Abstract
Keywords
Introduction
Clinical practice and research have shown how difficult it is to establish the diagnosis and prognosis of disorders of consciousness (DOC). Although several authors have emphasized the informative value of evoked potentials in the awake, there is evidence that sleep evaluation represents one of the most promising tools to assess residual brain function in order to improve DOC diagnosis.1-5
A number of genetic factors has been shown to be associated with different sleep traits and homeostasis, such as diurnal preference or electroencephalogram (EEG) characteristics.6,7 In particular, the coding region of human Period3 gene (Per3) contains a variable-number tandem repeat (VNTR) polymorphism (insertion of nucleotides 3031–3084, exon 18), a 54-nucleotide coding motif repeated in 4 (Per34/4) or 5 (Per35/5) units. The 4-repeats allele is the most common VNTR variant, whereas approximately 10% of the population is homozygous for the 5-repeats allele. 8 Moreover, the allelic frequency in African Americans and Caucasian/European Americans is similar, conversely to that reported between Asian and Caucasian ethnic groups. 9 Per3 gene, which is involved in the transcription of the molecular oscillators CLOCK and BMAL1, has been recognized to have a role in circadian rhythmicity generation. 10 It has also been associated with the response to total sleep deprivation in physiological conditions11,12 in patients with sleep6,13 and other neurological disorders. 14 However, the role of this molecular variant in DOC patients has never been investigated.
Assuming that particular genotypes may affect the performance and residual functions of DOC patients, the aim of this study was to explore the role of Per3 VNTR polymorphism in conditioning sleep patterns and to understand/analyze behavioral responses in a population of DOC patients in the Coma Research Centre (CRC) of the Neurological Institute “C. Besta” of Milan.15-17
The identification of an association between specific genetic factors and DOC patient functional recovery could be helpful in improving the knowledge of the molecular mechanism underlying DOC recovery and in better defining the diagnosis and prognosis of these patients. It could ultimately lead to the implementation of personalized rehabilitation and management.
Methods
Patients
This was an observational study conducted on adult DOC patients admitted to the CRC coordinated by the IRCCS Neurological Institute “C. Besta” of Milan. Its aim was a careful clinical, neurophysiological, and neuroradiological assessment of DOC patients.
The DOC patients were hosted at home or in 28 specialized, postacute rehabilitation or long-term care centers of the Lombardy Region, Italy. 17 The diagnosis of vegetative state/unresponsive wakefulness syndrome (VS/UWS) or minimally conscious state (MCS), according to the definition of the Aspen Neurobehavioral Conference Workgroup,18,19 was the mandatory criterion for the study.
Standard Protocol Approval, Registration, and Patient Consent
Informed written consent for study participation and DNA samples collection from all patients’ legal representatives/next-of-kin was mandatory for study inclusion. The study design was approved by the Ethics Committee of the IRCCS Neurological Institute “C. Besta” of Milan (report no. 10, December 15, 2010) and of each participating center. The study was performed in accordance with the World Medical Association Declaration of Helsinki. Since it was a purely observational study, patients underwent diagnostic procedures and received therapy according to local practices.
Privacy procedures were applied to protect patients’ personal identities.
Protocol and Definition of Outcome
Patients were hospitalized for 7 days at the IRCCS Neurological Institute “C. Besta”. During this period, a standard research protocol, administered by trained neuropsychologists and neurologists, was applied to all patients. It included collection of patients’ demographic (etiology, age at injury, gender, and acute Glasgow Coma Scale [GCS] score) and clinical data, information regarding acute events, cardiovascular risk factors (such as obesity, smoking, arrhythmias, and/or cardiomyopathy), and pharmacological treatments, from proxy and/or derived from clinical records.
The Coma Recovery Scale–revised (CRS-r) 20 was used as an outcome measure. A detailed methodological description for CRS-r is reported elsewhere. 17
A standardized instrumental examination, including EEG, evoked potentials (VEPs, SEPs, BAEPs and P300) and polysonnography, was also applied to assess cerebral residual functions and to confirm and/or revise the clinical diagnosis of VS/UWS or MCS.
The severity of coma at acute phase was derived from past clinical records (GCS
21
). The presence of
devices, infections, and decubitus ulcers was also registered. On the basis of aetiology,
patients were classified as “
VNTR Polymorphism Genotyping
Genomic DNA was obtained from patients’ blood using QIAamp DNA Mini Kit (QIAGEN, Hilden, Germany). Genotyping was performed by polymerase chain reaction (PCR) using Per3 specific primers as previously described. 14 The PCR products were analyzed by electrophoresis in order to separate 5 repeats allele (401 bp) from 4 repeats allele (347 bp).
Hardy-Weinberg Equation
The Per3 genotype allelic frequency of our DOC population was calculated by the
Hardy-Weinberg equation, to assess whether the genetic variation in a population remains
constant from one generation to the next in absence of disturbing factors. The
Hardy-Weinberg equation is expressed as:
Polysomnography
All DOC patients underwent a polysomnographic sleep evaluation (PSG), which started at
2:00
Polysomnography included 19 EEG channels with electrode array placed according to the 10-20 international system, 2 electro-oculogram channels, 1 electromyogram channel recorded from mylohyoid muscle, bipolar precordial electrocardiogram, and impedance thoracic pneumogram. PSG was recorded by means of Ag/AgCl surface electrodes (impedance <5 kohm) and acquired at a sampling rate of 256 Hz (Micromed SpA, Mogliano Veneto, Treviso, Italy).
