Abstract
Introduction
Deep brain stimulation (DBS) that targets the subthalamic nucleus (STN) of the basal ganglia has shown therapeutic potential as an adjunct to pharmacotherapy for alleviating motor symptoms of Parkinson’s disease (PD). 1 While the treatment appears effective in providing relief to the cardinal motor deficiencies of the disease (bradykinesia/akinesia, tremor, and rigidity), the outcomes on gait remain inconclusive.2,3 Evidence from long-term follow-up studies has shown that certain aspects of gait function improve initially postoperatively but then progressively worsen.4,5 It is estimated that gait deterioration occurs in about 25% of patients with PD (PwPD) after subthalamic deep brain stimulation (STN-DBS) 6 and a subset of them may experience fall.7,8 The causes of insufficient gait outcome are not well understood, and this is largely due to the paucity of objective investigations of gait characteristics in PwPD undergoing DBS. 9
Most long-term studies have investigated gait outcomes using Movement Disorder Society-Unified Parkinson’s Disease Rating Scale (MDS-UPDRS-III) clinical motor scores. 10 Although MDS-UPDRS-III is an internationally accepted and widely used clinical assessment scale (rapidly administered to measure clinically relevant outcomes), it only includes very few evaluations on walking ability or gait. The subtleties involved with controlling gait such as symmetry, variability, and coordination are important but remain unevaluated in standardized MDS-UPDRS-III assessments. Moreover, such objective gait characteristics could be used as biomarkers for reliable selection of DBS candidates, 11 adaptive stimulation, 12 and assessing treatment endpoints thereafter. 13
One of the defining symptomatic gait characteristics of PwPD is hypokinesia (slowness of walking) accompanied by small and variable step length. 14 The effects of STN-DBS on walking speed and step length were investigated initially by Allert and co-workers 15 in a pre-post study (follow-up assessments after 3 months) on 8 PwPD with severe disability. They found a significant increase in step length as well as gait velocity and subsequent studies have been able to corroborate this finding in both ON–OFF stimulation as well as pre-post designs.16-18 Previous evidence also suggests beneficial effect of STN‐DBS on hip, knee, and ankle joint range of motions.11,16 However, varying reports on other important gait parameters (e.g., cadence and dual limb support time) and a general lack of long-term objective follow-ups preclude concrete conclusions for the alleviation of gait deficits using STN-DBS in PwPD.2,3,9
One pertinent investigation of STN-DBS in PwPD that has not been adequately described is the effects on asymmetry and dyscoordination of gait. This is particularly important because asymmetrical gait characteristics are often the first motor symptoms seen in these populations, 19 demonstrated as a clinical marker of prodromal PD 20 and discriminate distinct motor stages of PwPD. 21 In clinics, the practice of using bilateral DBS lead implantation is quite common, with surgical targets and stimulation settings often optimized to achieve best effects on upper limb tremor, rigidity etc., while minimizing relevant adverse effects (can be referred to as ‘clinically determined settings’, evaluated using MDS-UPDRS I to IV). However, despite the prevalence of asymmetrical stimulation settings for bilateral DBS, asymmetry, particularly in the lower limbs has been shown to often persist 15 or increase at 6 months after surgery 11 for unknown reasons. Importantly, insufficient attention to asymmetry may be responsible for adverse events including freezing of gait (FOG) and fall episodes in a subgroup of patients after the surgery.22-24
As mobility and functional gait are critical for health and quality of life, an in-depth account of stimulation to gait and clinical outcome relations seems then essential for developing a comprehensive DBS therapy in PwPD. The current study is part of a larger research project designed to investigate the predictive value of objective gait measures for DBS therapy outcomes in PwPD. In an exploratory study design, we systematically investigated alterations in our primary outcome: MDS-UPDRS III and our secondary measures: gait asymmetry, dyscoordination, and walking speed 6 months post-DBS in PwPD. We tested the hypothesis that PD motor subtypes, including tremor dominant (TD), postural instability and gait disorder (PIGD), and indeterminate (neither tremor nor postural instability as defining features) types would show distinct therapeutic effects on clinical and gait outcomes.
Methods
Study Design
Baseline and Longitudinal Demographics and Clinical Characteristics.
