Abstract
Introduction
Different patterns of reorganization have been described during the acute and subacute phases of stroke in functional MRI and positron emission tomography studies. These include perilesional extension of activation, shifts from primary to secondary motor areas, and recruitment of motor areas of the nonaffected hemisphere.1-9 Using specific rehabilitation approaches in patients with chronic stroke, most studies have emphasized activation changes within the primary sensorimotor cortex (SMC) and premotor regions, with improvements that vary partly in relation to damage of the corticospinal tract.10-12 Fewer studies have focused on the selective role of the somatosensory cortex (S1) in motor recovery,13-15 but correlation analysis of the observed activation shift toward S1 in relation to increases in hand function have not been unsuccessful.14,15 It was suggested that the posterior shift of activation toward S1 has no clinical relevance. 13 To date, only 1 study correlated activation change patterns within the secondary somatosensory cortex and ipsilesional premotor cortex with improved hand function following a modified constraint-induced movement therapy (CIMT). 16
The lack of a relationship between activation within the S1 and motor score may be a result of several factors. Most of the patients studied had both paresis and numbness to differing extents. Therefore, divergent results were obtained from inhomogeneous patient groups. Furthermore, diverse imaging paradigms and tests of hand function were used, and several studies failed to perform a correlation analysis between the functional test and imaging brain activation.13-15 The present study aimed to investigate the association of changes in functional MRI (fMRI) activity within the S1 with improvement in hand function. Our study was designed to address important considerations. We selected chronic stroke patients with hand weakness but normal sensory examination. As the improvement of hand function can be influenced by spontaneous recovery 17 within the first months following stroke, we focused on chronic stroke patients with no motor improvement during the previous 3 months before beginning the study by testing hand function 3 months earlier. To intensively exercise the impaired hand, participants were trained with CIMT. fMRI activity of the sensory network was induced by a passive hand movement before and after CIMT. The advantage of using this paradigm is based on its independence from individual performance and clinical improvement. Consequently, it can be assumed that activation changes between baseline and post-CIMT (ΔfMRI) are induced by the training. Finally, to relate fMRI activity changes (ΔfMRI) to changes in hand function (Δtest), a correlation analysis between these parameters was performed. 18
Methods
Patients
Patients were recruited by advertising in newspapers in the context of general information about stroke therapy, by contacting local doctors, and by talking at local stroke groups. Our careful selection process took 3 years and screened 78 potential participants. Table 1 details the characteristics of our participants.
Characteristics of the 7 Selected Patients
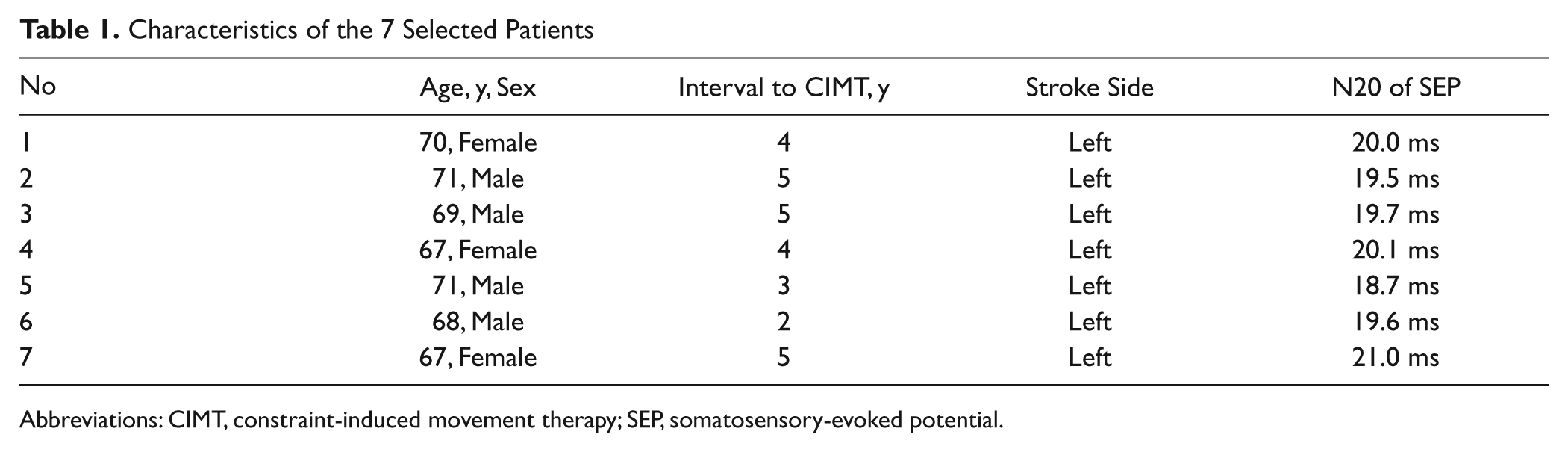
Abbreviations: CIMT, constraint-induced movement therapy; SEP, somatosensory-evoked potential.