In literature, data on differences between aware and “unaware” waking states are scant. In our DOC patients, we defined epochs as “waking state” if a sleep pattern was absent and PSG data were not polluted by artifacts. Sleep evaluation was performed on 30-second epochs, in accordance to the rules proposed by the American Academy of Sleep Medicine. 23 Moreover, sleep evaluation was interpreted by means of visual inspection by a certified/licensed sleep medical practitioner, blinded to the clinical assessment of DOC patients.
For each patient, total sleep time (TST; defined as the amount of actual sleep time during PSG recording) and rapid eye movement (REM), non-REM (NREM 2), and slow wave sleep (SWS) periods were calculated. We excluded stage 1 of sleep from the subsequent analysis because the standard description of this stage does not match with the altered behavior and PSG of DOC patients, 2 and because we could not clearly distinguish the latter from epochs of relaxed wake in most patients.
Statistical Analysis
Descriptive statistics are shown as median (range) and categorical variables are
expressed in percentage. Nonparametric tests were chosen because of the small subgroups
sample. Mann-Whitney test or Kruskal-Wallis test were used to evaluate differences between
2 or more groups, and a Bonferroni correction was used for post hoc tests. Chi-square
tests were used to compare proportions and to evaluate associations between categorical
variables. The relationship between CRS-r total scores (dependent variable) and age, sex,
etiology (traumatic vs vascular), acute phase severity, outcome evaluation time, was
assessed by multiple regression analysis. CRS-r score and outcome evaluation time were not
normally distributed, hence a logarithmic transformation (log 10×) was made in order to
obtain normal distribution. Tests were considered significant when the
Statistical analyses were performed using IBM SPSS Statistic 20.
Results
During the study period, January 2011 to May 2013, 153 DOC adult patients were admitted to the CRC of the IRCCS Neurological Institute “C. Besta” of Milan. 17 Since DNA collection started only after 6 months from the CRC onset, 110 patients were included in the present study. From this pool of patients, 17 subjects were further excluded due to diagnosis of severe disability, 14 for lack of DNA collection informed consent from a legal representative/next-of-kin, and 8 for inadequate DNA quality (Figure 1). However, demographic (etiology, age at injury, gender, and acute GCS score) and risk factors profile (cardiovascular risk factors, such as obesity, smoking, arrhythmias, and cardiomyopathy) of the excluded patients did not differ significantly from those of included subjects (data not shown).
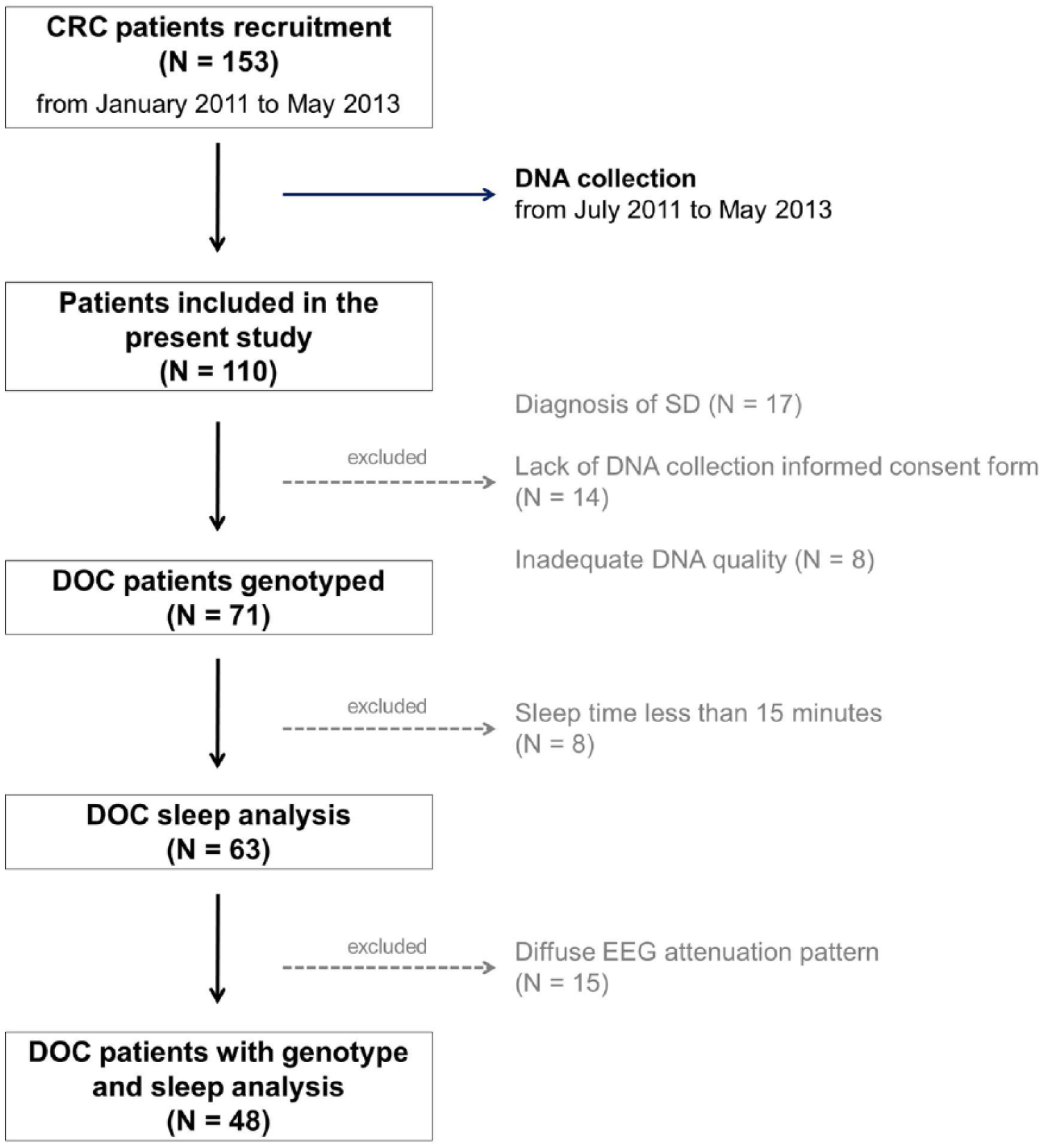
Flowchart of study selection. From January 2011 to May 2013, 153 adult patients with disorders of consciousness (DOC) were admitted at the Coma Research Centre (CRC) of the IRCCS Neurological Institute “C. Besta” of Milan. Since DNA collection started with a 6-month delay from the CRC onset (July 2011), 110 patients were included in the present study (for details, see text).
