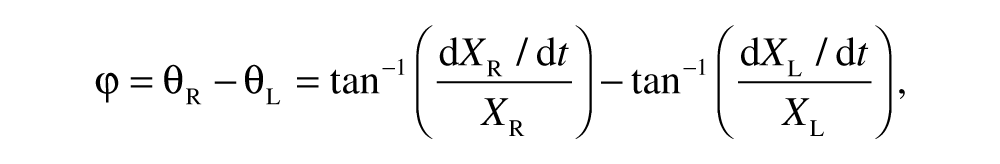Abstract
Background. Freezing of gait (FOG) is a significant clinical problem in Parkinson disease (PD). Similar freezing-like episodes occur during finger movements, but little is known about ongoing motor problems during repetitive hand movements. Objective. To investigate if the regulation of bimanual movements is impaired in those with FOG and if withdrawal of an auditory cue amplifies this problem. Methods. A total of 23 PD patients (11 with and 12 without FOG) and 11 controls (CTRLs) performed repetitive finger movements, either externally paced or following cue withdrawal. Movement frequency, amplitude, and coordination pattern were manipulated. The stability and accuracy of movement were evaluated after exclusion of freezing trials. Results. With auditory pacing present, movement performance was comparable between groups. Following cue withdrawal, motor control deteriorated in those with FOG, resulting in smaller and less stable amplitudes, hastened and more variable frequency, and decreased coordination stability. Conversely, the performance of those without FOG remained mostly similar to that of CTRLs. Conclusions. Compared with those without FOG, those with FOG show greater continuous dyscontrol of bimanual movements, similar to the continuous timing and scaling difficulties during locomotion. Those with FOG also benefit from auditory cueing during upper-limb movements, but these are highly cue dependent. This implies that internal timekeeping functions are more disturbed in those with FOG, who may require rehabilitation strategies for repetitive upper-extremity tasks that include cueing and imagery.
Introduction
Parkinson disease (PD) causes slow and hypokinetic movement.1,2 Freezing of gait (FOG) occurs frequently in the later stages 3 and is associated with fall events. 4 FOG is defined as a brief interruption of walking during which patients are unable to generate effective forward stepping movements despite the intention to walk. 5 The responsiveness of FOG to medical and surgical treatments is often limited. 6 Therefore, effective rehabilitation strategies are needed. Training patients to use external sensory cues that coincide with the appropriate stepping rhythm or stride length generally improves the gait pattern7-10 in PD. However, the beneficial effects of cues were not as obvious in those with FOG11-14 as in those without FOG and were more dependent on the presence of the cue. 14 Those with FOG (PD + FOG) and those without FOG (PD − FOG) may have distinct cognitive and motor profiles, even in the face of similar disease severity.15-21 FOG is associated with greater cognitive dysfunction, especially for executive functioning.15-17 PD + FOG patients also have a more severely disrupted gait pattern outside the momentary FOG episodes (hereafter referred to as ongoing or continuous abnormalities), including increased variability of step timing, 18 disordered bilateral coordination19,21 and a reduction in stride amplitude. 21 However, it is still unclear which of these abnormalities is most affected in patients with FOG.
Freezing-like motor blocks are not restricted to gait but also occur in other rhythmic tasks such as speech, 22 writing, manual tapping, tooth brushing, and foot wiping.23-26 We recently showed that upper-limb freezing episodes (FO-UL) were correlated to FOG and were also preceded by similar spatiotemporal changes—namely, a gradual decrease in movement amplitude and increase in frequency (hastening).25,26 Abnormal high-frequency components were also found to be common characteristics of both FO-UL and FOG. 26 Hence we argued that freezing is related to a general deficit in the organization of repetitive motion that is effector independent. 26 In contrast to the continuous gait difficulties described for those with FOG,18-21 little is known about motor problems during hand movements outside actual freezing episodes.24,25 It is also unclear whether these motor problems can be overcome by cueing as greater cue dependency may also decrease the efficiency of cueing for upper-limb movements in those with FOG, just like in gait. 14
Therefore, the first aim of the present study was to compare the general spatiotemporal characteristics of rhythmical finger movements between those with FOG, those without FOG, and controls (CTRLs) while movement amplitude, 21 frequency, 22 and coordination complexity 20 were systematically manipulated. In particular, we tested which of these determinants had the largest impact on movement quality in those with FOG. Second, we examined whether the performance of rhythmical finger movements is more sensitive to auditory cue withdrawal in those with FOG than in those without FOG and in CTRLs. 14 This research question has high relevance for rehabilitation because cueing for tasks other than gait has been an unexplored area. Furthermore, insight into the underlying motor control deficits of freezing will aid in refining current rehabilitation strategies for PD subgroups.
Methods
Participants
A total of 23 PD patients were recruited in the Movement Disorders Clinic of the University Hospital Leuven. Patients who had experienced FOG within 1 month before testing and thus scored positively on item 1 of the revised FOG Questionnaire 27 were classified as those with FOG (PD + FOG; n = 11). Those with FOG and those without FOG (PD − FOG, n = 12) were matched for age, sex, and disease severity. Patients were in Hoehn and Yahr stage II or III 28 during the on state. Exclusion criteria were as follows: (1) diagnosis of a neurological disease (other than PD), (2) presence of a deep brain stimulator, and (3) dementia (Mini-Mental State Examination [MMSE] score <24/30). A total of 11 age-matched, healthy CTRLs (male = 10) also participated. Participants gave informed consent consistent with the Declaration of Helsinki. Ethics approval was received by the Commissie Medische Ethiek Katholieke Universiteit Leuven.
