Abstract
Objective. To evaluate the effectiveness of a home-based cognitive rehabilitation (CR) program based on the video game Dr Kawashima’s Brain Training (DKBT; Nintendo, Japan), in improving attention, processing speed, and working memory of patients with multiple sclerosis (MS). Methods. This was a randomized, wait-list control study. Patients with MS and failure in at least one between Stroop Test (ST), Paced Auditory Serial Addition Test (PASAT), and Symbol Digit Modalities Test (SDMT) were submitted to an 8-week home-based CR program playing DKBT. Patients were evaluated at baseline and after DKBT by the aforementioned tests, by the Modified Fatigue Impact Scale (MFIS) and by the MS Quality of Life-54 questionnaire (MSQoL-54). Results. Fifty-two 52 patients were screened for eligibility; 35 (mean [standard deviation] age of 43.9 [8.4] years, median Expanded Disability Status Scale score of 2.0 (range = 2.0-6.0) were randomly assigned to the intervention group (n = 18) or wait-list control group (n = 17). ANCOVA analysis showed a significant effect of DKBT on ST (F = 5.027; P = .034; F2 = 0.210), SDMT (F = 4.240; P = .049; F2 = 0.177), and on some subscales of MSQoL-54. The PASAT and cognitive subscale of MFIS also showed an improvement, but this was just not significant (F = 4.104, P = .054, F2 = 0.171, and F = 4.226, P = .054, F2 = 0.237, respectively). Conclusion. We suggest that a home-based DKBT program may improve cognitive functions, some aspects of QoL, and cognitive fatigue in patients with MS.
Introduction
Cognitive impairment is a common and disabling symptom of multiple sclerosis (MS). 1 Although the profile of cognitive deficits varies among patients, the ability to maintain and manipulate information for a short-time period (working memory), and the speed at which that information is processed (processing speed) are 2 domains frequently affected in MS, even in the early stage of the disease. 2 Executive functioning, which refers to the cognitive ability for complex, goal-directed behavior and adaptation to environmental changes or demands, may also be affected.1-4
Rehabilitation of cognitive impairment in MS represents an emerging issue. 5 In recent years, encouraging results have been reported with computer-assisted training specifically focused on information processing, attention, and executive functions and performed with dedicated software.6,7 Unfortunately, the time and costs of these cognitive rehabilitative programs limit the possibility of use in real life; therefore, the development of home-based effective strategies should be desirable. Cognitive training through self-management programs is rapidly growing and may offer the opportunity to reach a high number of individuals and to treat patients whenever face-to-face treatment is not sought or obtainable. Till now, strategies based on vídeo games have not been tested for this purpose in patients with MS; however, some evidence suggests a possible benefit of Dr Kawashima’s Brain Training (DKBT; Nintendo, Kyoto, Japan) on executive functions, working memory, and processing speed in healthy people.8,9 It has been hypothesized that this effect is a result of the fact that most training games in DKBT entail processes recruiting the prefrontal regions or the precuneus, which are also involved in the executive functions, working memory, and processing speed. 9
Based on findings in healthy individuals, we aimed at evaluating the effectiveness of a home-based cognitive rehabilitation (CR) program using a video game console—that is, the Italian version of the DKBT—in improving executive functions, working memory, and processing speed in patients with MS.
Methods
Participants
A consecutive series of patients diagnosed as affected by MS according to revised McDonald criteria 10 and regularly attending the MS Centre of S. Andrea Hospital in Rome were recruited for this independent, randomized, wait-list control pilot study. The study protocol was approved by the local Ethical Committee, and each patient provided written informed consent before any study-related procedure.
Before study enrolment, a neurologist and a psychologist (LDG and VB) screened patients for eligibility. Inclusion criteria were relapsing-remitting MS course, age between 18 and 50 years, at least 8 years of education, and failure in at least 1 of the following tests: Stroop Test (ST), Paced Auditory Serial Addition Test (PASAT) 3-s presentation rate, and Symbol Digit Modalities Test (SDMT).11-13 Failure on PASAT and SDMT was defined as a score below the fifth percentile of normative data for the Italian population 14 and failure on ST as an equivalent score below 3. 11
Exclusion criteria were the following: disease exacerbation in the previous 3 months; any motor or visual condition that could interfere with the performance of training; history of seizures; presence of depression and/or anxiety assessed by the Hamilton Depression Scale (HAM-D) and the Hamilton Anxiety Scale (HAM-A), with cutoff scores for exclusion of 7 and 9, respectively.15,16 The Mini Mental State Examination (MMSE) was also performed to exclude severely cognitively impaired patients—that is, those with a score equal or below 24. 17 Patients with psychiatric illnesses, history of alcohol or substance abuse, history of medications that may interfere with attentional level, previous CR training or treatment with anticholinesterasics were also excluded. We also excluded left-handed patients to ensure uniformity in the performance of ST 18 ; handedness was assessed by means of the Edinburgh inventory. 19
Finally, patients enrolled in the trial also had to be willing to not change or start any disease-modifying drug or symptomatic medication for the entire duration of the study.
