Abstract
Objective. To investigate the safety and efficacy of 50-Hz repetitive transcranial magnetic stimulation (rTMS) in the treatment of motor symptoms in Parkinson disease (PD). Background. Progression of PD is characterized by the emergence of motor deficits that gradually respond less to dopaminergic therapy. rTMS has shown promising results in improving gait, a major cause of disability, and may provide a therapeutic alternative. Prior controlled studies suggest that an increase in stimulation frequency might enhance therapeutic efficacy. Methods. In this randomized, double blind, sham-controlled study, the authors investigated the safety and efficacy of 50-Hz rTMS of the motor cortices in 8 sessions over 2 weeks. Assessment of safety and clinical efficacy over a 1-month period included timed tests of gait and bradykinesia, Unified Parkinson’s Disease Rating Scale (UPDRS), and additional clinical, neurophysiological, and neuropsychological parameters. In addition, the safety of 50-Hz rTMS was tested with electromyography-electroencephalogram (EMG-EEG) monitoring during and after stimulation. Results. The authors investigated 26 patients with mild to moderate PD: 13 received 50-Hz rTMS and 13 sham stimulation. The 50-Hz rTMS did not improve gait, bradykinesia, and global and motor UPDRS, but there appeared a short-lived “on”-state improvement in activities of daily living (UPDRS II). The 50-Hz rTMS lengthened the cortical silent period, but other neurophysiological and neuropsychological measures remained unchanged. EMG/EEG recorded no pathological increase of cortical excitability or epileptic activity. There were no adverse effects. Conclusion. It appears that 50-Hz rTMS of the motor cortices is safe, but it fails to improve motor performance and functional status in PD. Prolonged stimulation or other techniques with rTMS might be more efficacious but need to be established in future research.
Keywords
Introduction
Gait difficulties in advanced Parkinson disease (PD) represent a primary cause of disability and a therapeutic challenge because of refractoriness to conventional therapy. Trials of noninvasive brain stimulation are promising. Meta-analyses have shown a modest efficacy of high-frequency repetitive transcranial magnetic stimulation (rTMS) on motor performance in PD.1,2 Controlled 25-Hz rTMS studies reported gait improvement,3,4 which may be potentiated by increasing stimulation frequency. 4
In this double-blind, randomized, sham-controlled study (RCT), we investigated the efficacy and safety of prolonged 50-Hz rTMS, which borders limits of safe use in humans, 5 and the technical possibilities of conventional rTMS for the treatment of gait difficulties and bradykinesia in PD.
Methods
Study Population
Patients aged 40 to 80 years with PD according to the UK-PD-Brain-Bank-criteria, Hoehn-Yahr stages 2 to 4 (“off” medication) were included; they had slowing of gait, defined as taking ≥6 s to walk 10 m. Severe freezing, inability to walk 10 m, or daily falls were exclusionary. We considered the optimal medication to correspond to a levodopa-equivalent dose of ≥300 mg/d in Hoehn-Yahr stages 2 to 4, and this was required to remain unchanged during the study period. Exclusion criteria were dementia (Mini-Mental State Examination score ≤24/30), significant medical or psychiatric illnesses, history of seizures, pregnancy, or metal devices in the head. Screening included electroencephalograms (EEGs) reviewed by epileptologists.
A power analysis yielded a sample size of 6 and 13 participants per arm for the on and off conditions, providing 80% power with a 2-sided α = .05 to detect a gait improvement (primary outcome measure) similar to the one we reported previously with 25-Hz rTMS. 3 We prospectively enrolled the target population of 26 patients (Figure 1). Randomization was based on a computer-generated block allocation schedule. The study period was from September 2009 to April 2010. The study was approved by the NIH institutional review board and registered (ClinicalTrial.gov: NCT00977184). All participants gave written informed consent.
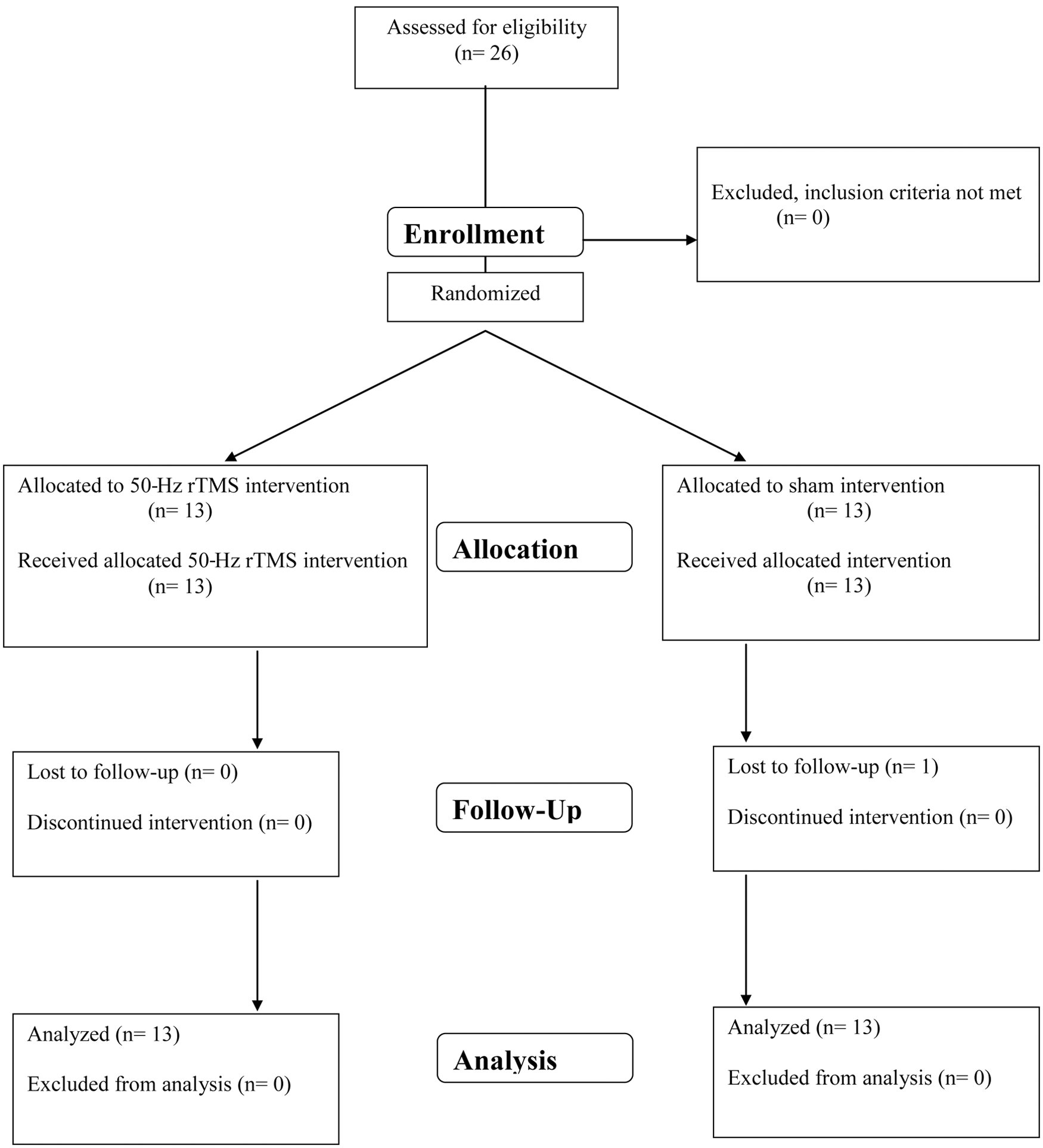
Flow diagram of patients with Parkinson disease (PD) enrolled in this therapeutic study. Abbreviation: rTMS, repetitive transcranial magnetic stimulation.
The 50-Hz rTMS Intervention
