Abstract
Introduction
In response to injury, the human brain possesses a remarkable ability to preserve or regain function.1-4 Two means by which this is accomplished are through functional reorganization and compensation. Functional magnetic resonance imaging (fMRI) has emerged as a valuable tool to observe this reorganization and compensation poststroke.5-7 For example, fMRI maps were more strongly correlated with behavioral outcome than total infarct volume determined by structural MRI. 8 Hence fMRI may be useful in the management of stroke patients during rehabilitation.
Typically, fMRI studies mainly focus on identifying brain regions that exhibit an increase in neuronal activity during the execution of a task or in response to a stimulus, in comparison to a baseline or control state. In fMRI studies of stroke resulting in motor deficits, hand and finger movements are often tasks of choice because impairments commonly involve the hand.9-11 Specifically, repetitive wrist and finger flexion elicit activity within cortical and subcortical regions involved in movement planning, initiation, execution, and control; these regions include the motor and sensory cortices, basal ganglia, brainstem, and cerebellum. 12
Another imaging approach examines the temporal fluctuations of the fMRI signal to identify brain regions that act in synchrony during task execution rather than identifying regions that simply become activated. The degree of synchrony among the brain regions is thought to indicate the strength of task-related functional connections. 13 During unilateral hand movements, within-hemisphere connections of the healthy brain act to facilitate activity within the contralateral cortex; across-hemisphere connections may inhibit activity in the ipsilateral cortex.14-16
Task-related fMRI, however, has not emerged in routine clinical assessment of stroke and its recovery. Results obtained from fMRI are highly dependent on patient effort during the performance of specific tasks that may not even interrogate all brain regions of interest. In addition, fMRI is limited to those who can perform the tasks; that is, patients with more severe deficits are often contraindicated for fMRI because they will not generate fMRI signals that can be measured. Changes in brain activity magnitude over repeated sessions may also be task specific and dependent on consistent patient effort and movement strategy over all sessions, which is unlikely for many stroke patients. In fact, even for simple hand movements and cognitive tasks, brain activity magnitude decreases in healthy controls over repeated imaging sessions, 17 perhaps because they become acclimatized to the MR environment. Finally, because the variability of fMRI activity magnitude over repeated sessions may be high, a detectable difference over imaging sessions may prove difficult for clinical application in individual patients. 18 Hence the reliability of single-patient fMRI maps of brain activity may not be sufficient for purposes of clinical application.
In the absence of a task—that is, during rest—temporal synchrony has also been demonstrated between distinct brain regions, consistent with our current understanding of anatomic connections within the brain.19,20 This includes regions involved in motor function, both within and across hemispheres.
21
These anatomic patterns of the resting state have been termed
Investigations of these resting-state networks in patient groups other than stroke patients have recently surfaced. Disruption in prefrontal, inferior temporal, and premotor resting-state connectivity (whether it is increased or decreased relative to normal) has been demonstrated in schizophrenia patients 28 as well as disruptions within the “default-mode” network,29,30 a collection of association regions that include the posterior and ventral anterior cingulate cortices. 31 Similar findings within the default-mode network have been observed in Alzheimers disease 32 and major depression. 33 Hence, alterations in the resting state appear to be associated with symptoms and deficits.
Resting-state connectivity studies have been reported in stroke populations34,35 that reveal disruptions in interhemispheric functional connectivity within attention and motor networks.36,37 Although cross-sectional studies may help determine the association between connectivity and motor performance, only repeated assessments of the same patient over days or weeks will successfully define the changes in connectivity associated with recovery. To address this, a recent longitudinal study of 10 young stroke patients (41-55 years of age) demonstrated that the reorganization of functional connections among preselected motor regions is less optimized following the recovery of moderate-to-severe motor deficits; that is, recovery was associated with a redistribution of motor function throughout the network rather than focused within a few regions, as is typically observed in the healthy brain. 35 In addition, the change in the strength of functional connections was found to correlate with the change in clinical assessment of motor function.
For resting-state fMRI to be truly useful for clinical investigations, it must be feasible to include resting-state fMRI in an existing acute stroke imaging protocol within the first hours poststroke, such that resting-state fMRI can contribute to clinical management. Subsequent imaging sessions would then be relative to this early time point for each patient, rather than a session conducted days or even weeks poststroke.35,37 This timing could greatly improve the determination of the dynamics of functional connections associated with recovery. For example, motor stroke patients require considerable effort to execute even simple hand movements. A recent fMRI study demonstrated that regions of the basal ganglia are associated with mental and physical effort. 38 Thus, it is possible that there may be an interaction between these regions and cortical regions of the motor network, either at the acute stage or during recovery, if more effort is required to execute the recovered movements.
We conducted a study in which we hypothesized that interhemispheric resting-state connectivity of the motor cortices will be reduced at less than 24 hours poststroke in patients with unilateral motor deficits compared with healthy participants as well as stroke patients with no motor deficits. Furthermore, we predicted that this disrupted connectivity between motor regions would be reestablished after complete recovery, but reduced functional connections remain between cortical motor regions and subcortical regions associated with effort. If these hypotheses are confirmed, it will demonstrate the potential of resting-state fMRI to understand the impact of stroke on the function of brain networks within hours of stroke and to assess and monitor the recovery of brain function over weeks and months. Such a tool could also have a significant impact on rehabilitation strategies and possibly on how health care resources are allocated.
