Abstract
Background. Patients in a vegetative state pose problems in diagnosis, prognosis, and treatment. Currently, no prognostic markers predict the chance of recovery, which has serious consequences, especially in end-of-life decision making. Objective. We aimed to assess an objective measurement of prognosis using advanced electroencephalography (EEG). Methods. EEG data (19 channels) were collected in 14 patients who were diagnosed to be persistently vegetative based on repeated clinical evaluations at 3 months following brain damage. EEG structure parameters (amplitude, duration, and variability within quasi-stationary segments, as well as the spatial synchrony between such segments and the strength of this synchrony) were used to predict recovery of consciousness 3 months later. Results. The number and strength of cortical functional connections between EEG segments were higher in patients who recovered consciousness (P < .05 to P < .001) compared with those who did not recover. Linear regression analysis confirms that EEG structure parameters are capable of predicting (P = .0025) recovery of consciousness 6 months postinjury, whereas the same analysis failed to significantly predict patient outcome based on aspects of their clinical history alone (P = .629) or conventional EEG spectrum power (P = .473). Conclusions. The result of this preliminary study demonstrates that structural strategy of EEG analysis is better suited for providing prognosis of consciousness recovery than existing methods of clinical assessment and of conventional EEG. Our results may be a starting point for developing reliable prognosticators in patients who are in a vegetative state, with the potential to improve their day-to-day management, quality of life, and access to early interventions.
Keywords
Introduction
The vegetative state (VS) or unresponsive wakefulness syndrome (UWS) is a complex neurological condition of “unawareness of self and environment in which the patient breathes spontaneously, has a stable circulation, and shows cycles of eye closure and opening which may simulate sleep and waking.” 1 Thus, VS/UWS is commonly agreed to be a state of “wakeful unconsciousness,” 2 whereas the type of consciousness researchers have in mind is phenomenal consciousness—the sort of awareness that there is something to enjoy, from the subject’s point of view. 3 According to the Royal College of Physicians, 1 a VS/UWS is classified as persistent when it lasts longer than a month with recovery rates approaching zero after 12 months for patients older than 40 years. 4 Nontraumatic brain injuries are considered to have a poorer prognosis. 5
Once VS/UWS is diagnosed, the chance of recovery is considered to be low-to-moderate and almost always involves some level of disability 2 ; sometimes VS/UWS lasts the whole life. 5 In any case, giving a precise estimate of the likelihood of further recovery remains difficult.
The first stage of recovery is characterised by a minimally conscious state (MCS), which is
a condition of severely altered consciousness in which minimal but definite behavioural evidence of self or environmental awareness is demonstrated. In MCS, cognitively mediated behaviour occurs inconsistently, but is reproducible or sustained long enough to be differentiated from reflexive behaviour.
6
Like VS/UWS, the MCS may be transitory and precede further recovery of consciousness or last the whole life. 2
An accurate and reliable judgment of VS/UWS patients’ awareness is of paramount importance for their diagnosis and prognosis. 7 Despite an increasing number of published scientific research in recent years, at present, we still do not have objective and validated prognostic markers that allow clinicians to predict the chances of recovery in VS/UWS patients. 8 Wrong estimation of the chances of recovery can lead to serious consequences, especially when end-of-life decision-making is concerned. 9 Therefore, objective measurement tools through which an individual’s level of retained awareness can be assessed (without explicit reports) are needed to achieve more accurate estimates of prognosis.
Potential Role of Electroencephalogram
An electroencephalogram (EEG), which permits bedside assessment, could be particularly helpful since, in contrast to functional magnetic resonance imaging, it is routinely available in most clinics/laboratories, is affordable, and allows direct and objective recording of spontaneous brain activity without the need of any behavioral response from the patient. 10 More specifically, EEG measures a highly organized macrolevel electrophysiological phenomena in the brain, which capture the operations of large-scale cortical networks (neuronal assemblies) and which are remarkably correlated with behavior, cognition,11-15 and consciousness.16,17
Despite compelling evidence demonstrating the usefulness of standard resting-state EEG in predicting recovery from coma,18,19 there are only scarce reports that show some promise of predicting VS recovery20-22 or overall survival. 23 Furthermore, the predictive value of individual resting-state EEG classifications has not been adequately addressed. 24 As a result, conventional resting-state EEG (based on spectral analysis) is typically used only for gross and qualitative analysis and is not practical for long-term patient monitoring nor as a sophisticated prognostic tool. 25
An alternative strategy to examining, EEG is offered by microstructural analysis of the signal.14,15,26 Using this strategy, recent studies have established that each EEG signal (channel) within the multichannel EEG recording could be represented as a sequence of quasi-stationary (nearly stable) segments. Throughout the duration of each segment, the neuronal assembly that generates the oscillation is supposed to be in steady, quasi-stationary state. The transition of one EEG segment to another reflects change in neuronal assembly microstate.14 -16 Temporal synchronization of quasi-stationary EEG segments among different EEG channels could thus represent synchronized activity of different local and transient neuronal assemblies that play an important role in cognition and consciousness.16,17 This strategy of EEG analysis has been validated in a number of electrophysiological, cognitive, and clinical studies and is proven to be robust, consistent and statistically reliable.14,27,28
In applying microstructural strategy of resting-state EEG analysis to VS/UWS and MCS patients, it has been suggested that the incapacity of VS/UWS patients to generate consciousness is most likely linked to disruptions in local and large-scale EEG structures.17,29 In particular, it has been found that the absence of consciousness in VS/UWS patients is paralleled by impairment in (a) EEG segmental characteristics (small amplitude, short duration and high amplitude variability) and (b) temporal synchronicity among EEG segments obtained from different EEG channels (decreased number and strength of functional connections). 17 At the same time, fluctuating (minimal) awareness in MCS patients was paralleled by partial restoration of EEG segmental characteristics (increased amplitude and duration as well as decreased amplitude variability) and their temporal synchrony parameters (increased number and strength of functional connections), approaching those found in healthy fully conscious subjects. 17 These findings are summarized schematically in Figure 1.