Acute Phase Statement
Of the 71 DOC patients, 26 (36.6%) were traumatic and 45 (63.4%) had vascular origin (43.5% hemorrhagic, and 56.5% postischemic brain damage). The patients median age at injury was 45 years (range 17-81 years), and 41 (57.7%) were males. The median GCS score derived by past clinical data was 3 (range 3-10).
Demographic and clinical data at the acute phase statement by genotype distribution are summarized in Table 1. Although we did not find any statistical differences in age, sex, GCS score, and etiology between genotypes, Per35/5 patients seemed to be younger (40 years, range 20-68), and more frequently females (66.7%) than other genotype subgroups.
Presenting Features of Disorders of Consciousness Patients in the Acute Phase Statement by Genotype Distribution. a
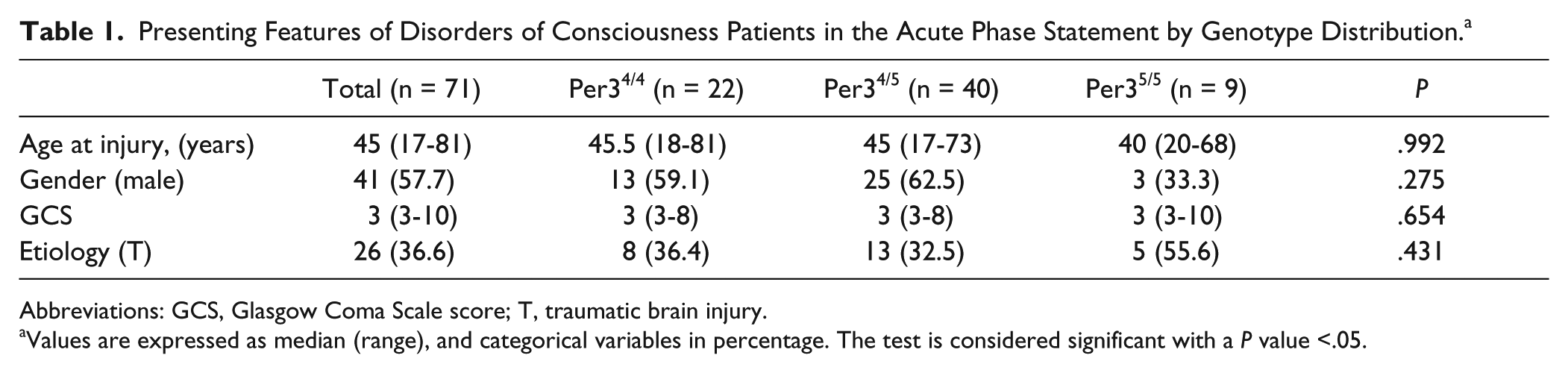
Abbreviations: GCS, Glasgow Coma Scale score; T, traumatic brain injury.
Values are expressed as median (range), and categorical variables in percentage.
The test is considered significant with a
Per3 Allele Distribution
The human Per3 clock gene contains a polymorphic domain expressing 4 or 5 copies of a
54-bp tandem repeat sequence. Homozygous carriers of the longer allele are referred as
Per35/5, homozygous for the short one as Per34/4, and carriers of
the heterozygous variant as Per34/5. By genotyping our 71 DOC samples, we found
a PER3 allele frequency of 0.59 (4-repeats allele) and 0.41 (5-repeats allele). The
genotype distribution was not in Hardy-Weinberg equilibrium (
Outcome Evaluation
The median time between onset and CRC evaluation (outcome evaluation time) was 4 years (range 2-22 years). According to the definition of the Aspen Neurobehavioral Conference Workgroup, 24 44 (62%) of our DOC patients were diagnosed as VS/UWS, whereas 27 (38%) as MCS. The overall CRS-r median score was 7 (range 4-16); in particular, 7 (range 4-9) in VS/UWS and 9 (range 7-16) in MCS patients.
Forty-four (62%) of our DOC patients were tracheostomized and/or had respiratory failure, while 8 (11.8%) were affected by arrhythmias and/or cardiomyopathy. A previous history of epilepsy was found in 9 (13.2%) of our DOC patients, whereas it was detected at follow-up examination in 44 (64.7%).