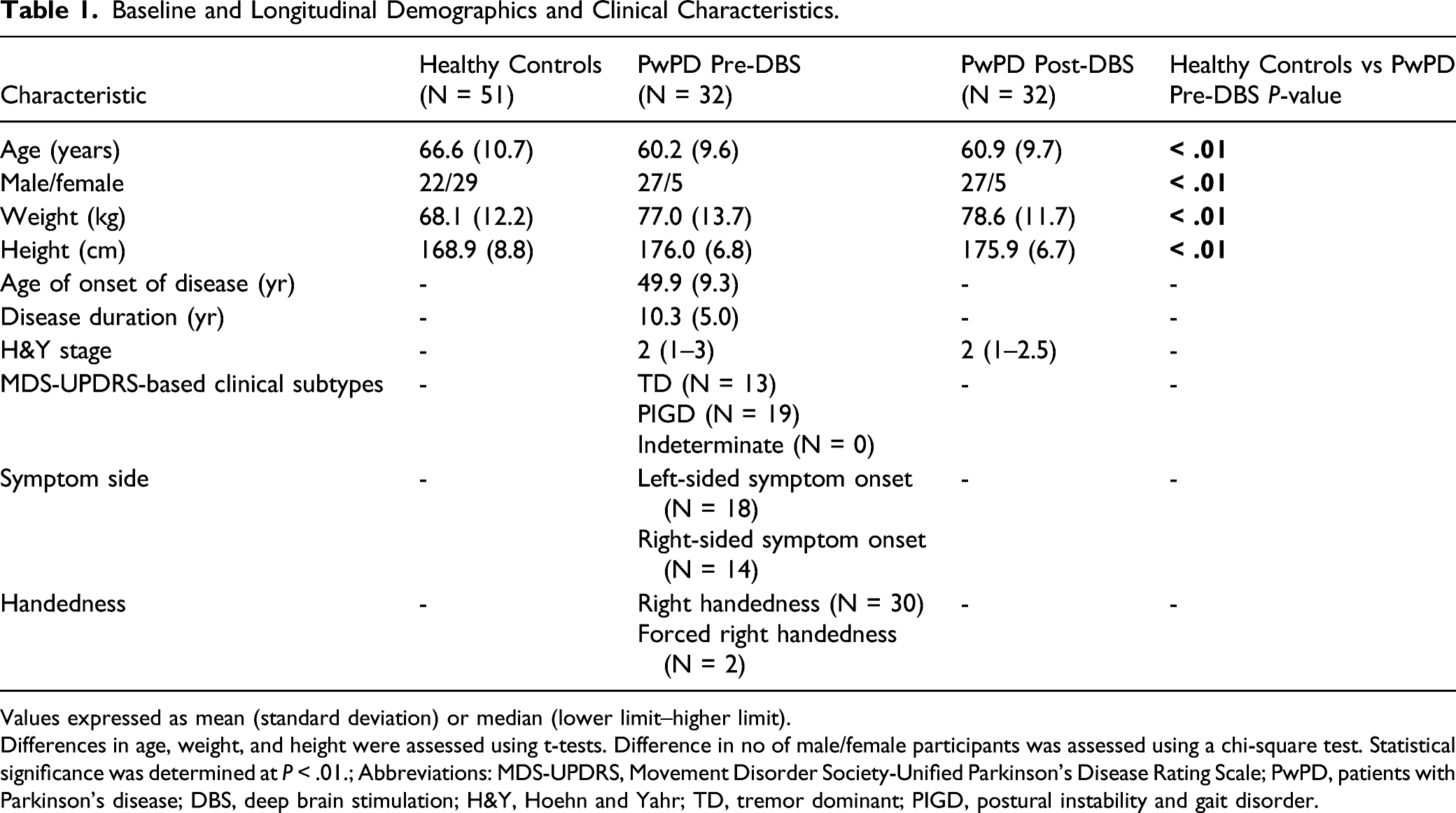
Values expressed as mean (standard deviation) or median (lower limit–higher limit).