Procedures
This was a randomized wait-list control study; participants in the wait-list served as a control group, and they had the option of entering the DKBT program at the end of the study.
Patients who met all eligibility criteria at screening were randomly assigned in a 1:1 ratio to 2 arms by computer-generated random numbers during the same visit. The randomization procedure was performed using computer-generated random numbers by an operator (LP) who was not involved in study measurements.
Patients allocated to the intervention group were asked to perform the DKBT and received the DBKT console to start the training the following day; patients allocated to the control group were observed for 8 weeks without any interference.
Evaluations
Complete neurological evaluations, including the Expanded Disability Status Scale (EDSS) score, 20 were performed at screening and after the 8-week period. Neuropsychological evaluations were performed at screening to test the patient’s eligibility and represented the baseline (T0) assessment for randomized patients. At the end of the study period (T1), patients included in either the intervention group or control group were retested; retest was performed within 3 days from the end of training for the intervention group and after 8 weeks (±3 days) for the control group.
All assessments were done by a trained neuropsychologist (VB) who was blinded to the patient’s group allocation. Cognitive evaluation included ST, PASAT, and SDMT.11-13 Alternative forms of PASAT and SDMT were used for all patients at T0 (version A) and T1 (version B) assessments to minimize the learning effect.
The Modified Fatigue Impact Scale (MFIS) was also administered at T0 and T1 to provide an assessment of the effects of fatigue in terms of physical (ph-MFIS), cognitive (cg-MFIS), and psychosocial functioning (ps-MFIS). 21
Finally, the 54-item MS Quality of Life questionnaire (MSQoL-54) was administered at T0 and T1 to assess the impact of the intervention on QoL; its relative subscales and summary scores were also calculated: physical function, role limitations–physical, role limitations–emotional, pain, emotional well-being, energy, health perceptions, social function, cognitive function, health distress, overall QoL, sexual function, physical health composite summary, and mental health composite summary scores. 22
Intervention
During the 8-week training period, participants allocated to the intervention group were trained in games of memory, attention, visuospatial processing, and calculations. The cognitive training was performed at home with the Italian version of the DKBT (http://www.nintendo.it/Giochi/Nintendo-DS/Brain-Training-del-Dr-Kawashima-Quanti-anni-ha-il-tuo-cervello).
Patients were instructed by a psychologist (FDL) on how to use the console and how to perform the training. They were required to play 30 min/d, 5 d/wk, for 8 consecutive weeks. They were required also to follow the instructions of the game provided during the training from a virtual guide and to experience all the puzzles proposed. The number of puzzles proposed increased through time. Table 1 summarizes the games proposed.
Description of Game Included in the “ Dr Kawashima’s Brain Training: How Old Is Your Brain?”

A second visit with the same psychologist was performed 2 weeks after T0 to check the correct use of the device and the correct execution of the training. In case of problems, the psychologist (FDL) planned a third visit with the same aim. Phone contacts were also scheduled every week during the intervention period to encourage patients to adhere to study protocol.
Moreover, during the intervention period, the virtual guide of the DKBT stimulated adherence, complimenting in case of perseverance or blaming in case of poor regularity at the beginning of each training session. At the end of the training period, the overall adherence was controlled through the data recorded on the device that allowed checking both the puzzles performed in a day and the scores obtained. Patients were also encouraged to contact a psychologist (FDL) in the study team in case of adverse events and for any question regarding the study protocol or technical problems.
End Points
Cognitive end points were measured as changes between T0 and T1 in ST, PASAT, and SDMT. Changes in MFIS total score and its subscales as well as changes in the MSQoL-54 questionnaire and its subscales were also considered.
Statistical Analysis
Given the exploratory nature of this pilot trial, no sample size estimation was performed. Data are presented as mean (standard deviation) or median (range), as appropriate. Whether the 2 treatment groups were well balanced after randomization was tested using the Mann-Whitney U and the Fisher exact tests for continuous and categorical variables, respectively.
For data analyses, normalized values of cognitive scores were obtained using Italian normative data. 12 The efficacy of home-based DKBT on the aforementioned end points was investigated by using ANCOVA models, which included as covariates age, gender, educational level, disease duration, EDSS, and baseline performance on cognitive tasks and patient-reported outcomes.
Effect sizes (estimated as Cohen’s F2) were also provided. The effect sizes were rated as small, medium, and large for F2 of 0.02, 0.15, and 0.35, respectively. 23 Pearson correlations between changes in neuropsychological tests and in patient-reported outcomes were also carried out.
