Abstract
Background. Few therapeutic options exist for the millions of persons living with severe arm impairment after stroke to increase their dose of arm rehabilitation. This study compared self-guided, high-repetition home therapy with a mechanical device (the resonating arm exerciser [RAE]) to conventional therapy in patients with chronic stroke and explored RAE use for patients with subacute stroke. Methods. A total of 16 participants with severe upper-extremity impairment (mean Fugl-Meyer [FM] score = 21.4 ± 8.8 out of 66) >6 months poststroke were randomized to 3 weeks of exercise with the RAE or conventional exercises. The primary outcome measure was FM score 1 month posttherapy. Secondary outcome measures included Motor Activity Log, Visual Analog Pain Scale, and Ashworth Spasticity Scale. After a 1-month break, individuals in the conventional group also received a 3-week course of RAE therapy. Results. The change in FM score was significant in both the RAE and conventional groups after training (2.6 ± 1.4 and 3.4 ± 2.4, P = .008 and .016, respectively). These improvements were not significant at 1 month. Exercise with the RAE led to significantly greater improvements in distal FM score than conventional therapy at the 1-month follow-up (P = .02). In a separate cohort of patients with subacute stroke, the RAE was found feasible for exercise. Discussion. In those with severe arm impairment after chronic stroke, home-based training with the RAE was feasible and significantly reduced impairment without increasing pain or spasticity. Gains with the RAE were comparable to those found with conventional training and also included distal arm improvement.
Introduction
There are more than 5 million stroke survivors living in the United States, with more than 650 000 people experiencing a new stroke each year. 1 An estimated 40% of this population live with moderate to severe impairment of their upper extremity,1-4 but evidence has shown that intensive rehabilitation exercise can reduce this impairment if an appropriate type and dose of therapy is used.5-10 Unfortunately, although there is not much work showing what the adequate dose of rehabilitation is, current methods likely provide repetitions that are an order of magnitude too few to be most effective, if animal models are used as a reference.11,12 Intensive one-on-one exercise with a therapist can significantly increase the amount of exercise performed, 13 but this is costly and may be impractical for many patients. Another approach is to develop home-based rehabilitation programs that allow patients to augment the amount of therapist-guided therapy they receive with additional self-guided exercise. However, self-guided rehabilitation programs have not been rigorously tested, 14 and individuals with severe arm impairment may be further limited by an inability to complete exercises at home without assistance.15,16
A machine-based approach could improve home therapy options by providing the motivation, real-time and long-term feedback, assistance, and automation needed to promote a large number of additional exercise repetitions. For example, the BATRAC system uses rhythmic auditory cueing to prompt hundreds of assisted reaching movements in a short amount of time.17-19 A similar approach has been used for exercise of the lower extremity. 20 However, although BATRAC has been successful in clinical trials, there has been no report to our knowledge of a controlled home-based test of BATRAC. Machine-based systems may also include virtual reality components, which have been shown to be motivating and thus might increase patient compliance with home-based therapy programs.21-23 Robotic rehabilitation devices can provide assistance and automate therapy, and they have been shown to improve outcomes, limit patient frustration, enhance sensory input, and promote self-efficacy.15,24-26 Unfortunately, because of the cost and complexity of robotic devices, they have not been rigorously tested outside of clinical settings and are largely unavailable for home use. Thus, although previous machine-based approaches are promising, there is still a need for more home therapy options. 27
We recently developed a device called the resonating arm exerciser (RAE) that might be an appropriate tool for improving self-guided home-based rehabilitation. 28 The RAE consists of a lever that attaches to the push rim of a wheelchair, a forearm support, and an elastic band attached between the lever and the wheelchair frame (Figure 1). A user operates the RAE by pushing and pulling on the lever, which rolls the wheelchair back and forth in place by 20 to 30 cm. Similar to the BATRAC system, the lever acts as a mechanical constraint that guides the arm through a movement with coordinated shoulder and elbow flexion and extension similar to reach-and-retrieval tasks. This configuration is also mechanically resonant, so if individuals push and pull on the lever at the system’s resonant frequency, they are rewarded with a larger movement. We found that this assistance amplifies the active range of motion of a user’s arm to approximately twice his unassisted range of motion, and every volunteer who tried the RAE in the clinic was able to exercise with it, regardless of their impairment level, weight, age, or other factors. 28 A similar type of exercise (using the impaired arm to rock in a rocking chair) has previously been shown to reduce long-term impairment after stroke.29,30 Furthermore, in a pilot study (n = 8), we found that chronic stroke patients who exercised with the RAE in a clinic performed thousands of repetitions, significantly increased their FM score and arm active range of motion, and had no increase in arm pain. 28
