Abstract
Introduction
Patients diagnosed as being in a minimally conscious state (MCS) demonstrate discernible behavioral evidence of consciousness but remain unable to reproduce this behavior consistently. 1 The MCS rarely changes to normal conscious behavior. Very few studies have focused on a therapeutic intervention in patients with consciousness impairment, specifically in MCS. 2,3 We investigated the effects of repetitive transcranial magnetic stimulation (rTMS) on the immediate behavior and electroencephalographic (EEG) activity of a patient in a MCS.
Babiloni et al 4 demonstrated a relationship between a responsive α EEG activity and conscious awareness. The α frequency band is predominant over the posterior (α rhythm) and rolandic (µ rhythm) sites. It is modulated by sensory input and motor output and reflects the spontaneous rhythm of sensory and sensorimotor areas. 5 Both α and µ rhythms indicate a healthy inhibitory state of the network necessary for blocking irrelevant information during focused attention and memory storage. 6,7
EEG is a widely accepted method for obtaining specific information on the level of cortical information processing and changes that occur during unconsciousness and varying states of conscious awareness. 8 Generally, more severe traumatic brain damage appears to correlate with increased power in slow bands. The power spectra and coherence analyses usually show a decreased EEG regional power for nearly all frequency ranges correlated with the underlying structural brain injury. 9 Therefore, a broad spectrum of pathological EEG features exist and include different connected brain networks. We may infer that an increase of the α frequency band is correlated with a functional improvement in the patient.
In the present study, rTMS was performed on the primary motor cortex (M1). This site of stimulation was chosen on the basis of previous investigations that applied repetitive TMS to the motor cortex and observed a transient increase of neuronal oscillations in the α and β frequency EEG bands. 10-12 In TMS-EEG studies, the synchronization of neuronal activity was found to show topographic specificity. 13 α and β reactivity is more prominent after magnetic stimulation of the sensory or motor cortex than after stimulation of the dorsal premotor cortex. TMS stimulation of the dorsal premotor cortex is more often associated with changes in power of the lowest frequency bands, θ and δ. 14,15 Finally, M1 is the only site at which the excitability of the cortex can be safely monitored in real time without inducing seizures. 16
As a control for the more direct cortical stimulation of rTMS, we used peripheral nerve stimulation because most of its stimulus parameters and the experimental condition are comparable with rTMS. Moreover, it activates the central motor system and may promote recovery in states of impaired consciousness. 17
The study was conducted with the approval of the local ethics committee with the precondition of the absolute safety of the rTMS procedure.
Methods
A 70-year-old man suffered sudden loss of consciousness and left hemiplegia after a large right thalamic hemorrhage 4 years earlier associated with hypertension. He also had a minor ischemic stroke in the left basal ganglia. Then, 1 month later, the patient recovered to a minimal level of consciousness; he opened his eyes spontaneously or in response to pinprick stimulation, turned his eyes toward the source of sounds, and visually tracked a moving object. Verbal communication or signing (ie, with eye movements) was absent. A prolonged EEG recording showed awake background, intermittent generalized background slowing, and periodic return of these patterns indicating a preserved sleep/wake cycle. His Glasgow Outcome Scale (GOS) score was 2, with a diagnosis of MCS. Over the following 4 years, the patient’s GOS score was reassessed at least every 6 months and remained unchanged. He was readmitted to our rehabilitation center for a trial of rTMS.
The JFK Coma Recovery Scale–Revised (JFK CRS-R) was used to evaluate the residual functions of the patient. It consists of 23 items in 6 subscales arranged hierarchically to reflect brain stem, subcortical, and cortical pathology. The lowest items on each subscale represent reflexive activity, and the highest items reflect cognitively mediated behaviors. 18 The patient had a score of 14. He opened his eyes spontaneously, localized sound and noxious stimulation withdrawing the respective limb from the pain source, visually pursued moving objects, and vocalized without comprehensible words.
An ABA experimental stimulation design was used. The patient participated in 3 different sessions with 3 days between sessions. In the first session (condition A), the patient underwent 10 trains of 20 Hz rTMS delivered through a focal, figure-of-eight coil connected to a Magstim Rapid2 transcranial magnetic stimulator (maximum output 2.2 Tesla) for 10 minutes. The stimulus intensity was set at 10% below the participant’s motor threshold at rest, with the coil placed on the left primary motor cortex (M1). In the second session (condition B), median nerve stimulation at the wrist was performed for 10 minutes. The stimulus intensity elicited sustained thumb twitching at a frequency of 3 Hz. Because both treatments involve the same amount of interpersonal attention, placebo effects should be comparable between condition A and B. Finally, the third block (condition A again) of stimulation consisted of a second session of rTMS to replicate any effects obtained previously.