Participants had to meet 5 criteria: right-handed, according to a modified Oldfield Handedness Questionnaire 19 ; onset greater than 1 year; no change in motor test values for 3 months prior to the study’s initiation in comparison with baseline (the initiation of CIMT)—tests were evaluated when patients fulfilled inclusion criteria at screening, and these tests were reevaluated 3 months later immediately before starting CIMT (see also behavioral investigation)—and able to voluntarily flex and extend the affected wrist at least 20° and each finger of that hand to at least 10°; normal sensory examination, comparing both arms and hands, for tactile contact, pinprick, and vibration; and Ashworth Scale ≤1. Patients were excluded from the study according to the following criteria: having a Motor Activity Log (MAL) score of 3.0 points or more, because their performance was expected to be too close to a ceiling effect with no substantial benefit from the therapy, and presenting any contraindication for a MRI investigation. Furthermore, patients with cognitive impairments, which made them unable to follow training instructions, were not considered. Persistent uncontrolled arterial hypertension was another exclusion criterion. Patients with hemodynamically relevant intracranial or extracranial artery stenosis, which can alter the BOLD signal, 20 as well as patients with stroke lesions involving the postcentral gyrus, thalamus, or brain stem retrospectively (at baseline a T1-weighted MRI sequence was acquired) were also excluded. Whenever the N20 latency and amplitude of the median nerve somatosensory-evoked potential (SEP) were outside the range of a control of 30 healthy persons with in-house values, the patient could not participate. Because age and arm length have an influence on SEP, the SEPs of 7 age-matched, right-handed individuals with matched arm length were selected for a further comparison, retrospectively. The parameters of the patients were as follows: mean age, 69.0 years; N20 of SEP = 19.8 ± 0.7 ms; N20 amplitude = 1.8 ± 0.4 µV; and N20/P25 peak-to-peak amplitude = 16.0 ± 13.5 µV. A description of the SEP procedure followed here can be found elsewhere.21,22
Behavioral Investigation
All participants underwent a physical examination to assess their eligibility 3 months prior to the initiation of the study. On fulfillment of the inclusion criteria, all the project details were clearly explained, and written informed consent was given by each patient. At this time, motor function tests were performed. The same tests were carried out again before starting CIMT (defined as the baseline) and after CIMT (post).