Design and Procedure
Prior to testing, the following clinical data and information were collected: the motor section of the Unified Parkinson’s Disease Rating Scale (UPDRS 29 ), FOG Questionnaire, MMSE, Hoehn and Yahr staging, and the cognitive section of the Scales for Outcomes in PD. 30 This clinical screening occurred at participants’ homes when in the on state.
The experimental session in the laboratory took place in the early morning during the “practically defined off phase”—that is, after omitting the morning dose of medication (12-15 hours after last intake). On this day, the UPDRS motor examination was repeated. Seated on a height-adjustable chair, participants performed a bimanual task consisting of rhythmic flexion and extension movement of the index fingers. We chose a bimanual rather than a unimanual paradigm because it allows well-controlled manipulations of coordination complexity within an experimental setting. Movement complexity, amplitude, and frequency were manipulated according to a 2 × 2 × 2 factorial design. Individuals in the 3 subgroups (PD + FOG, PD − FOG, and CTRL) performed either an easy coordination pattern requiring simultaneous finger movements (in-phase coordination) or a complex pattern requiring alternating movements (antiphase coordination). Before testing, a single trial of alternating movements lasting 30 s enabled the computation of participants’ comfortable amplitude and frequency. During testing, they were instructed to make movements at their comfortable amplitude or at a smaller amplitude (66% of the comfortable amplitude). Similarly, cycle frequency was either comfortable or fast (ie, 100% and 133% of the comfortable frequency). The size of the amplitude decrement and frequency increment was determined in a pilot study and chosen to challenge movement control, so that freezing-like movement breakdown could be triggered. Movement was initially guided by auditory pacing (first 6 movement cycles with cue present), after which it became internally generated (cue withdrawn). Participants performed 3 trials per condition. A square box covering both hands prevented visual feedback during movement. A Micro 1401 acquisition unit (Cambridge Electronic Device, UK) with Spike 2 software recorded angular displacements with temporal and spatial resolutions of 2000 Hz and 0.0001° through analogue encoders placed on the rotation axis of a device connected to each index finger. Participants were given sufficient time to practice and familiarize themselves with task requirements.
Data Processing
This study focused on ongoing motor regulation in those with FOG outside the freezing episodes. Therefore, we excluded trials in which FO-UL occurred for this analysis. FO-UL was defined as “a period of an involuntary absence of or markedly reduced cyclic movements.” 26 A detailed description of the freezing detection method is given elsewhere. 26
Kinematic time series were low-pass filtered (digital Butterworth filter with a cutoff frequency of 30 Hz) using Matlab (version 7.4). The movement parameters that were obtained are discussed below.
Within-limb movement amplitude
Movement amplitude was calculated based on point-by-point measures of the end-effector motions using the Hilbert transform.31,32 This way, mean movement amplitude and its coefficient of variability (COV), that is, the ratio of standard deviation to the mean expressed as a percentage, were obtained.
Within-limb frequency measures
Mean frequency, the deviation from the requested frequency (frequency error in Hz), and frequency COV (expressed as a percentage) were computed using peak-to-peak measures of the end-effector motions.
Consistent with previous research,25,27 results for the disease-dominant hand in patients and the nonwriting hand in CTRLs will be reported for within-limb amplitude and frequency parameters.
Interlimb coordination
Coordination parameters were based on the relative phase (RP), which corresponds to the difference in position of the left and right fingers in their movement cycle at a given moment. 33 RP is expressed in degrees and was calculated on a point-by-point estimation using the instantaneous phase of each signal, produced by the Hilbert transform.31,32 The following equation was used:
where XR and XL are the instantaneous displacements of the right and left end effectors, respectively, and dXR/dt and dXL/dt are the instantaneous velocities. Circular variability of RP (RPvar) was computed. Absolute error scores (RPerror) indicated the degree of deviation from the target RP (ie, 0° and 180° for in-phase and antiphase patterns, respectively).
Movement outcomes of trials within a same condition were averaged (3 trials per condition or fewer in the case of omitted freezing trials).
Statistical Analysis
Statistical analyses were performed using STATISTICA (8.0). Clinical parameters were compared between groups using a 1-way analysis of variance (ANOVA), nonparametric Mann-Whitney U tests (2 groups), or Kruskal-Wallis tests (3 groups) in the case of skewed distribution of the data. A repeated-measures ANOVA was used to test the influence of within-subject factors CUE (present, withdrawn), PATTERN (in-phase, antiphase), AMPLITUDE (comfortable, small), and FREQUENCY (comfortable, fast) in addition to the between-subject factor GROUP (PD + FOG, PD − FOG, and CTRL). The relative contribution of within-subject manipulations during movement after cue withdrawal was further explored in those with FOG using a 1-way ANOVA comparing the 2 levels of factors Frequency, Amplitude, and Pattern. Significance levels were set at .05. Significant effects were addressed by Newman-Keuls post hoc tests. Results are reported as means and standard errors of measurements.