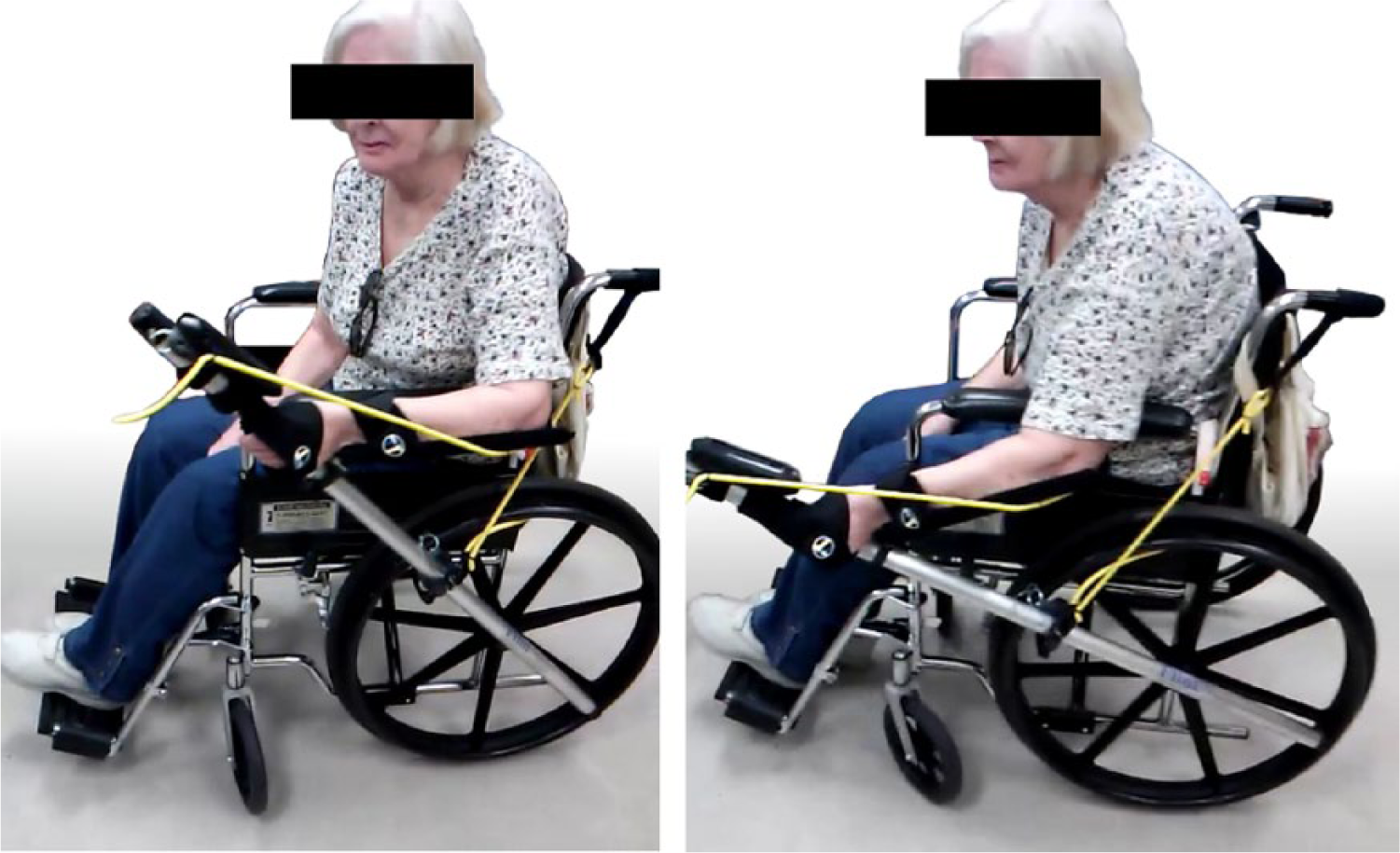
The resonating arm exerciser (RAE): the RAE quickly attaches onto the wheelchair push rim. The user exercises rhythmically with the device at about 1 Hz by extending the shoulder and flexing the elbow (left) and flexing the shoulder and extending the elbow (right) to activate the resonance of the device. The device rolls back and forth in place during the exercise.
The goal of this study was to test whether the RAE can provide a safe, motivating, and effective method of self-guided arm exercise to users at home and to determine whether home-based, assisted exercise with the RAE leads to greater reductions in arm impairment than conventional, self-guided, home-based exercises. To answer these questions, we performed a randomized controlled trial that compared self-guided home-based RAE therapy with conventional home-based therapy. We hypothesized that participants who performed RAE therapy would both exercise more and have significantly greater reductions in arm impairment than participants who performed conventional arm exercises, as assessed at a 1-month follow-up. We chose the 1-month follow-up as the primary outcome because persistent benefits are important to individuals with stroke.
Of additional interest was whether the RAE could be an appropriate tool for providing early, high-repetition arm therapy for individuals with subacute stroke (ie, <6 months poststroke). Therefore, we also performed a small pilot study of the RAE in a clinic with subacute stroke patients to gain the experience needed to design a larger efficacy study with this dynamic and complex population. Here, we hypothesized that the participants would be able to perform a large number of repetitions with the RAE without experiencing arm pain.
Methods
All experiments were approved by UC Irvine’s Institutional Review Board. Participants provided informed consent.
Chronic Stroke Trial in the Home
Study Design and Participants
The first study was a randomized controlled trial that compared self-guided, home-based exercise with the RAE to conventional self-guided, home-based therapy for individuals in the chronic phase of stroke. Inclusion criteria were the following: experienced one or multiple strokes more than 6 months previously, Upper Extremity Fugl-Meyer (FM) score <30 out of 66, absence of shoulder pain, ability to understand the instructions to operate the device, and age <80 years (because older age could be a confounding variable). We provided participants with a loaner manual wheelchair if they did not own one.
Outcome Measures and Data Collection
All assessments were performed at UC Irvine by a single blinded evaluator. The primary outcome measure was the Upper Extremity FM score. 31 The secondary measures included the Modified Ashworth Scale of spasticity, 32 the Visual Analog Pain Scale, the Motor Activity Log (MAL), 33 Box and Blocks score, 34 and quantitative measures of active range of motion at the shoulder and elbow acquired with a goniometer. For the goniometer measurements, only 1 measurement was taken to avoid fatigue. The single trained evaluator used the same goniometer and followed the same set of placement instructions for each participant.
Interventions
Participants returned 1 week after the initial evaluation to repeat the baseline clinical exam and verify whether they had a stable baseline. At this time, the supervising therapist placed the participants into either the RAE (n = 8) or conventional therapy (n = 8) groups via adaptive randomization based on their initial FM score. 35 That is, participants with a FM score >30 were alternately placed in the 2 groups, as were those with a FM score <30. Based on our pilot data, 28 8 participants would provide a 90% chance of detecting a significant difference of 8 points on the FM scale at the 5% significance level (the FM scale has a minimal clinically significant difference of 4.25 points 36 ). The participants in the RAE group were given a RAE device and instructed on how to use it safely. The participants in the conventional therapy group were given a booklet of exercises developed by experienced occupational therapists at the Rehabilitation Institute of Chicago for home therapy (see the online appendix). These exercises included passive range of motion, weight bearing, and active movement exercises for the shoulder and elbow, some of which used a tabletop for support. Both groups were instructed to exercise for 3 hours per week over at least 3 sessions per week for 3 weeks. This intensity has been shown to produce a therapeutic effect.9,37,38 An upper limit was not placed on the amount of exercise they could perform; however, the participants were instructed to stop exercising and consult the supervising therapist if they began to experience any pain or discomfort.