We performed real or sham 50-Hz rTMS in 8 sessions over 2 successive weeks, a session per day for 4 consecutive days per week. We applied 50-Hz rTMS to both primary motor cortices (M1) in each session, in an alternating order, at a stimulation intensity of 80% active motor threshold (AMT) for 6 s, amounting to the same number of stimuli as with 25-Hz rTMS that improved gait and bradykinesia. 3 We used the same circular 90-mm coil (parasagittal orientation, handle back) placed at the optimal position for motor-evoked potentials (MEPs) in the abductor pollicis brevis (APB) on each side. This setting provides extensive motor cortex stimulation, which may (transcallosally) spread to the contralateral hemisphere. The coil was connected to a Magstim-Rapid magnetic stimulator (Whitland, UK) inducing an anterior-posterior/posterior-anterior biphasic current. In the sham condition, we placed an inactive coil similarly to the active coil, and the active coil itself was oriented perpendicularly on top of the inactive coil, causing similar acoustic and vibratory sensations without exposing the patient to the magnetic field. Participants received interventions while on medication. The stimulating apparatus was set up out of sight of blinded investigators.
Safety Testing
We tested safety during the first intervention in both groups as a control and to maintain blinding as described. 5 We monitored patients for clinical and neurophysiological signs of a seizure. Electromyography (EMG) activity was recorded from the APB, extensor carpi radialis (ECR), biceps brachii (BB), and deltoid (DEL) muscles of either arm. We monitored the EMG for spread of excitation to more proximal muscles (ECR, BB, and DEL), which might indicate an increase in cortical excitability preceding epileptic activity and for activity outlasting stimulation, which might indicate afterdischarges or seizure. We performed EEGs immediately after the first and last interventions. Clinical assessment included Verbal Fluency (letters FAS or CJM; each for 1 minute), and the Frontal Assessment Battery (FAB) was repeated after the last intervention.
Clinical Assessment
Baseline and follow-up evaluations were performed before, 1 day, and 1 month after the last intervention. Primary outcome measures were the change in the timed test of gait in the on and off states 24 hours after the intervention period compared with baseline (evidence class I). We assessed gait by measuring the time to walk 10 m. Two trials were averaged. Patients were instructed to walk fast without taking the risk of falling, wearing the same shoes and consistently using assistive devices if needed. We assessed bradykinesia by the time to perform the following sequence 10 times: (1) hand closing and opening, (2) elbow flexion, (3) hand closing and opening, and (4) elbow extension. This is similar to a sequential task shown to correlate with bradykinesia. 6 Before baseline assessment, patients practiced this task until performance appeared not to get faster and, then, abstained from further practice to minimize learning effects, which were controlled by the study design. We chose timed tests because they are more sensitive in detecting changes than scores and are independent of subjective assessment. These motor tests and the Unified Parkinson’s Disease Rating Scale (UPDRS) were assessed in the “best on” and “practically defined off state” by the same blinded raters. Because the practically defined off state required overnight (≥12 hours) withdrawal of dopaminergic medication, assessment in the best on state followed, considered by the patients and blinded rater as the best response to their usual medication. Gait and bradykinesia were also timed immediately before and after each intervention session for acute effects.
Secondary outcome measures included the Falls and Gait Questionnaire containing the Freezing of Gait Questionnaire, 7 Beck Depression Inventory, and a Health Survey(SF-12v2), addressing the subjective perception of health and well-being. We tested visuomotor speed and procedural learning in the Serial Reaction Time Task (SRTT) as described, except for a shorter sequence of 8 instead of 12 items. 5
Neurophysiological Assessment
Resting motor threshold (RMT) and AMT were determined to the nearest 1% of the maximum stimulator output required to elicit an MEP of the APB ≥50 µV/≥100 µV in ≥5/10 trials during rest and weak voluntary contraction of 10% maximum quantitative EMG. We measured the MEP recruitment curve at rest and during weak contraction at stimulus intensities of 90%, 100%, 110%, 120%, 130%, 140%, and 150% RMT and AMT (8 pulses each every 6 s). We determined the cortical silent period (CSP) during weak voluntary contraction with a TMS pulse at 100% AMT and measured from MEP onset until return of voluntary EMG activity. Recruitment and CSP (right APB) were determined before and immediately after the first and 24 hours after the eighth intervention. All measurements were performed in the on and off states except for those after the first intervention performed only in the on state.
