Abstract
Background. Bimanual coordinated movements may be impaired after stroke, so an assessment of causes is necessary to optimize rehabilitation strategies. Objective. We assessed the role of afference-based sources of coordination, including phase entrainment and error correction based on visual and somatosensory feedback. Methods. In all, 10 persons with unilateral chronic stroke and 8 age-matched controls participated in a kinesthetic tracking protocol, in which the hemiparetic upper limb was passively driven by the machine. The task consisted of matching the trajectory of the driven limb as accurately as possible with the freely moving limb in 2 conditions: eyes closed and eyes open. We analyzed the continuous relative phase (CRP), the mean absolute difference between positions (ADP) between the positions of the 2 limbs, and the jerk of the matching limb. Results. Coordination instability (CRP standard deviation) and mean ADP were significantly higher for patients with eyes closed, compared with patients with eyes open, controls with eyes closed, and controls with eyes open. Moreover, the jerk was higher for the nonparetic limb of patients than for the control group. Thus, the nonparetic limb did not produce optimally smooth movements even as the motor-driven paretic limb did. Conclusion. Besides deficits caused by interhemispheric competition and motor execution of the paretic limb, somatosensory feedback is a limiting factor in bimanual coordination after stroke. The findings have clinical implications pertaining to the design and individualization of efficient bimanual movement therapy.
Keywords
Introduction
Stroke is the main cause of chronic sensorimotor deficits in adults, with about 30% to 50% of poststroke patients being affected by long-lasting impairment, especially hemiparesis or hemiplegia. Among them, 80% suffer from upper-limb impairment. In particular, impairment of bimanual movements can be considered as a major functional consequence of stroke because many everyday activities involve both hands and therefore require interlimb coordination. 1 Bimanual coordination is especially worth focusing on because it has emerged as a promising technique for stroke rehabilitation,2,3 not only within a functional task-oriented approach but, interestingly, also under the assumption that recovery of the paretic limb would be facilitated by coupling with the nonparetic limb. Bimanual movement therapy is thought to rely on a positive entrainment of the affected side by the unaffected side, notably by activation of similar neural networks in both hemispheres during coordinated performance.4,5 However, results on the effectiveness of “pure” bimanual rehabilitation (ie, without coupling bimanual movements to other techniques, notably rhythmic auditory cueing or electrical stimulation) are not conclusive.6-8 Bimanual coordination performance results from various interacting perceptual and control factors; understanding why and under which conditions bimanual rehabilitation may be the most beneficial requires unraveling the relative contribution of the different processes that are involved in interlimb coordination and likely affected after stroke.
A recent body of work building on the bimanual coordination paradigm 9 allows the following underlying control processes to be distinguished10,11: (1) The integrated timing of feed-forward signals to both limbs: these afference-independent processes have been located at the cortical level, notably the motor and premotor areas involved in the control of interhemispheric interactions.12,13 (2) Afference-based correction mechanisms: these refer to intentional processes involving the supraspinal level, which allow adjusting movements based on visual and somatosensory perception of the coordination (errors) performed. 10 Research has shown that visual feedback stabilizes bimanual coordination,14,15 whereas perturbed somatosensory feedback deteriorates coordination. 16 (3) Phase entrainment by contralateral afferences: phase entrainment refers to an involuntary, essentially peripheral reflex-like process, depending on the intensity of movement-elicited somatosensory afferences from the contralateral limb, which causes specific attraction toward in-phase (simultaneous activation of homologous muscle groups in the 2 limbs, ie, 0° relative phase) and antiphase (alternated activation of homologous muscles, ie, 180°) coordination patterns.17,18 Thus, somatosensory afferences are involved in both intentional correction mechanisms and phase entrainment. (4) Finally, following a more intuitive line of thought, properties of the movement trajectories with which participants are coordinating may also constitute a determinant factor for bimanual performance. Indeed, it seems obvious that it is easier to have 1 limb adapting to a smooth and highly predictable trajectory than to an erratic and unpredictable one, independently of any neurological consideration.
So far, the impairment of bimanual coordination after stroke has mainly been understood in terms of disruption of the interhemispheric balance, entailing an alteration of the feed-forward signals to the paretic limb. Indeed, in bimanual movements after unilateral stroke, the interhemispheric inhibition exerted by the unaffected hemisphere over the lesioned one is much stronger than the inhibition exerted by the lesioned hemisphere over the unaffected one, thus contributing to increased asymmetry in the feed-forward commands sent to both limbs. Consequently, one means advocated for rehabilitation consists of diminishing this interhemispheric competition by decreasing and increasing inhibitions of the lesioned and contralateral hemisphere, respectively.19-22 Considering the control processes outlined above, alteration of the feed-forward command sent to the paretic limb may, in particular, have 3 consequences: first, obviously, an impairment of the integrated timing of feed-forward signals, which cannot optimally contribute to coordination; second, the degradation of the paretic limb’s movement trajectories, becoming typically segmented, irregular, and with multipeaked velocity profiles, 23 which renders coordination more difficult to achieve; third, feed-forward and feedback processes are to some extent interrelated 24 ; for instance, it has been shown that error correction processes in bimanual coordination benefit from muscle activation on account of the prediction of sensory consequences of efferent signals.11,17 Thus, alterations of the feed-forward command to the paretic limb also likely affect somatosensory feedback and impair the afferencesbased processes involved in coordination. That is, although increased reliance on afference-based processes could be considered crucial to compensate for the alteration of efferent signals, afferences may also constitute a limiting factor of bimanual coordination.