All P values <.05 in either direction were considered as significant; given the exploratory nature of this study, no correction for multiplicity was applied. Statistical analyses were carried out by using a PC version of Statistical Package for Social Sciences, version 17.0 (IBM SPSS, Chicago, IL).
Results
A total of 52 patients with MS were recruited for the study and screened for eligibility (Figure 1). Of these, 17 did not meet the eligibility criteria: 7 did not present an impairment at screening, 5 showed presence of depression and/or anxiety using HAM-D and HAM-A, 4 had motor or visual conditions that could interfere with the performance of training, 1 had a MMSE score of 24. In all, 35 patients were randomly assigned to either the intervention group (n = 18) or the control group (n = 17). The population consisted of 26 women and 9 men, with a mean (standard deviation [SD]) age of 43.9 (8.4) years, a mean (SD) instruction level of 14 (3) years, a mean (SD) disease duration of 12.4 (7.8) years, and a median EDSS score of 2.5 (ranging from 2.0 to 6.0).
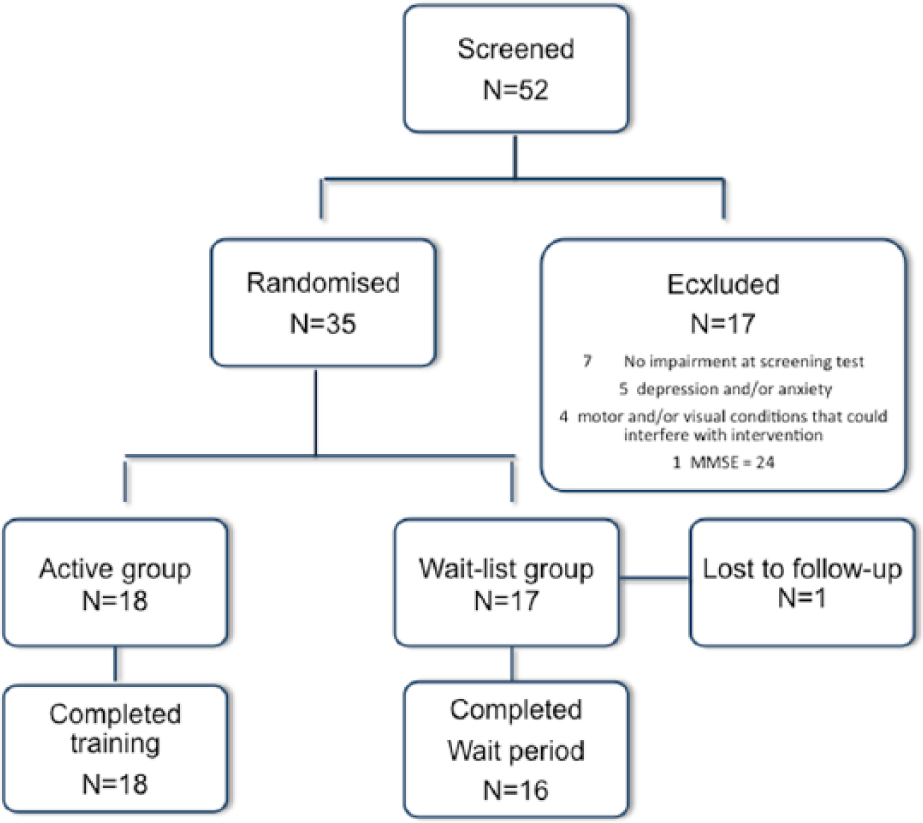
Study flow chart.
No differences were found at baseline (T0) between the 2 groups in terms of demographic, clinical, and neuropsychological characteristics as well as in MFIS and MSQoL-54 scores (Tables 2 and 3).