To monitor amount of use of the RAE, we mounted a smartphone to the main shaft of the device and developed an application that counts and logs the number of repetitions a user performs. The RAE group was instructed to run the application during each exercise session. The conventional group was asked to self-record the amount of time they spent exercising on a written log sheet; such self-report methods have been shown to be reliable. 39 The supervising therapist contacted all participants weekly by phone to ensure that they were not experiencing any difficulties with their exercise and to query about any adverse events or pain.
After the 3-week exercise period, the participants returned for posttherapy clinical assessments. At this assessment, participants also took the Intrinsic Motivation Inventory (IMI) to evaluate their perceptions of their therapy. The IMI is a series of questions answered on a scale from 1 to 7 that are related to perceived enjoyment, usability, stressfulness, motivation, and value of the exercises they performed. 40 We also retrieved the data from the smartphone on the RAE at this session. Participants returned 1 month later for follow-up assessments.
After the 1-month follow-up assessments, the participants in the conventional therapy group were also given a RAE device and instructed on how to use it safely. These participants then repeated the therapy regimen (3 h/wk for 3 weeks) using the RAE to exercise. At the end of this crossover exercise period, they again returned for posttherapy assessments. These participants also returned 1 month later for a final crossover follow-up assessment.
Data Analysis
We calculated individual differences in each outcome measure at the posttherapy and 1-month follow-up assessments from the average of the 2 baseline assessments because no significant differences were found between the 2 baseline assessments for any measure (see Results). We tested for significantly greater improvements in the RAE group compared with the conventional therapy group using a 1-tailed, nonparametric Wilcoxon rank sum test because normality is difficult to confirm with such a small sample size. We tested for significant within-group changes using a 2-tailed Wilcoxon signed rank test. Because exercise with the RAE primarily features proximal movements, we analyzed the FM results further by separating out the distal and proximal components. We also examined the absolute scores from the Visual Analog Pain Scale at the posttherapy and 1-month follow-up assessments because individuals with stroke may have difficulty reporting accurate changes in pain over time, 41 but the absolute scores are an accurate indicator of the intensity of an individual’s current pain. 42
In addition to this analysis, we decided post hoc to analyze the effect of RAE therapy on the combined RAE group and conventional therapy group during the crossover period (n = 16). We calculated individual differences for the crossover group compared to the assessment performed 1 month after the initial conventional therapy. These changes can likely be safely attributed to exercise with the RAE because there was no significant change in the assessment values at 1 month after conventional therapy compared with the assessments performed immediately after therapy. We again tested for significant differences between groups using a 1-tailed Wilcoxon rank sum test and for significant changes within the combined RAE group using a 2-tailed Wilcoxon signed rank test.
Subacute Stroke Study in the Clinic
We also performed a small pilot study of clinical RAE therapy with individuals in the subacute phase of stroke (ie, < 6 months poststroke). Inclusion criteria were the same as for the home-based study, except for the time since stroke, which was greater than 2 weeks and less than 6 months. The primary outcome measures were the Visual Analog Pain Scale to assess arm pain and the total number of exercise repetitions recorded by the smartphone on the RAE. Secondary outcome measures were Upper-Extremity FM Score and the Modified Ashworth Scale of spasticity.
The study followed a protocol similar to the home-based study (ie, 2 baseline assessments 1 week apart, exercise for 3 h/wk over 3 weeks, a posttherapy assessment, and a 1-month follow-up assessment). The only differences were that there was no conventional therapy group and the exercises were performed under partial supervision in a clinic. That is, caregivers set the RAE up on the participants’ wheelchairs and reminded them to exercise but then let them exercise on their own.
The data from this study were not analyzed for significance, but individual results are presented in full. Changes were measured from the second baseline assessment only because baseline values were not stable for this group.
Results
We recruited 17 participants with chronic stroke to participate in the home-based study (Figure 2). One dropped out for reasons unrelated to the study. No adverse events occurred during the course of the study. There were no significant differences between the 2 baseline measurements of each outcome measure taken 1 week apart for either group. There were also no significant differences between the groups’ baseline measures, except that the RAE group had a lower initial MAL score (Table 1).
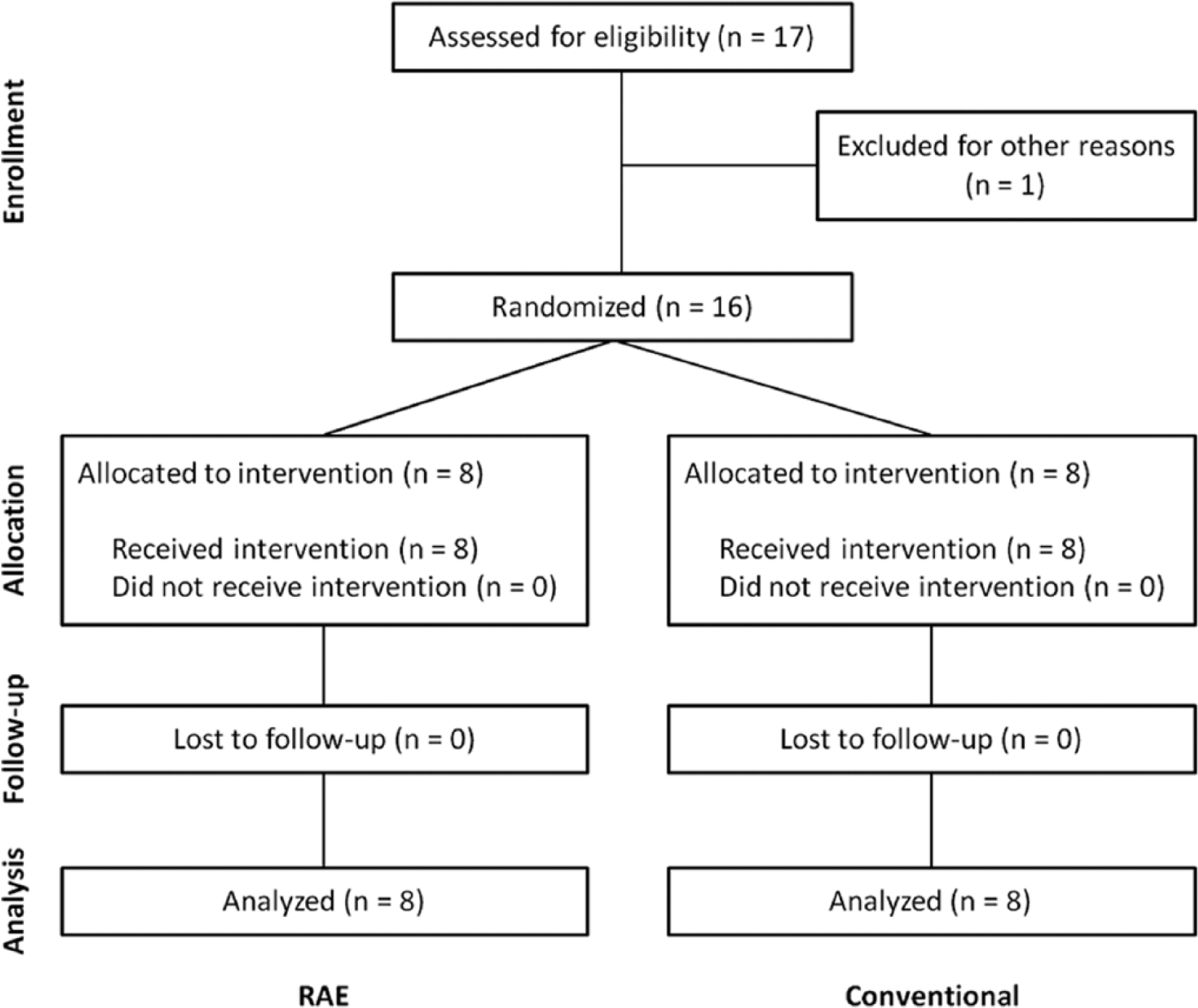
A flow of individuals participating in the study.