Per3 Genotype Distribution and Clinical Features
DOC patients’ demographic, clinical, and outcome CRC evaluation by genotype distribution are summarized in Table 2.
Disorders of Consciousness Patients Demographic, Clinical, and Outcome Evaluation by Genotype Distribution, Performed at the Coma Research Centre. a
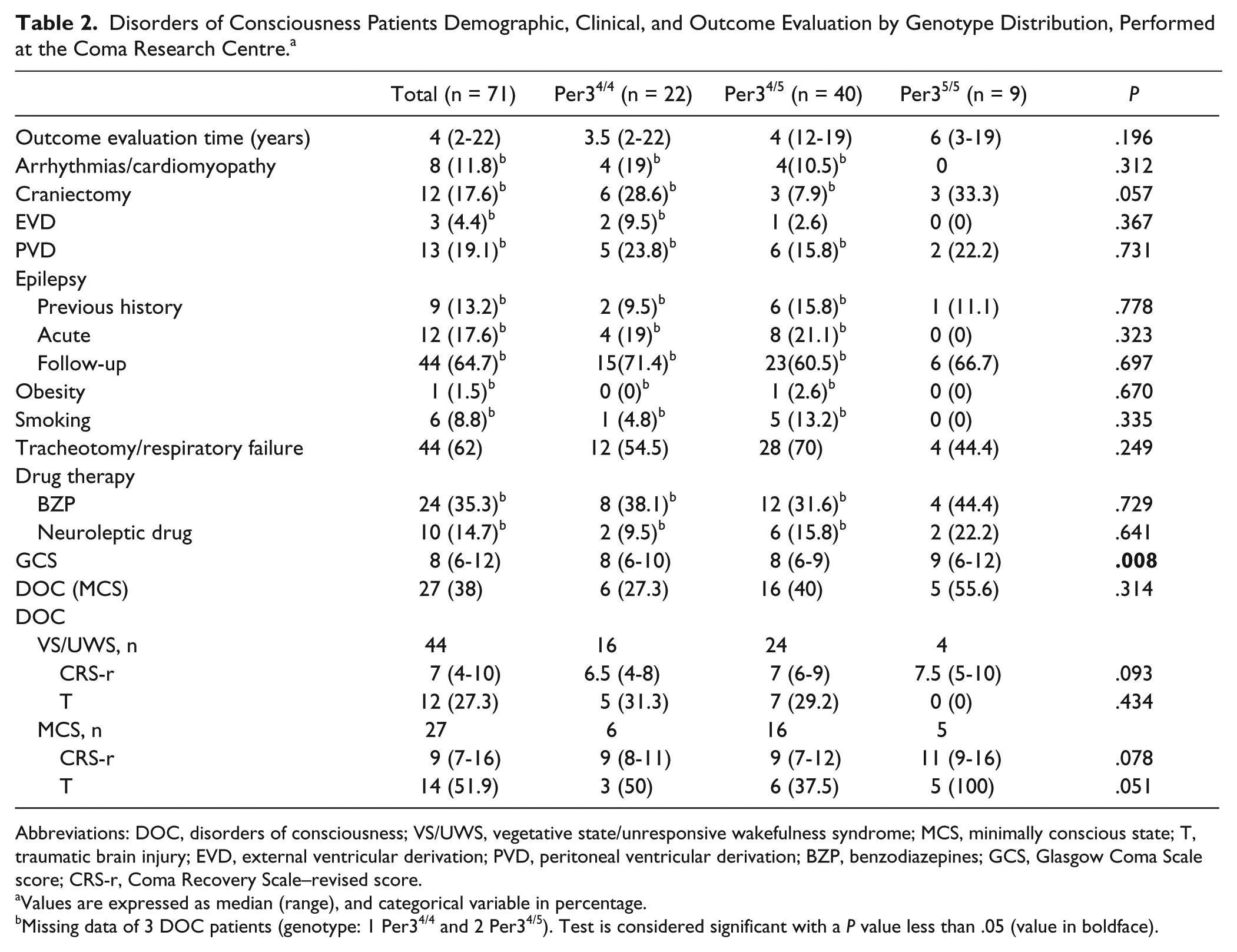
Abbreviations: DOC, disorders of consciousness; VS/UWS, vegetative state/unresponsive wakefulness syndrome; MCS, minimally conscious state; T, traumatic brain injury; EVD, external ventricular derivation; PVD, peritoneal ventricular derivation; BZP, benzodiazepines; GCS, Glasgow Coma Scale score; CRS-r, Coma Recovery Scale–revised score.
Values are expressed as median (range), and categorical variable in percentage.
Missing data of 3 DOC patients (genotype: 1 Per34/4 and 2
Per34/5). Test is considered significant with a
Twenty-two (31%) of our patients carried the Per34/4, 40 (56.3%) the
Per34/5, and 9 (12.7%) the Per35/5 genotype. The genotype
distribution was not significantly associated with DOC subgroups although
Per35/5 tend to be more represented in MCS patients (55.6%). Nine (13.2%) of
our DOC patients suffered from epilepsy before coma (3 treated with antiepileptic drugs).