Baseline Clinical, Neuropsychological, and Fatigue Data of Patients in Intervention and Control Groups. a
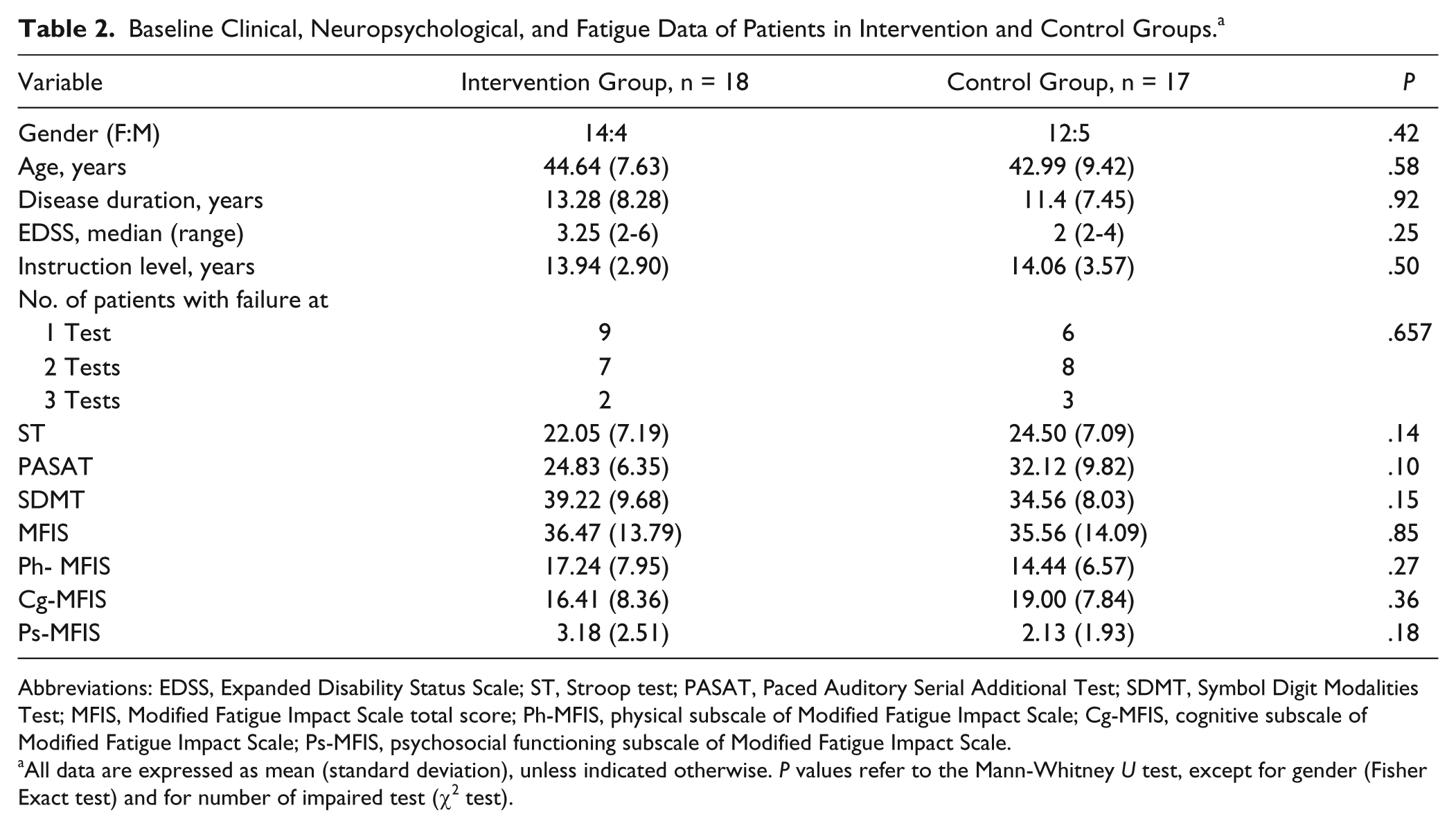
Abbreviations: EDSS, Expanded Disability Status Scale; ST, Stroop test; PASAT, Paced Auditory Serial Additional Test; SDMT, Symbol Digit Modalities Test; MFIS, Modified Fatigue Impact Scale total score; Ph-MFIS, physical subscale of Modified Fatigue Impact Scale; Cg-MFIS, cognitive subscale of Modified Fatigue Impact Scale; Ps-MFIS, psychosocial functioning subscale of Modified Fatigue Impact Scale.
All data are expressed as mean (standard deviation), unless indicated otherwise. P values refer to the Mann-Whitney U test, except for gender (Fisher Exact test) and for number of impaired test (χ2 test).