Materials and Methods
Participants
This study was approved by the Conjoint Health Research Ethics Board of the University of Calgary. Written informed consent was obtained from all participants prior to their participation. A total of 36 patients (18 men, 18 women; mean age, 67 ± 15 years) with first-ever ischemic stroke causing unilateral motor weakness and/or paresthesia (15 left hand, 21 right hand) were enrolled in the study. Stroke was confirmed by routine clinical X ray computed tomography images and neurological assessment. Deficits were confirmed by a score of greater than 0 on the motor subscale of the National Institutes of Health Stroke Scale (NIHSS), as determined by clinical neurological assessment. Also, 23 additional ischemic stroke patients (9 men, 14 women; mean age, 67 ± 13 years) with nonmotor deficits were enrolled as a comparative control group. In addition, 15 right-handed healthy participants were enrolled (7 men; 8 women; age, 64 ± 9 years) as another control group.
Image Acquisition
Imaging was performed using a 3-T MR scanner equipped with an 8-channel phased-array head coil (Signa Excite; GE Healthcare, Waukesha, Wisconsin). Foam padding was placed around the participant’s head to restrict head motion during scanning. Stroke patients were invited to participate in 3 imaging sessions (at <24 hours, 7 days, and 90 days). T2-weighted, flow-compensated, fast spin echo images with a phase acceleration factor of 2 (ASSET; GE Healthcare, Waukesha, Wisconsin; repetition time (TR)/echo time (TE) = 5200/102 ms; echo train length = 12; 24 × 24 cm2 field of view; 512 × 256 matrix; 5-mm thick slices numbering 28; receiver bandwidth = 31.25 kHz ) and fluid-attenuated inversion-recovery images (TR/TI/TE = 9000/2250/140 ms; 24 × 24 cm2 field of view; 256 × 192 matrix; 5-mm thick slices numbering 27; receiver bandwidth = 31.25 kHz) were also collected as part of a routine clinical MR imaging protocol. These images were used for anatomic registration of the resting-state data. Diffusion-weighted images with fat suppression were also acquired using a dual spin echo sequence (TR/TE = 16 000/73.1 ms;
Analysis