Statistical Analysis
Full-factorial repeated-measures analyses of variance (ANOVAs) were used to examine all outcome measures. Each model included a between-subjects factor for Treatment and a within-subjects factor for Time and Session (preintervention vs postintervention) when applicable. Omnibus main effects and interactions were examined post hoc using Bonferroni adjusted simple effects tests within the context of the ANOVA. A priori comparisons were made as specified. Levene test was used to verify the homogeneity of variance assumption, and Shapiro-Wilks test and the standardized residuals were examined to verify the normality assumption. Linear mixed-effects models were applied for the analysis of the recruitment curve at rest and weak voluntary contraction and for the CSP.
Because the assessment after the first intervention (time 2) was done in the on condition alone, we ran 2 models: (1) a model with the factor Medication (on condition vs off condition [plus factor Med]) without time 2 and (2) a model with the on condition alone at all time points. Given the concern about treatment effects being caused by baseline differences in CSP, we ran an additional mixed model introducing baseline as covariate, which we also reran without 90% intensity because of limited variance in the model at that intensity.
Significance was evaluated at P < .05, 2 tailed. Adjustments for multiple comparisons were made separately for primary and secondary measures using Bonferroni procedure. Cohen d effect sizes are reported to show the size of group differences, where differences are measures at the end point. Statistical analysis was done with SPSS version 19.0 (SPSS Inc, Chicago, Illinois).
Results
A total of 26 patients completed the study, but 1 patient withdrew from the 1-month follow-up assessment (secondary end point). Demographics and clinical findings did not differ between groups (Table 1) and neither did the outcome measures at baseline (all P > .19). No patient reported discomfort or other symptoms. We observed no adverse events. No changes in dopaminergic or other medications were reported in this 6-week study period.
Demographic and Clinical Findings in Patients With Parkinson Disease Receiving 50-Hz rTMS (n = 13) or Sham Treatment (n = 13) a
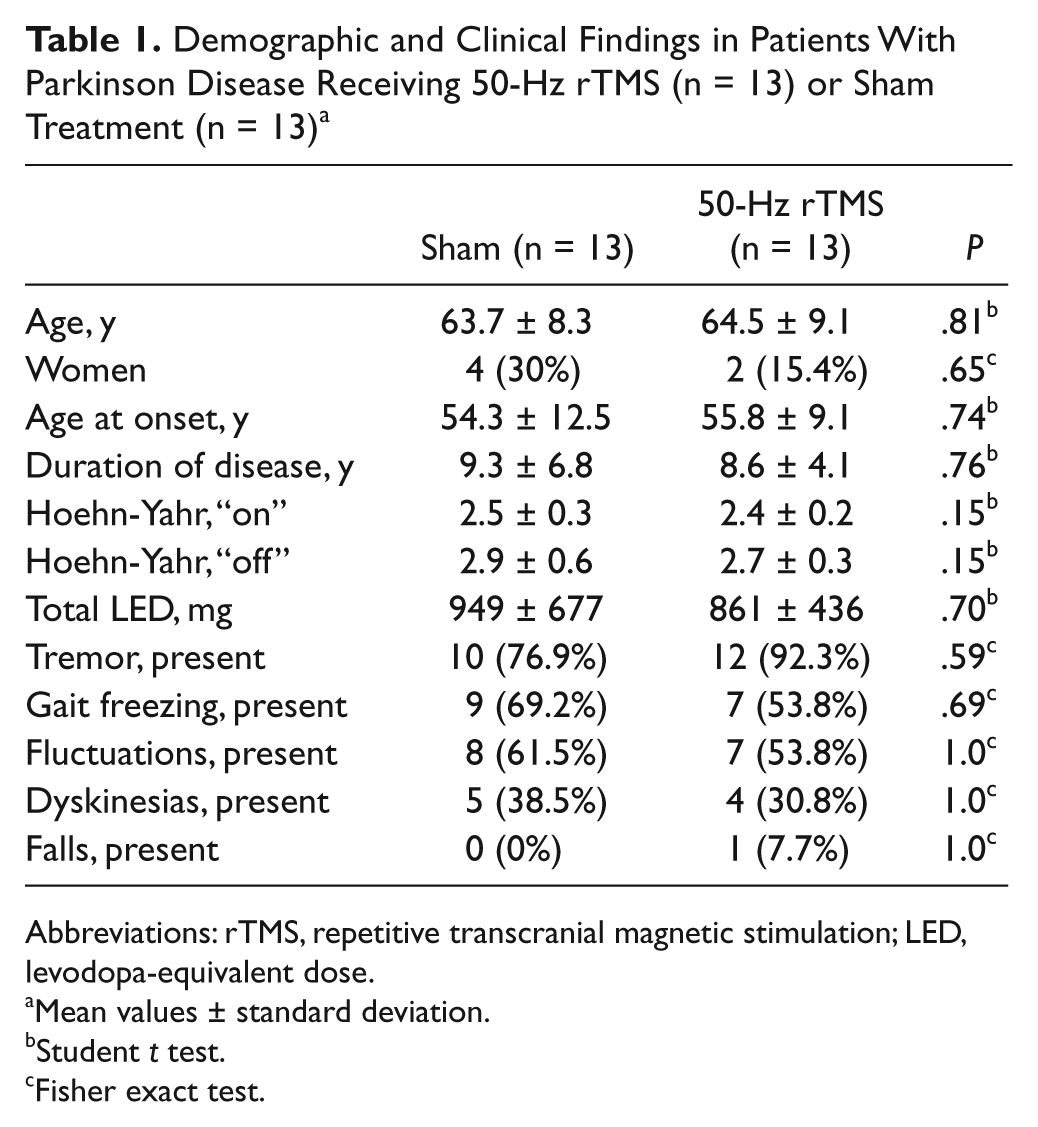
Abbreviations: rTMS, repetitive transcranial magnetic stimulation; LED, levodopa-equivalent dose.