Forty-four (64.7%) of them reported a medical history of seizure, status epilepticus,
and/or epileptic activity detected with EEG (spikes, spike-waves, periodic epileptiform
discharges, etc) in the acute phase (12 patients, only 1 treated with antiepileptic drugs)
or in the follow-up period (32 patients, all treated with antiepileptic drugs). Although
we did not identify any significant association between the considered risk factors and
genotype subgroups, Per34/5 carriers more frequently presented some
cerebrovascular risk factors such as obesity and smoking (1/40 and 5/40 respectively;
The total median CRS-r value was significantly different in the 3 subgroups
(
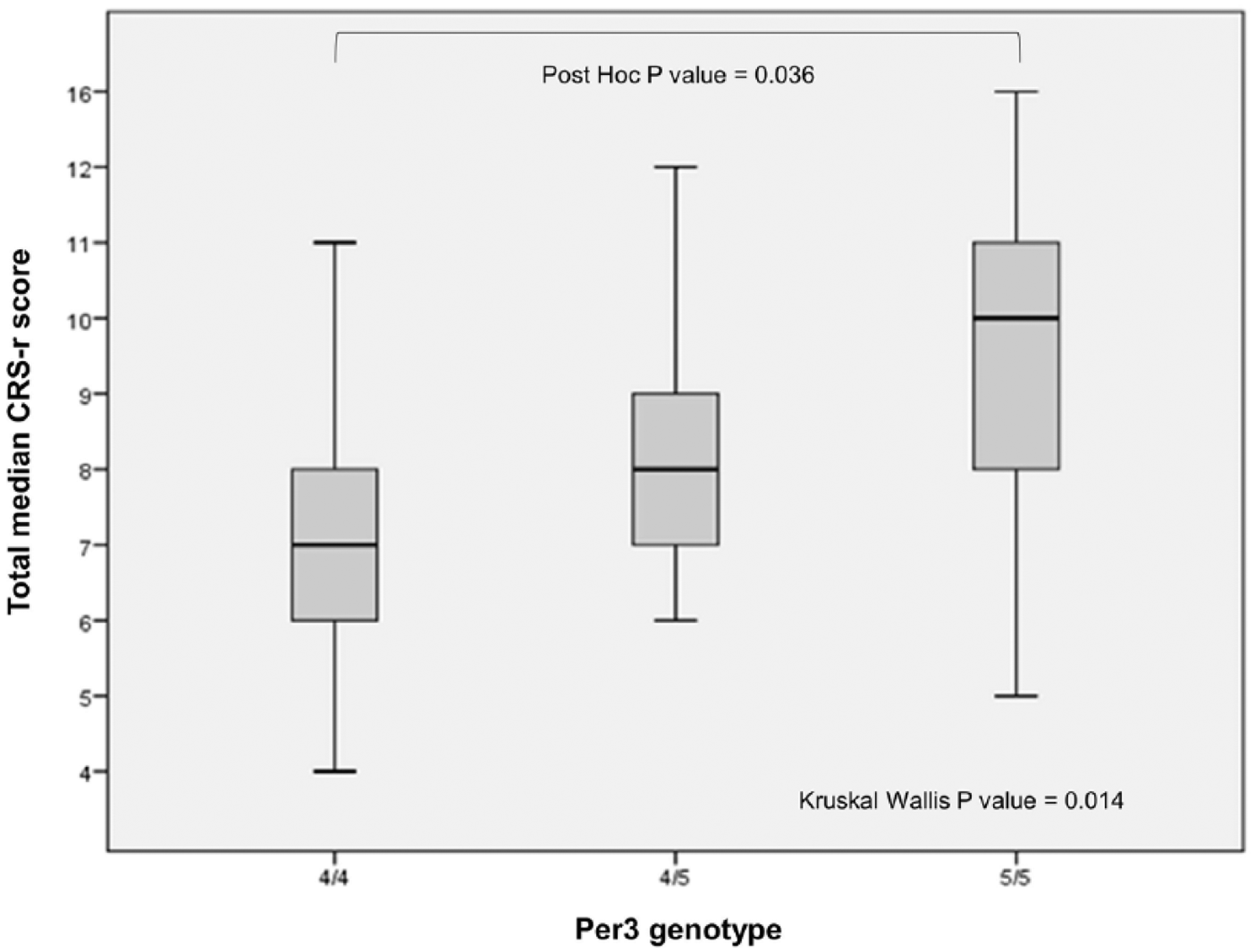
Total median Coma Recovery Scale–revised (CRS-r) score in relation to Per3 genotype
distribution. Total median CRS-r value was significantly different in the 3 genotype
subgroups (
The subanalysis of CRS-r subitems found significant dissimilarities for visual and
auditory scores by genotype distribution: visual Per35/5 3 (range 1-4),
Per34/4 1 (range 0-3), and Per34/5 1 (range 0-3),
Disorders of Consciousness Patients Outcome CRS-r Subitems Score by Genotype Distribution. a
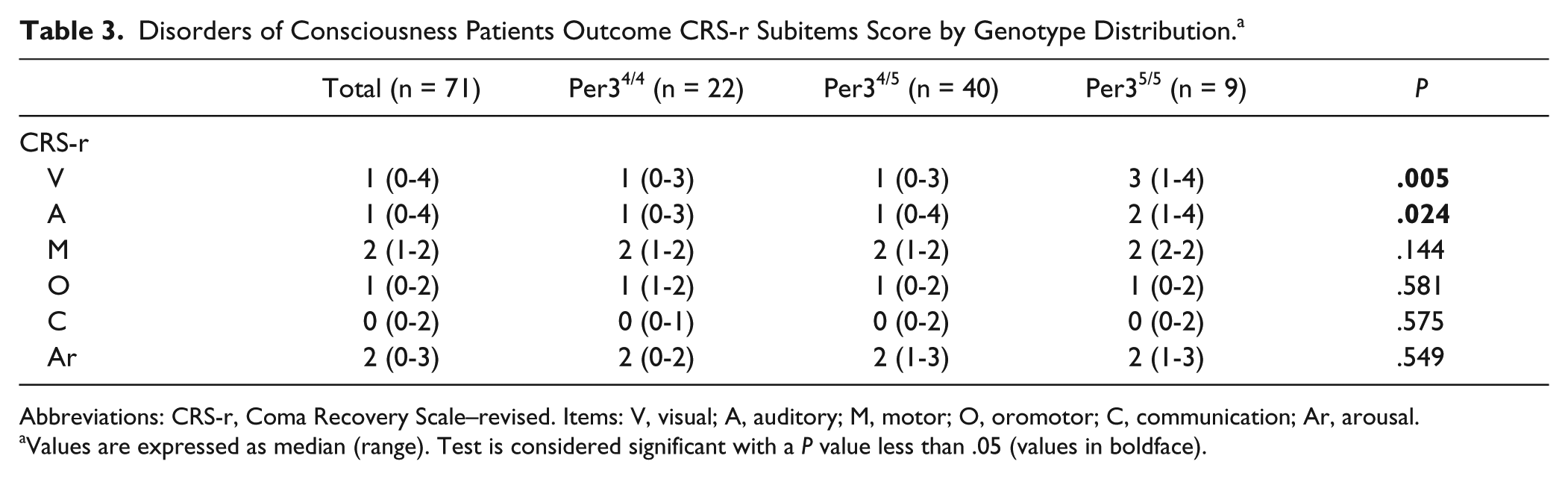
Abbreviations: CRS-r, Coma Recovery Scale–revised. Items: V, visual; A, auditory; M, motor; O, oromotor; C, communication; Ar, arousal.
Values are expressed as median (range). Test is considered significant with a
Sleep Patterns and Per3 Genotype
Although 44 patients had a previous history of epilepsy (see also clinical results for details), we recorded partial seizures in only 2 patients (diagnosis: 1 VS/UWS and 1 MCS). Epochs including seizure and/or artifacts polluting PSG data (eg, movements) were scored neither as “waking state” nor as REM/NREM sleep.