Participant Demographics.
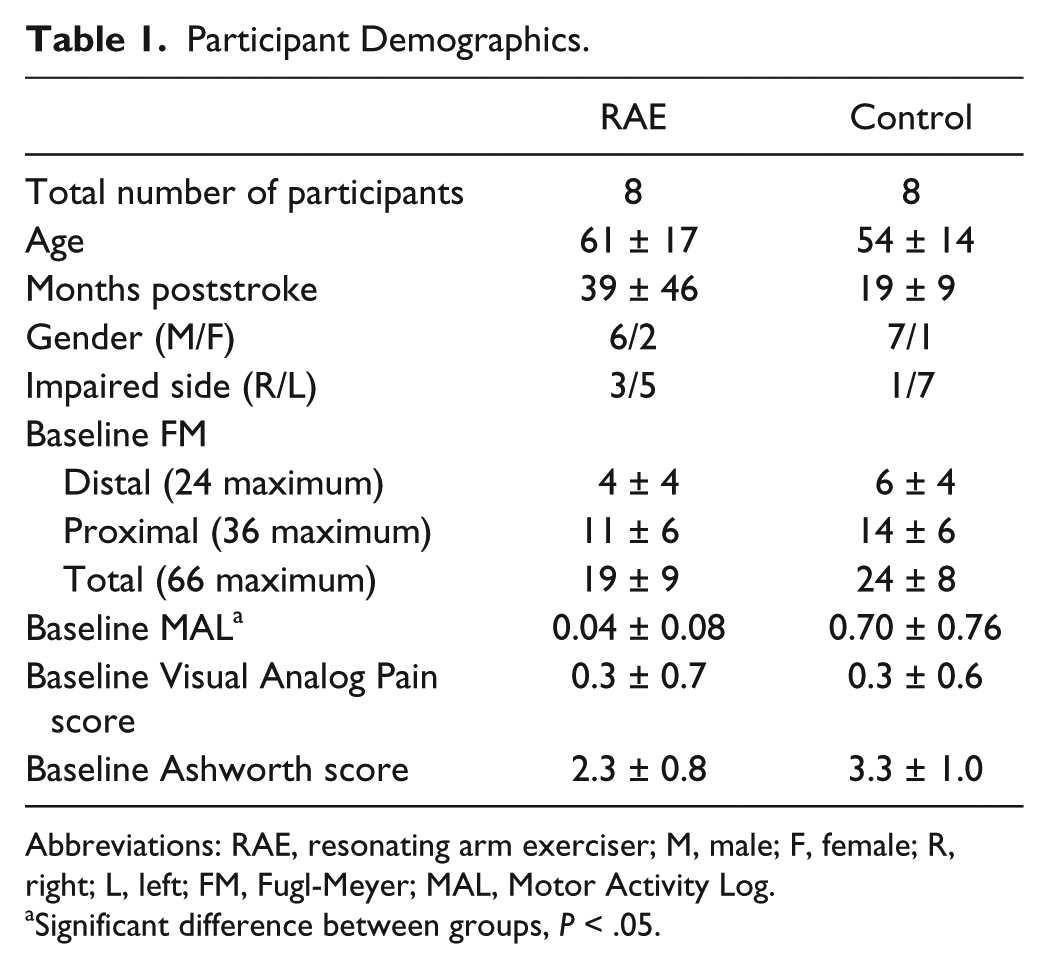
Abbreviations: RAE, resonating arm exerciser; M, male; F, female; R, right; L, left; FM, Fugl-Meyer; MAL, Motor Activity Log.
Significant difference between groups, P < .05.
A significant increase in FM score was found for both the RAE group and the conventional therapy group at the posttherapy assessment (changes of 2.6 ± 1.4 and 3.4 ± 2.4, P = .008 and .016, respectively, Figure 3). However, no significant change in FM score compared with baseline was found for either group at the 1-month follow-up assessment. At this assessment, the mean change in FM score for the RAE group was greater than that for the conventional group, with a difference of 1.56 points (95% CI = −3.50 to 6.62). However, the difference was not significant (Table 2). After crossing over to train with the RAE, the conventional therapy group had significant improvements in FM score at both the posttherapy and 1-month follow-up crossover assessments (changes of 3.9 ± 3.1 and 3.7 ± 2.7, P = .008 and .008, respectively).