The 3 sessions of stimulation were preceded and followed by 30 minutes of EEG; 20 minutes of resting EEG were performed hourly to examine for further clinical changes, for 8 hours after stimulation. A long-term follow-up was carried out 6 months later. The EEG was measured using a standard EEG amplifier (SD MRI 21) and a cap with 21 electrodes positioned according to the international 10/20 system. EEG signals were subdivided into regions of interest, such as frontal (electrodes F4/F8 and F3/F7), central (electrodes C4, Cz, and C3), parietal (electrodes P4, Pz, and P3), temporal (electrodes T4/T6 and T3/T5), and occipital regions (electrodes O2 and O1) according to the Brodmann areas and processed using wavelet-based spectral analysis. 19,20
Reference-free recordings were obtained by calculating the local average reference using the software EEGLAB, a Matlab toolbox 6. A notch filter (50 Hz) and a baseline correction were applied to all channels. EEG epochs with ocular, muscular, and movement artifacts were identified and rejected. Segmentation into nonoverlapping epochs of 2 s was applied. A fast Fourier transform was applied to nonoverlapping epochs each containing 2048 data points, with maximum resolution (0.5 Hz) and then averaged across epochs under the same conditions. Density power spectra Px(f) (µV2/Hz) were estimated for all frequencies between 0 and 50 Hz; then, relative power (%) was estimated for δ (1-3.5 Hz), θ (3.5-7.5 Hz), α (8-13 Hz), and β (14-30 Hz) frequencies. The bioengineers who analyzed the EEG data were unaware of the 3-session stimulation sequence.
The clinical assessment of consciousness was performed using the JFK CRS-R. Two blinded raters evaluated the patient immediately before and after each stimulation session and hourly for 8 hours. The EEG monitoring was performed briefly after each clinical assessment of the patient. We compared percentage changes across conditions (A, B) using a repeated-measures 1-way analysis of variance (main factor “stimulation”
Results
The EEG findings of the patient included diffuse slowing in the θ and δ frequency ranges and absence of reactivity of the dominant posterior rhythm. Power spectral analysis showed a reduction in power for all frequencies, with more marked reduction in the right hemisphere.
After median nerve stimulation (condition B), the patient did not exhibit any clinical or behavioral changes, and the EEG did not change significantly (Figure 1D). Immediately after the first rTMS session, however, the patient’s JFK CRS-R score changed from 13 to 19 (auditory, 4; visual, 5; motor, 5; oromotor/verbal, 2; arousal, 3). The patient was able to maintain continuous eye opening and to accomplish complex voluntary tasks on verbal command, such as to reach the right arm toward a glass of water, grasp it, lift it, and bring it to his lips without drinking the water (a video recording is available). Over the 6 hours after stimulation, the patient showed signs of increased arousal and awareness and was looking around and had some object manipulation spatial abilities. This behavioral change after the rTMS session suggested that the patient had regained, to some extent, the capacity to understand simple commands and to recognize, reach for, and use objects. The behavioral improvement, confirmed hourly by repeated JFK CRS-R assessment, lasted for 6 hours after the rTMS session. In the seventh hour assessment, the patient appeared less reactive, not able to follow simple commands, and showed a JFK score of 14. The second session of rTMS (condition A again) was administered 2 days later and yielded similar results. The patient manifested some behavioral changes such as shaking hands on command and following the examiner with his gaze. The JFK-CRS-R score improved from 14 to 19.