From a clinical point of view, motor control impairment in stroke patients is often related to deficits of integration of somatosensory and/or visual afferences, independently of elemental sensory deficits. 25 It can be related to various cognitive impairments such as visuospatial deficits, neglect, or other attentional deficits. The gathering of visual information has been shown to be responsible for spatial estimation deficits in hemiplegic stroke patients, for example, during a visual reaching task. 26 Somatosensory deficits are considered to be a common consequence of stroke, although there is no consensus on their exact prevalence,27,28 and disorganized sensations may produce disorganized movements in spite of an intact motor system. 29
Based on these considerations, the purpose of the present study is to tease apart the role of afference-based processes in the impairment of bimanual coordination after unilateral stroke. In particular, we hypothesized that somatosensory deficits in the paretic limb contribute to the impairment of bimanual coordination. Assessing the influence of factors other than the paretic limb motor performance may not only contribute to better understanding the mechanisms of bimanual coordination in stroke patients but also have important implications for the design of efficient rehabilitation strategies. To do so, we used a kinesthetic tracking task in which the paretic limb of stroke patients was passively driven by the machine. The integrated timing of feed-forward signals has been shown to be a predominant source of coordination in active bimanual movement 30 while being negligible in coordination with 1 passively driven limb. 10 In contrast, the role of afference-based processes has been shown to be much stronger in the case of passive movement coordination than when the 2 limbs are active. 10 Finally, passive driving of the paretic limb allows for controlling the effect of unpredictable and irregular movement trajectories. Thus, the present task design should allow us to determine whether afference-based processes are partly responsible for impaired bimanual performance. With respect to the above hypothesis, somatosensory deficits would affect both the intentional corrections based on somatosensory afferences and phase entrainment. The expected result was thus an impairment of bimanual coordination in stroke patients compared with control participants, especially when performing the task with eyes closed.
Methods
Participants
Two groups of participants were involved in the experiment. The experimental group was composed of 10 hemiparetic chronic stroke patients (9 men; mean age, 62 ± 13 years). All were involved in the experiment between 12 and 55 months after the onset of a primary unilateral stroke. Patients were mild to moderately impaired, with upper-extremity Fugl-Meyer scores 31 ranging between 40 and 61 out of 66. Two patients were left-hand dominant before stroke; 5 of 10 patients had motor-dominant-side stroke. The main inclusion criteria were notably a spasticity score inferior or equal to 3 of 4 (modified Ashworth scale 32 ) for elbow and wrist flexors and extensors, the absence of orthopedic limitations on elbow and wrist, the absence of neglect (Bergego scale <5), the absence of aphasia (Boston scale >5), and the absence of global cognitive disability (Mini-Mental State Examination score > 22/30). Table 1 summarizes the main clinical characteristics of patients.