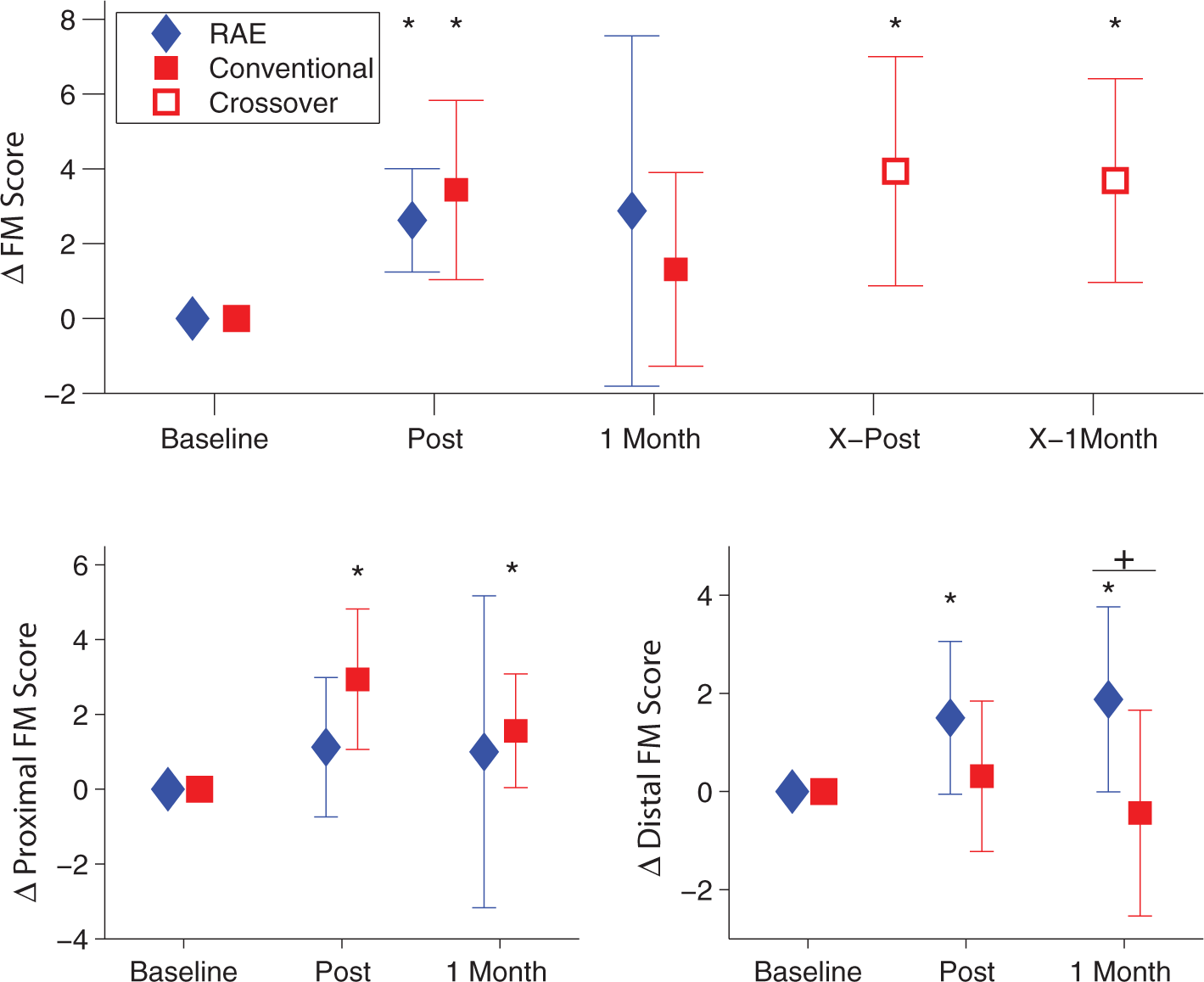
Top: change in FM score for both groups throughout the experiment (n = 8). The diamonds (RAE group) and squares (conventional) show the mean change in FM score at the postexercise and 1 month follow-up evaluations. The open squares denote the crossover period for the conventional group and mark the mean change in FM score for that group immediately after exercise with the RAE and 1 month after exercise with the RAE. Bottom: change in FM score between the 2 groups separated into proximal (bottom left) and distal (bottom right) components. There was a significantly greater increase in distal FM score for those who exercised with the RAE compared with the conventional group (P = .02, shown with a “+”).a
Home-Based Study Results. a
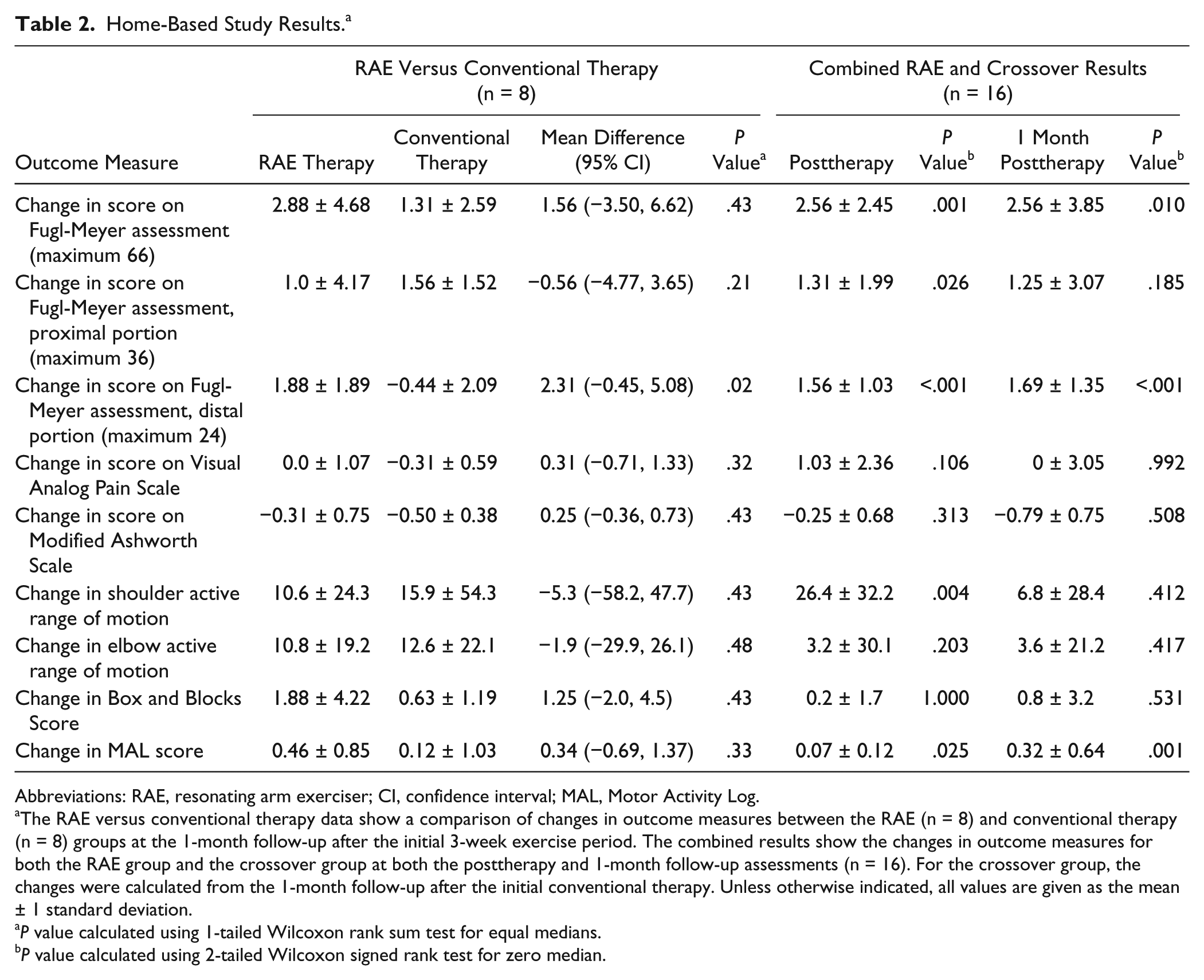
Abbreviations: RAE, resonating arm exerciser; CI, confidence interval; MAL, Motor Activity Log.