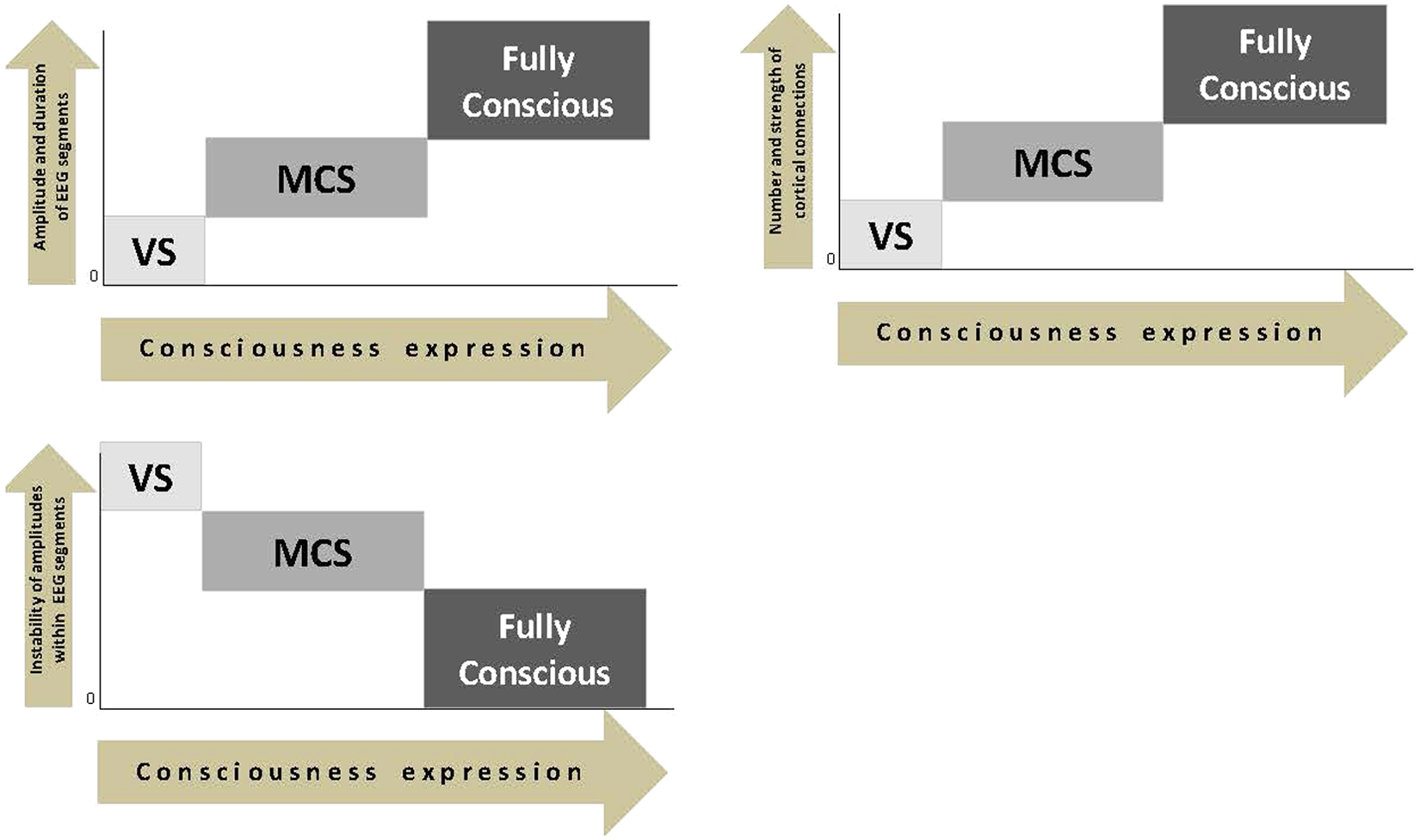
Schematic presentation of relations between consciousness expression and EEG-based segmental characteristics and functional connectivity (this scheme is based on data published in Fingelkurts et al 17 ). Abbreviations: EEG, electroencephalography; VS, vegetative state; MCS, minimally conscious state.
Aim of the Study
Here, we investigated whether the aforementioned characteristics of resting-state EEG segments (amplitude, duration, and variability), as well as the spatial synchrony of EEG segments and the strength of such synchrony could predict the recovery of conscious awareness after 6 months postinjury in a sample of fourteen patients who were diagnosed to be persistently vegetative on the basis of repeated and thorough clinical evaluations within 3 months following brain damage.
Methods
Patient Cohort
We recruited patients with traumatic and nontraumatic brain injury admitted to the neurorehabilitation unit at the Fondazione Istituto “San Raffaele–G. Giglio” who met the currently accepted international definition of persistent VS/UWS.1,5,30 Additionally, the Levels of Cognitive Functioning Scale (LCFS) score 31 was assessed on the day of admission (about 3 months postinjury) and 3 days later when an EEG was registered, to estimate the stable expression of clinical consciousness.21,22 The LCFS has a linearly graded scale ranging from 1 to 8 (1 = patient is unconscious; 8 = patient is self-oriented and conscious of the environment) and is well correlated with resting-state EEG abnormalities in patients with brain damage.21,22 At the time of EEG scanning, all patients had a LCFS score of 1 or 2 (1.6 ± 0.5).
