Abstract
Background. Repetitive transcranial magnetic stimulation (rTMS) of the brain has been shown to modulate cortical excitability. Combinations of rehabilitation therapies with rTMS might enhance the therapeutic effects. Objective. The purpose of this study was to investigate the effects of high-frequency rTMS followed by treadmill training on cortical inhibition and walking function in individuals with Parkinson disease (PD). Methods. A total of 20 patients with PD were randomized into an experimental group and a control group. Participants received rTMS (experimental group) or sham rTMS (control group) followed by treadmill training (30 minutes) for 12 sessions over 4 weeks. Repetitive TMS was applied at a 5-Hz frequency over the leg area of the motor cortex contralaterally to the more affected side for 6 minutes. Outcomes, including corticomotor inhibition and walking performance, were measured before and after training. Results. The results showed significant time effects on almost all corticomotor and functional variables. There are significant interaction effects between group and time of evaluation on the motor threshold, duration of the cortical silent period, and short interval intracortical inhibition of the contralateral hemisphere relatively to the more affected side as well as on the fast walking speed and timed up and go. Conclusions. The findings suggested that combination of rTMS and treadmill training enhances the effect of treadmill training on modulation of corticomotor inhibition and improvement of walking performance in those with PD.
Keywords
Introduction
According to the basal ganglia-thalamocortical circuit model, 1 the degeneration of dopaminergic nigrostriatal pathways results in functional deafferentiation of the frontal cortex, including the primary motor cortex. This could contribute to the pathophysiology of motor disturbances in people with Parkinson disease (PD). These disturbances can be revealed by changes in cortical excitability, as assessed by transcranial magnetic stimulation (TMS). 2 Enhanced corticospinal motor output was observed at rest in PD by decreased motor threshold (MT) and increased amplitude of motor evoked potential (MEP) at rest.3-5 Changes in the excitability of inhibitory drive seem also involved in the pathophysiology of PD. The duration of cortical silent period (SP) was found to be shorter and intracortical inhibition to be reduced in people with PD compared with healthy people.4,6-8 In general, rather consistent results have been reported for cortical excitability studies in PD.9,10 The changes in cortical excitability support the use of cortical stimulation as a therapeutic strategy for PD.
Repetitive TMS (rTMS) of the brain has been shown to modulate cortical excitability and cortical inhibition in healthy people, with the effect depending mostly on the stimulation frequency. Low-frequency stimulation induced de-pression of cortical excitability, whereas high-frequency stimulation induced facilitation.11,12 These changes in cortical excitability are transient, lasting 10 to 30 minutes.13,14 The similar frequency-dependent effects of rTMS also showed in people with stroke. 15 However, such effects on patients with PD are not well established. A recent meta-analysis concluded a significant positive effect of high-frequency rTMS and little effect of low-frequency rTMS on motor performance in PD. 16 High-frequency stimulation of cortical motor areas was also found to increase SP duration.17-19 Butler and Wolf further viewed rTMS as a tool that helps the brain to reach an optimal state of learning to facilitate subsequent training effects. 20 The principle of activity-dependent neuroplasticity also suggests that combinations of rehabilitation therapies with rTMS might have promise in facilitating beneficial neuromodulation. 21 However, few studies have yet been conducted to examine this possibility.
Gait hypokinesia is one of the most incapacitating symptoms of PD. 22 Antiparkinsonian medicines ameliorate many of parkinsonian symptoms, including balance and gait disturbances, to some degree, but they are not very effective in treating all gait abnormalities. There is growing evidence in the therapeutic use of the treadmill walking in PD. A systemic review reported that the use of treadmill training may improve gait parameters, such as gait speed and stride length, of people with PD at Hoehn and Yahr stages I to III. 23 Treadmill training is currently being studied as a treatment modality for promoting activity-dependent neuroplasticity by lengthening SP in early PD. 24 Repetitive TMS is a potential approach to maximize motor recovery in a short-term period. In a previous study, we found that rTMS enhances the effect of task-oriented training in those with chronic stroke. 25 It is possible that modulating cortical excitability by rTMS before an exercise training would enhance therapeutic effects.