Using each participant’s anatomic images, a region was defined within each hemisphere using the drawing tool of FSLview, part of FSL (FMRIB’s Software Library, http://www.fmrib.ox.ac.uk/fsl), to include the primary motor cortex (ie, the precentral sulcus—Brodmann area [BA 4]) and the primary sensory cortex (the postcentral gyrus—BAs 3, 1, and 2). This region extended from the lateral surface to the midline and from the vertex of the brain to the level of the superior aspect of the lateral ventricles caudally. 39 This area was then reduced to a volume of 10 cm3 using a procedure based on intravoxel temporal cross- correlation of the resting-state data, 40 which defines a region of contiguous image voxels whose resting-state signals are the most synchronous. The final region in each hemisphere was defined as the primary and sensorimotor cortex (SM1).
Resting-state images were preprocessed using FEAT (FMRI Expert Analysis Tool), version 5.4, part of FSL. Preprocessing steps included motion correction using MCFLIRT,
41
slice timing correction to the center slice, non–brain removal using FSL’s Brain Extraction Tool (BET),
42
6-mm spatial filtering, high-pass temporal filtering (Gaussian-weighted least-squares straight line fitting, with σ = 50.0 s), and grand mean intensity normalization. Images were then entered into a general linear model using the estimates of motion as regressors, resulting in a set of residual images with movement-related intensity changes from the fMRI time series removed.
43
Interhemispheric SM1 connectivity was calculated relative to the connectivity of ipsilesional SM1 with itself (ie, relative connectivity or RelCon),
44
which has been shown previously to be independent of image signal-to-noise ratio and is more sensitive to the presence of reduced connectivity. In this method, the temporal cross-correlation between each and every possible interhemispheric SM1 voxel pairs is computed, and the Pearson correlation coefficients are averaged after Fisher transformation. The same is computed for all voxel pairs within ipsilesional SM1. Interhemispheric resting-state SM1 connectivity is then computed as the average interhemispheric correlation coefficient divided by the average within-ipsilesional SM1 correlation coefficient. Connectivity was then compared between stroke groups and with healthy controls using Student