Main Characteristics of Patients. a
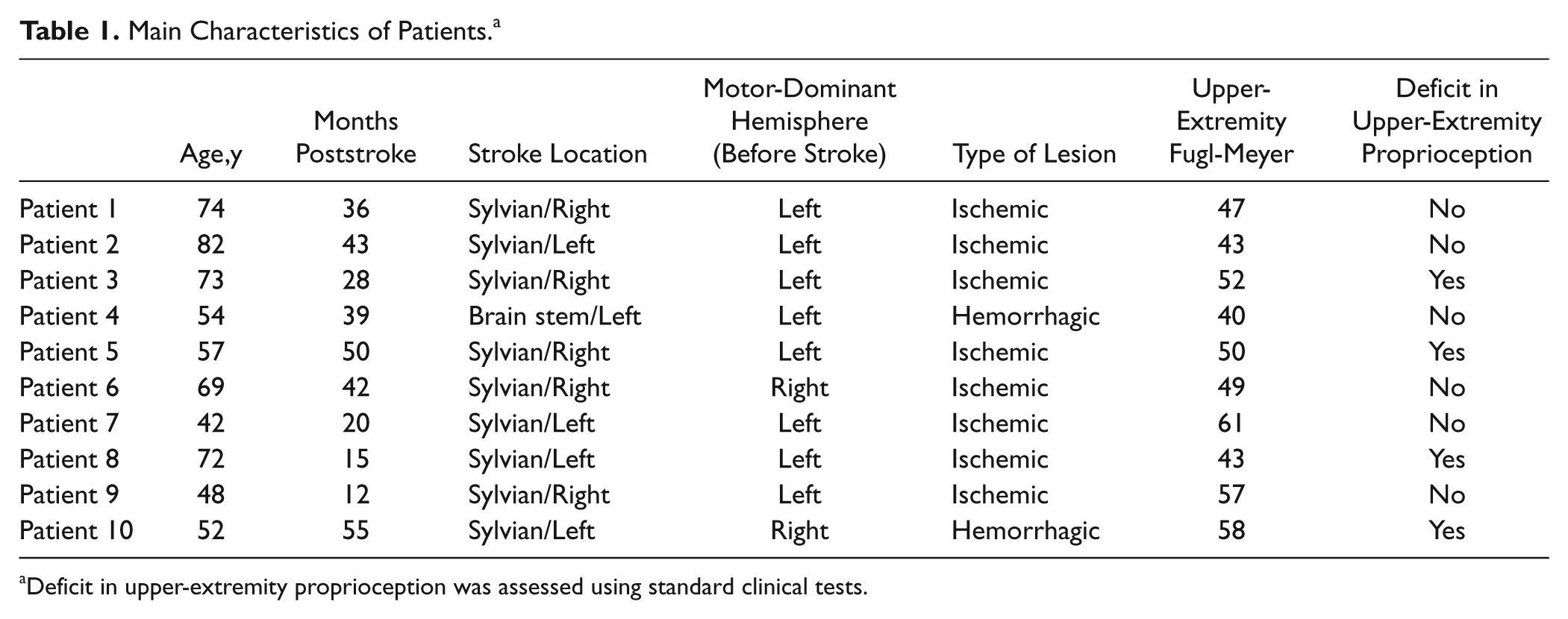
Deficit in upper-extremity proprioception was assessed using standard clinical tests.
The control group consisted of 8 healthy individuals (6 men) matched for age (mean age, 57 ± 4.6 years). They declared neither recent upper-limb traumatism nor previous stroke. One participant was left-handed.
The study protocol (EudraCT Number: 2009-A01297-50) was approved by the Ethical Committee South Mediterranea III and conducted according to the Declaration of Helsinki. Participants gave written consent before participating.
Apparatus
We used a Con-Trex MJ isokinetic machine (Con-Trex MJ, CMV AG, Dübendorf, Switzerland), which offers a continuous passive motion mode notably used for mobilization of stroke patients. 33 Participants were seated on the machine’s chair, with the arms alongside the trunk. The forearm of the passively driven limb was rested on the Con-Trex ad hoc manipulandum, designed for implementing flexion-extension movements of the elbow in the sagittal plane. The forearm was attached to the manipulandum using straps, and participants placed their hand on the handle of the manipulandum. The alignment between the rotational axes of the dynamometer and the elbow joint was checked by the experimenter for each trial. The elbow of the matching limb was rested on a padded cast, so that participants could freely perform flexion-extension movements. With the free hand, participants held a handle equivalent in size to the manipulandum handle. To minimize unwanted body movements during task recording, straps were also applied across the chest. For each participant, the parameters of the isokinetic machine were set so as to produce 10 successive flexion-extension cycles without arrest. The total movement amplitude was 50° around the elbow joint, starting from a minimum position of 100° flexion to a maximum position of 50° flexion. Movement velocity prescribed by the machine was set at 20/s, which corresponds to an oscillation frequency of 0.2 Hz (5-s cycle periods). Movement amplitude and velocity were set in absolute rather than relative values to allow for proper comparisons because it has been shown that both oscillation frequency and amplitude are likely to affect the relative strengths of the different control processes involved in coordination.10,17
Movement trajectories of the 2 hands were recorded using a 3D motion capture system (Zebris CMS-20, Medical GmbH, Isny, Germany). The system uses miniature transmitters (markers) emitting ultrasonic impulses. A measuring device composed of 3 microphones allows one to capture the 3-dimensional coordinates of the markers. The measuring device was placed in front of the participant, at a distance of about 150 cm from the Con-Trex chair. A marker was placed on each of the 2 handles held by the participants, so as to face the measuring device. Three-dimensional trajectories of the markers were recorded with a sampling frequency of 50 Hz. Figure 1 schematizes the experimental device.