We chose tests that had previously been applied by various clinical CIMT studies23-25—namely, the Wolf Motor Function Test (WMFT)26,27 and the MAL.28,29 Their reliability and validity have previously been determined. For the MAL, the amount of use (MAL-AOU) and the quality of movement (MAL-QOM) of the affected arm were rated by the patient. For the WMFT, the functional ability (WMFT-FA) and the number of seconds required for these tests (WMFT-sec), measured with a stopwatch, were conducted by the physiotherapist. For the WMFT-sec, the average number of seconds for the subtests was calculated (for all the tests used, see Table 2). For the WMFT-FA, video sequences were recorded and presented for evaluation to a second physiotherapist, who was not aware of the time of recording (ie, whether the recordings were made before or after CIMT), as described previously.24,30 To test clinical efficacy, we compared MAL-QOM, MAL-AOU, WMFT-FA, and WMFT-sec using the nonparametric Wilcoxon test. We compared test results 3 months before CIMT with baseline and baseline with post results. The threshold was set at
Tests Used Throughout the Study With Group Results (Mean) and SD
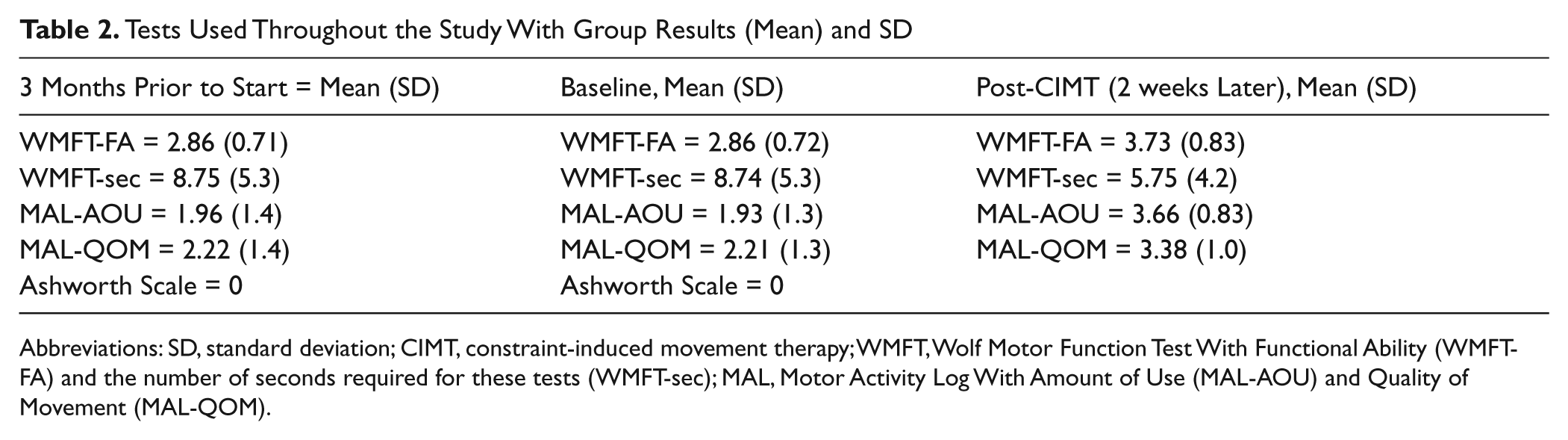
Abbreviations: SD, standard deviation; CIMT, constraint-induced movement therapy; WMFT, Wolf Motor Function Test With Functional Ability (WMFT-FA) and the number of seconds required for these tests (WMFT-sec); MAL, Motor Activity Log With Amount of Use (MAL-AOU) and Quality of Movement (MAL-QOM).
Training
CIMT was performed for 6 hours daily over 2 consecutive weeks within the facilities of the Physical Therapy Unit of the Department of Neurology. The aim of training was to force the patient to use the affected hand. CIMT is intended to help the patient overcome learned nonuse of the paretic arm. 31 To avoid use of the less-affected hand during a training period, it was placed in a splint for 90% of the waking hours. The intensive training tasks were tailored toward the activities of daily living undertaken by the patients (eg, eating and opening/closing jars and clothes pins). Patients received intensive daily motor activity training with shaping techniques according to previous protocols.23-25
Functional MRI
The following conditions were investigated at baseline and post-CIMT with the patient’s eyes closed. Rest scans served as a low-level baseline condition (REST). Two of the conditions included right and left passive wrist joint movements. All conditions were presented in a pseudorandomized order. There were 4 cycles, each containing the experimental conditions alternated with the rest conditions, with no gap between cycles. Each condition lasted 31 s. The right and left hands were alternately moved by manual exercise conducted by an examiner in the scanner room. Dorsal extensions and plantar flexions of the wrist (0°-50°) were performed 3 times (with a tone to inform the examiner) in 3.1 s, 30 times within each cycle of 31 s. This was followed by a rest condition of 31 s. The signal for a hand movement was given by a signal via headphones to the examiner who moved the hand. This signaling task was controlled by a PC running Presentation software (Neurobehavioral Systems, http://www.neurobs.com). The amplitude of the passive hand movement was limited by the physiological dorsal extension of the hand (50°), in the absence of spasticity. The forearm was fixated, and only a dorsal extension of the hand was performed. Before scanning, participants underwent passive hand movements in the MR environment to learn to avoid active movements of the hand and artifacts of head movement. Surface EMG electrodes were positioned on the dorsal interosseous muscle I and on the extensor digitorum communis muscle. Contraction of arm or hand muscles activated an alarm. MRI was acquired while patients relaxed their arms and hands.