In addition to the region-of-interest approach, a whole-brain voxel-by-voxel analysis was performed for each participant using the average time course from ipsilesional SM1 as the signal of interest in a general linear model. A group voxel-based analysis across participants was then conducted using a general linear mixed model after transformation of the image data into the standardized space of the Montreal Neurological Institute brain using FLIRT. This analysis generated whole-brain connectivity maps for each of the motor stroke, nonmotor stroke, and healthy participant groups as well as a difference map between the motor-stroke group and healthy controls. The left and right hemispheres were exchanged for some patients, so that the ipsilesional hemisphere was common to all patients for group analysis. All maps were generated at a corrected
Results
Data from 5 motor stroke patients and 3 nonmotor stroke patients were excluded from group analysis because of excessive head motion during scanning. As a result, data from 31 motor stroke patients (16 men, 15 women; mean age, 65 ± 15 years; 15 left-side deficit, 16 right-side deficit; time of imaging poststroke, 13 ± 7 hours) and 20 nonmotor stroke patients (8 men, 12 women; mean age, 65 ± 12 years; time of imaging poststroke, 14 ± 7 hours) were analyzed. The stroke patient groups did not differ in age (
Motor stroke patient characteristics are summarized in Table 1, and the locations of stroke for all patients are depicted in Figure 1. Of the 31 patients with a motor deficit, 17 had subcortical or white matter lesions, 11 had cortical lesions, and 3 had cerebellar or brainstem lesions. Interhemispheric SM1 connectivity was 0.80 for motor stroke patients, which was significantly less than the value of 0.94 for healthy controls (
Characteristics of Patients With Motor Stroke (Dashes Indicate a Patient Did Not Participate)
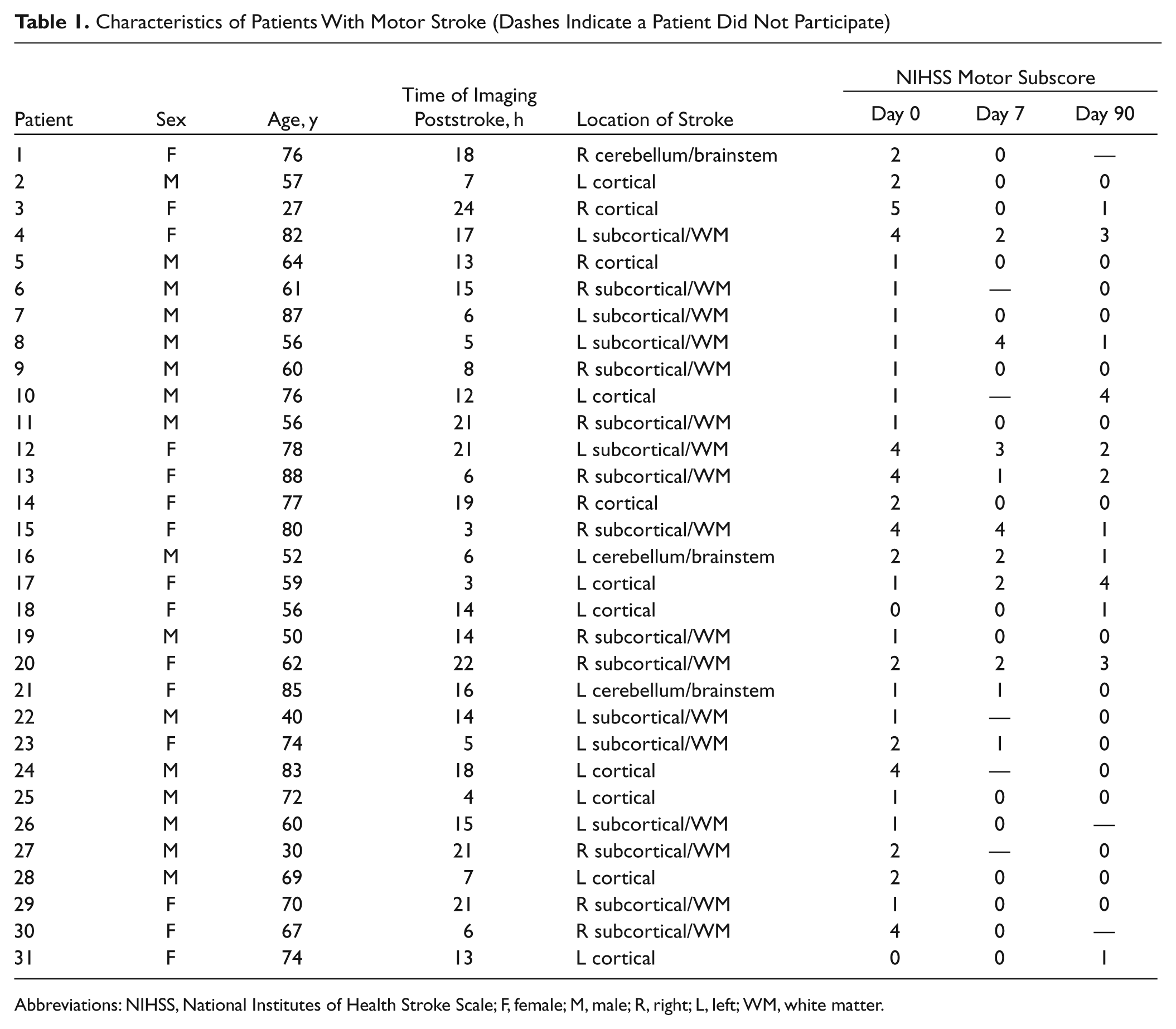
Abbreviations: NIHSS, National Institutes of Health Stroke Scale; F, female; M, male; R, right; L, left; WM, white matter.
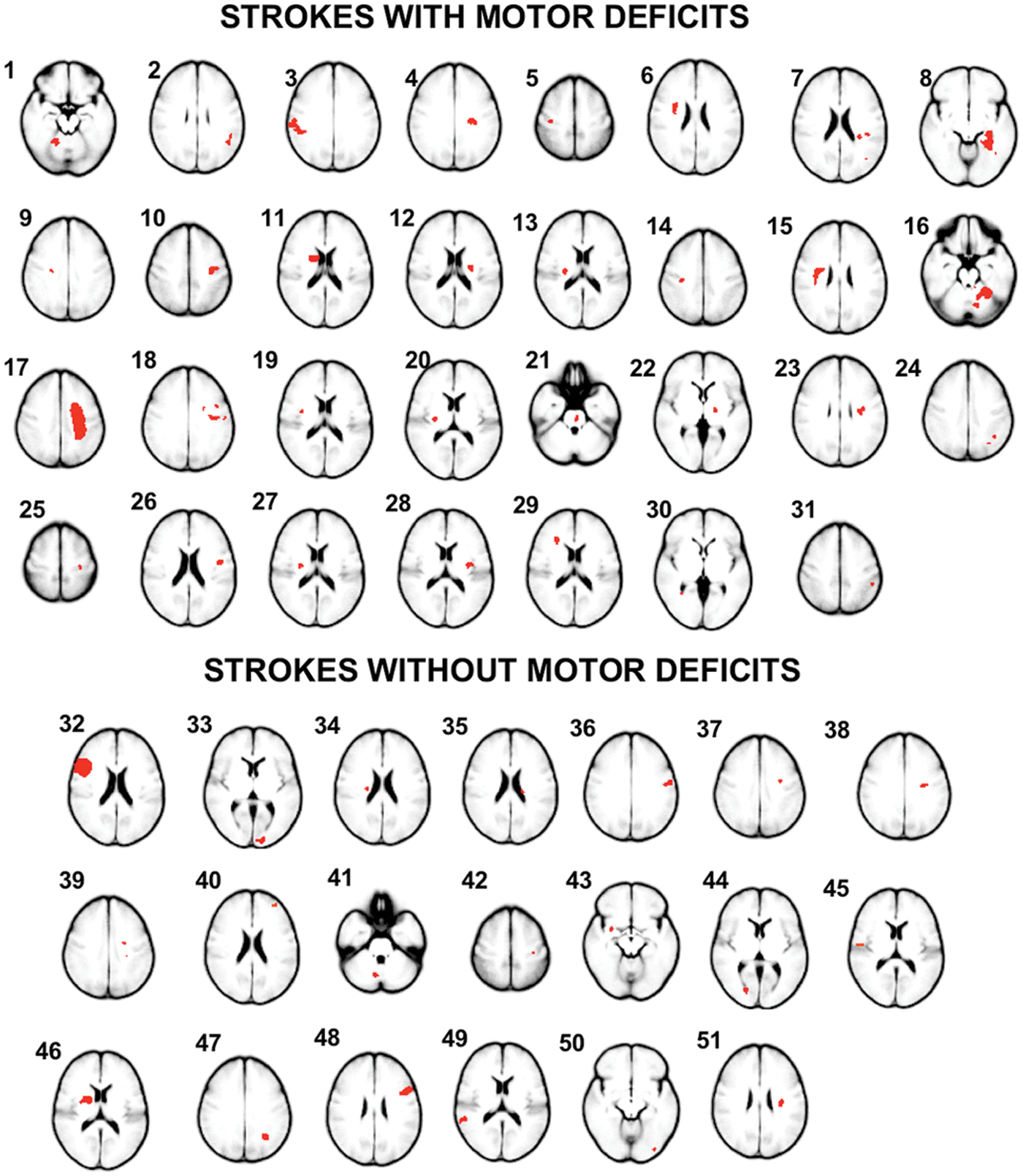
Images depicting the location of ischemic stroke (in red) in patients with motor deficits (top) and without motor deficits (bottom), as determined from acute diffusion-weighted imaging.
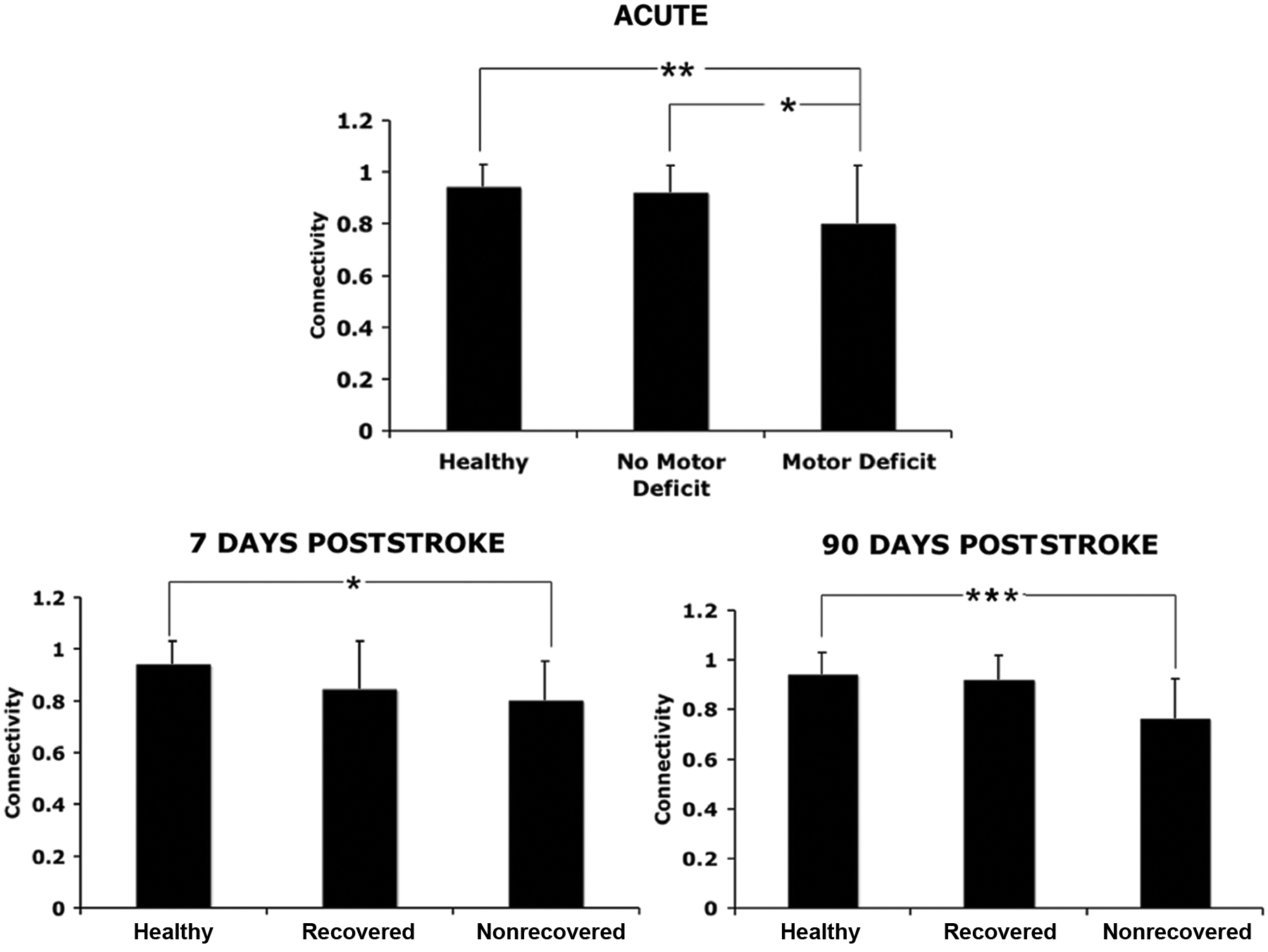
Top: interhemispheric SM1 connectivity during acute stroke (<24 hours); patients with motor deficits exhibit significantly lower connectivity than healthy controls (**