Eight patients were excluded from sleep evaluation because they slept less than 15
minutes during polygraphic recordings (DOC diagnosis: 2 VS/UWS and 6 MCS). In the
remaining 63 DOC patients, the total sleep time (TST) was 172 minutes (range 25-515
minutes). Particularly, TST was significantly higher in MCS (201 minutes (range 88-515
minutes) versus VS/UWS patients (159.5 minutes, range 25-340;
Fifteen VS/UWS patients showed a diffuse EEG attenuation (amplitude <20 µV) as a predominant/unique sleep pattern (all with low total CRS-r scores; data not shown). Since it is possible to speculate that the severe cortical and/or brainstem damage minimized the role of Per3 gene polymorphism in the preservation of residual sleep, we further excluded these patients from advanced sleep stage analysis.
In all the 48 residual DOC patients, we found the presence of NREM 2 stage of sleep,
whereas SWS stage was found in 41 (85.4%: 20 MCS and 21 VS/UWS patients) and REM stage in
22 (45.8%: 13 MCS and 9 VS/UWS patients) DOC patients. The subgroup analysis between MCS
and VS/UWS patients revealed a significant difference only in NREM-2 + SWS duration (NREM
total; MCS > VS/UWS,
Sleep Evaluation by Disorders of Consciousness Subgroups Distribution. a
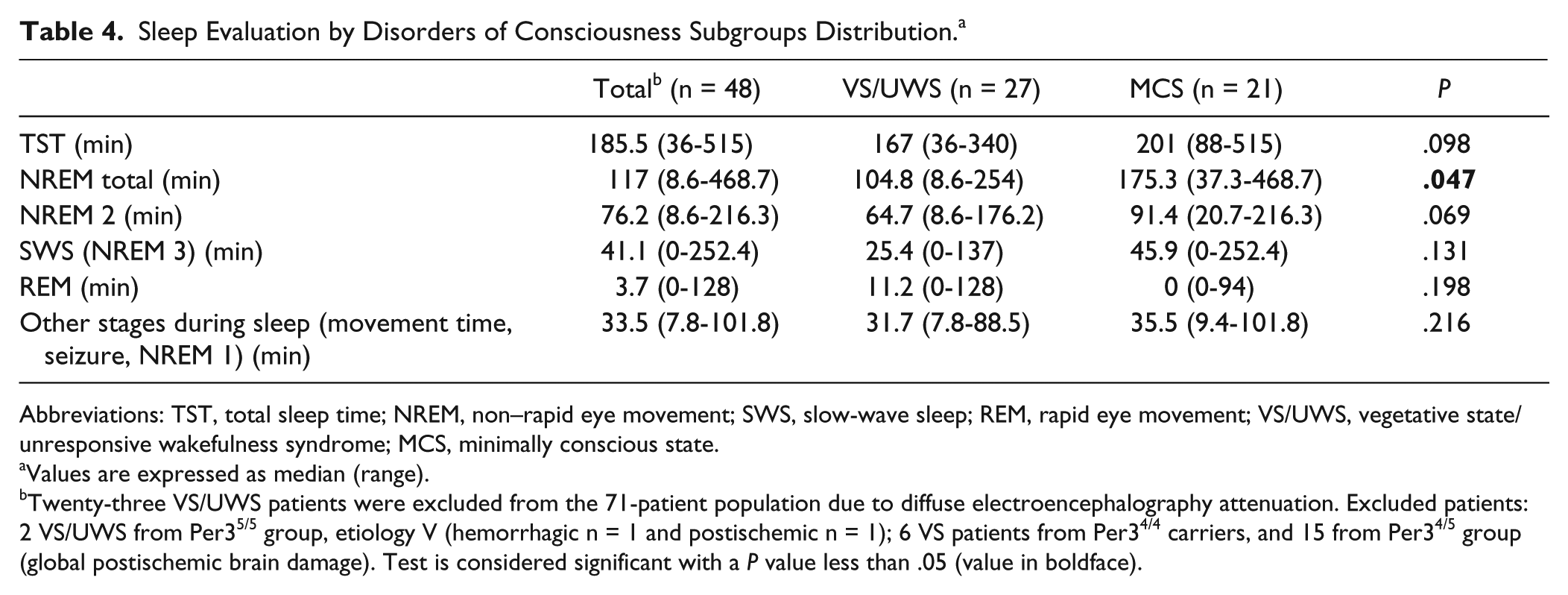
Abbreviations: TST, total sleep time; NREM, non–rapid eye movement; SWS, slow-wave sleep; REM, rapid eye movement; VS/UWS, vegetative state/unresponsive wakefulness syndrome; MCS, minimally conscious state.