Data Acquisition
BOLD-sensitive T2*-weighted functional magnetic resonance images were acquired using a 3T whole-body MRI system (TIM TRIO, Siemens, Erlangen, Germany) equipped with a standard head coil. We used an 8-channel receive-only head coil. Contiguous multislice echoplanar images (TE = 60 ms) were obtained in axial orientation. A total of 30 slices (3-mm thickness) were acquired every 3.1 s. The voxel size was 3.28 × 3.28 mm2 (64 × 64 pixels), giving a total field of view of 210 × 210 mm2. In total, 160 volumetric images were acquired in the first experiment. For each participant, a T1-weighted MRI containing a voxel size of 1 × 1 × 1 mm3 (256 × 256 mm2 field of view; 160 sagittal slices; flip angle 15°; repetition time 8.9 ms; inversion time 800 ms) was acquired.
Data Processing and Statistics
fMRI data processing was performed with SPM8 (Wellcome Department of Cognitive Neurology, London, UK) running under Matlab7a (MathWorks, Sherborn, Massachusetts). The first 4 images of each run were discarded to allow equalization of longitudinal magnetization.
Calculation of motion- and distortion-corrected fMRI volumes was performed at the scanner. 32 The resulting volumes were spatially normalized to a symmetric template based on the Montreal Neurological Institute reference brain using the normalization parameters estimated during segmentation of the T1-anatomic scan. 33 Normalized fMRI images were then smoothed with an isotropic 6-mm full-width at half-maximum Gaussian kernel to allow valid statistical inference, according to the Gaussian random field theory. 34 Because normalization in patients with a large lesion might lead to incorrect normalization, a mask of the lesion was made and included in this procedure. The mask of the lesion was created on the basis of the coregistered T1-weighted MRI sequences. These sequences revealed (in the case of a chronic stroke lesion) different contrasts and were therefore suitable for delineating the infarction with a customized SPM-based tool. 35 Low-frequency components of fMRI time series were removed by high-pass filtering to stabilize the MR signal.
A general linear model approach was used based on a model of the time course and a hemodynamic response function. Each voxel was analyzed in the context of Gaussian random fields
36
designed to detect significant regional differences. To explore the primary effects of active conditions, a random effect analysis was performed. Stimuli onsets were convolved with a canonical hemodynamic response function, as implemented in SPM8.