Mean values ± standard deviation.
Student t test.
Fisher exact test.
Safety Testing
We observed no clinical or neurophysiological signs of impending or actual epileptic activity during or after the first intervention and no EEG changes after the last intervention. We found no worsening after the first intervention (Supplementary Table 1) in gait (Treatment, P = .56) and sequential hand and arm movements (Treatment, P = .26), but all improved despite practice before enrollment (Time, P = .021 and <.001). UPDRS motor score (Treatment, P = .86), verbal fluency (Treatment, P = .45), and FAB performance (Treatment, P = .33) remained unchanged. In the SRTT, reaction time shortened slightly (Time, P = .004) and similarly in both groups (Treatment, P = .44).
Gait
No participant depended on assistive devices. Walking time decreased in on and off states (Figure 2A; Table 2; Time, P < .001 and P = .002), but 50-Hz rTMS had no effects on gait in either state (Treatment, P = .40 and P = .29; Treatment-Time, P = .85 and P = .98). We found no changes in the Falls and Gait Questionnaire and in the Freezing of Gait Questionnaire (Table 3; Treatment, P = .5 and P = .57; Treatment-Time, P = .12 and P = .073; Time, P = .76 and P = .83), either.
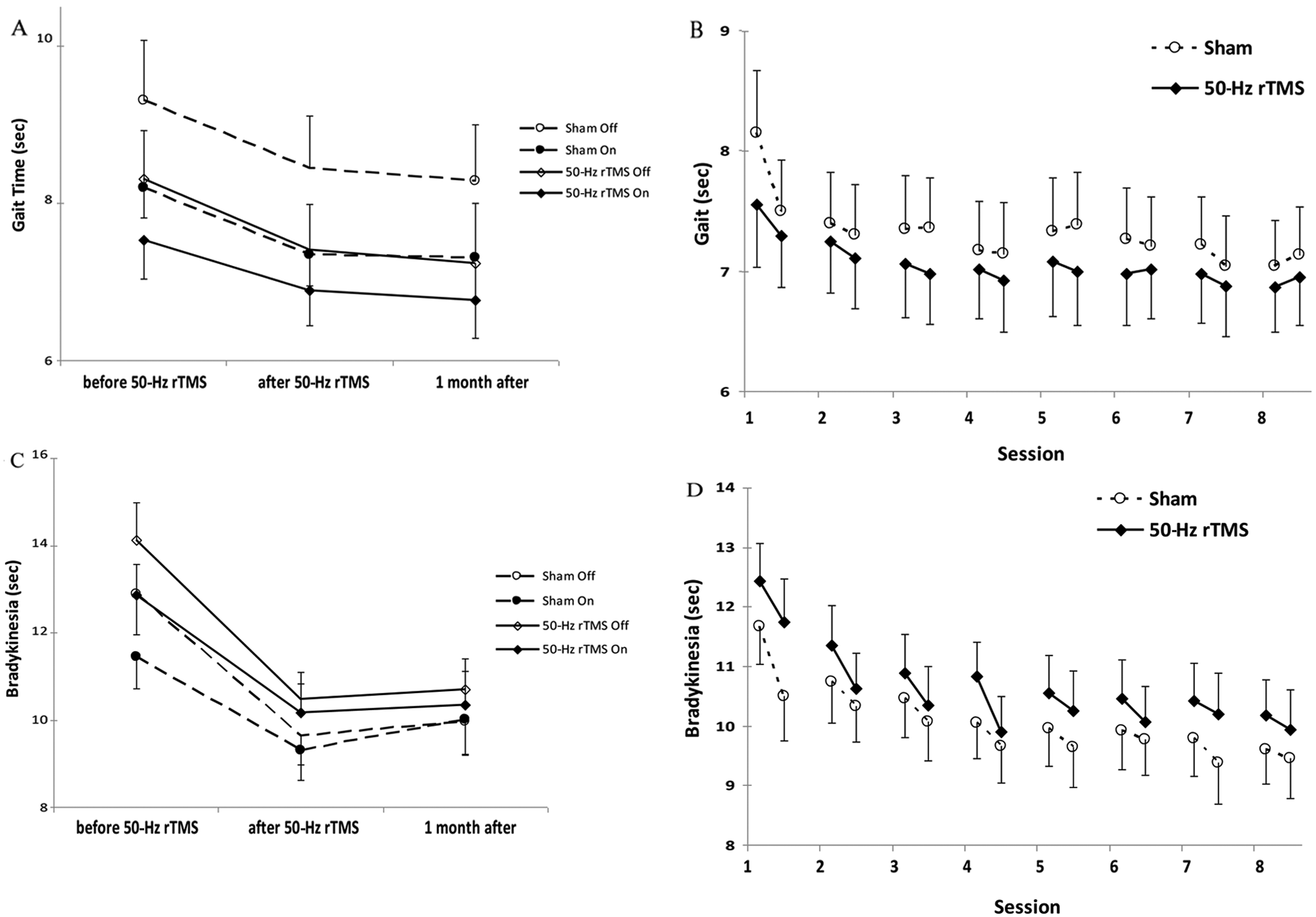
Gait and sequential hand and arm movement time: A. Gait time before, 1 day, and 1 month after the last intervention (mean ± standard error). The figure shows the time needed to walk 10 m in the on and off states. Abscissa indicates the time of measurement and ordinate indicates the gait time. The solid lines and diamonds indicate the 50-Hz rTMS (n = 13) and the dashed lines and circles the sham group (n = 13). Open symbols indicate the off (medication) condition, and filled symbols indicate the on condition measurements. B. Gait time before and after each intervention (mean ± standard error). The figure shows the time needed to walk 10 m. Abscissa indicates the time of measurement; ordinate indicates the walking time. The solid lines and filled diamonds indicate the 50-Hz rTMS (n = 13), and the dashed lines and open circles the sham group (n = 13). At baseline, gait time did not differ between groups. C. Sequential hand and arm movement test before, 1 day, and 1 month after the last intervention (mean ± standard error). The figure shows the time needed to execute the sequential hand and arm movement test in the on and off states. Measurements for the left and right hands were pooled. Abscissa indicates the time of measurement. Ordinate indicates the execution time. The solid lines and diamonds indicate the 50-Hz rTMS (n = 13) and the dashed lines and circles the sham group (n = 13). Open symbols indicate the off (medication) condition and filled symbols indicate the on condition measurements. D. Sequential hand and arm movement test before and after each intervention (mean ± standard error). The figure shows the time needed to execute the sequential hand and arm movement test. Measurements for the left and right hands were pooled. Abscissa indicates the time of measurement; ordinate indicates the execution time. The solid lines and filled diamonds indicate the 50-Hz rTMS (n = 13), and the dashed lines and open circles the sham group (n = 13). At baseline, sequential hand and arm movement times did not differ between groups. Abbreviation: rTMS, repetitive transcranial magnetic stimulation.