The RAE versus conventional therapy data show a comparison of changes in outcome measures between the RAE (n = 8) and conventional therapy (n = 8) groups at the 1-month follow-up after the initial 3-week exercise period. The combined results show the changes in outcome measures for both the RAE group and the crossover group at both the posttherapy and 1-month follow-up assessments (n = 16). For the crossover group, the changes were calculated from the 1-month follow-up after the initial conventional therapy. Unless otherwise indicated, all values are given as the mean ± 1 standard deviation.
P value calculated using 1-tailed Wilcoxon rank sum test for equal medians.
P value calculated using 2-tailed Wilcoxon signed rank test for zero median.
No significant difference was found between the changes in the 2 groups in secondary outcome measures at 1 month. The RAE group showed nonsignificant decreases in score on the Visual Analog Pain Scale (change of −0.69 ± 3.53 points) and Modified Ashworth Scale (change of −0.25 ± 0.89).
We further analyzed the data by separating out the distal and proximal components of the FM exam. Notably, exercise with the RAE led to significant improvements in distal FM score at both the posttherapy and 1-month follow-up assessments (changes of 1.5 ± 1.56 and 1.88 ± 1.89, P = .047 and .047), whereas the conventional exercises had no apparent effect on distal FM score, and there was a significant difference (P = .02 at 1-month follow-up, Figure 3).
When we analyzed the combined data for all participants before and after RAE therapy, we found significant increases in FM score and MAL score at both the posttherapy assessment and the 1-month follow-up (FM changes of 2.56 ± 2.45 and 2.56 ± 3.85, P = .001 and .01; MAL changes of 0.07 ± 0.12 and 0.32 ± 0.64, P = .025 and .001) and a significant increase in active range of motion at the shoulder at the posttherapy assessment (change of 26.4° ± 32.2°, P = .004, Table 2). Again, no significant increase in pain or spasticity was found after therapy. One participant reported a moderate level of arm pain after exercise with the RAE, but this did not persist at the 1-month follow-up.
Based on the data from the smartphones, participants performed an average of 383 repetitions per exercise session with the RAE and about 6000 repetitions during the entire study. However, they did not use the smartphone for every exercise session, either because they found the interface confusing or because they forgot to turn on or charge the smartphone. Based on the self-recorded exercise logs, the participants in the conventional therapy group performed about 10 hours of exercise on average during the 3-week exercise period. This translates to about 500 total repetitions, using a rough estimate of 50 repetitions per hour, which is based on the types of exercises and durations prescribed in the booklet of exercises these participants followed. Of the 5 qualities of exercises assessed by the IMI, participants found exercise with the RAE to be significantly less stressful than the conventional exercises (P < .001, Figure 4). One participant in the conventional group did not provide responses to the IMI.
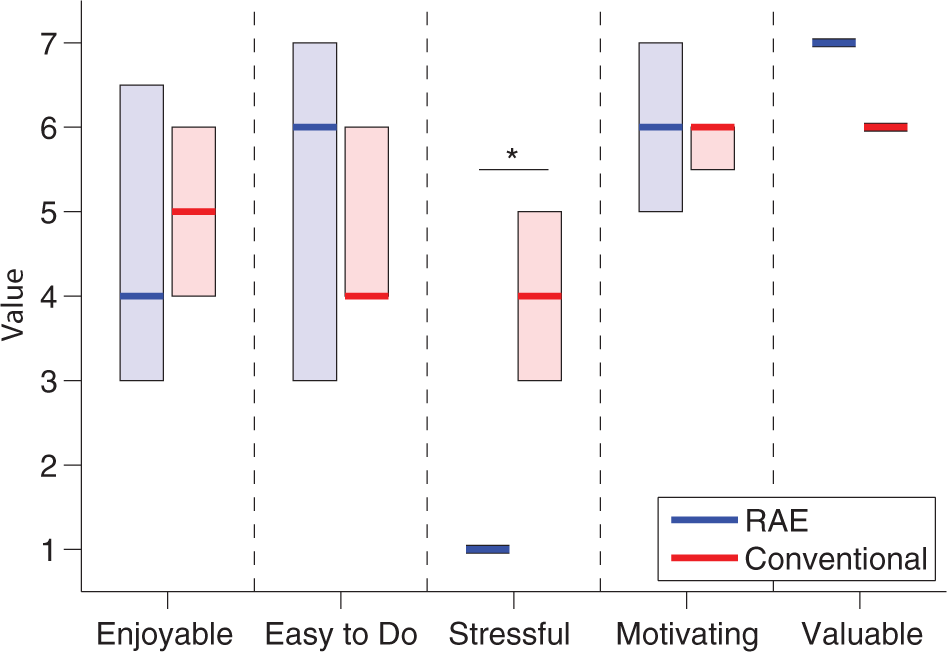
Results from IMI assessment for the RAE group (n = 8, left bars) and conventional group (n = 7, right bars). Solid lines denote the median values, and the shaded boxes show the interquartile range. Participants in the RAE group found the exercise to be significantly less stressful than conventional exercises (*P < .001).