To characterize white matter integrity of the afferent somatosensory tracts in the 7 stroke patients, their lesions were overlaid with results of a probabilistic fiber tensor imaging. The latter was performed in 50 healthy individuals (mean age, 30 years; range, 25-73 years). Therefore, we used a diffusion-weighted spin-echo, echo planar imaging sequence to examine the anatomic contribution between 2 regions of interest (ROIs) selected within the thalamus and S1 (the probabilistic diffusion tensor imaging–based fiber tracking). The whole brain was covered with contiguous 2-mm slices with an in-plane resolution of 2 × 2 mm2. The diffusion encoding was performed in 61 different directions with an effective
The anatomic description was based on the probabilistic cytoarchitectonic maps as implemented in the SPM Anatomy Toolbox (http://www.fz-juelich.de/SharedDocs/Downloads/INM/INM-1/DE/Toolbox/Toolbox_18.html). 40
Results
Behavioral Effects of CIMT
Motor hand function improved in all patients after CIMT. Following multiple comparison correction (Bonferroni correction), MAL-AOU (
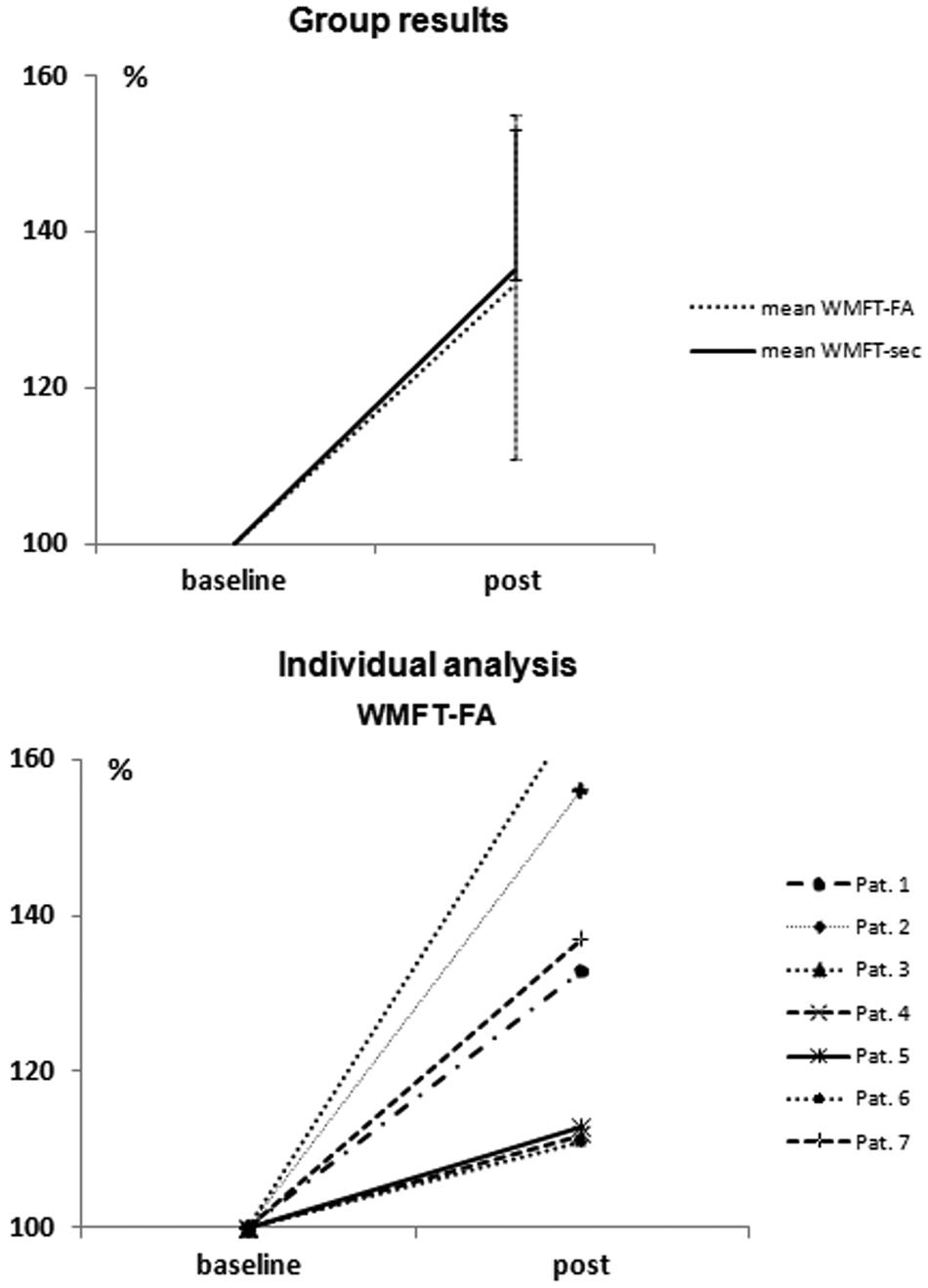
Hand function improvements are presented as mean group results in the upper diagram (error bars represent SD). In the lower diagram, the individual WMFT-FA are shown. The WMFT-sec and WMFT-FA of the affected right hand are normalized, by percentage, to baseline: (Affected handpost/Affected handbaseline) × 100. Abbreviations: WMFT-FA, Wolf Motor Function Test with functional ability; WMFT-sec, number of seconds required for the WMFT tests; SD, standard deviation.