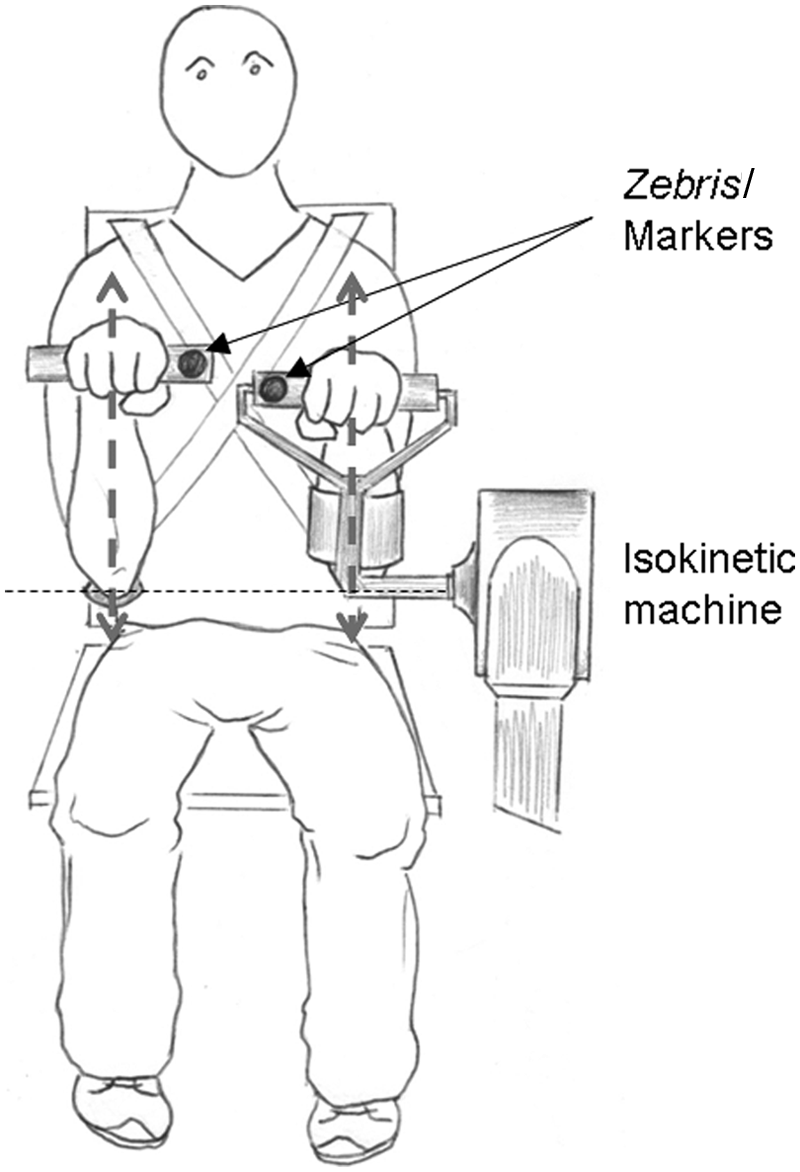
Schematic representation of the experimental device.
Task and Procedure
After a short familiarization phase, the experimental task was as follows: participants were instructed to match with their free hand the trajectory of the passively driven hand as accurately as possible, during 10 flexion-extension cycles. In addition, they were asked to keep the muscles of the passively driven limb as relaxed as possible. The task was performed under 2 conditions: (1) with eyes open (Eye-O) and (2) with eyes closed (Eye-C). The order of the 2 conditions was randomized.
For stroke patients, the hemiparetic limb was always the passively driven limb. To test for any effect of hand dominance on the variables analyzed, the control group performed the 2 task conditions both with the dominant and the nondominant hands being passively driven by the machine (D-D and ND-D). The 4 conditions were assigned in a pseudorandom order. All participants performed 1 trial in each experimental condition, and a minimum resting period of about 2 minutes was given between each trial.
Data Reduction and Analysis
We applied a second-order bidirectional low-pass Butterworth filter (cutoff frequency 2 Hz) to the right- and left-hand vertical movement trajectories and analyzed 9 successive movement cycles for each trial. The first cycle was discarded from analysis to avoid any effect caused by delayed adaptation to the task. To assess the capacity of the freely moving limb to coordinate with the passively driven limb, we focused on the following variables:
The mean and standard deviation (SD) of continuous relative phase (CRP) between the 2 oscillating limbs: bimanual coordination performance has usually been assessed through the CRP, where mean CRP indicates the accuracy of coordination (for mean CRP = 0° the 2 limbs move perfectly in phase), and the SD assesses the stability of coordination. 9 CRP (in degree units) is given by
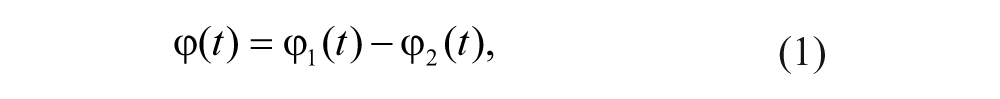
where
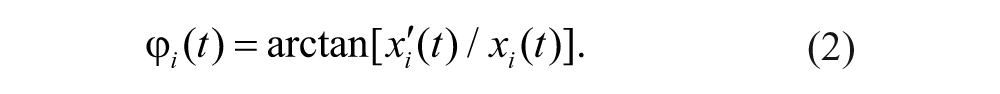
2. The mean of absolute differences between positions (ADP) of the 2 limbs is given by
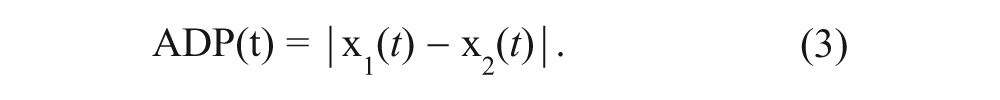
The mean ADP was computed over all successive ADP values. The main difference between CRP and ADP is that CRP assesses the difference between the relative progression of the 2 limbs within their current movement cycles, independently of the amplitude of oscillations (and thus of movement velocity), whereas ADP assesses the accuracy of trajectory matching, including movement amplitude (and thus velocity).