Results
Participants
Data of 1 patient from the PD + FOG group were excluded from the analysis because freezing episodes occurred in all but 1 trial. Apart from severe FOG, this patient showed no other signs of greater disease severity or cognitive impairment than the other patients. Also, 8 others with and 2 without FOG demonstrated FO-UL, resulting in a total of 64 trials with FO-UL (on average 6.4 [1.4] per person). These trials were excluded from analysis. Details about the occurrence and characteristics of freezing episodes can be found elsewhere. 26 All but 1 freezing episode occurred after cue withdrawal. Table 1 represents the clinical characteristics of the remaining 10 individuals with FOG, 12 individuals without FOG, and 11 CTRLs. Age, gender distribution, and comfortable movement frequency and amplitude measured before actual testing were similar in the 3 groups. Mean disease duration, UPDRS motor scores in on and off states, Hoehn and Yahr staging, daily levodopa-equivalent dose, and MMSE scores were not significantly different between the PD + FOG and PD − FOG groups, indicating comparable disease profiles. Scores for the cognitive section of the Scales for Outcomes in PD were lower in those with FOG than in those without FOG and in CTRLs but fell within the range of normal cognition. 34
Demographic and Clinical Characteristics of 12 Participants Without FOG, 10 With FOG, and 11 Controls (Median and Interquartile Ranges)
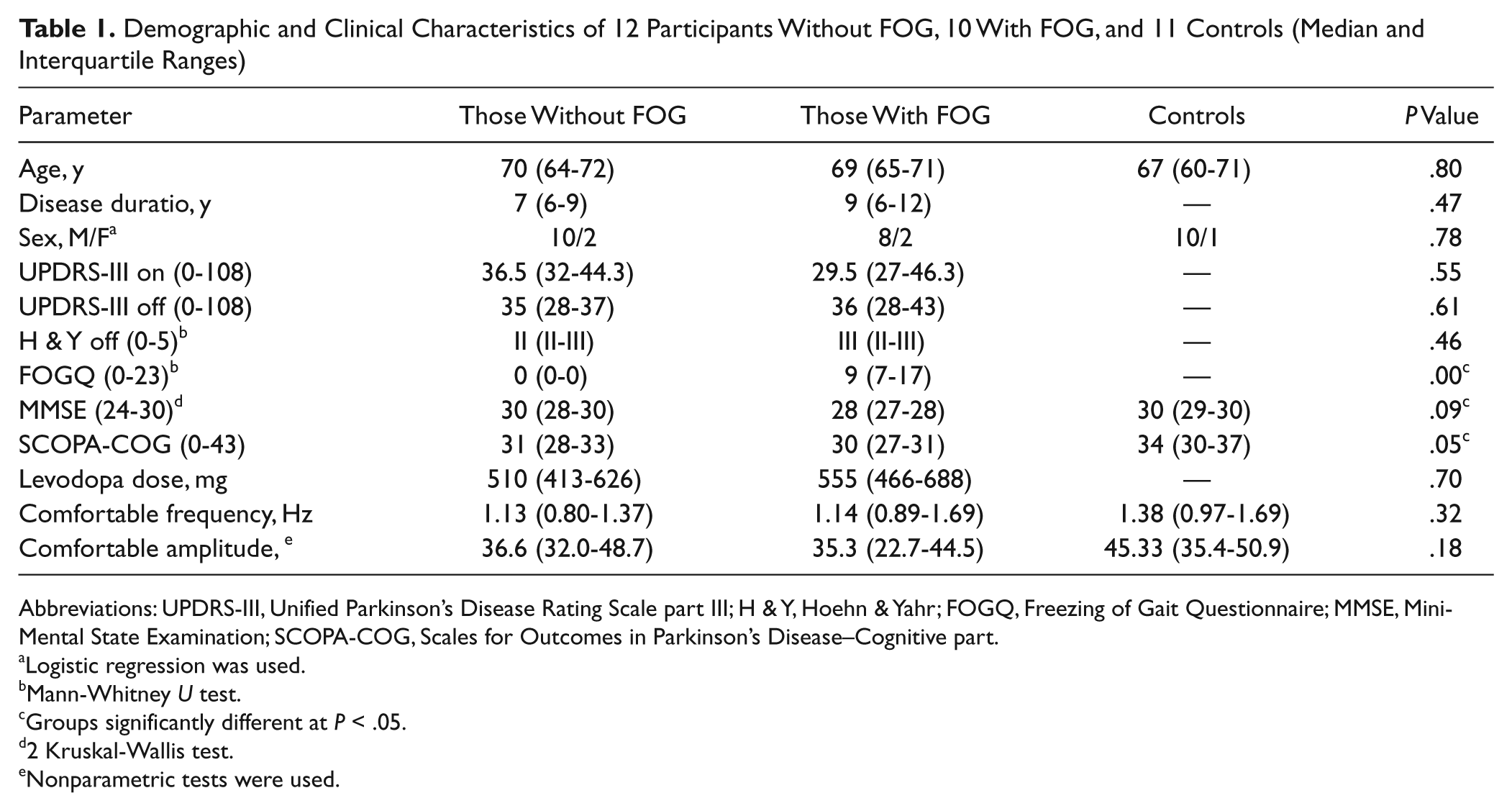
Abbreviations: UPDRS-III, Unified Parkinson’s Disease Rating Scale part III; H & Y, Hoehn & Yahr; FOGQ, Freezing of Gait Questionnaire; MMSE, MiniMental State Examination; SCOPA-COG, Scales for Outcomes in Parkinson’s Disease–Cognitive part.
Logistic regression was used.
Mann-Whitney U test.
Groups significantly different at P < .05.
2 Kruskal-Wallis test.
Nonparametric tests were used.