Values are expressed as median (range).
Twenty-three VS/UWS patients were excluded from the 71-patient population due to
diffuse electroencephalography attenuation. Excluded patients: 2 VS/UWS from
Per35/5 group, etiology V (hemorrhagic n = 1 and postischemic n = 1); 6
VS patients from Per34/4 carriers, and 15 from Per34/5 group
(global postischemic brain damage). Test is considered significant with a
Table 5 summarizes the sleep stages analysis by genotype distribution. The sleep analysis by genotype distribution did not reveal any significant difference in median sleep measures at PSG examination between Per3 genotypes. However, although not significant, the TST tends to be higher in Per35/5 patients: 221 minutes (range 88-515 minutes), in comparison with the Per34/4 and Per34/5 patients (151.5 minutes, range 36-477 minutes and 188 minutes, range 44-422 minutes, respectively). Moreover, SWS phase also seems to be higher in Per35/5 patients (97.3 minutes, range 0-252.4 minutes), in comparison with the Per34/4 and Per34/5 patients (23.7 minutes, range 0-132.8 minutes and 44.3 minutes, range 0-202.6 minutes, respectively).
Sleep Stages Evaluation by Genotype Distribution. a
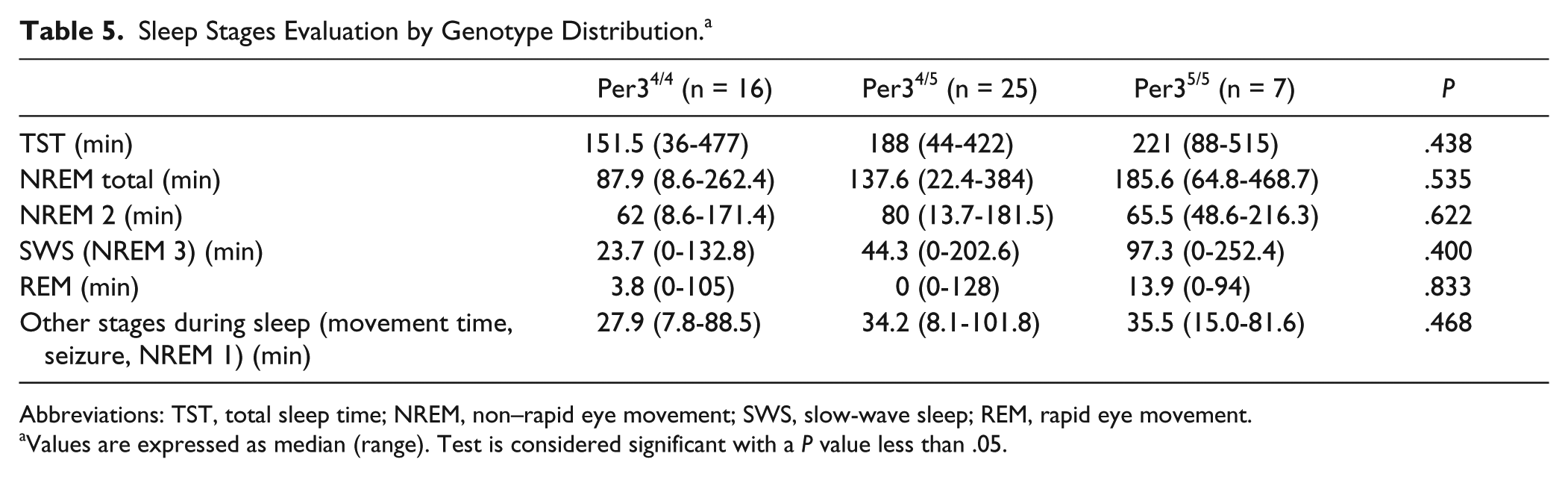
Abbreviations: TST, total sleep time; NREM, non–rapid eye movement; SWS, slow-wave sleep; REM, rapid eye movement.