The purpose of this study was to investigate the effect of 5-Hz rTMS followed by treadmill training on cortical excitability as measured through TMS in people with PD and to examine whether normalizing cortical excitability is accompanied by improving gait performance.
Methods
Participants
Participants were recruited from a medical center in Taipei. Eligible participants had been diagnosed with idiopathic PD according to the United Kingdom Brain Bank Criteria 26 by a neurologist. Information about age, gender, the more affected side, and disease duration were obtained through patient interviews and from medical charts. All participants met the following inclusion criteria: (a) Hoehn and Yahr stages II through III, (b) ability to walk independently, (c) stable medication usage, (d) freedom from any other problems that might affect training, and (e) a score of more than or equal to 24 on the Mini-Mental State Examination. The exclusion criteria were as follows: (a) the MEPs could not be induced by TMS, (b) unstable medical conditions, (c) histories of other diseases known to interfere with participation in the study, (d) past history of a seizure, and (e) use of a cardiac pacemaker.
Finally, 29 individuals were identified as potential participants for this study. Of these, 22 participants gave informed consent and participated in the study (Figure 1). This study was a double-blinded, randomized controlled trial. Both the assessor and the participant were blinded. Block randomization was used to assign participants to either the experimental or the control group; this was done by an independent person who selected one of a set of sealed envelopes 30 minutes before the intervention began.
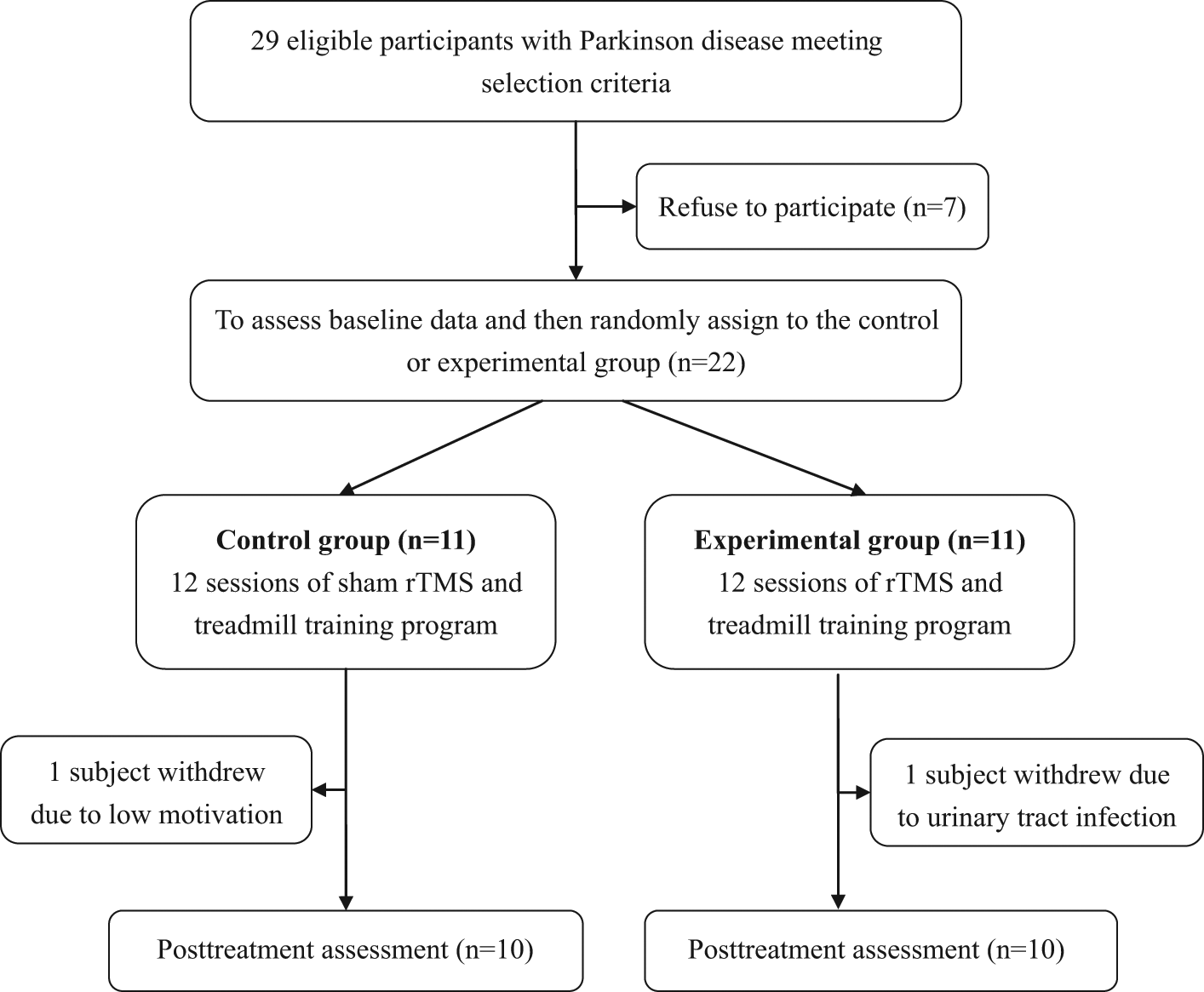
Flow chart of participants recruited in this study.Abbreviation: rTMS, repetitive transcranial magnetic stimulation.
Study Protocol
The study protocol was approved by the institutional review board of Taipei Veterans General Hospital and explained to all included participants before their participation. All participants underwent a double-blind, placebo-controlled study where they received a 6-minute active or sham rTMS followed by a 30-minute treadmill training for 12 sessions (3 sessions a week) over a 4-week period. Outcomes, including corticomotor excitability and walking performance, were measured before intervention (pre) and after completing a 12-session training (post) by an assessor who was blinded to participant’s group. The measurement and intervention were conducted with the patients in the “on” state when they were moving freely and easily without dystonia, excessive rigidity, or tremor.