Baseline Quality-of-Life Data of Patients in Intervention and Control Groups. a
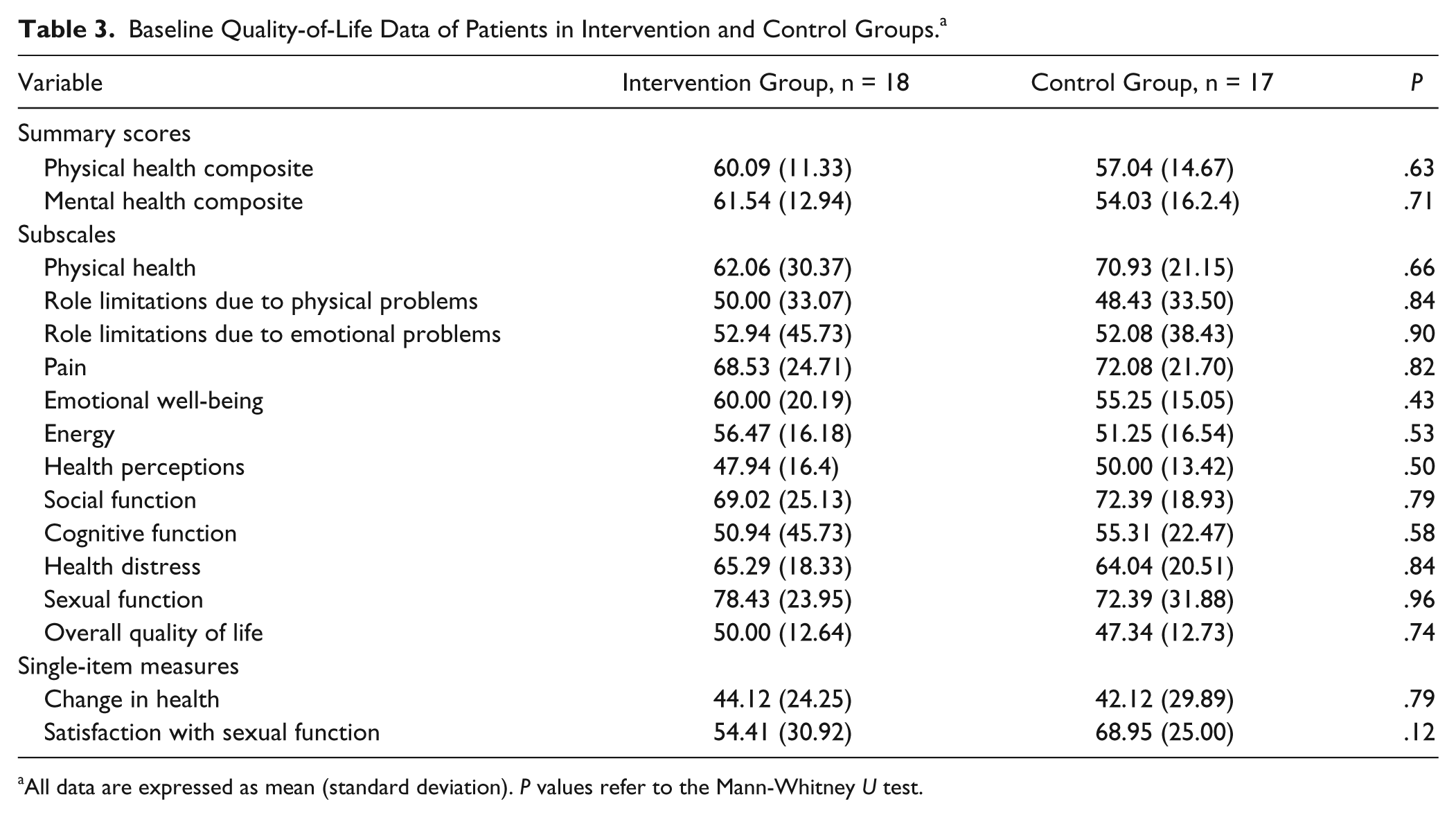
All data are expressed as mean (standard deviation). P values refer to the Mann-Whitney U test.
A patient in the control group moved to another center; therefore, he was considered lost to follow-up, and data of this patient were not included in the analysis. No clinical relapses or EDSS worsening were observed during the study in either group. The treatment adherence, evaluated as number of days in which the patient performed the training/total number of days required, was high (mean = 96%, ranging from 80% to 100%).
ANCOVA analysis showed that DKBT had a significant effect on ST (F = 5.027; P = .034; F2 = 0.210) and SDMT (F = 4.240; P = .049; F2 = 0.177) compared with no intervention. The PASAT and cognitive subscale of MFIS also showed an improvement (Table 4), but this was just not significant (F = 4.104, P = .054, F2 = 0.171, and F = 4.226, P = .054, F2 = 0.237, respectively).