Inclusion criteria for the patients included (a) confirmation of diagnosis of VS/UWS according to the diagnostic criteria,1,5,30 (b) within 3 months after acute brain event onset, (c) first-ever acute brain event, and (d) stable LCFS score during 3 days. Exclusion criteria comprised (a) any acute comorbidity or unstable vital signs, (b) obvious communicating or obstructive hydrocephalus, (c) a history of neurological disease before admission, and (d) severe spasticity (causing constant electromyography artifacts). Demographic information is summarized in the Table 1.
Basic Demographic and Clinical Characteristics of Patients
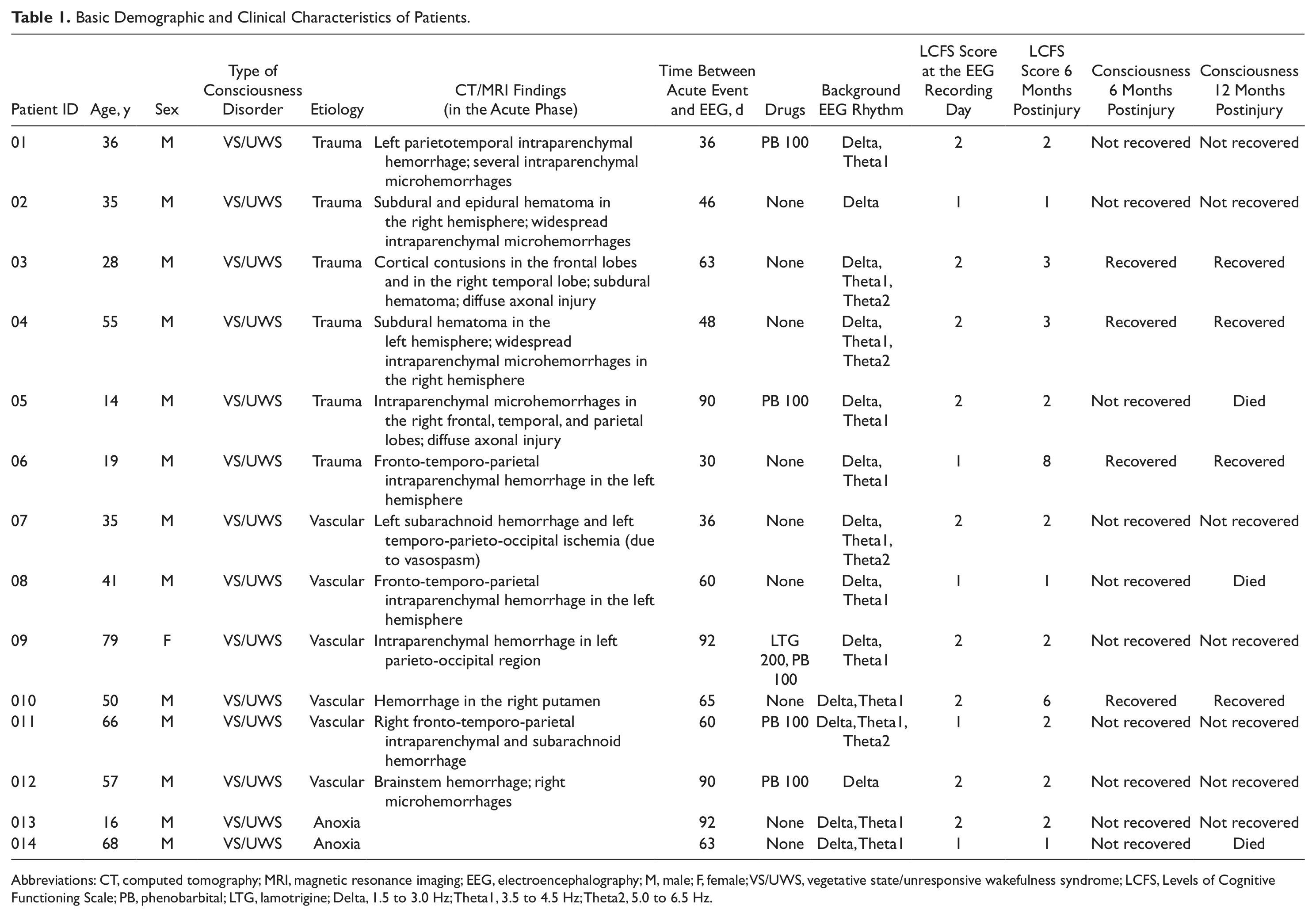
Abbreviations: CT, computed tomography; MRI, magnetic resonance imaging; EEG, electroencephalography; M, male; F, female; VS/UWS, vegetative state/unresponsive wakefulness syndrome; LCFS, Levels of Cognitive Functioning Scale; PB, phenobarbital; LTG, lamotrigine; Delta, 1.5 to 3.0 Hz; Theta1, 3.5 to 4.5 Hz; Theta2, 5.0 to 6.5 Hz.