Functional MRI
All patients completed the fMRI examination successfully. None of the participants had to be excluded on the grounds of active hand movements or head movements (the estimated residuary head movement did not exceed 2 mm) during image acquisition. Comparison of the group analysis between both investigation days (postintervention vs preintervention and vice versa) did not reveal any significant difference in fMRI activation. Single-participant analysis demonstrated an increase of activation within the SMC in 5 patients and a decreased activation within the SMC in 2 patients.
A positive correlation was evident between ΔfMRI and the hand function improvement ΔWMFT-FA within a maximal peak voxel within the ipsilesional S1 (
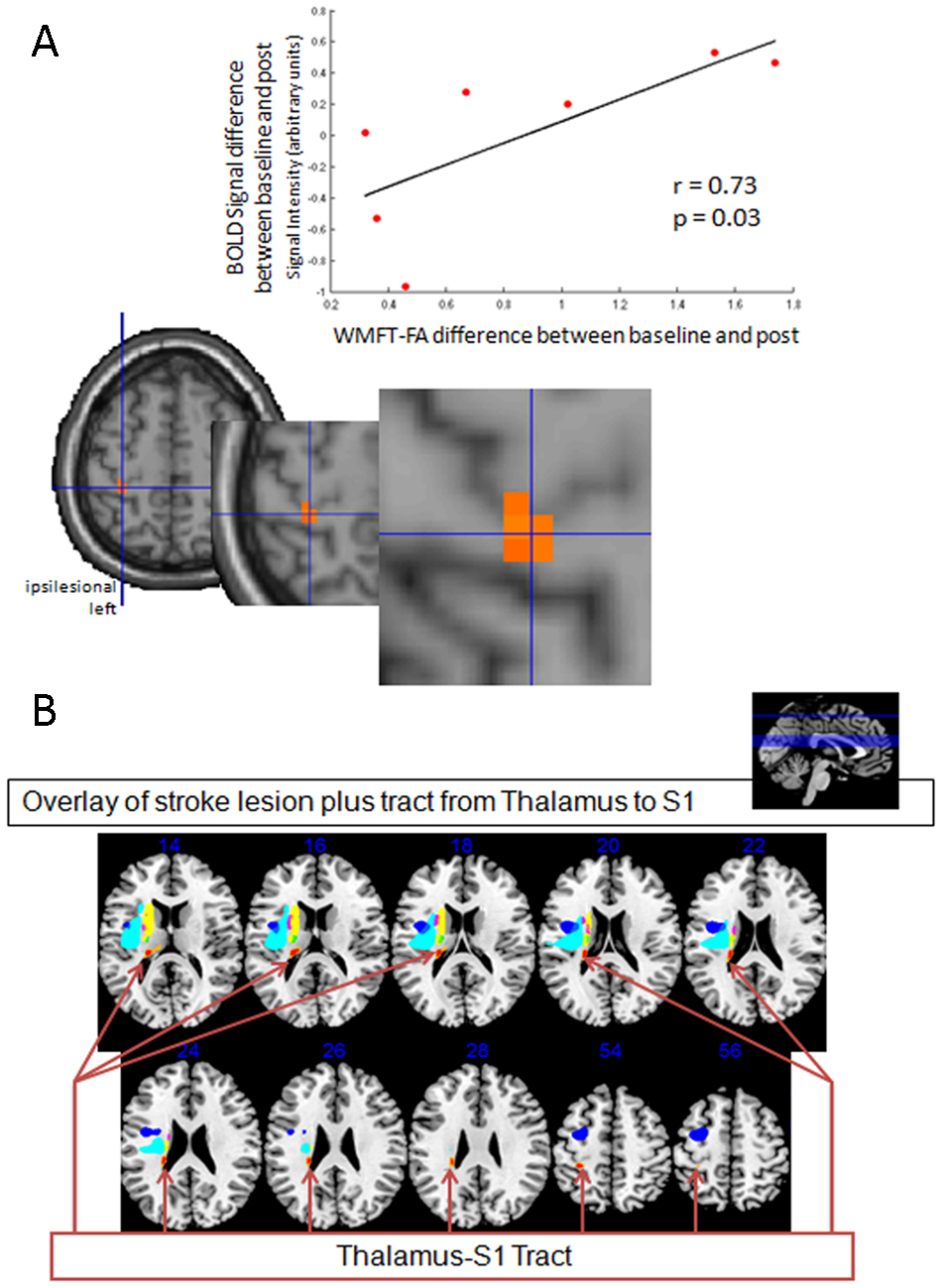
A. The correlation analysis between activation changes (post vs baseline CIMT of the affected hand; ΔfMRI) and ΔWMFT-FA (WMFT-FA post vs baseline CIMT) showed a close relationship within the ipsilesional S1. The curve represents the correlation analysis within the ipsilesional S1 between changes in fMRI activation peak and changes in hand motor function (
In addition, the fMRI activation at time point baseline was correlated retrospectively with changes in the clinical assessment of WMFT-FA (post vs baseline). This analysis demonstrated an activation peak within the ipsilesional S1 (
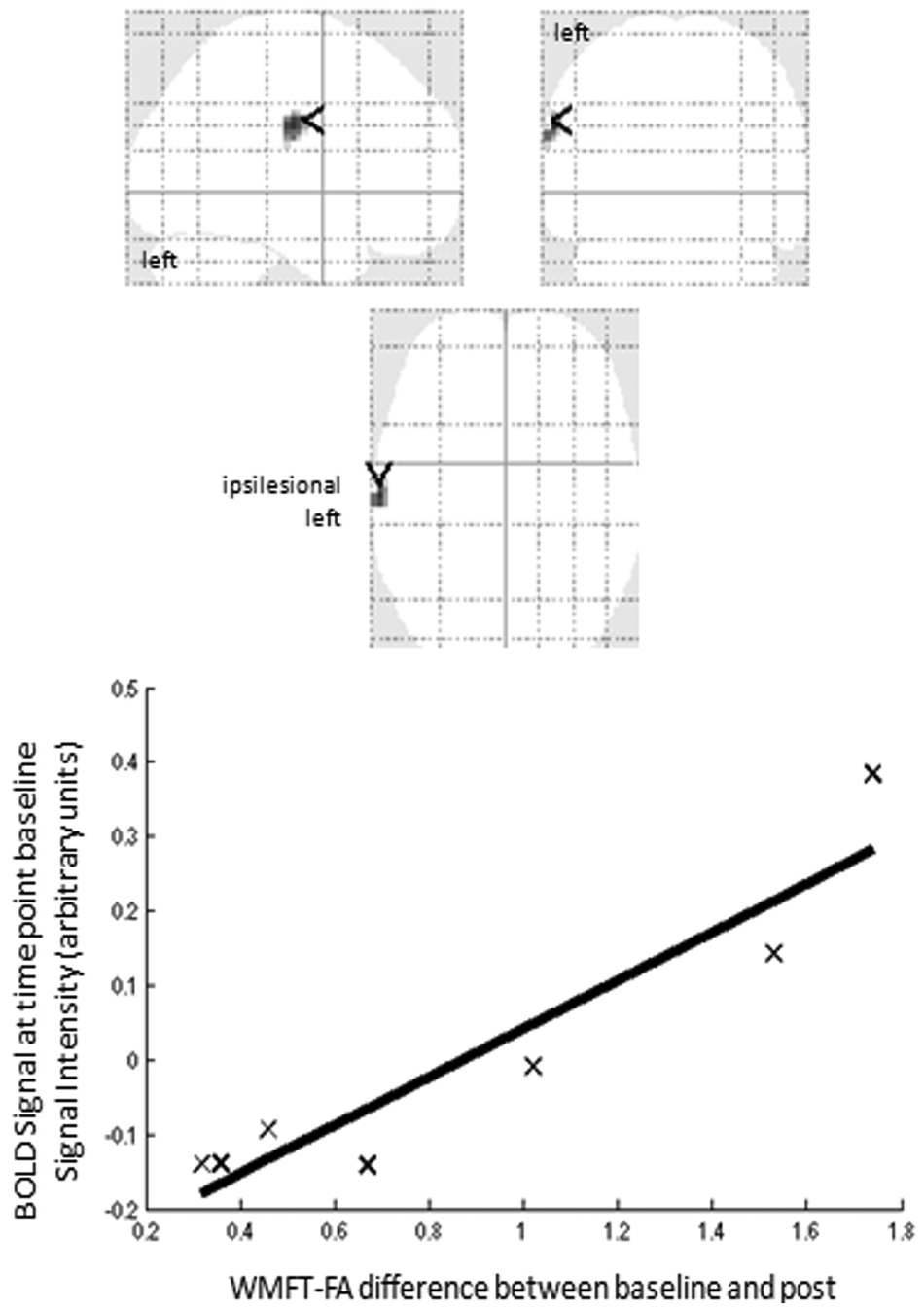
fMRI activation at time point baseline was correlated with changes of WMFT-FA (post vs baseline). The curve represents the trend line of this correlation analysis. This analysis demonstrated that with a higher activation peak within the ipsilesional S1 (
Discussion
