate a highly selective, homogeneous sample. However, exclusion criteria limited generalizability of the results. The next steps in this line of research need to explore the validity of the findings in a larger sample of patients, with the less stringent criteria. Despite the sample size limitation, all patients in the active group, and only half of the patients in the sham group, improved in motor performance after 1 month of treatment. Even though we cannot exclude an α-error because of small sample size, this finding along with the ease and favorable safety profile of the active intervention strongly encourage the design of future trials addressing efficacy of home-based nerve stimulation. To avoid ceiling effects or fatigue in association with repeated performance of JTT tasks, studies might focus on task-related training with shaping.14,27,28
The magnitude of improvement in motor performance in the active group was comparable with improvements described in previous reports on RPSS in the chronic phase after stroke (5% to 15%).9-12 The present study extends these previous reports and indicates, for the first time, that active home-based RPSS associated with training leads to persistent improvement in motor function in patients in the chronic phase after stroke and that this improvement is maintained for at least 4 months after completion of treatment.
Results of proof-of-principle studies indicate that active RPSS enhances motor performance or effects of motor training in patients with stroke9-14 and that this improvement might be delivered through the mechanisms of practice-induced functional reorganization. Thus, we theorize that RPSS augments somatosensory input, which, combined with the sensorimotor stimulation induced by the motor training, drives the optimization of the neural networks controlling affected limb movements. These practice-related changes in the brain strengthen motor cortical function and the effects of motor training. Data from healthy volunteers support this theory and show that RPSS enhances functional magnetic resonance imaging task-related activation in the primary motor, primary sensory, and premotor cortices.7,8 The relevance of afferent information is further highlighted by well-established deterioration in performance of motor tasks by deafferented limbs in humans and animals 29 and by reports of enhanced motor cortical excitability or use-dependent plasticity after one session of RPSS in healthy volunteers5,6 and chronic stroke patients. 9
Our results support the concept that enhanced somatosensory input over several weeks, delivered by a portable stimulator, can potentiate motor training in patients in the chronic phase after stroke and mild to moderate hand paresis. This finding provides a crucial step to close the gap between translational, proof-of-principle studies on the effects of RPSS in stroke rehabilitation, and appropriately powered clinical trials to evaluate RPSS effects. Considering that even mild motor hand weakness can be very decapacitating for patients and can compromise their ability to live independently,4,30 a home-based alternative to enhance the recovery of hand function can substantially affect stroke rehabilitation.
This study pioneered home-based RPSS combined with motor training without direct supervision of a therapist. The relatively low costs, favorable safety profile, and technical simplicity of home-based RPSS make this intervention an appealing neuromodulation tool to decrease disability not only in developed but also in developing countries where most strokes occur.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the São Paulo State Foundation for Research Support (FAPESP), Grant No. 07/53123-1, and by the Brazilian National Council for Scientific and Technological Development (CNPq), Grant No. 477916/2006-6.