At day 7, 28 patients who had acute motor deficits and 17 patients who had no acute motor deficits participated in the scanning session. Of the patients who had acute motor deficits, 18 had recovered (ie, NIHSS motor score of 0), and 10 still exhibited an NIHSS motor score greater than 0. As Figure 2 also shows, connectivity in recovered stroke patients did not differ from that in healthy volunteers (
At 90 days, 19 patients who had acute motor deficits and 11 patients with no motor deficits participated in the scanning session. Of the 19 patients who had acute motor deficits, 12 had recovered, and 7 still had a NIHSS motor score greater than 0. As Figure 2 also shows, recovered stroke patients did not differ from healthy volunteers (
Figure 3 shows brain regions exhibiting significant connectivity for healthy controls, acute nonmotor stroke patients, and acute motor stroke patients. These brain regions are summarized in Table 2 for healthy controls and motor stroke patients (nonmotor stroke patients exhibited no significant difference from healthy controls). Healthy controls and acute nonmotor stroke patients demonstrate the same and typical pattern of connectivity—that is, there were strong connections with the contralateral motor cortex as well as with bilateral subcortical regions, including the motor thalamus. For acute motor stroke patients, however, connectivity with the contralateral motor cortex appears reduced, and there were no significant connections with subcortical regions. The ratio of significant right-to-left sensorimotor cortex volume was 0.92 for healthy controls and 0.94 for nonmotor stroke patients, demonstrating a bilateral pattern of connectivity between the 2 hemispheres. For motor stroke patients, however, this ratio was 0.51, demonstrating a more unilateral and within-hemisphere pattern of connectivity. Figure 3 also shows brain regions exhibiting significantly greater connectivity in controls relative to acute motor stroke patients. These brain regions are also summarized in Table 2. Controls possess stronger connectivity with contralateral sensorimotor and premotor cortices as well as the contralateral thalamus and putamen.
Brain Regions Exhibiting Significant Connectivity With Left Sensorimotor Cortex at Less Than 24 Hours for Motor Stroke Patients as Well as for Healthy Controls
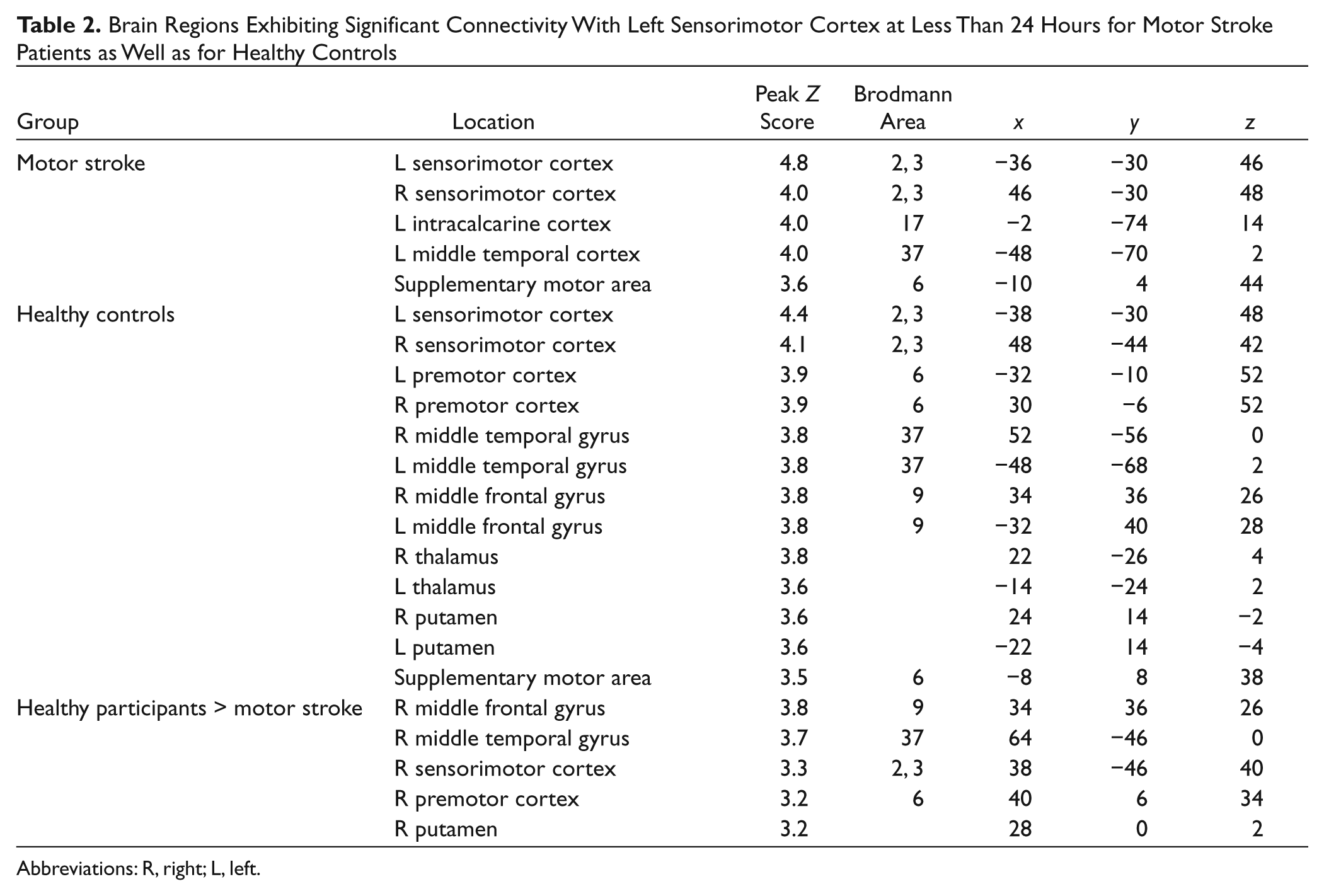
Abbreviations: R, right; L, left.