Based on clinical evaluations at 3-month follow-up after the EEG registration (thus 6 months postinjury), patients were retrospectively divided into 2 groups: 10 unrecovered—continued to be vegetative (VS-Pers patients; age, 44.7 ± 22 years; follow-up LCFS score = 1.7 ± 0.7) and 4 recovered—classified as minimally conscious (VS-MCS patients; age, 38 ± 17.3 years; follow-up LCFS score = 5 ± 2.4). Patients with severe brain damage have recovery chances within a year after injury,2,4 and therefore some patients in the unrecovered group at 6 months postinjury might in fact recover some level of consciousness 1 year postinjury; as it is evident from the Table 1, none of the VS-Pers patients recovered consciousness after 1 year, and 3 patients died. Hence, we used for our analysis the 6-month postinjury assessment as a cutoff point in time for prediction of recovery of consciousness.
The study was approved by the local institutional ethics committee and complies with good medical practice. Informed and overt consent of patients’ legal representatives, in line with the Code of Ethics of the World Medical Association (Declaration of Helsinki) and standards established by the Fondazione Istituto “San Raffaele–G. Giglio” Review Board were acquired. Data use was authorized by means of written informed consent of the VS/UWS patients’ caregivers.
EEG Registration
Waking resting EEG was recorded (0.5-70 Hz bandpass; 200 Hz sampling rate; ~30 minutes) with a Neuropack (Nihon Kohden, Tokyo, Japan) from 19 electrodes positioned according to the International 10-20 system. The impedance was below 5 kOhm. An electro-oculogram (0.5-70 Hz bandpass) was also collected.
EEG recordings were started if patients had their eyes open spontaneously, the eyelids were then closed by hand and kept closed until the end of registration. At the end of the recordings all patients opened their eyes spontaneously, suggesting an unchanged vigilance level throughout EEG registration. The presence of an adequate EEG signal was determined by visual inspection of the raw signal.
Prior to analysis, each EEG signal was bandpass-filtered (Butterworth filter of the sixth order) in the alpha (7-13 Hz), beta-1 (15-25 Hz), and beta-2 (25-30 Hz) frequency bands. Phase shifts were eliminated by forward and backward filtering. The mentioned frequency bands were chosen based on our previous study 17 ; only these frequency oscillations have shown dynamics consistent with the analytical consciousness model. 29
EEG Segmentation
In short, the adaptive segmentation algorithm can be described in 2 main stages (for details, see Fingelkurts and Fingelkurts 28 and Figure 2): (a) preliminary identification of the boundaries using automated algorithm that moves a double window screening along each separate EEG channel and (b) selection of actual (real) boundaries based on the steepness of previously detected EEG amplitude changes and Student criteria. Three EEG segment attributes were further estimated—(a) average amplitude within each segment (microvolts), (b) average length of segments (milliseconds), and (c) coefficient of amplitude variability within segments (%). These attributes inform about different features of neuronal assemblies: size, life span, and stability.11,15,28
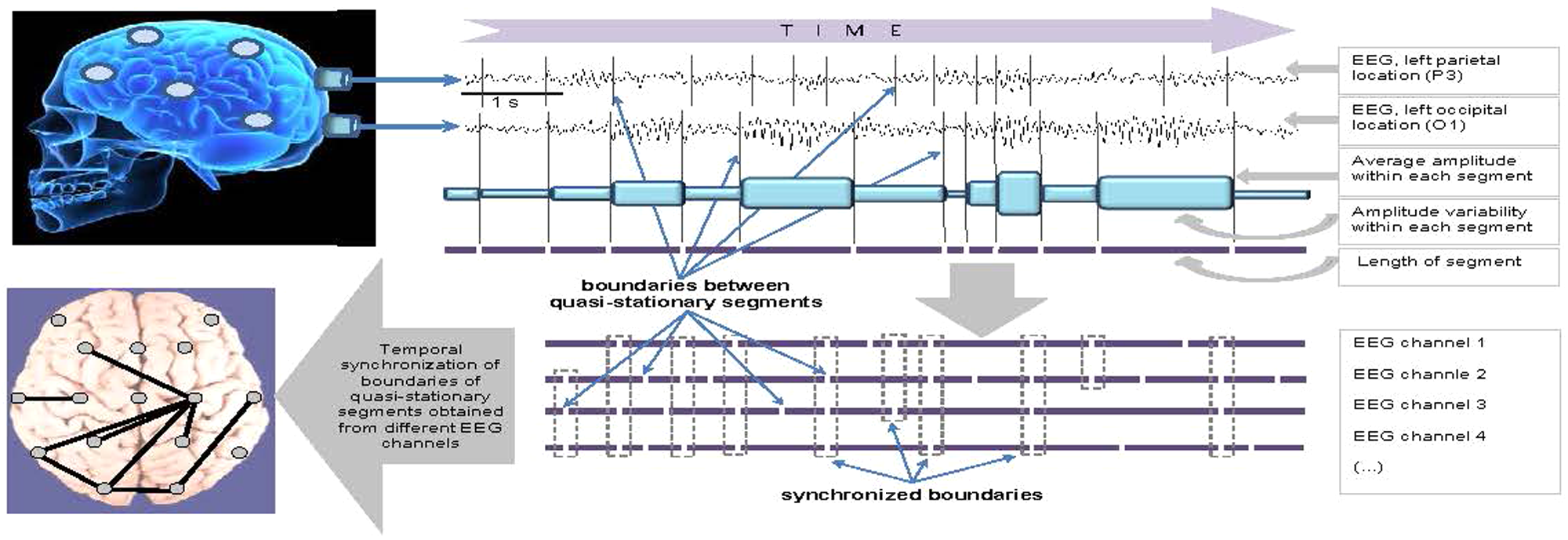
Schematic presentation of microstructural strategy of EEG (electroencephalogram) analysis (see text for explanation).