Gait and Sequential Hand and Arm Movement Time (mean ± standard error) at Baseline, a 1 Day, and 1 Month After the Last 50-Hz rTMS and Sham Intervention
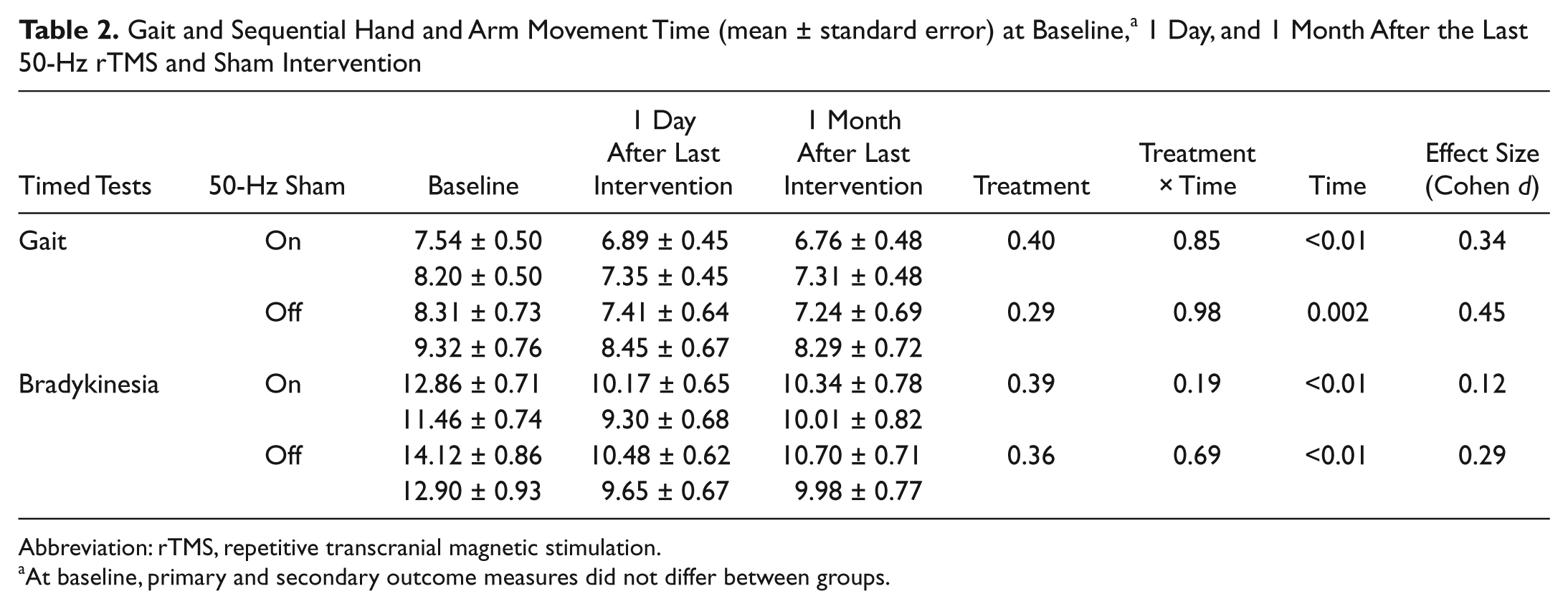
Abbreviation: rTMS, repetitive transcranial magnetic stimulation.
At baseline, primary and secondary outcome measures did not differ between groups.