Ongoing Movement Performance (Independent of Freezing Episodes)
Omitting trials in which freezing occurred led to missing values in 6 patients of the PD + FOG group (none in the PD − FOG group). Data were therefore pooled for the levels of the factor Frequency. The factor Frequency was preferred over factors Pattern and Amplitude because (1) it eliminated all missing values in contrast to pooling for the other factors and (2) it was considered a less-determining factor than Amplitude 21 and Pattern. 20 The results reported below are based on repeated-measures ANOVA with Cue, Pattern and Amplitude, and Group as factors. Similar results were obtained when data were pooled over other factors.
Movement amplitude
Amplitude requirements were based on subject-specific comfortable amplitudes, which were similar between groups (Table 1). All participants made smaller movements in the small-amplitude condition than in comfortable-amplitude conditions (P < .0005). The significant Group × Cue × Amplitude interaction (P = .044) showed that only in the PD + FOG group, the amplitude decreased after withdrawal of auditory pacing in comfortable- amplitude conditions (from 47.14 [5.64] to 42.36 [5.33]; P = .039). In contrast, CTRLs increased their amplitude after cue withdrawal in the comfortable- (64.70 [3.08] vs 69.15 [3.94]; P = .00023) and small-amplitude conditions (40.58 [3.12] vs 43.52 [3.46]; P = .016). This resulted in a significantly smaller amplitude in those with FOG (42.36 [5.33]) than in CTRLs (69.15 [3.91]) during uncued, comfortable-amplitude conditions (P = .039). No group differences were found when the cue was present.
We found a significant Group × Cue interaction effect (P = .00043) on amplitude variability. For externally guided movement, no group differences were found (Figure 1). However, during internally guided movement, amplitude COV (which corrects for differences in mean amplitude) was higher in those with FOG than in CTRLs (P = .044). A cue effect was only found in those with FOG where amplitude variability was significantly higher in the absence of the cue compared with while the cue was present (P = .000521).
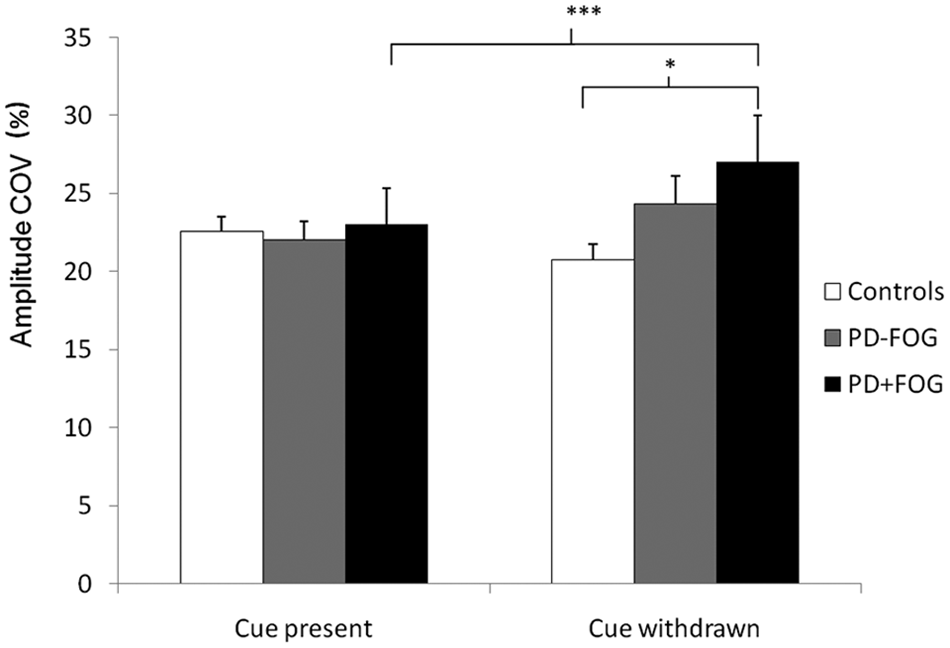
Amplitude coefficient of variability (COV) as a percentage in controls (CTRLs), those without FOG, and those with FOG during movement guided by auditory pacing (cue present) and after cue withdrawal (cue withdrawn). After removal of the cue, the amplitude COV of those with FOG significantly increased and became higher than that of CTRLs. Vertical bars represent standard error of measurements. Abbreviations: PD, Parkinson disease; FOG, freezing of gait.
Movement frequency
Frequency requirements were based on subject-specific comfortable frequency, which was comparable between groups (Table 1). The mean frequency increased when auditory pacing was removed (P = .0052). When we explored the almost significant Group × Cue interaction (P = .088), the post hoc test suggested that this hastening effect after cue withdrawal occurred mainly in those with FOG (P = .0053). Unlike CTRLs, both patient groups moved faster during small-amplitude conditions than comfortable-amplitude conditions (1.24 [0.13] Hz during comfortable- and 1.43 [0.16] Hz during small-amplitude conditions for the PD − FOG [P = .00027] and 1.55 [0.17] Hz and 1.74 [0.20] Hz for the PD + FOG [P = .00036] groups).
Main effects of the factors Amplitude and Cue showed that frequency error increased during small-amplitude compared with comfortable-amplitude conditions (P = .044) and after cue withdrawal (P = .012). The Group × Cue × Amplitude interaction (P = .023) revealed that an increase in frequency error following cue withdrawal occurred only in PD + FOG during small-amplitude conditions (from 0.22 Hz [0.070] to 0.44 Hz [0.11]; P = .00016). The frequency error increase in small- versus comfortable-amplitude conditions was only significant in those with FOG when the cue was withdrawn (from 0.28 Hz [0.079] in comfortable- to 0.44 Hz [0.11] in small-amplitude conditions; P = .00045).