Differences in age, weight, and height were assessed using t-tests. Difference in no of male/female participants was assessed using a chi-square test. Statistical significance was determined at
Data Collection
Clinical Assessment
Motor symptoms were assessed using MDS-UPDRS III, evaluated twice pre-surgery—in the ON and OFF medication conditions (as part of the levodopa challenge test used to screen DBS candidates that demonstrates levodopa responsiveness 25 )—as well as once around 6 months post-surgery in the medication and stimulation ON condition. MDS-UPDRS I (non-motor symptoms), II (activities of daily living), and IV (dyskinesias and motor fluctuations) were also performed once (OFF medication condition) before surgery and once after surgery. In addition, dopaminergic treatment was recorded as levodopa equivalent daily dose. The clinical subtypes of PwPD such as TD, PIGD, and indeterminate types were also identified. 26 The predominant symptom side was identified by medical history and during the clinical examination and substantiated using the MDS-UPDRS asymmetry: difference between left and right motor scores of MDS-UPDRS Part III (items 3.3–3.8 and 3.15–3.17). 27
Objective Gait Measures
PwPD were tested prior to surgery in the medication ON state, and once around 6 months after the surgery in the ON medication and ON stimulation condition. Here, most subjects in the post-condition were still taking a clinically adapted dose of medication (Supplementary Methods 2), except for 3 subjects that were completely OFF medication. The evaluators who performed the gait analysis were not blinded to the group status (e.g., asymptomatic controls vs PwPD). All participants were instructed to walk (barefoot, self-selected speed, and without any aid or assistance) continuously for 10 minutes in an eight-shape around 2 marked spots 10 m apart. The use of an “8-walk” protocol allows the collection of a high number of consecutive gait cycles during overground walking that is necessary in order to reliably assess gait performance. 28 A three-dimensional motion capture camera system (10 cameras; 61 markers; 100 Hz; Vicon Nexus, version 2.3/2.8.2, Oxford Metrics, United Kingdom) was used to record the movements. Before each data collection, a calibration routine was performed across the entire capture volume (calibration error threshold set at 0.5 mm). In addition, we followed the published recommendations for minimizing error associated with marker placement. 29 The trajectories of the heel (calcaneus), toe (metatarsal head III), and sacrum (midpoint between left and right posterior superior iliac spines) markers were used to extract gait kinematics.
Data Analyses
Pre-processing
The raw kinematic data (from 2 × 7-m straight sections of the 8-walk) were low pass filtered (zero phase fourth order Butterworth with cut-off frequency of 25 Hz). The gait events (heel strikes and toe-offs) were automatically extracted using a custom algorithm based on foot velocity. 30
Asymmetry ratio
Spatiotemporal gait measures of step length, step time, swing time, and stance time (Supplementary Methods 2) were evaluated for each foot separately. All outliers defined as intervals outside ±4 median absolute deviation (MAD) away from the median were eliminated. The number of outliers eliminated by this process across all parameters and participant groups was 15 (maximum) out of 221 (average) walking steps. For each subject, we then determined which foot had the larger vs smaller amplitude (e.g., longer vs shorter mean step length) for evaluating the asymmetry ratios
31
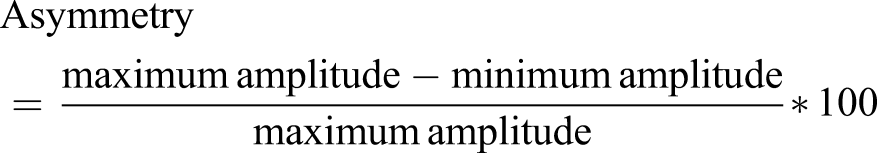
Asymmetry ratio of zero percentage indicates perfect symmetry.
Phase coordination index
Bilateral dyscoordination in left–right stepping phase was analyzed using the phase coordination index (PCI, 32 Supplementary Methods 2). Lower PCI values reflect a more accurate and consistent left–right stepping phase generation.
Statistical Analyses
For the comparison of
Clinical scores
Changes in the overall clinical scores were computed as: [(Pre-surgery scores – Post-surgery scores)/Pre-surgery scores] × 100%. The Wilcoxon signed-rank test was used to determine difference between the scores. Significance was set at
Changes to individual items (and composite of sub-items) were computed as mean differences [95% confidence interval]. The procedure for obtaining bootstrap confidence interval 36 is detailed in Supplementary Methods 3. These changes are not tested for significance because of large number of ties (no differences) in the data.
Gait characteristics
For the investigation of the neuromodulatory effects of DBS treatment on asymmetry and dyscoordination, effect sizes (ES) (Hedges’ g
36
) with 95% confidence interval CI (Supplementary Methods 3) were calculated using bootstrap resampling. The 95% CI on the effect size gives a measure of precision and confidence about our estimate and is not sensitive to the distribution of observations or underlying populations.
37
The rationale for including a control group is to demonstrate the importance of the direction of intervention effects (e.g., modification in asymmetry with respect to physiological asymmetry observed in asymptomatic controls without PD). The
All analyses were conducted in MATLAB (v2019a, The Mathworks Inc., USA) and R (version 1.2.5033, The R Foundation for Statistical Computing, Austria).