Secondary Outcome Measures (Mean ± Standard Error) at Baseline, a 1 Day, and 1 Month After the Last 50-Hz rTMS and Sham Intervention
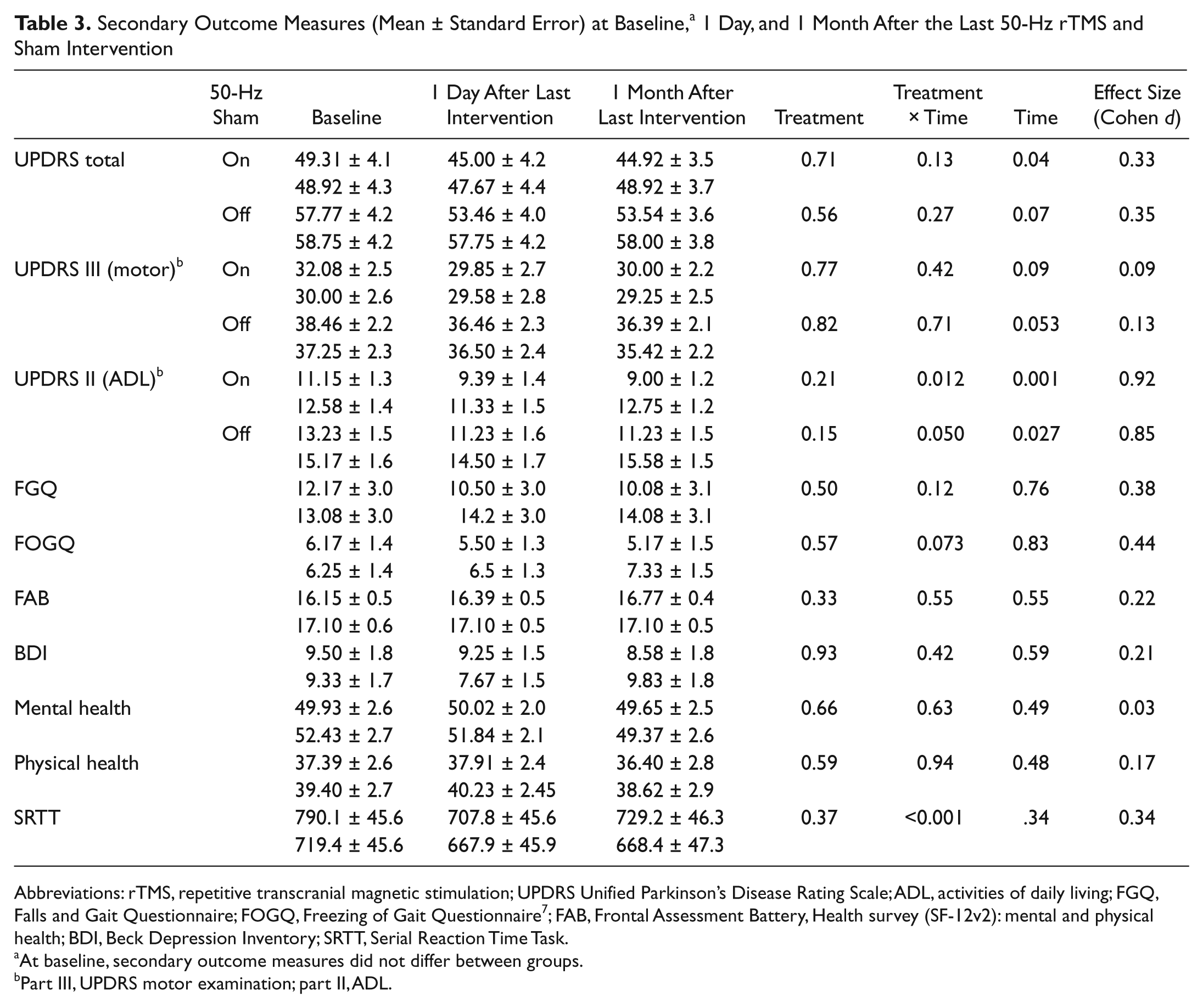
Abbreviations: rTMS, repetitive transcranial magnetic stimulation; UPDRS Unified Parkinson’s Disease Rating Scale; ADL, activities of daily living; FGQ, Falls and Gait Questionnaire; FOGQ, Freezing of Gait Questionnaire 7 ; FAB, Frontal Assessment Battery, Health survey (SF-12v2): mental and physical health; BDI, Beck Depression Inventory; SRTT, Serial Reaction Time Task.
At baseline, secondary outcome measures did not differ between groups.
Part III, UPDRS motor examination; part II, ADL.
Walking became faster after each session (Figure 2B; Supplementary Table 2; Session, P < .001), but there was no discernible effect of 50-Hz rTMS (Treatment, P = .66; Treatment-Session, P = .64; Treatment-Session-Time, P = .5).
Bradykinesia
Sequential hand and arm movements became faster (Figure 2C; Time, P < .001 on and off), but no effect of 50-Hz rTMS could be discerned in the on or off states (Treatment, P = .39 and P = .36; Treatment-Time, P = .19 and P = .69). Movement time decreased after every intervention session (Figure 2D; Supplementary Table 2; Session, P < .001) without differences between groups (Treatment, P = .51; Treatment-Session, P = .63; Treatment-Session-Time, P = .33).
Unified Parkinson’s Disease Rating Scale
The 50-Hz rTMS had no effects on UPDRS scores in the on and off states, including motor examination (Table 3; part III of UPDRS; Treatment, P = .77 and P = .82; Treatment-Time, P = .42 and P = .71) and total score (Treatment, P = .71 and P = .56; Treatment-Time, P = .13 and P = .27). A day after the intervention period, an improvement in activities of daily living was reported (part II of UPDRS; Time, P = .001 and P = .027, resulting in reduction in the total UPDRS score: Time, P = .04 and P = .066), and this was significantly more in the 50-Hz rTMS group on medication (Treatment-Time, P = .012, post hoc, P = .038; in the off state, P = .05), but these effects had disappeared at 1 month (Treatment, P = .21 and P = .15).
In the SRTT, reaction time shortened (Time, P < .001) without differences between groups (Treatment, P = .37, and Treatment-Time, P = .48). The 50-Hz rTMS did not improve sequence-specific learning or learning rate (Treatment, P > .42 and P = .30; Treatment-Time, P > .44 and P = .61).
The 50-Hz rTMS had no effects on depression scores (Treatment, P = .93; Treatment-Time, P = .42) and mental (Treatment, P = .66; Treatment-Time, P = .63) and physical well-being (Treatment, P = .59; Treatment-Time, P = .94).