Values are expressed as median (range). Test is considered significant with a
Discussion
Because of medical progress in intensive care, the number of DOC patients is increasing. However, although some data highlight the role of transcranial magnetic stimulation, electroencephalography and neuroimaging studies as possible tools to assess DOC residual functions, the reliability of these methodologies has yet to be fully established. 25 Previous studies described a wide spectrum of sleep patterns ranging from normal to severe architecture disorganization in DOC patients, supporting the importance of sleep assessment in evaluating the integrity of brain function.2,3,26 Similar to previous reports, our MCS patients have a longer total sleep time and NREM (NREM 2 + SWS) phase, in comparison with VS/UWS subjects. This finding suggests that MCS patients have a better preservation of sleep rather than VS/UWS patients. Cologan et al 2 emphasized the difference between MCS and VS/UWS in REM sleep, advancing the idea that the REM phase may be useful to differentiate the VS/UWS patients who present the persistence of minimal consciousness features. Conversely, we found that VS/UWS patients tend to have a longer duration of REM sleep compared to MCS, although not statistically significant. This could be partially explained by the exclusion from our analysis of VS/UWS patients with the most severe sleep pattern (diffuse EEG attenuation).
We observed that Per35/5 carriers present a significantly higher median CRS-r score in comparison to Per34/4 (10 vs 7). Despite not being statistically significant, this effect seems to be confirmed in both VS/UWS (7.5 vs 6.5 vs 7, Per35/5, Per34/4, and Per34/5, respectively) and MCS patients (11 vs 9 vs 9, Per35/5, Per34/4, and Per34/5, respectively) suggesting that, independently from the severity, Per35/5 genotype could be associated with a better outcome. Although no data on the specific role of Per3 gene on DOC patients’ functional and cognitive outcome are available so far, our results seem not to be in line with previous authors, who reported that Per35/5 genotype is associated with the worst performance, particularly in executive subjective alertness, and sustained attention tasks during sleep deprivation (also in physiological condition).11,12 In these studies, the worst performance of Per35/5 carriers has been related to the different cerebral-related task activation patterns during working memory assignment. In fact, after sleep deprivation, Per35/5 carriers, differently from Per34/4 subjects, were observed to have widespread reduced cortical activation and supplemental cortical region recruitment. 27
Better Per35/5 CRS-r performances in our DOC patients may be explained by the
restorative function of sleep, since total sleep time tends to be longer in
Per35/5 patients (221 minutes, range 88-515 minutes), in comparison with
Per34/4 and Per34/5 carriers (151.5 minutes, range 36-477, and 188
minutes, range 44-422, respectively). Thus, the higher TST in Per35/5 DOC
patients could be associated with a
Our study takes advantage of a high number of well-phenotyped patients (among which the majority are of vascular etiology, quite rare in literature), as well as the length of sleep recording and outcome survey. However, it is limited by some methodological biases mostly due to the small sample size and the hospital frame sampling, which does not consider patients emerging from coma or those who are dead. Moreover, the Per35/5 subgroup is impaired by the presence of a combination of sleep promoting factors (ie, increased frequency of benzodiazepines and neuroleptic use, lower number of tracheotomies) that, despite not being statistically significant, might influence the higher sleep quantity and, therefore, the better level of recovery.
Conclusions
Although our findings have to be confirmed both in longitudinal and in a larger series of DOC patients, we have shown for the first time a possible association between Per3 VNTR polymorphism and cognitive and functional impairment of DOC patients. Even though the exact molecular mechanism by which this polymorphism influences DOC performance has yet to be understood, we speculate that its effect is mediated by higher total sleep time and slow wave sleep that would improve preservation of the main cerebral connections, rather than by an alteration of sleep homeostasis. Finally, the higher susceptibility to sleep pressure found in Per35/5 carriers would suggest the introduction of some management measures, such as the preservation of Per35/5 carriers from sleep deprivation, due to the need for more efficient sleep time to preserve cognitive functions.
Supplemental Material
Supplementary_Table – Supplemental material for Is Period3 Genotype Associated With Sleep and Recovery in Patients With Disorders of Consciousness?
Supplemental material, Supplementary_Table for Is Period3 Genotype Associated With Sleep and Recovery in Patients With Disorders of Consciousness? by Gloria Bedini, Anna Bersano, Davide Rossi Sebastiano, Davide Sattin, Francesca Ciaraffa, Valentina Tosetti, Greta Brenna, Silvana Franceschetti, Emilio Ciusani, Matilde Leonardi, Jesus Vela-Gomez, Giorgio B. Boncoraglio and Eugenio A. Parati in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to thank all the patients’ families for their kind cooperation in this study. The present article was submitted on behalf of Coma Research Centre (CRC) multidisciplinary team. The authors would like to acknowledge the contributions of the following members: Dr A. Andronache, Dr R. Benti, Dr D. Caldiroli, Dr P. Fazio, Dr G. Marotta, Dr F. Molteni, Dr M. Pagani, Dr F. Panzica, Dr B. Reggiori, Dr C. Rosazza, Dr G. Varotto, Dr E. Visani, Dr D. Duran, Dr A. M. Giovannetti, Dr V. Covelli, Dr M. G. Bruzzone, Dr S. Ferraro, Dr A. Nigri, Ms E. Gioffredi. We thank A. Smith for the English review of the article. We also thank Dr M. Ranieri and Dr M. Dossena for the critical reading of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project CRC, “Coma Research Centre” was supported by Grant No. IX/000407–05/08/2010 awarded by Regione Lombardia. The study was conducted in collaboration with European Federation of Biomedical Research, FERB.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