Results
Patient and Demographic Data
The overall subject dropout rate was 13.5% (5 PwPD). One participant dropped out before surgery and 4 participants dropped out after surgery as presented in Supplementary Figure 1. There were significant differences (
Clinical Characteristics
Effect of Bilateral Stimulation of the Subthalamic Nucleus on MDS-UPDRS scores.
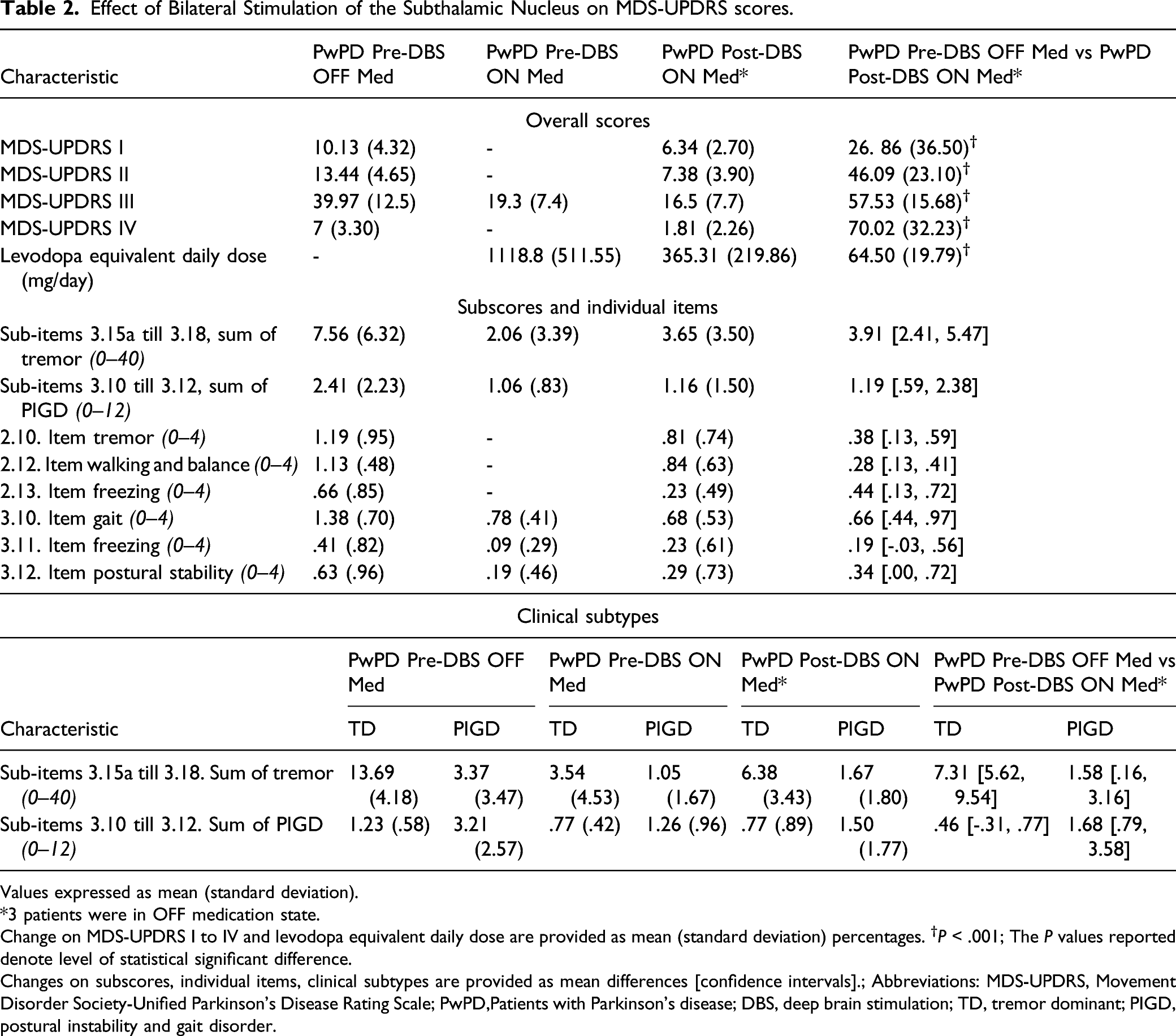
Values expressed as mean (standard deviation).
*3 patients were in OFF medication state.