Variability of movement frequency increased in PD patients when small-amplitude compared with comfortable-amplitude movements were required (in PD − FOG, P = .00015; in PD + FOG, P = .017; Group × Amplitude interaction, P = .039). Figure 2 illustrates the significant Group × Cue interaction (P = .0023). Frequency variability was similar between groups when external cueing was present. However, after cue withdrawal, variability increased significantly in those with FOG only (P = .0028), resulting in a higher frequency COV (frequency COV = 13.47% [1.93]) compared with the PD − FOG group (10.19% [1.61]; P = .012) and CTRLs (6.80% [0.63]; P = .00017).
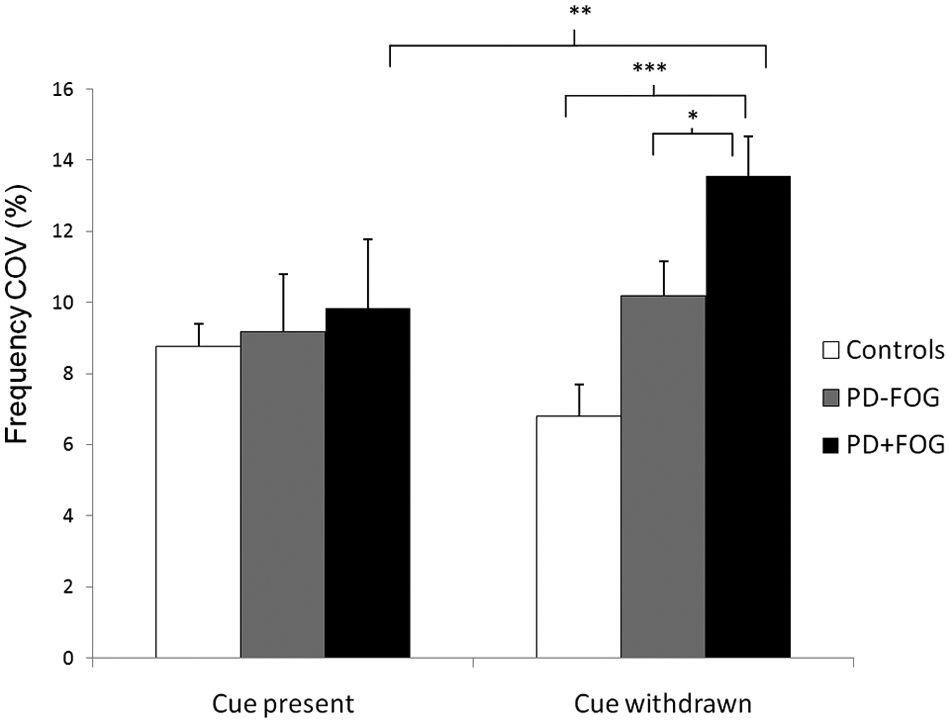
Variability of movement frequency (COV) in CTRLs, those without FOG, and those with FOG during movement guided by auditory pacing (cue present) and after cue withdrawal (cue withdrawn). Withdrawal of external cueing resulted in significantly increased frequency COV in those with FOG than in those without FOG and CTRLs. Vertical bars represent standard error of measurements. Abbreviations: PD, Parkinson disease; FOG, freezing of gait.
Interlimb coordination
Although the main effect of group failed to reach significance (P = .088), those with FOG showed a clear trend to have a higher RP error compared with CTRLs (P = .069). In all participants, RP error was lower while the cue was present than after cue withdrawal (P = .041).
Coordination variability was higher during antiphase than in-phase coordination trials (P < .000005). The significant Group × Cue × Pattern interaction is depicted in Figure 3 (P = .018). In those with FOG, RPvar increased following cue withdrawal (P = .055 and P = .00012 for in-phase and antiphase conditions, respectively). In those without FOG, this cue effect was only present during antiphase conditions (P = .012). In the most complex condition—namely, antiphase coordination after cue withdrawal—RPvar was significantly higher in those with FOG than in those without FOG (P = .0024) and CTRLs (P = .00027).