Interventions
Participants in the experimental and control groups received rTMS and sham rTMS for 6 minutes, respectively, followed by a 30-minute treadmill training for 12 sessions. A well-trained and qualified physical therapist delivered rTMS and treadmill training.
Repetitive transcranial magnetic stimulation
Repetitive TMS was delivered to the scalp over the leg area of the motor cortex contralaterally to the more affected side by using a MagStim SuperRapid stimulator (MagStim Co, Ltd, Carmarthenshire, Wales, UK) and a figure-of-eight coil. Real rTMS involved an intensity of 100% resting MT and a train of 1200 pulses (5 Hz) for 6 minutes during each session while the participants were at rest. The train duration was 10 seconds with 5-second intertrain interval. The motor cortex was stimulated by holding the figure-of-eight coil tangentially over the leg area at the optimal site for response from the tibialis anterior muscle. The location of the stimulation site was marked, referenced by vertex-centered coordinates, and recorded to maintain consistency among sessions. The coil was oriented at about 0° over the transverse plane with handle pointing laterally to induce a lateral-to-medial current flow in the cortex. Sham rTMS was performed with the coil angled at 45° from the plane tangential and touched the scalp by one wing of the figure-of-eight coil, 27 using the same stimulation parameters (noise, time, and frequency) as used for real rTMS.
Treadmill training
All participants walked on a motorized treadmill (Biodex, Shirley, New York) after the rTMS. The participants wore a safety harness without body weight support during walking on the treadmill to prevent falls. We started the training program of each participant by setting the treadmill speed to 80% of his/her overground comfortable walking speed and increased by an increment of 0.2 km/h per 5 minutes as tolerated. The training program was designed with positive feedback and reenforcement of the participant’s performance by an experienced and qualified physical therapist. During the training, the physical therapist encouraged the participant to walk with large strides and correct posture. The participants were instructed to refrain from holding a handrail if possible.