Neurophysiology
Rest and active MEP recruitment curves and CSP were similar in the on and off states (P = .68, 0.98, and 0.70). In both recruitment curves, MEP amplitudes increased as did CSP duration with stimulation intensity in all conditions (all P < .001). The 50-Hz rTMS had no effects on rest and active MEP recruitment curves (Treatment, P = .37 and 0.58 [plus factor Med] and P = .40 and 0.31 [no factor Med]) but prolonged CSP (Treatment, P = .003 and 0.04: real > sham stimulation). This effect remained near significant when correcting for baseline (Treatment, P = .078) and fully significant without 90% intensity (Treatment, P = .049), prior to correction for multiplicity (see supplementary file).
Discussion
In this double-blind, randomized, sham-controlled study, repeated 50-Hz rTMS of the motor cortex (M1) did not improve gait, upper-extremity bradykinesia, or global motor performance in PD. There appeared a short-lived on-state improvement in activities of daily living (UPDRS II) but without changes in functional status or self-reported well-being. Conversely, the intervention appears to be safe, extending current safety limits of subthreshold 50-Hz rTMS 5 to a duration of 6 s.
The absence of effects on motor function matches findings in our therapeutic trial of intermittent θ-burst stimulation (iTBS). 8 Consequently, there is currently no evidence for therapeutic efficacy of 50-Hz rTMS, neither continuous nor patterned as in iTBS, which consists of bursts of 50-Hz rTMS. In contrast to iTBS, 8 we found no beneficial effects on mood with 50-Hz rTMS. One reason could be that we did not stimulate the dorsolateral prefrontal cortex (DLPFC), which has been found to be comparable to antidepressants in PD patients with depression. 9 We had refrained from this stimulation for concerns over safety of 50-Hz rTMS, which we had solely established for M1. 5 On the other hand, stimulation of DLPFC may not contribute to improvement in motor function. It was found that 25-Hz rTMS of M1 and DLPFC 3 was not superior to stimulation of M1 alone, 4 but these were not directly compared, and an investigation of 10-Hz rTMS targeting the left DLPFC and/or M1 for treatment of freezing of gait was prematurely terminated because of inefficacy. 10 We focused on speed but also assessed gait disturbances with a questionnaire focusing on freezing, which is inherently difficult to ascertain in laboratory conditions. An observation of reduced freezing in the iTBS study suggested potential responsiveness to high-frequency rTMS, which remained unsubstantiated in both 50-Hz rTMS and iTBS trials. 8
Irrespective of type of intervention, we found comparable improvement in rTMS- and sham-treated patients. We presume these to result from motor learning, most manifest in the sequential hand and arm movements and familiarization with the test setting or to reflect a substantial placebo effect. Acute improvement after rTMS and sham intervention could result from striatal dopamine release caused by high-frequency rTMS 11 and can even arise from expectation alone because sham-rTMS had the same effect. 12 The latter could also mediate the placebo response. These effects corroborate findings in the tDCS 13 and iTBS trials 8 and underline the importance of a controlled study design. Conversely, both 50Hz-rTMS and iTBS 8 did not enhance motor learning, in contrast to tDCS. 13
This absence of cumulative effects on gait and bradykinesia with repeated interventions contrasts with the efficacy of 25-Hz rTMS.3,4 Methodological reasons limit comparability between different stimulation patterns, but standardizing changes in the UPDRS motor score in various trials yielded a larger effect size of conventional rTMS 2 than 50-Hz rTMS and iTBS. 8 The discrepancy between significant improvement with 25-Hz rTMS and absence of effects with patterned and continuous 50-Hz rTMS raises questions as to whether and how stimulation patterns might vary in their mechanism of action. The controlled 25-Hz rTMS trial providing the rationale for the current studies had applied the same coil and targets, 3 underlining the specificity of the stimulation pattern and precluding adverse effects of potential simultaneous stimulation of the contralateral hemisphere. But even though we had twice the statistical power to detect a therapeutic efficacy of 50-Hz comparable to 2-Hz rTMS, 3 we found no effects in the best on state that would constitute the reason for an add-on intervention.
We found no effects on recruitment curves either, but cannot exclude a potential effect on the CSP (Figure 3), which needs to be further studied. The CSP is thought to reflect excitability of the motor cortex and to involve inhibitory circuits, which may be mediated by dopamine. 14 Deep brain stimulation (DBS) 15 and conventional rTMS16-18 modulate CSP suggesting that 50-Hz rTMS may activate the same mechanism, which differs from iTBS. 8 The functional significance of CSP remains unknown, and CSP may not correlate with the motor function.19,20 The lack of clinical efficacy precludes conclusions about the role of physiology in mediating the effects of noninvasive brain stimulation. In this and our previous study, 8 CSP did not differ in the on and off conditions, which is probably because we tested lower stimulation intensities.21,22 Few therapeutic trials have looked at changes in physiology and their correlation with clinical outcome. Increase of MEP amplitudes correlated with clinical improvement with repeated 25-Hz rTMS 3 but not with iTBS, 8 questioning a causal effect. These findings suggest persistent effects, implying that changes were induced in synaptic strength underlying plasticity that remains preserved in PD.23-25