We also recruited 4 participants with subacute stroke (3 male) to participate in the clinical pilot study of the RAE for this dynamic population. The average time since stroke was 3.3 ± 2.1 months. The participants completed thousands of repetitions with the RAE without an increase in arm pain or spasticity (Table 3).
Results of Pilot Feasibility Study With Individuals With Subacute Stroke. a
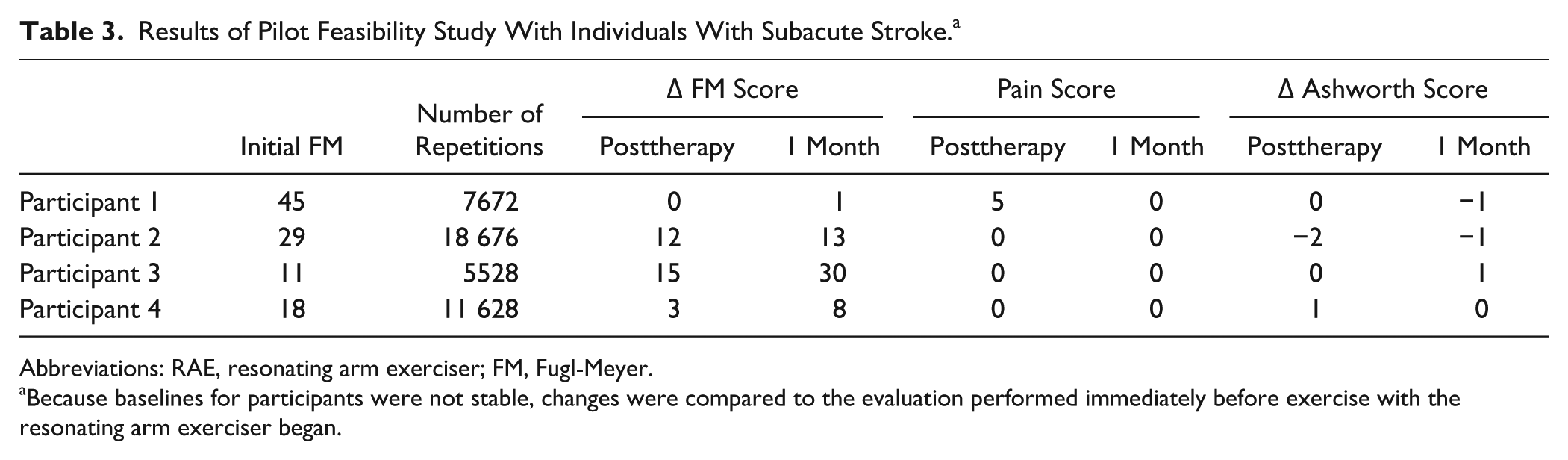
Abbreviations: RAE, resonating arm exerciser; FM, Fugl-Meyer.
Because baselines for participants were not stable, changes were compared to the evaluation performed immediately before exercise with the resonating arm exerciser began.
Discussion
Our first key question was whether exercise with the RAE is a safe, motivating, and effective method of home-based rehabilitation. The RAE did not significantly increase arm pain after exercise, and only 1 participant reported a moderate level of arm pain after exercise with the RAE. Furthermore, no adverse events occurred during the study. The IMI indicated that the participants found exercise with the RAE to be easy to do, valuable, and significantly less stressful than conventional exercises. When we analyzed all participants before and after RAE therapy, we observed a significant increase in FM and MAL scores compared with baseline at both the posttherapy and the 1-month posttherapy evaluations. These results suggest that self-guided, home-based exercise with the RAE is safe, motivating, and effective at reducing arm impairment for severely impaired individuals, although the magnitude of that reduction was moderate.
Our second question was whether home-based, assisted exercise with the RAE would lead to greater reductions in arm impairment than conventional exercises. Contrary to our initial hypothesis, exercise with the RAE did not improve outcomes compared with conventional exercises, except for distal impairment reduction. We first discuss these results as they relate to home-based therapy for individuals with severe impairments, then with respect to the current idea of task-specific rehabilitation. We conclude by addressing limitations and suggesting several directions for future research.
Machine-Based Home Therapy for Individuals With Severe Impairment
Few studies have explored home-based therapy for individuals with severe upper-extremity impairment, presumably under the assumption that these individuals have limited potential for future recovery or would not be able to perform the exercises. Yet the gains we observed in the present study are similar to those achieved by moderately impaired individuals after intensive exercises.24,43 Furthermore, participants in the conventional therapy group performed their exercises at home without human assistance, suggesting that conventional home-based therapy is feasible for this population. However, machine-assisted exercise has numerous potential advantages, including motivational aspects, the ability to measure changes, telerehabilitation capabilities, and the addition of video games, a possibility that we have begun to explore with the RAE. 44 Furthermore, the RAE group in the present study reported their therapy to be significantly less stressful than the conventional therapy group, which suggests that a machine-based approach may also reduce patient anxiety during exercise. In a separate study with nonimpaired users, the participants perceived time as moving more quickly when exercising with the RAE, highlighting again possible beneficial psychophysical effects of resonance entrainment. 45
Machine-based approaches also allow for large numbers of practice repetitions. Based on the smartphone data, the RAE group performed an average of 383 exercise repetitions per session during the study. This compares favorably with the 400 to 600 repetitions per day used in animal studies to induce plasticity12,46,47 and is an order of magnitude greater than the normal amount of repetitions performed in conventional, supervised table-top therapy. 11 Given the dose-dependent nature of rehabilitation outcomes,5,37,48,49 the ability to both perform and track such a large number of movement repetitions is notable and may help prevent learned nonuse (and a subsequent decline in mobility) after rehabilitation is complete.48,50,51 Indeed, once spontaneous recovery plateaus after 3 months,52-56 a large number of repetitions is likely required to elicit further recovery through motor learning or other plasticity mechanisms.48,49,57 The observation that the participants in the conventional therapy group likely performed an order of magnitude fewer repetitions than the participants in the RAE group during the study may then explain the significant difference in distal impairment reduction between the 2 groups at the 1-month follow up. With respect to proximal recovery, the present results suggest that a large number of repetitions of a single, stereotyped shoulder/elbow movement may produce therapeutic benefits that are comparable to what is obtained with fewer repetitions of more varied movements when the interventions are approximately time matched.