Outcome Measures
Corticomotor excitability
The MT at rest, SP duration, short interval intracortical inhibition (SICI), and long interval intracortical inhibition (LICI) of the tibialis anterior muscle elicited by TMS were used to evaluate motor cortex excitability bilaterally. In this study, we used the more affected side to describe the tested hemisphere. The ipsilateral hemisphere relatively to the more affected side was abbreviated as IHMA, and the contralateral hemisphere relatively to the more affected side was abbreviated as CHMA.
The MEPs of the bilateral tibialis anterior muscles were recorded by an electromyographic (EMG) machine in response to TMS (Magstim 200 electromagnetic stimulation) delivered through a figure-of-eight coil placed on the contralateral motor cortex. During measurements, participants sat comfortably in a chair with hips and knees in 90° flexion and wore a fitted cap marked with a coordinate system (distance, 1 cm). Muscle activity was carefully monitored by real-time EMG. The coil was positioned parallel with the transverse plane and the handle of the coil pointed outward to induce a lateral-to-medial current flow in brain.
The optimal scalp location (hot spot) was determined by moving the TMS stimulator over the scalp in 1-cm steps. Once the hot spot was identified, a single-pulse TMS was delivered to the location to determine the resting MT, defined as the lowest stimulus intensity necessary to elicit MEPs greater than 0.05-mV peak-to-peak amplitude in at least 5 of 10 consecutive stimuli. The threshold intensity was expressed as a percentage of maximum stimulator output. The intensity used in the postassessment was the same as that used in the preassessment.25,28-30 The SP duration was determined during isometric voluntary contraction of the contralateral tibialis anterior muscle. Voluntary contraction started several seconds before TMS. EMG was played through a loudspeaker as a feedback to the participants. Five magnetic stimuli were applied at an intensity of 120% resting MT while the participant performed maximum voluntary contraction. The EMG traces were rectified and averaged. The SP duration was determined from the MEP onset to the recurrence of at least 50% of EMG background activity. 17
Paired-pulse paradigm was performed in the relaxed tibialis anterior muscle to assess the SICI and LICI according to Kujirai’s procedure, 31 using a BiStim module and a figure-of-eight coil. The intensity of the conditioning stimulus was set at 80% resting MT with short interstimulus interval (2 ms) and at 120% resting MT with long interstimulus interval (100 ms) for SICI and LICI, respectively. The test stimulus was always delivered at 120% resting MT. For each condition, 10 trials were collected and averaged. The amplitude of the conditioned MEP was expressed as a percentage of the mean unconditioned MEP amplitude.
Gait performance
Ten-meter walk test and timed up and go test were measured to evaluate the gait performance. For the 10-m walk, a 10-m distance was marked on the floor. The participant began this test 1 m before the starting line and completed the test 1 m after the finish line. Time was recorded from the time when the participant crossed the starting line to the time when the participant crossed the finish line. The participant was instructed to walk at a comfortable pace for 3 trials and then to walk at a faster pace for 3 trials. The time between trials was 1 minute. Walking speed was derived from dividing the distance by the time (in seconds) required to walk the 10 m. The data were averaged from the 3 trials at both the comfortable speed and the fast speed. The reliability and stability of the 10-m walk test for people with PD have been established. 32 For the timed up and go, the participant was asked to stand up from a chair, walk 3 m, turn around, return to the chair, and sit down. The time taken to complete this task was measured with a stopwatch. This test has previously demonstrated high reliability in people with PD. 33
Statistical Analysis
All analyses were performed using the SPSS 17.0 statistical package. Descriptive statistics were generated for all variables, and distributions of variables were expressed as mean ± standard deviation. Intergroup differences among baseline characteristics were evaluated using an independent t test or χ2 analysis. A 2-way analysis of variance with repeated measures was used to determine differences of each dependent variable. The between-subjects factor was the group, and the within-subject factor was the time interval. The Bonferroni correction for multiple comparisons was used for the post hoc test. The statistical significance was set at P < .05.