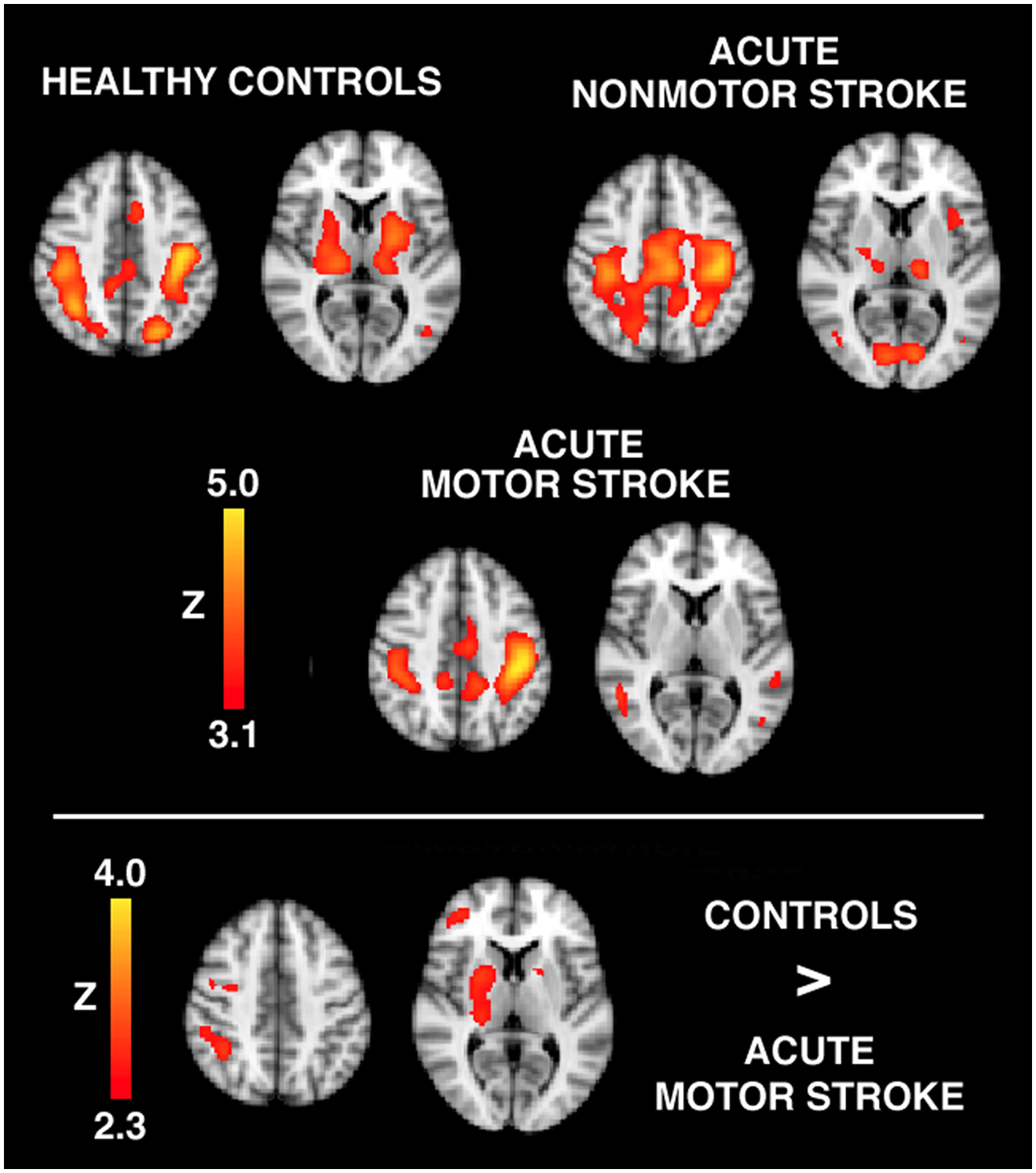
Top: whole-brain maps of connectivity with left SM1 at the acute stage of stroke for slice locations through regions of the motor network; the ipsilesional hemisphere is on the left. The color bar indicates the significance of the correlation with left SM1, expressed as a
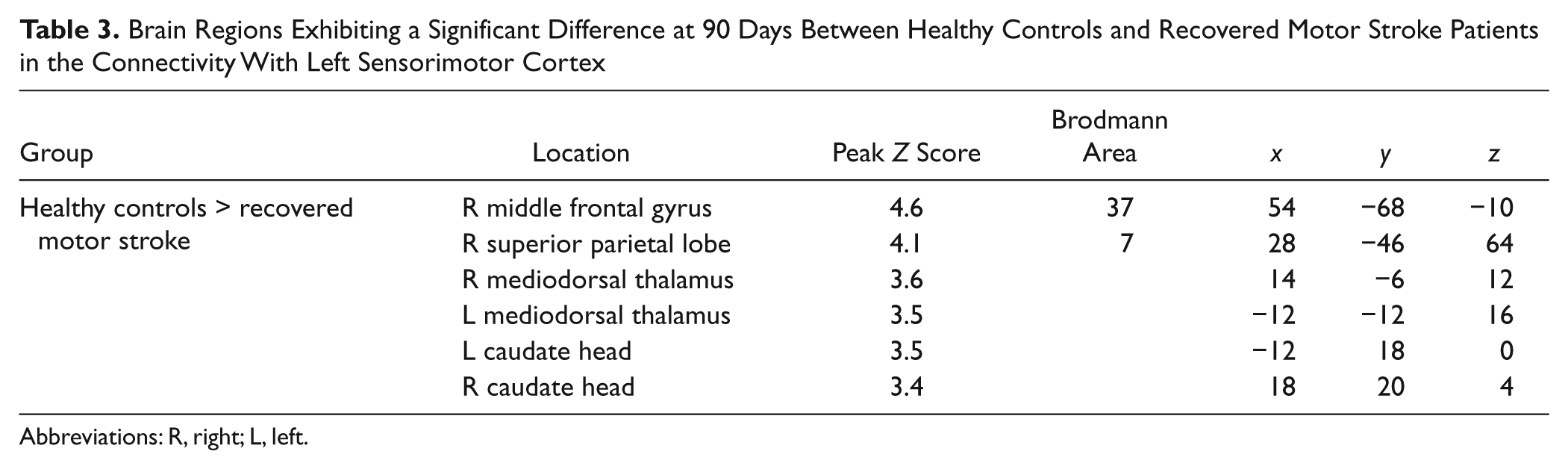
Figure 4 shows brain regions exhibiting significant connectivity for motor stroke patients who had recovered their function by 90 days and those who had not, in comparison with healthy controls. These brain regions are summarized in Table 3. Recovered motor stroke patients exhibit a bilateral pattern of connectivity in the motor cortices as well as in the thalamus, similar to controls; however, there was an absence of connectivity with the bilateral caudate and putamen. Nonrecovered motor stroke patients, however, still exhibited reduced connectivity with the contralateral motor cortex; however, connectivity with the bilateral thalamus appears to be restored. When directly comparing controls to recovered patients in a whole-brain analysis (see also Figure 4 and Table 3), controls exhibited greater connectivity with the head of the caudate as well as bilateral mediodorsal thalamus.