Synchronization of EEG Segments Among EEG Channels
Index of Structural Synchrony (ISS; for details see Fingelkurts and Fingelkurts 28 ) estimates synchronization of EEG quasi-stationary segments obtained from different brain locations. In brief, each boundary in one EEG channel (from any pair of EEG channels) was surrounded by a short (milliseconds) “window.” Any boundary from another channel was considered to coincide if it fell within this window (Figure 2). To arrive at a direct estimate (5% level) of statistical significance (P < .05) of the ISS, computer simulation of boundaries coupling was undertaken based on random shuffling of segments (500 independent trials) for each pair of EEG channels. 28 As a result of this procedure, the stochastic levels of RTP coupling (ISSstoch), together with the upper and lower thresholds of ISSstoch significance (5%) were calculated. Where there is no synchronization, the ISS tends toward zero, whereas positive (higher than upper stochastic level) or negative (lower than low stochastic level) values are indicative of synchronization (coupling of EEG segments is observed significantly more often than expected by chance as a result of random shuffling during a computer simulation) or desynchronization (coupling of EEG segments is observed significantly less than expected by chance as a result of random shuffling during a computer simulation), respectively.15,28 The strength of EEG structural synchrony is proportional to the actual value of ISS in each pair of EEG channels: The higher this value, the greater the strength of functional connection. The number of connections corresponds to a number of pairs of EEG channels with a statistically valid ISS.
Conventional Power Spectral Analysis
For conventional power spectrum estimates artifact-free EEG signals were filtered in the 1- to 30-Hz frequency range. Individual power spectra were calculated in the range of 1 to 30 Hz with 0.5-Hz resolution, using a fast Fourier transform with a 2-second Hanning window shifted by 50 data samples (0.39 seconds) for each channel of 1-minute EEG. After calculation of EEG short-term power spectra they were averaged within each EEG channel, then across all 1-minute EEGs for each patient. Spectral power was integrated within the following frequency bands: delta (1.5-3.0 Hz), theta (3.5-6.5 Hz), alpha (7-13 Hz), beta-1 (15-25 Hz), and beta-2 (25.5-30.0 Hz).
Results
Demographic and Clinical Information
Six of the patients had sustained a traumatic brain injury (TBI), whereas the remaining 8 had sustained a nontraumatic brain injury (non-TBI). There were no significant differences between the 2 groups (VS-Pers—unrecovered and VS-MCS—recovered) at the time of EEG registration in terms of length of time since brain injury (Student t test, P = .262), patients’ age (Student t test, P = .599), LCFS score (Student t test, P = .733), or hemispheric localization of brain damage (χ2 test, P = 1.000). However, 50% of patients from the VS-Pers group were administered medication, whereas no one from the VS-MCS group was medicated; this difference was statistically significant (χ2 test, P = .0000001).
All patients in both groups underwent the same rehabilitation treatment during hospitalization, consisting of daily verticalization in the standing position (30 minutes) and kinesitherapy (2 hours), as well as regular bed mobilization and chair transfers.
Prognostic Value of EEG Segments’ Attributes
Consistent with expectations based on our previous study, 17 we found that average amplitude within EEG quasi-stationary segments and the length of these segments were larger, whereas the amplitude variability within EEG segments was lower in VS/UWS patients who recovered some level of consciousness 3 months later (thus 6 months postinjury), when compared with unrecovered VS/UWS patients (Figure 3). This observation was similar for all 3 frequency bands (alpha, beta-1, and beta-2).