Task-Specific Rehabilitation
The results of this study also have scientific interest with respect to the idea of task-specific rehabilitation, which suggests that patients must practice the specific tasks they want to relearn for rehabilitation to be effective. Although there is substantial evidence supporting this claim,11,58-62 it may be an oversimplification because it fails to take into account neural plasticity that could generalize to a wide array of tasks. Indeed, the movements performed during exercise with the RAE are arguably nonfunctional and stereotypical (ie, users only practiced a single movement pattern, repeatedly). Yet this stereotypical movement practice led to a reduction in impairment across multiple movements as assessed by the FM scale and to a significant improvement in ability to perform activities of daily living as assessed by the MAL score, which is a valid measure of functional arm use. 63 The RAE would likely function best as part of a comprehensive, task-specific rehabilitation program that included repetitive single movement training as well as practice of whole tasks, 58 but the present results suggest that a sole focus on whole task training is incomplete, a possibility other recent studies have also begun to note.29,64-67 A key remaining question is what types of single movement training will maximize transfer to real-world tasks. Or, perhaps, the kinematics of the machine are less important than the dose of movement it provides.
Exercise with the RAE also led to distal gains in addition to proximal gains. From the task-specific rehabilitation perspective, this result is again unexpected because exercise with the RAE primarily involves the shoulder and elbow. However, the positioning of the wrist and hand that the RAE provides is out of the flexor synergistic movement pattern common after stroke, and participants may have sometimes tried to actively grip the RAE as they exercised, which may have contributed to the observed distal recovery. A similar result was also found with BATRAC: participants who performed 6 weeks of bilateral arm training showed significant improvements in a finger-tapping task that was not explicitly trained. 68 The authors of that study suggest that this was a result of the rhythmic nature of the training, which could have affected a central neural control mechanism. Because exercise with the RAE is rhythmically cued via resonance entrainment, this could also explain the result observed here. In any case, this result should be studied further because distal function is crucial for many activities of daily living, yet is difficult to practice directly after injury.
Limitations and Future Directions
Although the observed benefits of at-home exercise with the RAE are encouraging, there are limitations to the device. First, the RAE requires a wheelchair to use, which may prevent individuals who no longer rely on a wheelchair from using the device; using a chair again might also have negative psychological ramifications. However, there are many stroke survivors who do regularly use wheelchairs (eg, >70% of subacute patients 69 ). If a potential user does not own a wheelchair, they must purchase one, but basic chairs only cost about $100, and in our experience, the individuals who did not regularly use wheelchairs did not mind using a wheelchair as an arm exercise device. Second, the type of floor the wheelchair was placed on affected the operation of the device by either increasing friction or causing the wheels to slip; this was a solvable problem for all our participants, but it did lead to some initial frustration. Third, clinicians and users desired the ability to move the wheelchair while the RAE was attached, which is not possible with the present design. Fourth, although participants did not get motion sickness, some reported being annoyed by the movement of the chair.
We have recently addressed these issues by coupling the lever to the wheel through a custom transmission that allows the lever to be rotated independently of the wheel (thus eliminating movement of the wheelchair and floor-surface dependence). By weighting the lever, we still achieved similar resonance-based assistance without rolling the chair. This transmission also includes a 1-way bearing that, when engaged, allows individuals to ambulate overground with the device. The resonance of the lever reduces the average force needed to roll forward by about 40%. 44 In pilot studies, we found that even severely impaired individuals were able to ambulate at least 100 feet in a straight line using this lever drive. 70
Other limitations of the study include use of self-report to measure the dose of the therapeutic interventions, a relatively small sample size, and limited improvement in functional ability. Finally, further studies are needed to fully understand the motivational aspects of the RAE.
The present study also explored whether the RAE could be an appropriate tool for providing arm therapy in individuals with subacute stroke (ie, <6 months poststroke), who may have a greater potential for use-dependent plasticity.55,71,72 The RAE was indeed a safe tool for the participants in this population because none reported increased arm pain, experienced motion sickness, or exhibited increased spasticity after thousands of repetitions with the device. A larger study is needed to examine the therapeutic benefits of exercise with the RAE for this population, but the ability to use it with minimal therapist supervision makes it an attractive tool for providing an early-stage, high-repetition, quantifiable therapeutic intervention, similar to what was done with a rocking chair and air splint, to positive benefit.29,30 Future studies should also directly examine the potential negative psychological ramifications of asking individuals who are recovering from stroke to return to a wheelchair if they no longer use one for mobility.
Conclusion
The RAE was a safe and effective device for individuals with severe impairments to use at home that in this study led to a reduction of arm impairment comparable to conventional exercise. Because of its simplicity, exercise with the RAE is limited to the practice of a single movement pattern, but this movement pattern is an important one, and repeating it a large number of times led not only to proximal impairment reduction but also to greater improvements in distal movement ability than conventional exercises. The RAE would perhaps best be used as an add-on to existing home-therapy programs, rather than as a replacement for them, as a method of increasing the dose of a particular exercise. Indeed, for individuals with severe impairments, the options for rehabilitation are limited, and any additional tool they can use to exercise will increase their opportunities for recovery.
Footnotes
Acknowledgements
Thank you to the UC Irvine Institute for Clinical and Translational Science for their support in the study.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Daniel Zondervan, Nizan Friedman, and David Reinkensmeyer have a financial interest in Flint Rehabilitation Devices, LLC, a company that develops rehabilitation devices. The terms of this arrangement have been reviewed and approved by the University of California, Irvine, in accordance with its conflict of interest policies. The remaining authors declare that they have no competing interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by the Department of Education through NIDRR grant H133S120032. This study is registered at ClinicalTrials.gov under identifier NCT01769326.