Results
Participants
Of 22 participants, 11 were randomly assigned to the control group, and the other 11 were randomly assigned to the experimental group. Of 22 participants, 2 did not complete the intervention (1 in the control group and 1 in the experimental group; Figure 1). The 20 participants who completed the intervention attended all treatment sessions. None of the participants reported any adverse events. Demographic characteristics of participants in both groups are presented in Table 1. The differences in the demographics of the 2 groups were insignificant. Moreover, differences in all the preintervention-selected outcome measures of the 2 groups were insignificant (Tables 2 and 3).
Demographic Characteristics of Included Participants in Both Groups a
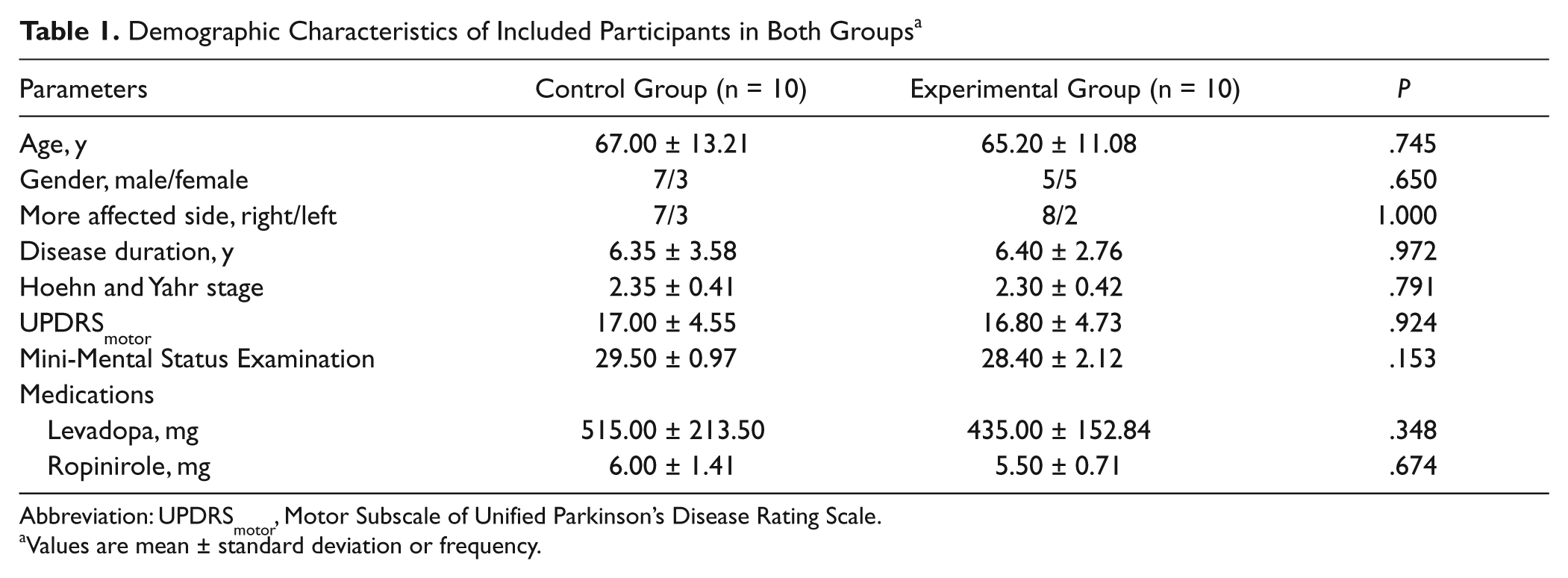
Abbreviation: UPDRSmotor, Motor Subscale of Unified Parkinson’s Disease Rating Scale.
Values are mean ± standard deviation or frequency.