3. To assess smoothness of the matching limb movements, we computed a dimensionless jerk measure, as recommended by Hogan and Sternad 34 :
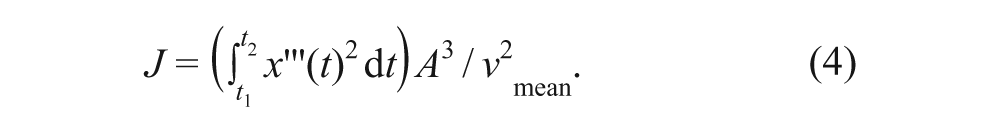
In this equation x″′ is the third derivative of the limb’s position series, A is the total amplitude of movement, and vmean is the mean velocity. This computation was done separately within each semicycle, so that the mean jerk for each trial was obtained by averaging the successive measures. (For more details on computation of CRP, ADP, and jerk, see online supplementary material.)
Statistical analyses were run on the 4 variables under focus: mean CRP, CRP SD, mean ADP, and mean jerk. For the control group, we first tested for an effect of hand dominance on each variable, using a 2-way analysis of variance (ANOVA) Driven limb (2) × Feedback (2), with repeated measures on both factors. For all variables, the analysis showed no difference between the dominant and nondominant hands being passively driven (mean CRP: F(1, 7) = 0.897, P = .375; CRP SD: F(1, 7) = 1.866, P = .214; mean ADP: F(1, 7) = 0.427, P = .534; mean jerk: F(1, 7) = 2.384, P = .166), no significant difference between the Eye-O and Eye-C conditions (mean CRP: F(1, 7) = 4.942, P = .062; CRP SD: F(1, 7) = 0.571, P = .474; mean ADP: F(1, 7) = 0.021, P = .888; mean jerk: F(1, 7) = 0.371, P = .562), and no significant interaction (mean CRP: F(1, 7) = 0.338, P = .579; CRP SD: F(1, 7) = 0.027, P = .875; mean ADP: F(1, 7) = 0.018, P = .898; mean jerk: F(1, 7) = 0.023, P = .883). Consequently, for each variable, we averaged the values of each participant from the Eye-O dominant and nondominant driving conditions and from the Eye-C dominant and nondominant conditions. The average values of the control group were then compared with the patient group.
To test our main hypothesis we applied a 2-way ANOVA Stroke (2) × Feedback (2), with repeated measures on the second factor, on each variable. Tukey honestly significant difference (HSD) post hoc comparisons were used to localize the differences in case of a significant effect of interaction between the 2 factors.
Results
All trials taken together, the mean of mean CRP was −6.13° (SD = 10.82), the mean SD of CRP was 13.64° (SD = 4.74), the mean of mean ADP was 36 mm (SD = 14), and the mean of mean jerk was 9.38 × 105 (SD = 4.65 × 105). Figure 2 shows a representative example of CRP and ADP series obtained from 1 patient trial.
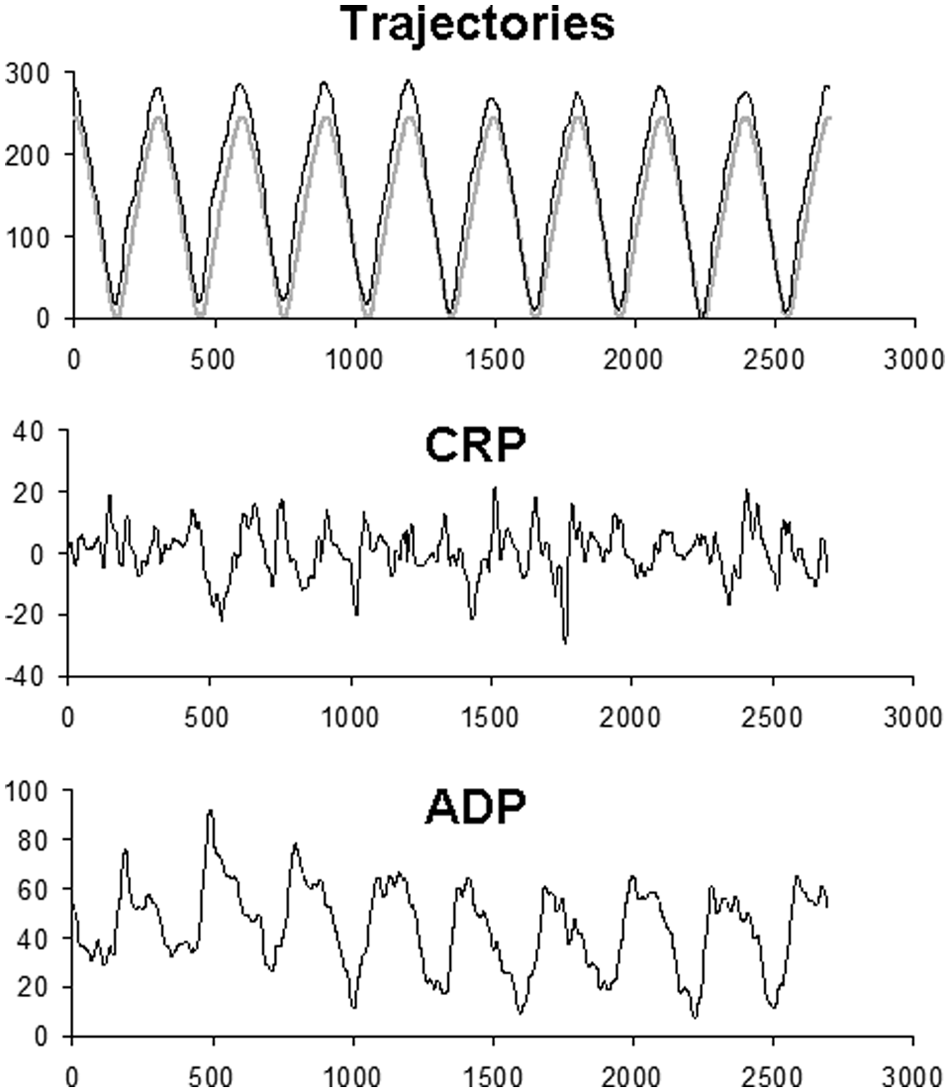