Brain Regions Exhibiting a Significant Difference at 90 Days Between Healthy Controls and Recovered Motor Stroke Patients in the Connectivity With Left Sensorimotor Cortex
Abbreviations: R, right; L, left.
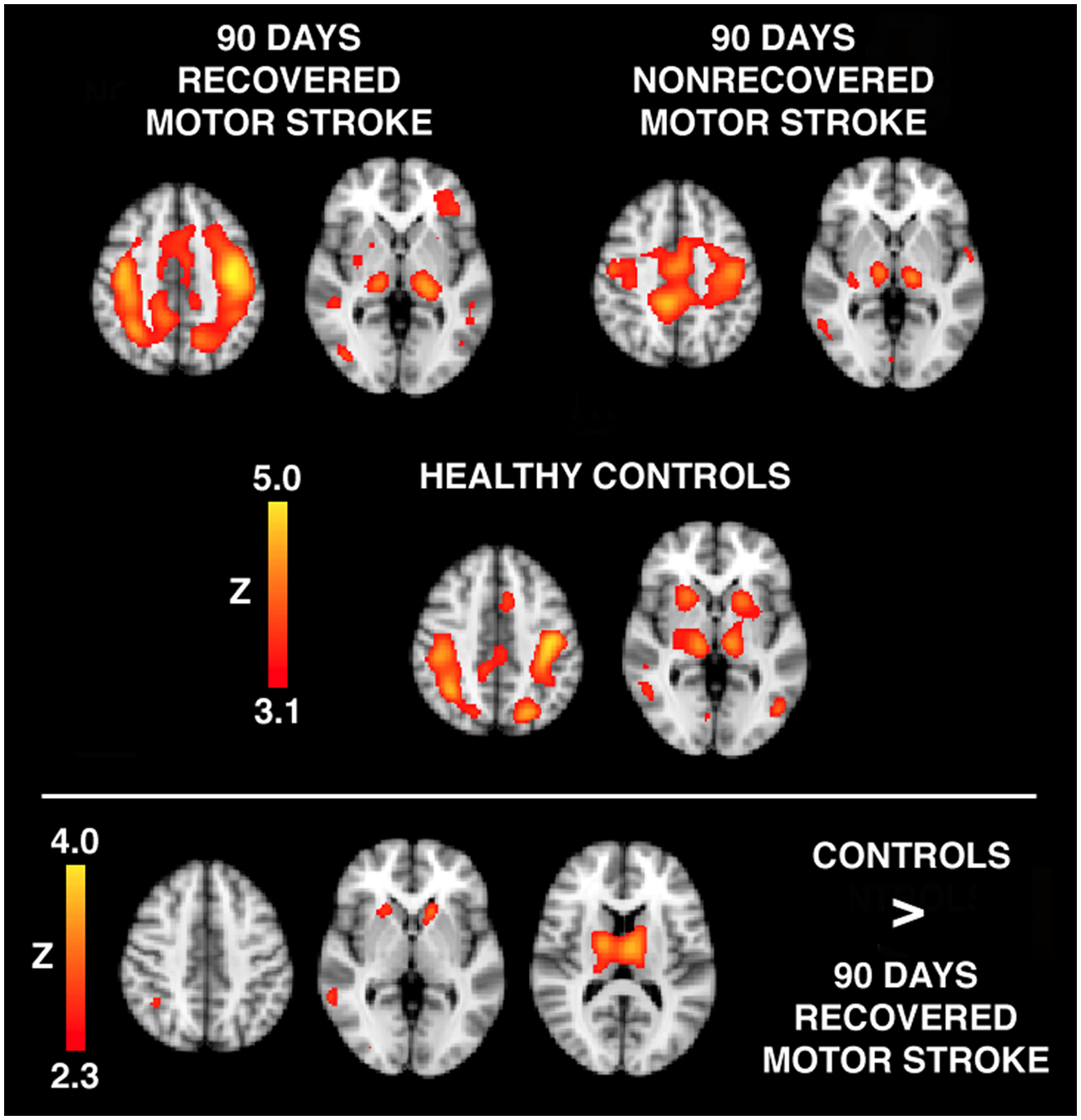
Top: whole-brain maps of connectivity with left SM1 at 90 days poststroke for slice locations through regions of the motor network. The ipsilesional hemisphere is on the left. The color bar indicates the significance of the correlation with the left SM1, expressed as a
Discussion
Our findings demonstrate that acute ischemic stroke patients experiencing mild-to-moderate motor deficits exhibit reduced interhemispheric resting-state connectivity between the motor cortices, in agreement with previous studies.35,36 Our findings also demonstrate that there are changes in functional connectivity associated with recovery, consistent with previous reports.35,37 Our study, however, demonstrates the utility of resting-state fMRI within hours poststroke. This has important clinical implications because an assessment of the early connectivity changes associated with stroke has the potential to affect stroke treatment and provides a more accurate baseline with which to compare the efficacy of rehabilitation.