Comparison of Cortical Measures a
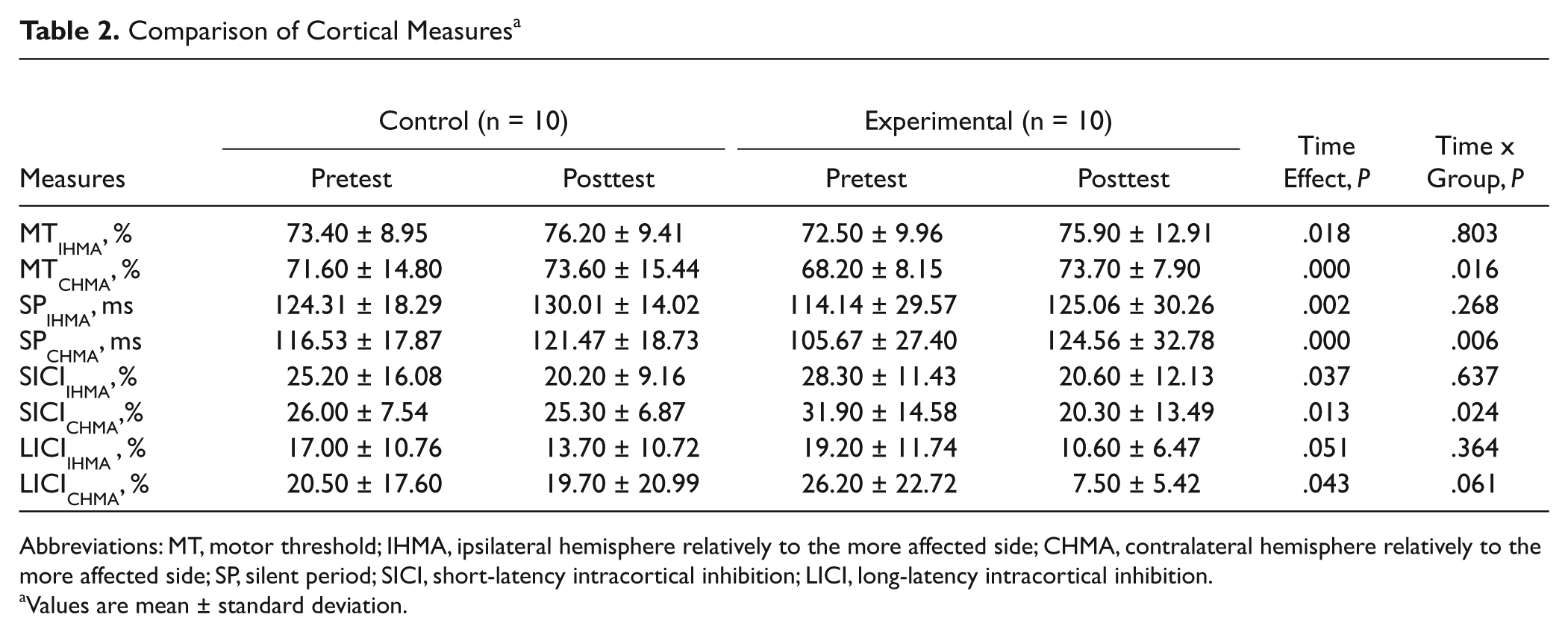
Abbreviations: MT, motor threshold; IHMA, ipsilateral hemisphere relatively to the more affected side; CHMA, contralateral hemisphere relatively to the more affected side; SP, silent period; SICI, short-latency intracortical inhibition; LICI, long-latency intracortical inhibition.
Values are mean ± standard deviation.