Representative example of the trajectories produced by the 2 limbs (gray line: paretic/driven limb; black line: freely moving limb) and the CRP (in degree units) and ADP (mm) series obtained in 1 experimental trial (patient 7, Eye-O). Abbreviations: CRP, continuous relative phase; ADP, absolute difference between positions.
For mean CRP, the ANOVA showed no significant difference between patients and control group (F(1, 16) = 0.161, P = .693, ηp2 = 0.010, 95% confidence interval [CI95] = 0.00-0.25, power = 0.066), a marginally significant difference between the Eye-O and Eye-C conditions (F(1, 16) = 4.305, P = .054, ηp2 = 0.212, CI95 = 0.00-0.52, power = 0.496), and no interaction between the 2 factors (F(1, 16) = 0.008, P = .931, ηp2 = 0.000, CI95 = 0.00-0.11, power = 0.051).
For CRP SD, the ANOVA showed a significant effect of interaction between Stroke and Feedback (F(1, 16) = 7.689, P = .014, ηp2 = 0.325, CI95 = 0.02-0.60, power = 0.740) and marginally significant effects of Stroke (F(1, 16) = 3.441, P = .082, ηp2 = 0.177, CI95 = 0.00-0.49, power = 0.414) and Feedback (F(1, 16) = 3.162, P = .094, ηp2 = 0.165, CI95 = 0.00-0.48, power = 0.387). Tukey HSD post hoc analysis demonstrated that CRP SD was significantly higher for stroke patients in the Eye-C condition (mean = 16.87°) than in the 3 other conditions (patients Eye-O: 13.63°, control Eye-C: 11.27°, and control Eye-O: 11.98°).
For mean ADP, the analysis showed a significant effect of Feedback (F(1, 16) = 5.594, P = .031, ηp2 = 0.259, CI95 = 0.00-0.55, power = 0.603) and a significant interaction between Stroke and Feedback (F(1, 16) = 4.973, P = .040, ηp2 = 0.237, CI95 = 0.00-0.54, power = 0.554) but no main effect of Stroke (F(1, 16) = 1.76, P = .203, ηp2 = 0.099, CI95 = 0.00-0.41, power = 0.239). Tukey HSD tests showed that mean ADP was significantly higher for stroke patients in the Eye-C condition (47 mm) than in the 3 other conditions (patients Eye-O: 31 mm, control Eye-C: 32 mm, and control Eye-O: 32 mm).
Finally, for the mean jerk, we found a significant difference between patients (11.85 × .105) and control group (6.3 × .105, F(1, 16) = 11.52, P = .004, ηp2 = 0.419, CI95 = 0.07-0.66, power = 0.890) but no significant effect of Feedback (F(1, 16) = 0.62, P = .443, ηp2 = 0.037, CI95 = 0.00-0.32, power = 0.115) nor interaction between the 2 factors (F(1, 16) = 1.51, P = .236, ηp2 = 0.086, CI95 = 0.00-0.39, power = 0.212). All results are summarized in Figure 3.