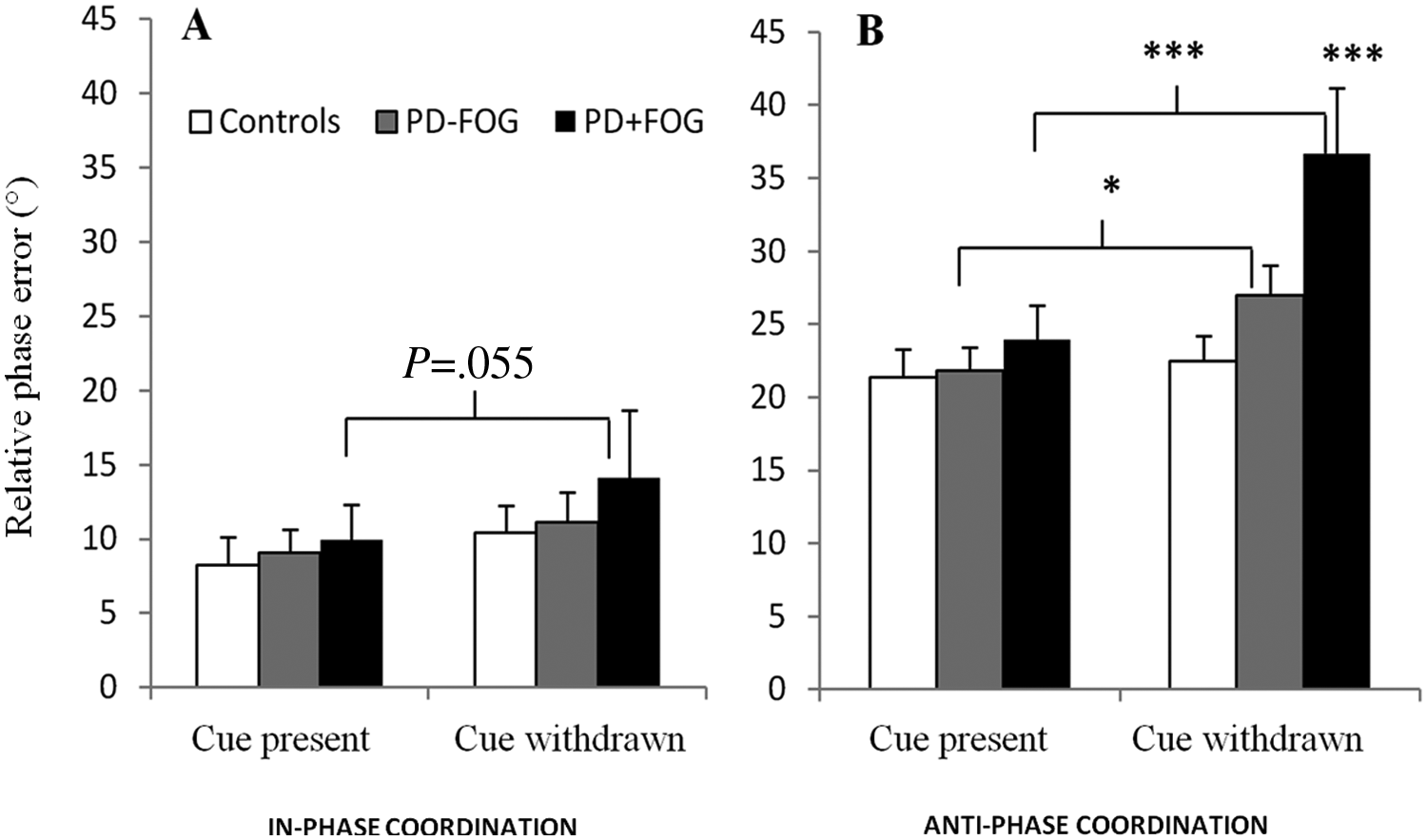
Variability of relative phase (RP) in CTRLs, those without FOG, and those with FOG during movement guided by auditory pacing (cue present) and after cue withdrawal (cue withdrawn) during (A) in-phase coordination and (B) antiphase coordination. RP variability during in-phase coordination was higher in those with FOG after cue withdrawal compared with when the cue was present. During antiphase coordination, variability increased after cue withdrawal in those without FOG and even more strongly in those with FOG, resulting in significantly higher RP variability than in those without FOG and CTRLs. Vertical bars represent standard error of measurements. Abbreviations: PD, Parkinson disease; FOG, freezing of gait.
Exploratory Subanalysis in Patients With FOG
We further explored which movement constraint (amplitude, frequency, and pattern) had the most deteriorating influence on uncued motor performance in those with FOG (Table 2). We were especially interested in the influence of a given factor (eg, Amplitude) on outcome parameters that did not directly depend on it (eg, on frequency outcomes instead of mean and variability of amplitude). Unlike frequency and pattern constraints, the manipulation of amplitude affected outcomes within the scaling domain as well as frequency and coordination measures. In small-amplitude conditions, the frequency of those with FOG became hastened (11.78% increase in mean frequency from 1.65 [0.18] Hz to 1.86 [0.21] Hz; P = .0086) and deviated more from the requested frequency (83.03% increase in frequency error from 0.28 [0.067] Hz to 0.44 [0.11] Hz; P = .013) than under comfortable-amplitude conditions. Similarly, intermanual coordination in those with FOG became worse in small-amplitude compared with comfortable-amplitude conditions, resulting in a 52.26% increase in RP error (from 9.99 [1.45] to 15.06 [2.67]; P = .018) and a 22.58% increase in RP variability (from 22.57 [2.49] to 27.41 [2.91]; P = .0026).