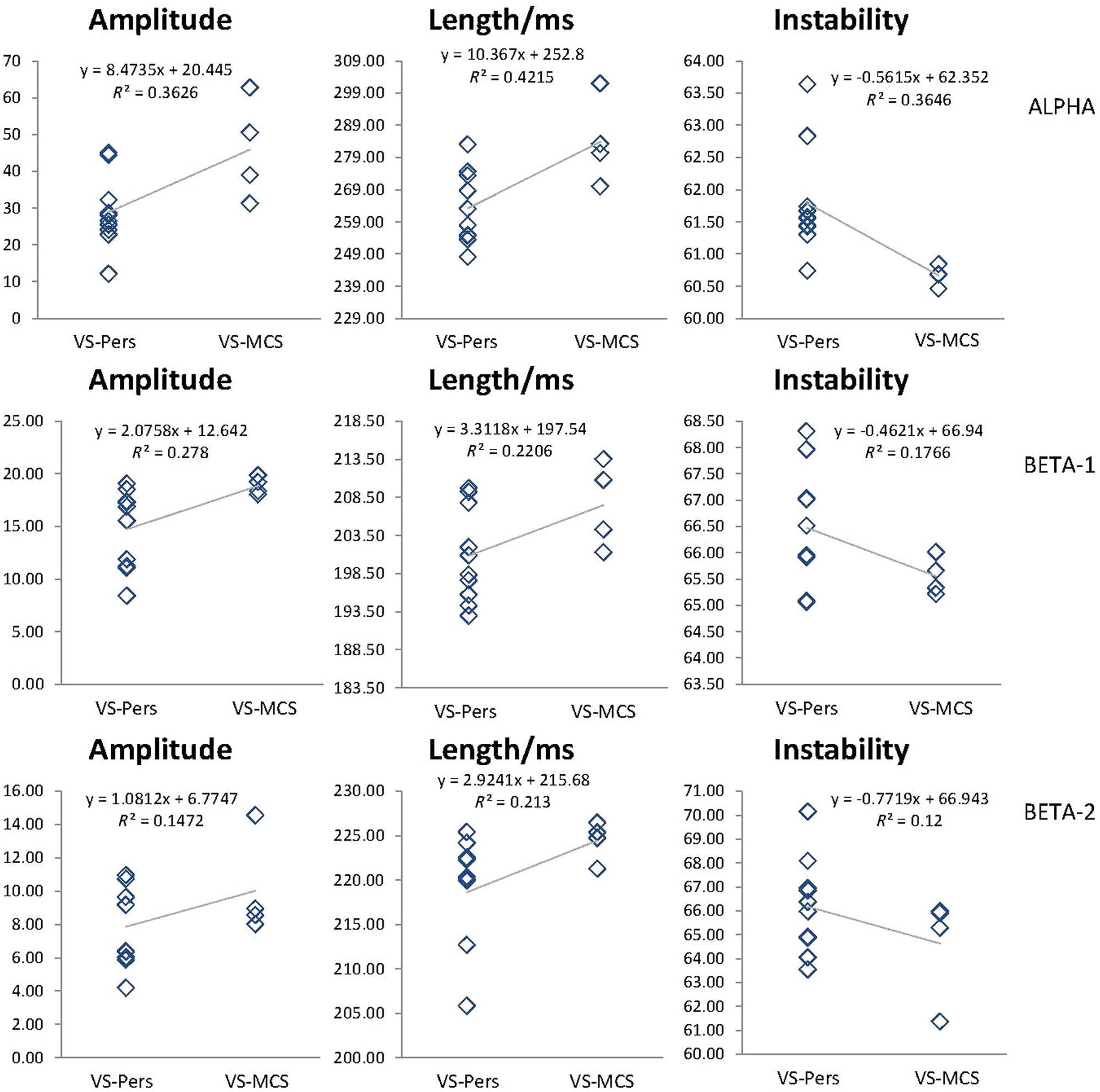
EEG segment attributes. Data averaged across all 1-minute EEG epochs and all EEG channels for each subject within each group: unrecovered (VS-Pers; n = 10) and recovered (VS-MCS; n = 4). The mean values of segment attributes indicated by the Y-axis: amplitude within each segment (microvolt); length of segments (milliseconds); coefficient of amplitude variability within segments (%). Lines in the graphs represent the tendency, which is presented by the linear equation and R2. Alpha—EEG rhythm within 7 and 13 Hz, Beta-1—EEG rhythm within 15 and 25 Hz, Beta-2—EEG rhythm within 25 and 30 Hz. EEG, electroencephalography; VS-Pers, persitent vegetative state; MCS, minimally conscious state.
To determine whether the 2 groups of patients differed significantly, group-EEG segment-attribute averages and respective standard deviations were calculated for the whole pull of correspondent 1-minute EEGs. A comparison of the same segment attributes between VS-Pers and VS-MCS groups was performed using Wilcoxon t test. We found no evidence of statistically significant differences between the 2 groups of patients (Alphaamplitude, P = .297; Alphalength, P = .364; Alphainstability, P = .179; Beta-1amplitude, P = .811; Beta-1length, P = .189; Beta-1instability, P = .33; Beta-2amplitude, P = .909; Beta-2length, P = .959; Beta-2instability, P = .388), though such differences have consistent pattern for all studied frequency bands (Figure 3). Therefore, these results suggest that the studied parameters of EEG segments may indicate a potential predictive trend and do not carry a predictive value.
Prognostic Value of Functional Connections and Their Strength
The number and strength of EEG segmental synchrony were assessed using an ISS index (see Methods section and Fingelkurts and Fingelkurts 28 for details). The differences in number and strength of ISS patterns between the 2 groups (VS-Pers and VS-MCS) were assessed using Wilcoxon rank t test. At first, all statistically valid EEG functional connections were averaged for each group and the whole pull of correspondent 1-minute EEGs within 9 categories of functional connectivity (shortleft/right, shortanterior/posterior, longleft/right, longanterior/posterior, and longinterhemispheric). The same procedure was done separately for the number and strength of these functional connections. Since the absolute number of possible functional connections within each category was different, their per-category percentage was calculated. During the final stage, an average of all 9 categories was calculated. Thus, average values of functional connections for the whole cortex were used for further analysis.
Statistically higher values for the number and strength of functional connections were found during the first assessment of VS/UWS patients (3 months postinjury) who showed good outcome (recovered) at 6 months postinjury, in comparison with unrecovered VS/UWS patients (Figure 4, Alphanumber, P = .01; Alphastrength, P = .028; Beta-1number, P = .007; Beta-1strength, P = .038; Beta-2number, P = .038; Beta-2strength, P = .021). Similar differences were observed in all 3 (alpha, beta-1, and beta-2) frequency bands (Figure 4). These results suggest that the number and strength of cortical functional connections recorded at third month following brain damage provide potentially useful information on the outcome of persistent VS/UWS patients 3 months later (thus 6 months postinjury).