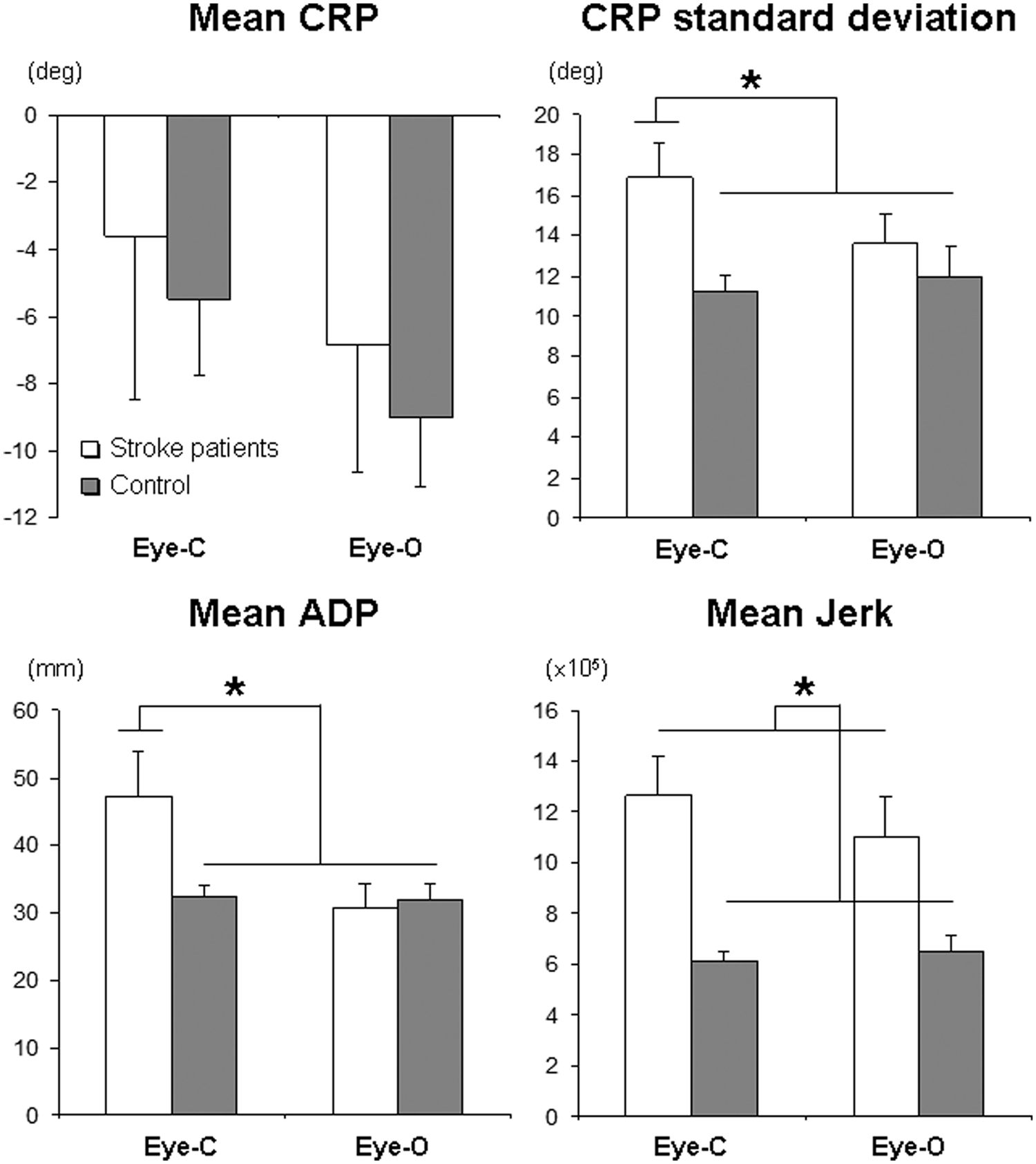
Summarizes the significant results obtained on CRP standard deviation, mean ADP, and mean jerk. Error bars represent standard errors. Abbreviations: CRP, continuous relative phase; ADP, absolute difference between positions; Eye-C, with eyes closed; Eye-O, with eyes open.
Discussion
This study intended to assess the role of afference-based sources of coordination in the impairment of bimanual coordination after stroke. Considering all the experimental conditions taken together, we found that the mean CRP was globally negative, which (given the convention used) indicates that the freely moving limb was slightly leading the driven limb (Figure 3). Such a phase shift has been reported by several studies on sensorimotor coordination and was accounted for by asymmetry between the 2 limbs, caused by, for instance, different eigenfrequencies of the 2 oscillators, handedness, or laterally focused attention. 35 In the present study, the global phase shift may in particular be a result of laterally focused attention inherent to the task requirements because 1 limb had to actively coordinate with the other passively driven limb. It may have been accentuated by the nonnatural low movement frequency used in the present experiment.
As mentioned in the introduction, somatosensory afferences are involved in both intentional correction mechanisms and phase entrainment. Phase entrainment has been shown to have a stabilizing effect for both in-phase and antiphase coordination patterns, at the expense of any other phase relationship. 18 Accordingly, differential strengths of phase entrainment between stroke patients and controls should primarily appear in the stability of the coordination performed, that is, in CRP variability. Analyses showed a significant difference between CRP SD in patients in the eyes-closed condition compared with that in patients with eyes-open, controls with eyes closed, and controls with eyes open, suggesting that phase entrainment is less effective after stroke. The same pattern of results was obtained on mean ADP, showing that trajectory matching performance was significantly degraded when patients performed the task without visual information compared with patients with eyes-open, controls with eyes closed, and controls with eyes open (Figure 3). Together these results converge toward the idea that somatosensory-based processes constitute a limiting factor of bimanual coordination after stroke.