Cognitive and Fatigue Outcomes. a
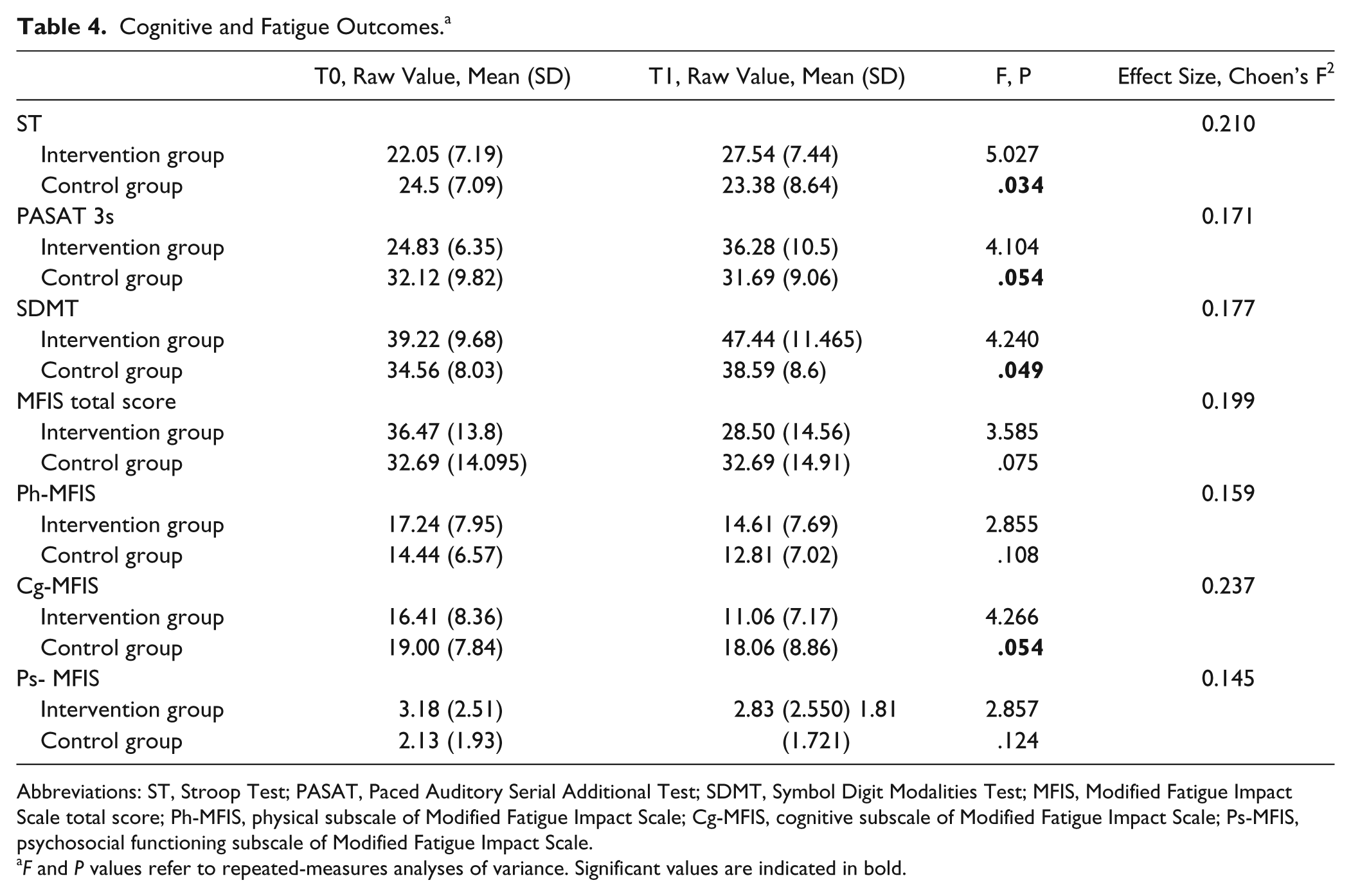
Abbreviations: ST, Stroop Test; PASAT, Paced Auditory Serial Additional Test; SDMT, Symbol Digit Modalities Test; MFIS, Modified Fatigue Impact Scale total score; Ph-MFIS, physical subscale of Modified Fatigue Impact Scale; Cg-MFIS, cognitive subscale of Modified Fatigue Impact Scale; Ps-MFIS, psychosocial functioning subscale of Modified Fatigue Impact Scale.
F and P values refer to repeated-measures analyses of variance. Significant values are indicated in bold.
Finally, the intervention group also showed improvement in some aspects of QoL, such as mental health composite score (F = 13.452; P = .002; F2 = 0.791), role limitations due to emotional problems (F = 6.803; P = .018; F2 = 0.378), emotional well-being (F = 5.456; P = .031; F2 = 0.303), cognitive function scores (F = 7.244; P = .015; F2 = 0.402) and health distress (F=4.904; P= 0.040; F2= 0.272) compared with the control group (Table 5). No significant effects were found on the other MSQoL-54 subscales. Pearson analyses did not show significant correlation between changes in neuropsy-chological tests and changes in patient-reported outcomes.