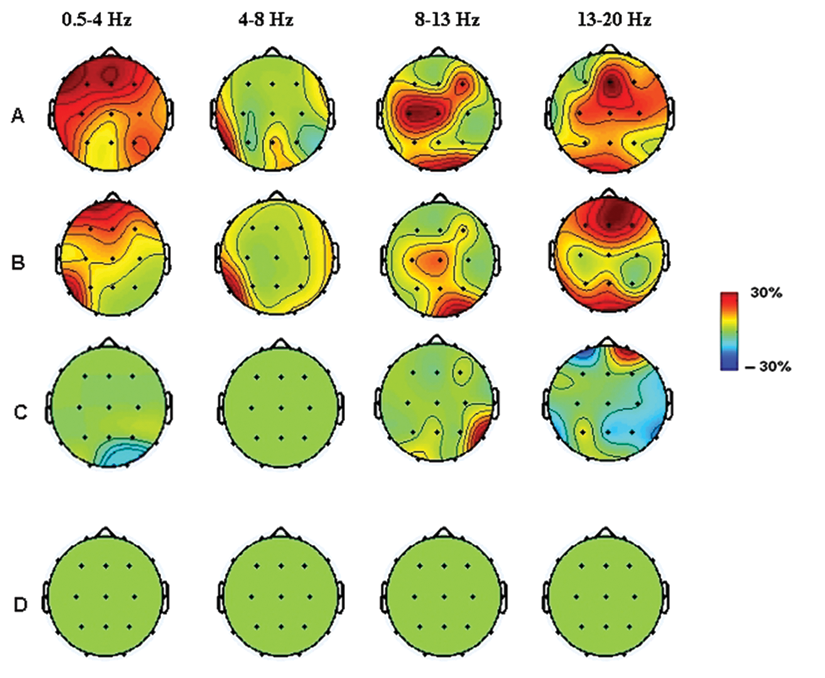
Topographic maps of cortical activation immediately after (A) and six and seven hours after (B,C) the session of rTMS. Cortical activation related to peripheral electrical stimulation (D)
The clinical improvements within 6 hours were accompanied by significant changes in the EEG power spectra. Figures 1A, 1B, and 1C illustrate the topographic maps of cortical activation related to rTMS. Distinct patterns were represented in 4 frequency ranges (0.5-4 Hz, 4-8 Hz, 8-13 Hz, 13-20 Hz). The differences between before and immediately after magnetic stimulation were significant in the α band (8-13 Hz), in the low and high β bands (13-20 Hz), and in δ frequencies (0.5-4 Hz;
Discussion
rTMS, in which intracranial currents are induced by repetitive stimulation higher than 5 Hz, is known to depolarize neurons under the stimulating coil and indirectly affect areas related to cognition and behavior. 21 A single session of rTMS delivered over M1 induced temporary behavioral improvements and an increase of EEG oscillatory cortical (α-β rhythms) and likely subcortical (δ activity) patterns in patients in a MCS. Although the stimulation with rTMS was focal over the M1, the observed effects were widespread, including many recording sites. This might be explained by volume conduction because of the known poor spatial resolution of EEG or because of generalized arousal. The proximity and connection between the stimulation areas of M1 and Dorsolateral Prefrontal Cortex (DLPFC) might be the reason for the increase in the δ band. Indeed, Okamura et al 14 and Griskova et al 15 found an increase in power of the lowest frequency bands after rTMS stimulation of the DLPFC.
The augmentation of oscillatory activity in the α range may have been because of improved cortical–thalamic interactions. 22 The observed widespread changes in δ power together with the increase in the other frequency bands may represent a change of metabolic activity in the brain. As reported in other studies, the application of high-frequency rTMS may induce an increased release of dopamine, which may modulate δ activity. Although isolated dominance of δ activity usually indicates compromised consciousness and awareness, the correlated improvement with the higher frequency band may index relative normalization of the metabolic situation. 21 The variations in cerebral activity among patients in a MCS or vegetative state indicate that some cerebral regions can retain partial function in patients with compromised consciousness. These regions, however, are not integrated in the complete network of information processing. The brain activity of different brain regions in patients in a vegetative state and MCS is not functionally connected because of the presumed interruption between these different brain areas and the thalamus. 23 Although we cannot unequivocally conclude that rTMS induced improved awareness, the patient had a very low probability to recover consciousness because his clinical condition had not changed in 4 years. His clinical improvements were similar to those reported in a MCS patient who was treated with bilateral electrical deep brain stimulation (DBS) within the central thalamus using frequencies of 50 to 100 Hz. 24 DBS, however, is invasive and not suitable for routine treatment because it is unclear which MCS patients would benefit; so the risk and cost may deter use of the procedure. Moreover, caregivers and legal representatives often do not give their consent for the implantation of DBS electrodes, as happened in our case. The patient’s wife refused further rTMS stimulation trials because of the marked unresponsiveness of the patient after the stimulation.
Our results suggest that the therapeutic effect of rTMS on the partially disconnected and lesioned brain might be associated with the restoration of the rhythmic activity of neurons in the thalamic pacemaker and consequent improvement of functional connectivity. 25,26 Although family members refused continuation of the experiment, rTMS may be useful in identifying subgroups of MCS patients who could benefit from more invasive stimulation strategies such as DBS and may help select patients suitable for brain computer interface–based communication. 27
Conclusions
We report a case study in which rTMS triggers transient neuronal oscillations in EEG frequency bands and behavioral changes in MCS. Patients responding to excitatory TMS might be candidates for permanently implanted DBS, and we do not exclude the possibility that transcranial stimulation might have a therapeutic role, promoting cognitive recovery. Future research that includes a large sample should be undertaken to confirm this novel hypothesis.
Footnotes
Acknowledgements
We would like to thank Professor Niels Birbaumer for his important assistance, suggestions and editing throughout the study.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