Comparison of Clinical Measures a
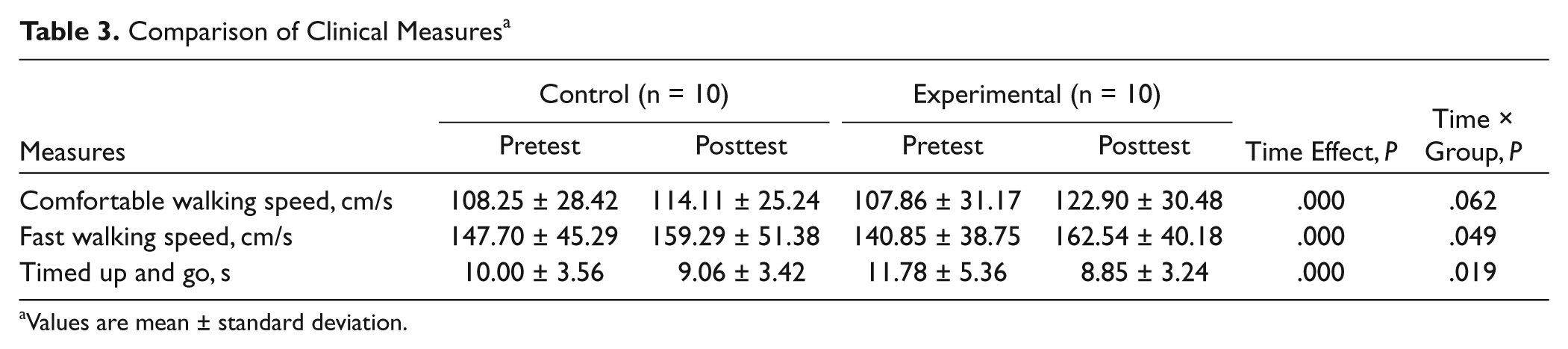
Values are mean ± standard deviation.
Treadmill Speed
The mean treadmill speed was increased from 3.06 ± 0.87 to 5.10 ± 1.30 cm/s in the control group and from 3.02 ± 0.89 to 5.09 ± 1.21 cm/s in the experimental group through the 4-week treatment period. The differences in the initial treadmill speed and the final treadmill speed of the 2 groups were insignificant.
Corticomotor Excitability
The results of corticomotor excitability are shown in Table 2. Analysis of variance with group and time interval as between-subjects factor and within-subject factor, respectively, showed a significant main effect of time on all TMS variables except LICIIHMA. The result of time effect showed a marginal effect (P = .051) on LICIIHMA. The analysis also showed a significant 2-way interaction effect of time × group on MTCHMA (P = .016), duration of SPCHMA (P = .006), and SICICHMA (P = .024). Also, the result of interaction effect showed a marginal effect (P = .061) on LICICHMA.
Walking Performance
The results of walking performance are presented in Table 3. Analysis of variance with group and time interval as between-subjects factor and within-subject factor, respectively, showed a significant main effect of time on comfortable walking speed, fast walking speed, and timed up and go. The analysis also showed a significant 2-way interaction effect of time × group on fast walking speed and timed up and go. Also, the result of interaction effect showed a marginal effect (P = .062) on comfortable walking speed.
Discussion
The main finding of this study is that, for people with PD whose MEPs could be induced by TMS, the combination of high-frequency rTMS and treadmill training significantly increased corticomotor inhibition as indexed by significant increase in resting MTCHMA, prolongation of SPCHMA duration, improvement in SICICHMA, and a trend toward an improvement in LICICHMA compared with treadmill training only. Concurrently, this modulation of corticomotor excitability is accompanied by improvement in walking performance. As far as we are aware, this is the first study to examine the therapeutic effect of combined intervention with rTMS in PD.