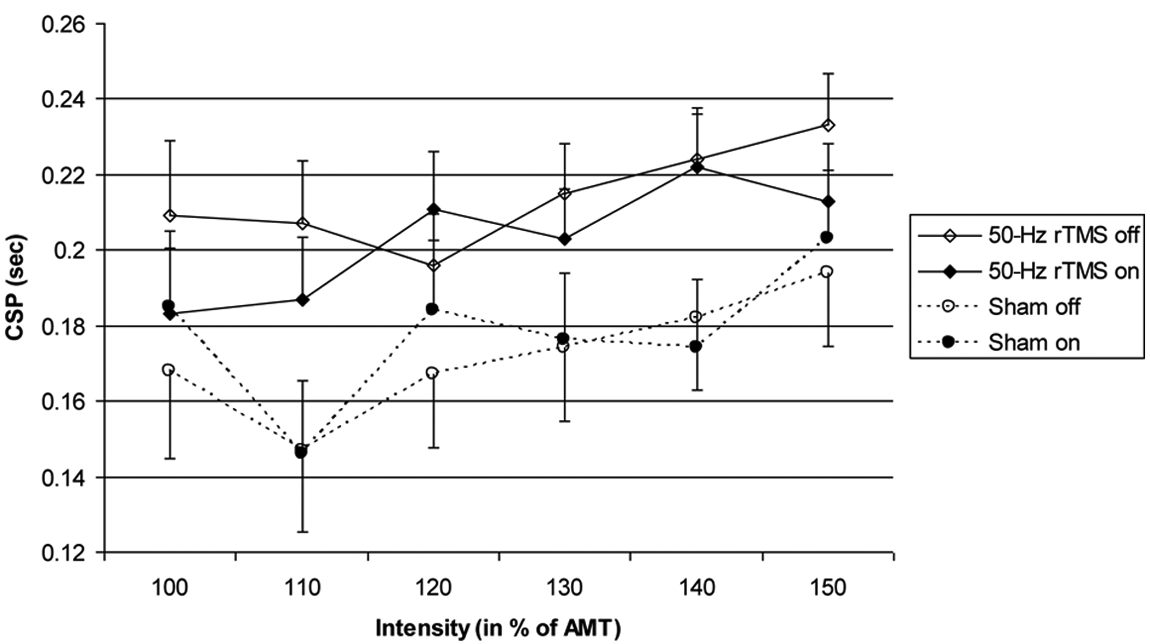
CSP after 8 interventions: CSP 1 day after the last intervention (mean ± standard error). The figure shows the duration of CSP in seconds (abscissa) at different stimulation intensities (in percentage of active motor threshold [ordinate]). The solid lines and diamonds indicate the 50-Hz rTMS (n = 13) and the dashed lines and circles the sham group (n = 13). Open symbols indicate the “off” (medication) condition and filled symbols indicate the “on” condition measurements. In this mixed-model analysis, in which baseline was introduced as covariate and 90% intensity not included because of limited variance (therefore, not shown in the figure), the effect of 50-Hz rTMS on CSP (Treatment, P = .049) is significant prior to correction for multiplicity. Abbreviations: CSP, cortical silent period; rTMS, repetitive transcranial magnetic stimulation; AMT, active motor threshold.
Beyond the postulated potentiation of efficacy by increasing stimulation frequency and, thereby, delivering more power, that is, rate of energy transfer, 4 there might be a rationale specific for 50-Hz rTMS. This arises from the hypothesized role of oscillatory activity in the motor cortex and basal ganglia in motor control and in the pathogenesis of motor disorders and also the speculation about the possibility of modulating this activity. In PD, in the medication off condition, pathological oscillatory activity in the β-frequency range (10-30 Hz) predominates. 26 This β activity decreases in response to dopamine 27 and high-frequency (130 Hz) DBS, 28 whereas γ activity (>30 Hz) emerges along with clinical improvement. 29 Further support comes from β frequency (20 Hz) stimulation of the subthalamic nucleus, which enhances bradykinesia, 30 indicating a potential contribution of β activity to bradykinesia and rigidity in PD. 26 This shift in power of β to γ activity might underlie the effects of dopamine and DBS. 31 rTMS might entrain oscillatory activity, 32 and 50-Hz rTMS might, thereby, induce the hypothesized “prokinetic” γ frequency while suppressing the “akinetic” β frequency. 26 Further research needs to confirm these observations. Safety concerns limit clinical applicability of high-frequency rTMS, and safety of 50-Hz rTMS for longer than 2 s 5 needed to be established first. Longer stimulation might be efficient considering that efficacy of DBS depends on chronic stimulation. This “entrainment” differs from the presumed mechanism in iTBS, which is intended to imitate normal firing patterns in the hippocampus by coupling γ frequency bursts (50 Hz) with a θ rhythm (5 Hz). This is supported by the induction of long-term potentiation and depression, which constitute mechanisms of plasticity in an animal model. 33
This study has limitations. Time demand and safety concerns may have biased patient selection, although few declined and hardly any were disqualified, indicating that this intervention may potentially be applied to most patients. One sham-treated patient withdrew from the 1-month follow-up, but this was a secondary end point. Participants’ reports and robust placebo response suggested blinding was maintained, facilitated by subthreshold rTMS and similar acoustic sensation during sham stimulation. Using different methods of sham stimulation does not appear to influence the outcome of rTMS studies and placebo response. 2
This study provides no evidence for a therapeutic potential of 50-Hz rTMS at the current stimulation parameters, but safety concerns and technical limitations precluded prolonging stimulation. Future protocols can further explore mechanisms of rTMS to establish more powerful stimulation parameters.
Footnotes
Acknowledgements
We thank Elaine Considine and Nguyet Dang for help in the research.
Authors’ Note
The authors’ contributions are as follows: David H. Benninger, design and conceptualization of the study, analysis and interpretation of the data, and drafting and revising the manuscript for intellectual content; Kazumi Iseki, conceptualization of the study, analysis and interpretation of the data, and revising the manuscript for intellectual content; Sarah Kranick, conceptualization of the study, analysis of the data, and revising the manuscript for intellectual content; David A. Luckenbaugh, analysis and interpretation of the data and revising the manuscript for intellectual content; Elise Houdayer, conceptualization of the study and revising the manuscript for intellectual content; Mark Hallett, design and conceptualization of the study, analysis and interpretation of the data, and revising the manuscript for intellectual content.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Hallett has received personal compensation or travel expenses for activities with Neurotoxin Institute, Parkinson’s and Ageing Research Foundation, Columbia University, Blackwell Publishers, Cambridge University Press, Springer Verlag, Taylor & Francis Group, Oxford University Press, John Wiley & Sons, and Elsevier as an advisory board member, an editor, a writer, or a speaker. He has also received license fee payments from the National Institutes of Health for the H-coil, a type of coil for magnetic stimulation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Intramural Research Program of the NINDS, NIH, and in part by a grant from the USAMRMC (W81XWH-06-1-0534).