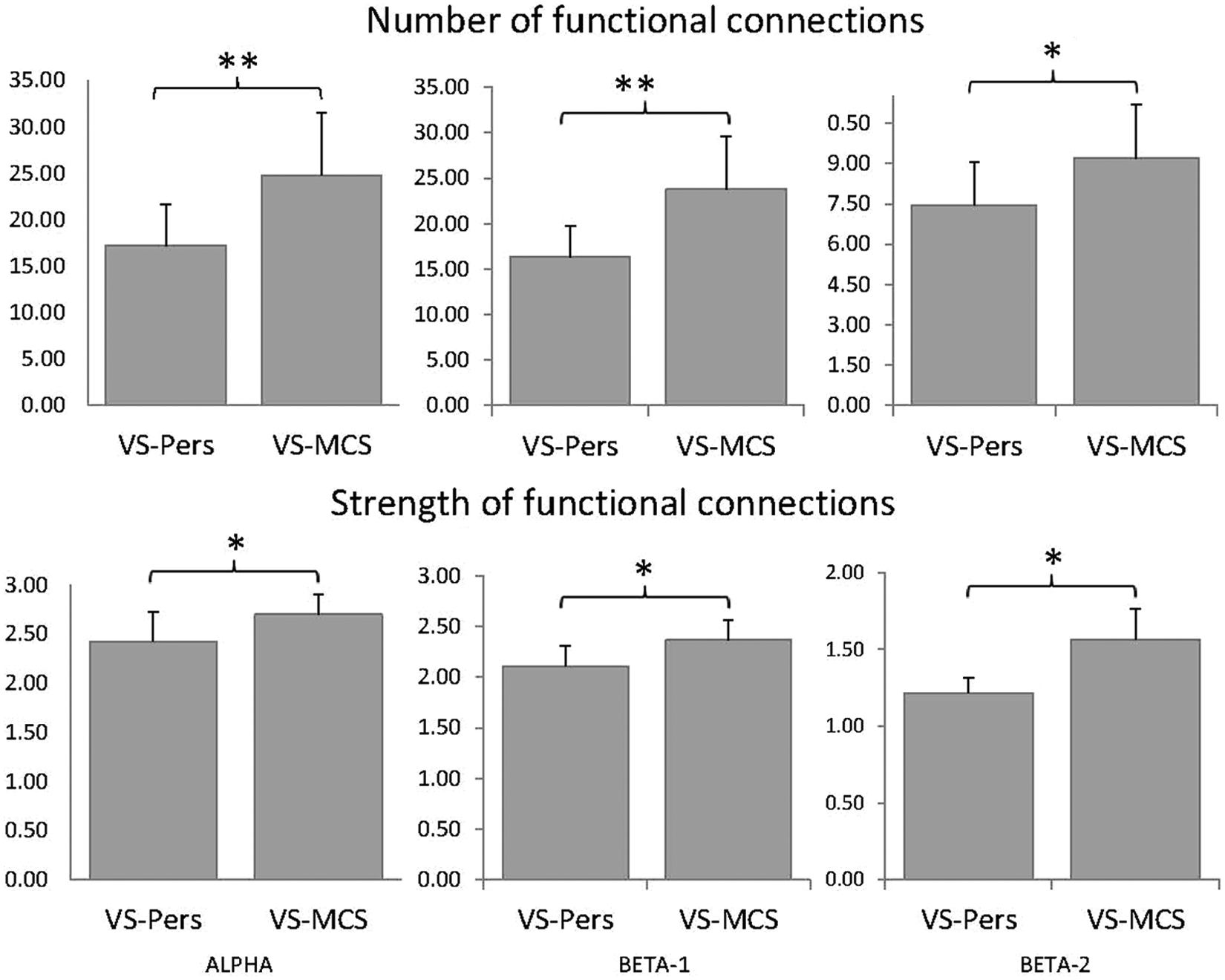
EEG structural synchrony. Data averaged across 9 connectivity categories for all pairs of EEG channels within each category and all subjects within each group. The Y-axis presents mean values of either number or strength of functional connections (n = 9 categories). Alpha—EEG rhythm within 7 and 13 Hz; Beta-1—EEG rhythm within 15 and 25 Hz; Beta-2—EEG rhythm within 25 and 30 Hz. Bars represent means ± SEM. *P < .05 and **P < .01. EEG, electroencephalography; VS-Pers, persitent vegetative state; MCS, minimally conscious state.
To determine whether an increase in the number and strength of cortical functional connections has a true predictive value of patients’ recovery, compared with clinical history parameters, we used a stepwise linear regression analysis. For large-scale parameters of resting-state EEG structure, 2 factors were used for each frequency band (alpha, beta-1, and beta-2): (a) number of functional connections and (b) strength of functional connections. For the clinical history parameters, the following 4 factors were used: (a) age at time of injury (years), (b) time since injury (days), (c) LCFS score, and (d) hemispheric localization of brain damage (left/right). For conventional EEG spectrum power analysis, the following 5 factors were used: (a) delta frequency band, (b) theta frequency band, (c) alpha frequency band, (d) beta-1 frequency band, and (e) beta-2 frequency band.
Linear regression analysis confirms that large-scale parameters of resting-state EEG structure (number and strength of functional connections) could successfully discriminate between recovered and unrecovered VS/UWS patient groups (F = 12.61, P = .0025), whereas the same analysis failed to significantly predict VS/UWS patient outcome based on aspects of their clinical history alone (F = 0.245, P = .629) or conventional EEG spectrum power (F = 0.754, P = .473). These results indicate that in contrast to the parameters of clinical history and conventional EEG analysis, the large-scale parameters of resting-state EEG structure measured 3 months postinjury can predict the recovery outcome of persistent VS/UWS patients 6 months postinjury. In other words, the higher the number and the strength of cortical functional connections in persistent VS/UWS patients, the higher their chance to recover some level of consciousness in the future.