In a selected population of chronic stroke patients with moderate hand paresis and normal sensory examination and SEPs, CIMT was used to study the underlying cortical plasticity within the S1 during motor recovery. The extent of improved hand function closely correlated with the activation peak changes within the ipsilesional S1. The more the hand function improved, the more peak activation within the S1 changed. It should be noted that 2 out of the 7 patients studied showed decreased S1 activation. This divergent course of activation within the S1 is in line with previous findings for the SMC, in which decreased activation was more pronounced with greater corticospinal tract lesions,10,11 although in the current study, we did not apply techniques like transcranial magnetic stimulation to investigate the integrity of the descending corticospinal fibers. Another study correlated hand functional improvement with activation changes after CIMT, but the investigators found an association with the secondary somatosensory cortex, among other regions. 16 Design differences may be responsible for this variance: An active fMRI motor paradigm was used, CIMT was performed in a modified manner (2 weeks of home-based training with 30 minutes of a predefined exercise program with the affected arm twice daily), and stroke patients were selected according to different criteria.
Earlier studies have shown an activation shift to the postcentral gyrus following stroke13-15,41 but failed to demonstrate a close relationship between this activation shift and hand motor recovery. We suggest that these previous results may have been influenced by interindividual variability. The variation among studies may be a result of the exact lesion location and the spared pathways but may also result from the outcomes used as well as the level of motor control and the time prior to starting treatment. 