Quality-of-Life Outcomes. a
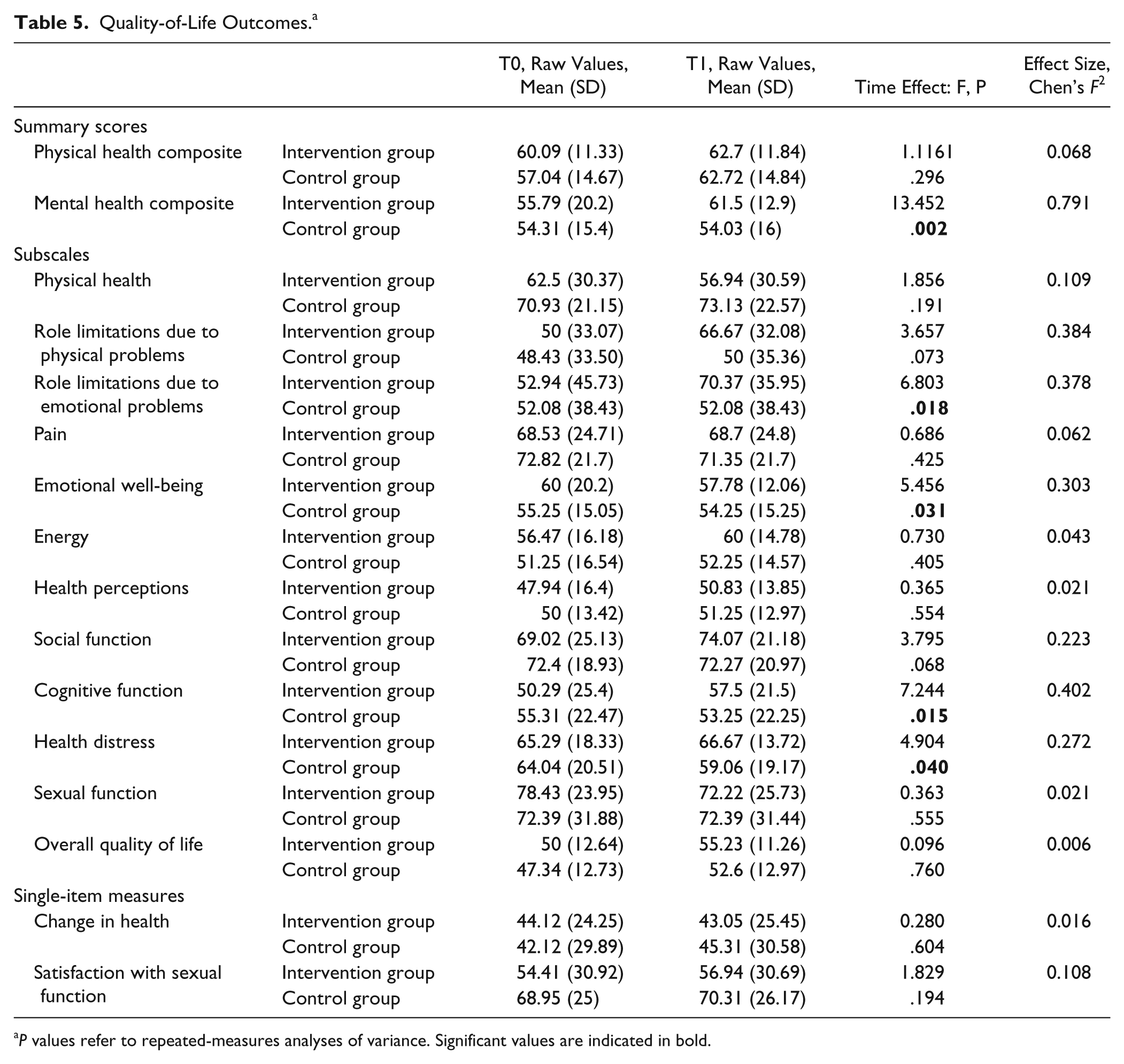
P values refer to repeated-measures analyses of variance. Significant values are indicated in bold.
Discussion
The main findings of our study are that, compared with no intervention, a home-based DBKT CR program improved information processing speed and executive function, as assessed by SDMT and ST, respectively. The improvements found in specific domains of cognitive functions also led to improved patient-reported outcomes—for example, in some aspects of QoL such as mental health composite score, role limitations due to emotional problems, emotional well-being, and cognitive function.
A recent Cochrane Analysis showed preliminary positive findings for the effects of neuropsychological rehabilitation in MS; authors recommended low-level evidence for positive effects, indicating that cognitive training can improve memory functions in addition to attention, whereas for other cognitive functions evidence is still poor. 24 Our results are in line with studies published in very recent years suggesting that rehabilitation of attention abilities may provide the most evident beneficial effects.6,7 In our study, the ST was the cognitive test for which treatment showed a larger significant effect. This is not surprising because the ST mainly involves sustained attention, the ability to elaborate relevant/irrelevant dimensions in parallel (to screen out nontarget information), and some aspects of executive functions, such as the ability to inhibit an automatic response while performing a task based on conflicting stimuli. 11 Compared with the control group, for patients in the intervention group, there was a significant effect on SDMT and a trend on PASAT, which are measures of working memory, sustained attention, and concentration.12,13
Home-based computer-assisted rehabilitative programs in MS provided promising results. Brenk et al 25 evaluated the effects of a short-term nonspecific home-based cognitive training on patients with MS compared with healthy controls. The authors reported an improvement in short-term and working memory, complex attention function, and visuoconstructive performance in both groups. 25 More recently, Amato et al 26 showed a significant improvement in PASAT and, marginally, in SDMT after the use of the Attention Processing Training program—that is, a computer-assisted program focused on sustained, selective, alternating, and divided attention training, performed at home under caregiver supervison.