Exploratory Analyses of the Relative Influence of Amplitude, Frequency, and Pattern Manipulations on Motor Performance in Those With FOG After Cue Withdrawal a
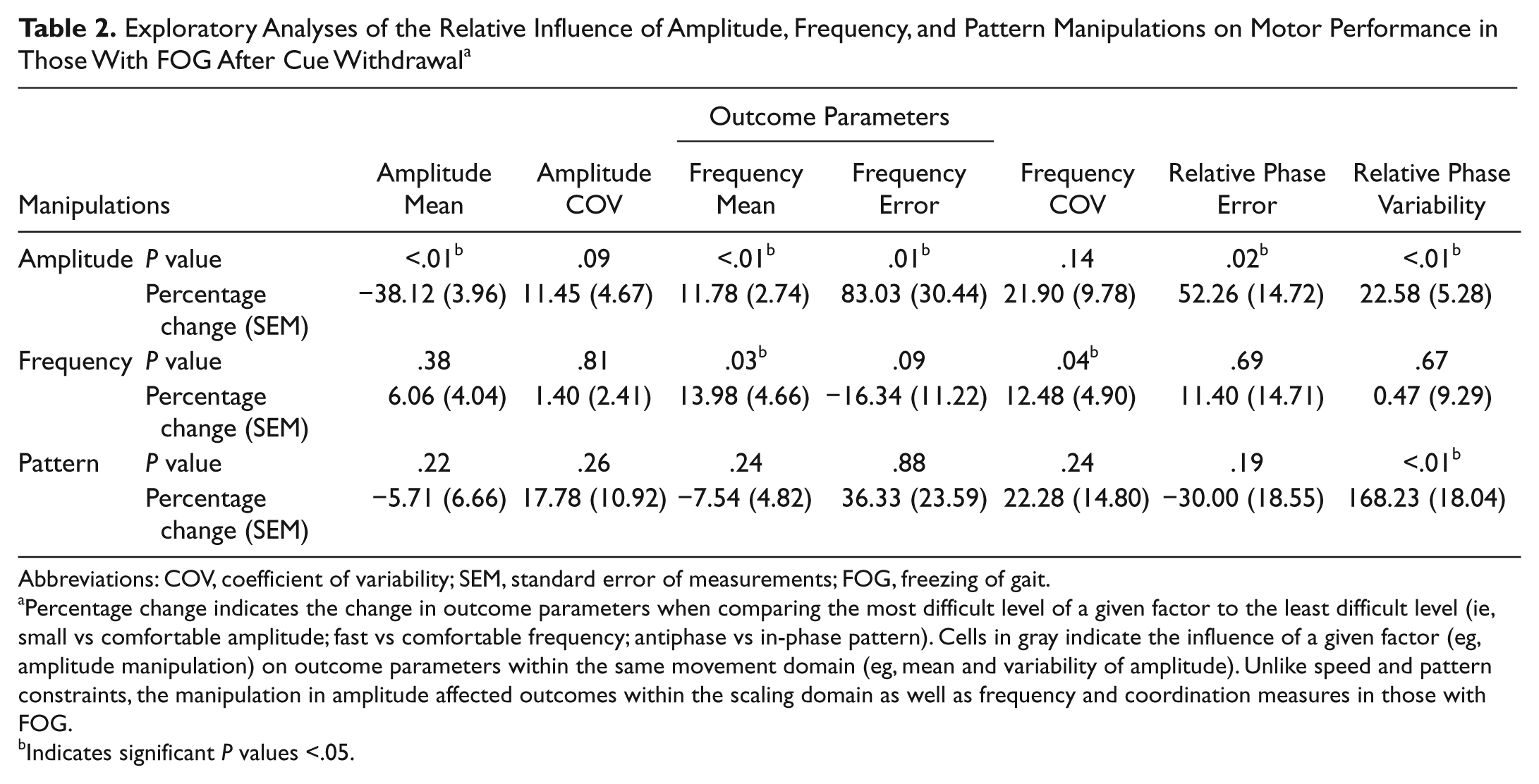
Abbreviations: COV, coefficient of variability; SEM, standard error of measurements; FOG, freezing of gait.
Percentage change indicates the change in outcome parameters when comparing the most difficult level of a given factor to the least difficult level (ie, small vs comfortable amplitude; fast vs comfortable frequency; antiphase vs in-phase pattern). Cells in gray indicate the influence of a given factor (eg, amplitude manipulation) on outcome parameters within the same movement domain (eg, mean and variability of amplitude). Unlike speed and pattern constraints, the manipulation in amplitude affected outcomes within the scaling domain as well as frequency and coordination measures in those with FOG.
Indicates significant P values <.05.
Discussion
The aim of the study was to investigate motor abnormalities in those with FOG and those without FOG during ongoing repetitive upper-limb movements and how these are influenced by cue withdrawal. We found that within-limb spatiotemporal and coordinative control of internally generated finger movements is much more affected in those with FOG than in those without FOG. These findings coincide remarkably with the pronounced FOG-related motor abnormalities present during gait.20-23 Second, the dramatic effect of cue withdrawal indicates that those with FOG benefit from external guidance but show increased cue dependency. This suggests that internal motor control is more affected in those with FOG than in those without FOG, 35 which is important for rehabilitation.
Motor Abnormalities in Patients With FOG
Internally generated finger movements in those with FOG were characterized by a small and unstable amplitude; a variable, hastened frequency; and decreased coordinative stability. In contrast, movement in those without FOG did not differ significantly from CTRLs in most parameters. The fact that visual feedback was absent and interlimb coordination was required may explain why our paradigm revealed a faulty scaling timing mechanism in those with FOG, which was not clearly shown previously.24,25
Although correlations were found between FOG and FO-UL,25,26 the generic nature of the freezing phenomenon is still a matter of debate. We previously showed that changes in the kinematic signals prior to a freezing episode are quite similar in gait and upper-limb movement. 26 FOG has been related to a continuously disrupted gait pattern even outside freezing episodes.18-21 The present results therefore expand the similarity between FOG and FO-UL, relating them to a faulty organization of ongoing movement, and add strength to the conceptualization of freezing as a generic spatiotemporal motor control problem. 26 It is plausible that this background of abnormal motor physiology culminates in episodic breakdown during gait and finger movement.
FOG is known to occur in the face of heightened attentional or perceptuomotor demands, for example when turning, 36 passing through a narrow door,37,38 or performing a secondary task while walking. 36 Here, freezing-related motor abnormalities such as hastening were triggered without additional cognitive, limbic, or postural load but were amplified in conditions where small-amplitude finger movements were requested. Though based on exploratory analyses, this finding is important because it is congruent with the dramatic effect of ongoing amplitude adjustments on cadence regulation during straight line walking 21 and turning 36 and with the triggering effect of small-amplitude movements on freezing episodes during finger movement. 26 These results underscore the core role of impaired amplitude regulation in the freezing phenomenon.21,26 Hence, reteaching patients to maintain a regular amplitude may improve writing skills and other repetitive upper-limb movement and prevent hastening or freezing episodes, but this awaits further study.