Treadmill walking has been proposed to be an effective means of promoting walking in people with PD. 34 A few studies have investigated the use of treadmill training for gait rehabilitation in PD. The findings sometimes suggest that treadmill training may modestly improve gait speed and stride length.23,35 A positive therapeutic effect of treadmill training was often obtained after a 6- to 8-week training program.23,35 Recently, a study by Picelli et al 36 used the Gait Trainer to provide a robotic gait training for 4 weeks in patients with PD. Their results showed that robotic-assisted gait training is more effective than conventional physiotherapy to improve walking capacity. However, the statistically significant improvement in gait speed (0.12 m/s for 10-m walk test at the fastest speed) by robotic-assisted gait training may not be clinically significant. 36 Our results are consistent with the results of Picelli et al. 36 The participants in the control group obtained 0.12 m/s increase in fast walking speed after treadmill training only. There is emerging evidence that rTMS may have therapeutic applications in neurological disease. 37 High-frequency rTMS has been suggested to have a benefit for improving gait speed in PD. 38 The present results showed that the participants in the experimental group gained 0.22 m/s in fast walking speed after combined rTMS and treadmill training. This gain in fast walking speed is quite close to a clinically significant change of 0.25 m/s for a 10-m walk test in PD. 39 Furthermore, our results showed that additional 5-Hz rTMS in the experimental group enhanced the improvement in speed during comfortable walking as well as for the timed up and go test (Table 3).
Both high-frequency rTMS17-19 and treadmill training 24 have been reported to have an effect on prolongation of SP duration in PD. Therefore, interventions may improve functional performance, concomitantly with restoring cortical inhibition. An important objective of this study is to investigate if rTMS and treadmill training may induce the restoration of cortical inhibition in PD. We found lengthening of SP duration, increase in SICI and LICI, as well as increase in resting MT in both groups that was consistently observed in bilateral hemisphere (Table 2). This finding suggests that either treadmill training only or combined intervention with rTMS and treadmill training facilitated the activation of inhibitory cortical pathways in PD. Our finding in intervention-induced lengthening of SP duration is consistent with previous observation with intervention of either high-frequency rTMS or treadmill training.17-19,24 Since the SP duration is a well-established measure of intracortical inhibition, this finding indicates that rTMS is capable of inducing an increase in intracortical inhibition in PD. 17 To our knowledge, modification of SICI and LICI by treadmill training is not previously reported in PD. There is only one study that investigated the effect of high-frequency rTMS on SICI and LICI in “on-drug” people with PD and resulted in no significant change in SICI and LICI. 40 In addition to methodological differences of rTMS, activity-dependent neuroplasticity from treadmill training might explain the different finding between our results and previous results. Moreover, there are no studies on the effects of high-frequency rTMS or treadmill training on resting MT in “on-drug” people with PD. In the current study, we found an intervention-induced increase in resting MT. The level of excitability within the motor cortex is a balance between excitation and inhibition. Therefore, an intervention that increases the intracortical inhibition such as lengthening of SP duration and increase in SICI and LICI may contribute to increase in resting MT. Some evidence suggest that high-frequency rTMS over supplementary motor area improves motor symptoms in patients with PD.41,42 Supplementary motor area is close to the motor area of the tibialis anterior muscle. We cannot rule out the possibility that partial benefit might originate from a spread of current.
Each corticomotor excitability measure tests different aspects of excitability. SP duration and LICI have been related to inhibition from GABAB receptors. 43 SICI has been related to inhibition from GABAA receptors.6,44 GABA is the major inhibitory neurotransmitter in the basal ganglia, and abnormalities of GABAergic transmission are key aspects of pathophysiology of movement disorders that involve the basal ganglia. 45 These 2 GABAergic receptor subtypes are both activated at the same time by thalamocortical inputs and intracortical fibers. 46 Consistently, our results showed that SP duration, LICI, and SICI were modulated in the same direction after intervention. Previous studies have shown a loss of cortical excitation and inhibition balance in PD, most likely the result of reduced intracortical inhibition.2,4,47 Therefore, interventions that restore intracortical inhibition may help normalize corticomotor processing and motor function.
Our results also showed that additional rTMS enhanced cortical inhibition especially in the hemisphere contralateral to the more affected side (Table 2). We determined the more affected side of each participant by clinical observation. This interhemisphere difference disappeared after additional rTMS.
Because the sample size was small, the therapist not blinded, and all participants had measureable MEPs, the results cannot be generalized to all persons and stages of PD. Our results suggest, however, that rTMS combined with treadmill training may benefit walking in people with PD by enhancing cortical inhibitory circuits. Larger trials seem warranted.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