In our study, we report similar results by using a different approach, which proposes a new strategy based on a very popular and widespread video game and does not require the presence of a caregiver. Patients were asked to follow game instructions and were guided and motivated by a virtual character created by the video game, so that they were completely independent of others. Furthermore, the console allows checking for patient’s compliance, because it records and stores the dates of training sessions and scores obtained in each puzzle.
Mechanisms underlying the clinical improvement observed in our patients following DKBT are difficult to identify. Studies based on functional magnetic resonance imaging (fMRI) revealed the occurrence of functional reorganization in specific brain regions following rehabilitative programs focused on attentional and executive functions.7,27,28 A structural reorganization after rehabilitation has also been described even after short treatment periods. 29 We may hypothesize that similar mechanisms were involved in our study population; however, functional and structural modifications associated with video game–based rehabilitative programs remain to be investigated.
It is interesting to note that we found a significant effect of DKBT on some aspects of QoL such as mental health composite score, role limitations due to emotional problems, emotional well-being,cognitive function and health distress. Because the presence of depression and anxiety was an exclusion criterion in our study, we can consider changes in these subscales to be a favorable result of the DKBT, thus reinforcing the validity of this CR approach in improving the general well-being of individuals affected by MS. However we did not find a correlation between changes in cognitive scores and improvement in QoL subscales. The small sample size and the short observation period may also be responsible for the scarcity of results. On the other hand, we can also hypothesize that the efforts spent on cognitive problems together with the possibility of performing a home-based rehabilitation program by means of a portable device reduced the patients’ stress, with beneficial effects on the physical and emotional components of daily living. The introduction of a game activity into the daily routine may also have contributed to this positive effect.
Finally, we report a trend for a beneficial effect of DKBT on self-reported cognitive fatigue. The efficacy of some nonpharmacological treatment approaches, such as energy conservation management and climbing, in reducing cognitive fatigue has been reported.30,31 However, this is the first study suggesting a possible benefit of a CR program on cognitive fatigue. Neurophysiological and fMRI studies suggest that the perception of fatigue may be the result of an increased effort required by the MS-damaged brain to reach the required functionality, such as cognitive or motor activity32,33; alternatively, it may reflect an impaired brain synchronization resulting from the disruption of brain networks. 34 Damage to the frontostriatal circuit and/or of parietal areas involved in attentional processes has been hypothesized to be crucial in the genesis of cognitive fatigue in MS.35,36
The positive effect of CR on both cognitive performance and cognitive fatigue that we reported in the present study supports the idea that an increased efficiency of networks involved in attentional processes may reduce perception of cognitive fatigue; however, the present study was not specifically designed to address this question.
In discussing our data, some important caveats need to be considered. First, we must mention some metho-dological limitations: because of the exploratory nature of this study, no primary measurement of outcome was identified, and no sample size estimation has been made. For the same reason, no correction for multiplicity was applied. Then, we have to consider the major limitation of this study, which is the lack of an adequate sham intervention: we cannot discount the fact that caring about cognitive issues may have a positive impact per se on patient mood and perceived functional outcome. Another limitation of the present study is the lack of a complete cognitive assessment, which does not allow the evaluation of far-transfer effects of such cognitive training on other cognitive functions. Furthermore, the small sample size limits the interpretation of the results. Finally, the follow-up for cognitive assessment is too short to establish the long-term efficacy of DKBT in relation to improved cognitive function. Therefore, the relevance of our finding for clinical daily practice aiming to improve cognitive functions and QoL of patients with MS has to be further investigated.
Rehabilitation of cognitive functions is a primary objective in people affected by MS; the use of a home-based strategy may lead to advantages in costs and consuming structures, allowing CR to merge with real life. Here, we tested a new strategy based on a very popular and widespread video game, suggesting the clinical benefit of DKBT on the rehabilitation of attention, information processing, and executive functions in patients with MS.
In conclusion, this pilot study suggests that a home-based DKBT program may improve cognitive function, some aspects of QoL, and cognitive fatigue in patients with MS. Future trials are needed to establish the present findings as well as the effectiveness of this practical approach in the long-term and to elucidate the functional substrates underlying the cognitive improvement.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: LDG, FDL, VB, and PP have nothing to disclose. LP received consulting fees from Merck Serono, Bayer Schering, and Biogen Idec and speaker honoraria from Biogen Idec, Teva, and Novartis. GB received consulting fees from Merck Serono and speaker honoraria from Bayer Schering, Biogen Idec, Teva, and Novartis. CP received consulting and lecture fees from Sanofi-Aventis, Biogen Idec, Bayer Schering, Merck Serono, and Novartis; he also received research funding from Sanofi-Aventis, Merck Serono, Bayer Schering, and Novartis.