To focus on ongoing motor abnormalities, we excluded trials in which FO-UL occurred, inherently leading to missing values in the PD + FOG group. Pooling data for both levels of the factor Frequency eliminated all missing values but may have influenced the present results. This methodological concern may be avoided by incorporating more repeated trials per condition in future research.
Neuroanatomic Correlates of Freezing
Bilateral coordination of automated movement sequences requires a tight connection of brain areas within a distributed network.39,40 The basal ganglia are crucial in regulating movement amplitude.39-44 As a result of striatal dysfunction, people with PD tend to shift from automatic to controlled movement, associated with decreased brain activity in the striato–supplementary motor areas and increased activation of the cerebellum and premotor-parietal areas.40,45,46 The distinct motor profile in those with FOG shown here suggests a more profound striatofrontal disruption that does not allow further tapping into compensatory neural reserves and may as such cause motor breakdown. A recent study 47 comparing brain activity during motor imagery in those with FOG and those without FOG supports this idea. The authors found similar striatal activation levels in both groups but relative underactivation in the supplementary motor area in those with FOG, which they related to stride length dyscontrol. Those with FOG did not show increased cerebellar and premotor-parietal activations but demonstrated increased activation in the mesencephalic locomotor region (MLR), a densely connected region that is thought to drive gait via central pattern generations in the spinal cord.48,49 Gray matter and connectivity changes in the MLR were also found in those with FOG.47,50 Further study is needed to understand whether the MLR is a similar key player in FO-UL or whether it is gait specific.
Cue Dependence in Patients With FOG
The use of sensory cues is an efficient rehabilitation tool in PD because it bypasses the deficient striatofrontal system in favor of cerebello-parietal-premotor pathways.10,51 As such, scaling and timing of steps can be improved in PD and translate to increased functional mobility after a training period.7-10 In a systematic review in 2008, Nieuwboer 11 concluded that the benefits of sensory cueing were less obvious in those with FOG than in those without FOG, indicating reduced capacity for compensation. The present findings propose that auditory cues can partially normalize motor abnormalities during upper-limb movement in those with FOG while they are in the off phase of the medication cycle. This is in line with recent evidence of improved gait parameters and turning behavior in those with FOG using rhythmic cueing.52-54 Few studies focused on cueing of movements in the upper extremities in PD, yielding inconsistent results. In contrast to earlier work,24,55 Ringenbach et al 56 found that timing, scaling, and coordination during a drawing task improved with auditory pacing, but the benefits of visual cueing were less evident. However, augmented visual feedback enhanced upper-limb motor learning in PD.57,58 The effect of cueing on bimanual movements in subgroups of those with FOG and those without FOG was recently addressed for the first time. 25 Visual target lines aided both groups in achieving a more stable and accurate drawing performance but increased the RP variability in those with FOG. In our study, movement parameters, including coordination stability, improved in those with FOG when auditory cueing was present but dropped dramatically after cue withdrawal.
These results strongly suggest reduced retention of external cueing following cue withdrawal in those with FOG. A previous study demonstrated short-term carryover effects of sensory cueing during walking in those with FOG when optimally medicated. 52 However, congruent with the results of Willems et al, 14 our results showed the opposite. This implies that those with FOG may suffer from a specific deficit in automating cued responses. Cued conditions were short and nonrandomized, which is a drawback of the study. As an order effect cannot be ruled out, the findings need replication by future studies.
The neural processes underlying internal continuation of bimanual movements after removal of an auditory cue have been investigated recently. 59 This functional MRI study showed no neurofunctional differences between the 2 timing phases in PD and matched CTRLs. However, in both the synchronization and continuation phases, compensatory activity increased in the cerebellothalamic pathway in PD patients, associated with motor performance similar to that of the CTRL group. It is therefore possible that this compensatory neural reserve is insufficient or not sustained in those with FOG, resulting in a continuous need for external information to drive the motor network. Motor learning strategies aimed at internalizing external information, for example using mental practice, may be especially relevant in those with FOG. Motor imagery was recently shown to be feasible in patients with PD, in general, 60 and in those with FOG, in particular.47,61 Moreover, the mutual influence of mental practice and external cues 62 may form a promising new direction in the development of rehabilitation strategies for repetitive upper-limb tasks in those with FOG. The feasibility of this motor learning approach should be examined.
Conclusions
Our findings of exaggerated dyscontrol of repetitive upper-limb movements in those with FOG corroborate results of previous studies on FOG-related abnormalities during walking. We showed for the first time that those with FOG may benefit from external sensory cueing to normalize scaling, timing, and coordination difficulties in an upper-limb repetitive task, but they show increased dependence on the presence of the cue. These findings can be taken into account for rehabilitation interventions for tasks such as writing, stirring, wiping, and tapping in patients with freezing episodes.
Footnotes
Acknowledgements
We thank all participants.
Declaration of Conflicting Interests
Wim Vandenberghe is a senior clinical investigator of the Research Foundation Flanders (FWO).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support was provided by the Research Council of Katholieke Universiteit Leuven, Belgium (Contract OT/07/074) and the Flanders Fund for Scientific Research (Project G0691.08).