However, the distribution of patients who had sustained a TBI was not equal in the VS-Pers (30%, n = 3/10) and VS-MCS (75%, n = 3/4) groups (χ2 test, P = .000001). This difference may have influenced the results since it is considered that patients with TBI have better prognosis. 5 To check whether the larger ratio of TBI patients in the VS-MCS group was influencing the main result of this study, we repeated the EEG segmental synchrony analysis for TBI patients only (n = 3 for VS-Pers and n = 3 for VS-MCS). This analysis resulted in the same significant differences between VS-Pers and VS-MCS groups for all studied frequency bands (Alphanumber, P = .008; Alphastrength, P = .045; Beta-1number, P = .045; Beta-1strength, P = .045; Beta-2number, P = .045; Beta-2strength, P = .045) as in the full sample groups. Therefore, we could conclude that the increased ratio of TBI patients in VS-MCS group was not affecting our results.
There was another potential confounding factor: 50% of patients from the unrecovered group (VS-Pers) were treated with phenobarbital (PB) and lamotrigine (LTG), whereas none of the patients from the recovered group (VS-MCS) were administered medications. These treatments may have influenced the EEG structure, giving rise to the observed differences between VS-Pers and VS-MCS groups. However, both drugs either have no effect or may cause increased coupling among EEG channels.32,33 In the present study, increased EEG synchrony was observed in the nonmedicated group, therefore it is unlikely that the PB and LTG treatments affected the result of the study; in fact they may have diminished the discovered statistically significant difference between the groups (such as in the analysis of EEG segmental characteristics).
Discussion
Prognostic accuracy for patients in persistent VS/UWS poses serious medical and ethical concerns because treatment decisions typically include the possibility of life-support being withdrawn.34,35 Currently, prognosis of the outcome is determined primarily through diagnosis (VS/UWS or MCS) and also by etiology of brain injury (traumatic, vascular, or anoxic) as well as the age of the patient. 2 However, misdiagnoses of VS/UWS are very common and have been shown to be as high as 37% to 45% if not using an appropriate behavioral scale.36-39
Here we have demonstrated that certain parameters of large-scale resting-state EEG structure could predict future recovery of some level of consciousness in persistent VS/UWS patients. Indeed, 29% (4/14) of patients who appeared to be persistently vegetative on the basis of repeated clinical assessment at the time of admission to the rehabilitation unit, but recovered consciousness 3 months later (thus 6 months postinjury), had a similar number and strength of cortical functional connections to those found in MCS patients from our previous study. 17 At the same time, 71% (10/14) of persistent VS/UWS patients who did not recover demonstrated an impaired large-scale structure of EEG as predicted based on our previous observations. 17 These results emphasize the modern understanding that intact communication between brain areas, namely, the coherent dynamic binding of operations performed by multiple neuronal assemblies, which are organized within a nested hierarchical brain architecture, is a basic requirement for consciousness.15-17,40,41 Furthermore, our results extend several lines of evidence on the strong implication of cortical alpha and beta rhythms on human higher functions and consciousness.17,29
We cannot draw any certain conclusions about the inner subjective experiences in this cohort of patients at the time of admission to the rehabilitation unit based solely on EEG structure analysis. However, our results may indicate that at least 4 VS/UWS patients had a resting-state EEG architecture compatible with partial preservation of awareness 17 despite fulfilling the clinical criteria for a diagnosis of persistent vegetative state.1,5,30 To determine whether these patients may be exhibiting partially preserved conscious processing, though not expressed behaviorally, more research is needed, and this is an objective for future studies. Currently, we can only state that prognosis for future recovery of consciousness in persistent VS/UWS patients can be determined accurately on the basis of large-scale resting-state EEG structure analysis alone at the time when patients meet all clinical criteria for the persistent VS1,5,30 (they show no signs of awareness). To fully appreciate the value of this result, it is necessary to consider that a clear-cut difference in resting-state EEG segment synchrony between the 2 groups (recovered and unrecovered) occurred at the early stage when reliable communication with patients could not yet be established and before spontaneous EEG showed significant modifications in the conventional (based on spectral analysis) parameters (see Table 1). Thus, the result of this study demonstrates that microstructural strategy of resting-state EEG analysis is better suited for providing prognosis of consciousness recovery than existing methods of clinical assessment and of conventional EEG.
Even though the findings of this preliminary study are quite promising, the limitation is that data for the analysis are based on 14 patients only with varied medical conditions, which is typical for this kind of population. Therefore one should take with caution the above-stated conclusion, and to confirm the results presented in this article, future studies that include a larger group of patients are warranted. Additionally, the results of this study are restricted by the fact that predictive value of the structural EEG measures were not compared with measures other than conventional (based on spectral analysis) measures, for example, event-related EEG measures, and that such a comparison should be an object of future studies.
Conclusion
As EEG (a) is inexpensive, portable, and available in most hospitals; (b) can be performed at the bedside; and (c) can be used in patients who have metal implants, the results of this preliminary work could have a direct clinical significance following further validation: They may establish a starting point in the development of reliable early markers for VS/UWS patient prognosis. This could contribute to improving day-to-day management, access to early interventions, and quality of life in such patients.
Footnotes
Acknowledgements
The authors thank C. Prestandrea (neurophysiology technician) for EEG recordings; C. Neves (computer science specialist) for programming, technical, and IT support; and D. Skarin for English editing.
Authors’ Note
The views expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the BM-Science Centre or the Fondazione Istituto “San Raffaele–G. Giglio”.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