In our study, we included healthy, age-matched controls because aging has been demonstrated to affect resting-state connectivity within the motor network.45,46 This is possibly the result of white matter degradation with age, 47 leading in turn to the recruitment of additional brain regions or activity to compensate for reduced motor control function often observed in older individuals.48-49
Unlike previous studies, we also demonstrated that interhemispheric resting-state connectivity between the motor cortices is not affected by nonmotor stroke deficits. Our whole-brain connectivity analysis also demonstrated that even though patients may recover motor function, there remains reduced resting-state connectivity between sensorimotor cortex and subcortical regions—namely, bilateral caudate head and anterior thalamus. These regions have been associated with mental and physical effort in healthy individuals. 38 In addition, the caudate head has been associated with executive functioning50,51 and the anterior thalamus with episodic memory. 52 Brain regions associated with cognitive function have not been implicated in previous longitudinal studies of motor functional connections in stroke patients.35,37 Our results suggest that higher level cognitive processing and effort associated with motor functions remain affected after recovery and play an important role during recovery; this may not be evident on neurological examination. Hence whole-brain or multinetwork analysis may be necessary to fully describe the dynamic nature of functional reorganization or recovery of connections in the brain poststroke, rather than restricting analysis to predefined motor regions as in a recent study. 35 Furthermore, initial assessment of functional connections days or weeks poststroke 37 may not be sufficient to capture the cognitive and effort contributions to recovery. Our findings also suggest that full neuropsychological assessment may be required in future studies to identify the existence of cognitive deficits related to motor functions.
The present study had a number of limitations. No task-related fMRI was performed at the time of study as a means to determine the relationship between brain activity during motor tasks and resting-state connectivity. In addition, no assessment of motor function other than NIHSS was performed. A NIHSS score of 0 does not necessarily mean that patients are fully recovered. Patients may still experience deficits in grip strength and finger dexterity that are not assessed by the NIHSS motor score. Comprehensive motor assessment was not feasible for the current study; we were afforded the opportunity to implement resting-state fMRI into an MR imaging protocol for the assessment of stroke within the first hours. Hence the purposes of the current study were to demonstrate the feasibility of short-duration resting-state fMRI within a clinical stroke imaging protocol and to determine if brain regions not previously implicated during recovery can be identified when acute assessment is available. Now that the feasibility and utility of resting-state fMRI have been demonstrated for motor stroke patients, a comprehensive investigation of the interrelationship between behavioral assessment of motor as well as cognitive function, task-evoked brain activity, and the resting state in the presence of stroke is certainly warranted.
Because our stroke patient population included those with cortical, subcortical, and cerebellar strokes, we could not definitively determine the differential impact of type and severity of stroke on resting-state connectivity. Hence from our current results, we cannot fully determine the mechanisms underlying reduced resting-state connectivity following stroke as well as its impact on brain function and patient behavior; further study is required.
In conclusion, resting-state connectivity is reduced at less than 24 hours in ischemic stroke patients experiencing motor impairments and resolves as these resolve. Our findings also suggest a cognitive component to recovery and that the assessment of cognitive function and cognitive brain networks should be included. These findings have potential applications for studies from stroke onset through rehabilitation because resting-state connectivity alleviates the need for patients to perform tasks. This removes the influence of patient effort and changes in movement strategy on fMRI findings. Resting-state studies also permit the assessment of all stroke patients, not just those who can perform motor tasks adequately, consistently, and without excessive head movement.
Footnotes
Acknowledgements
The authors would like to thank the Heart and Stroke Foundation of Alberta, NWT and Nunavut for funding the VISION-2 study.
Authors’ Note
The VISION-2 Study Group comprises the following: the Department of Clinical Neurosciences (A. Y. Poppe, S. Tymchuk, C. O’Reilly, T. Green, S. Idris, E. Stewart, C. Stephenson, C. Kenney, P. N. Sylaja, M. D. Hill, S. B. Coutts, and A. M. Demchuk) and the Department of Radiology (P. Sharma, M. Eesa, and M. Goyal) in the Seaman Family MR Research Centre, Foothills Medical Centre of the Calgary Health Region and the University of Calgary, Calgary, AB, Canada.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grant funding from the Canadian Institutes for Health Research (MOP-118096) and the Heart and Stroke Foundation of Alberta, NWT and Nunavut. Acute stroke imaging was also supported by the Alberta Heritage Foundation for Medical Research.