With regard to visually based error correction, our results showed no significant difference between patients and controls in the eyes-open condition, neither in mean CRP, nor CRP SD, nor mean ADP. These results suggest that the availability of visual feedback on the performed coordination allows stroke patients to produce efficient bimanual movements. That is, the integration of visual information may not constitute a limiting factor responsible for the impairment of bimanual coordination after stroke. In contrast, results suggest that the use of visual information when available is likely to compensate for deficits in somatosensory-based processes.
Finally, we found that the mean jerk was dramatically increased for stroke patients compared with the control group (Figure 3). It has been argued that deterioration of the unaffected limb kinematics in bimanual movements could be a result of adaptation to irregular paretic limb kinematics, thus being a consequence of the persistence of coupling after stroke. 36 In the present task, however, the paretic limb was motor driven. Therefore, the present result is rather consistent with previous studies suggesting motor deficits in the presumed-to-be-unaffected limb, which are apparent even in unilateral performance. 37 This contralateral deficit seems to persist even when coordinating with perfectly regular and controlled trajectories in the paretic limb. It might be accentuated especially with the low movement frequency imposed in the present experiment because evidence has shown that participants spontaneously tend to move close to their preferred frequency, avoiding slow movements. 38 The increased jerk in stroke patients might thus reflect the greater difficulty they have than control participants in moving the limb that is presumed to be unaffected at low frequency. Altogether, our results underline the fact that impaired bimanual performance cannot be exclusively attributed to the impaired motor capacities of the paretic limb and disruption of the interhemispheric balance but should also be related to the diminished capacity of the nonparetic limb to adapt by means of contralateral afference-based processes.
The significance of our present study needs to be discussed with regard to some methodological considerations. First, during the experiment, the chest of participants was strapped and the elbow of the freely moving limb fixed in position. These methodological precautions did not create an ecologically valid task because they restricted possible compensations by trunk and shoulder movements. However, they were a necessity to rigorously test the hypothesis of this study. In particular, ADP measures could have been biased by uncontrolled shifts in position of the freely moving limb. Second, muscle activity in the passively driven limb was not EMG monitored. Actually, participants were very likely to activate the muscles of the driven limb, even if not required, as evidenced previously in similar kinesthetic tracking experiments.10,11,17 With respect to the hypothesis of the present study, this would not constitute an artifact calling into question our conclusion. Indeed, external driving of the (paretic) limb rules out the effect of integrated timing of feed-forward signals and erratic movement production on coordination performance, thus allowing assessment of the effect of afference-based processes in the different experimental conditions. However, as mentioned in the introduction, afference-based coordination processes may benefit from muscle activation on account of the prediction of sensory consequences of efferent signals.11,17 Therefore, one may consider that different levels of muscle activation in the paretic (driven) limb could be one of the neurophysiological underpinnings of somatosensory deficits. Further studies that include EMG monitoring are required to address this issue in the context of stroke. Third, the effect sizes of the significant results are medium (for CRP SD and ADP) to large (jerk), allowing our results to be considered with confidence. Note, however, that the CIs are relatively large, which may be a result of the relatively small sample sizes. Larger samples would allow the estimation of effect size to be refined. Finally, patients involved in the present study were mostly mild to moderately impaired. Although studies addressing the effect of bimanual therapy after stroke have involved patients with very heterogeneous levels of initial impairment, a recent review suggests that bimanual training may provide greater benefit to mildly and moderately impaired patients. 6 In view of these methodological considerations, our present study thus allows for reasonable support of the hypothesis that somatosensory deficits in the paretic limb contribute to the impairment of bimanual coordination and may yield some useful implications for bimanual rehabilitation.
To conclude from a clinical point of view, our results raise the following implications and perspectives: First, they stress the need to control the quality and congruence of movement trajectories of the 2 limbs to maximize the efficiency of bimanual rehabilitation through interlimb coupling processes. 4 The role of robot-assisted training appears crucial in this respect: especially in an active-assisted mode, it allows consistent and appropriate somatosensory afferences for “optimal” movement kinematics, thereby improving the ability to rely on somatosensory feedback for the control of single-limb as well as bimanual movements.39-41 Second, these results highlight the advantage of protocols that associate bimanual training with modulation of somatosensory afferents using, for instance, electric stimulation or augmented feedback through virtual reality.6,40,42-44 Moreover, because visual feedback allows patients to compensate for somatosensory deficit, bimanual movement therapy should (at least partly) be conducted with eyes closed, so as to maximally trigger the use of somatosensory afferents. Finally, inconclusive results on the efficiency of “pure” bimanual training could be partly explained by the fact that the different control processes involved in coordination are impaired to different extents according to each patient’s clinical profile. Thus, targeting individual patients’ profiles with respect to the relative contribution of the different processes is one promising avenue of research.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the University Hospital of Montpellier (AOI 2009).