18 For example, in a recent study by Schaechter and colleagues, 14 although patients’ lesions spared the precentral and postcentral gyrus, the threshold of tactile detection of 2 out of the 9 patients studied was outside of the control group’s threshold, implying inhomogeneity in this group of patients with regard to sensation. This may explain why their cross-sectional study found a nonsignificant trend toward an increased activation within the S1 of patients with good compared with poor motor outcome of the affected hand. Because hand function in their patients improved following acute stroke, they proposed that functional changes within the S1 represent motor recovery. 14 Our data demonstrating the association of activation changes within the S1 with training-induced hand function improvement support this view. It has been suggested that a posterior shift reflects an increased proprioceptive attentional process to offset the motor impairment. 15 We cannot fully exclude an attentional influence, but we chose an fMRI paradigm that was independent of individual performance and clinical improvement.
It has been suggested that the posterior shift represents recruitment of corticospinal fibers originating in the postcentral gyrus 42 to compensate for the limitation of recruitment of the motor cortex. 15 The involvement of S1 in motor recovery may be based on recruitment of corticospinal fibers or on information processing between the S1 and the remaining M1 and premotor neuron. 43
If plasticity within the ipsilesional S1 in association with motor recovery is specific to the current group of patients, then perhaps they will benefit from a specific rehabilitation approach, such as electrical stimulation to enhance the effects of CIMT. 44 However, the small, although relatively homogeneous, sample size should be considered as a limitation of the study.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