Change on MDS-UPDRS I to IV and levodopa equivalent daily dose are provided as mean (standard deviation) percentages. †
Changes on subscores, individual items, clinical subtypes are provided as mean differences [confidence intervals].; Abbreviations: MDS-UPDRS, Movement Disorder Society-Unified Parkinson’s Disease Rating Scale; PwPD,Patients with Parkinson’s disease; DBS, deep brain stimulation; TD, tremor dominant; PIGD, postural instability and gait disorder.
A subgroup analysis was performed on MDS‐UPDRS III items that address clinical subtypes. The average
Gait Characteristics PwPD
PwPD had higher asymmetry ratios (highest effect size reported for step length asymmetry, ES [CI]: .56 [.06, 1.03],
Effect of Bilateral Stimulation of the Subthalamic Nucleus on Gait Characteristics.
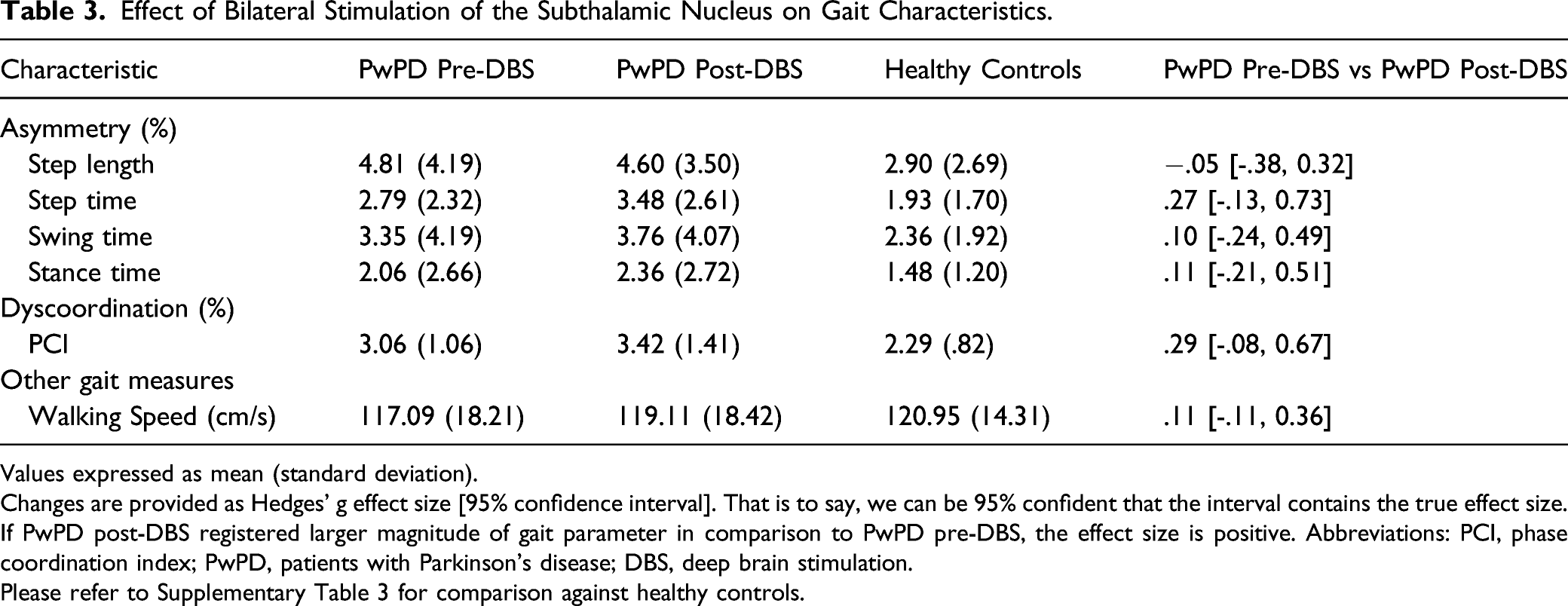
Values expressed as mean (standard deviation).
Changes are provided as Hedges’ g effect size [95% confidence interval]. That is to say, we can be 95% confident that the interval contains the true effect size.
If PwPD post-DBS registered larger magnitude of gait parameter in comparison to PwPD pre-DBS, the effect size is positive. Abbreviations: PCI, phase coordination index; PwPD, patients with Parkinson’s disease; DBS, deep brain stimulation.
Please refer to Supplementary Table 3 for comparison against healthy controls.
Gait Characteristics Clinical Subtypes
Between subtypes
Effect of Bilateral Stimulation of the Subthalamic Nucleus on Gait Characteristics: Clinical Subtypes.
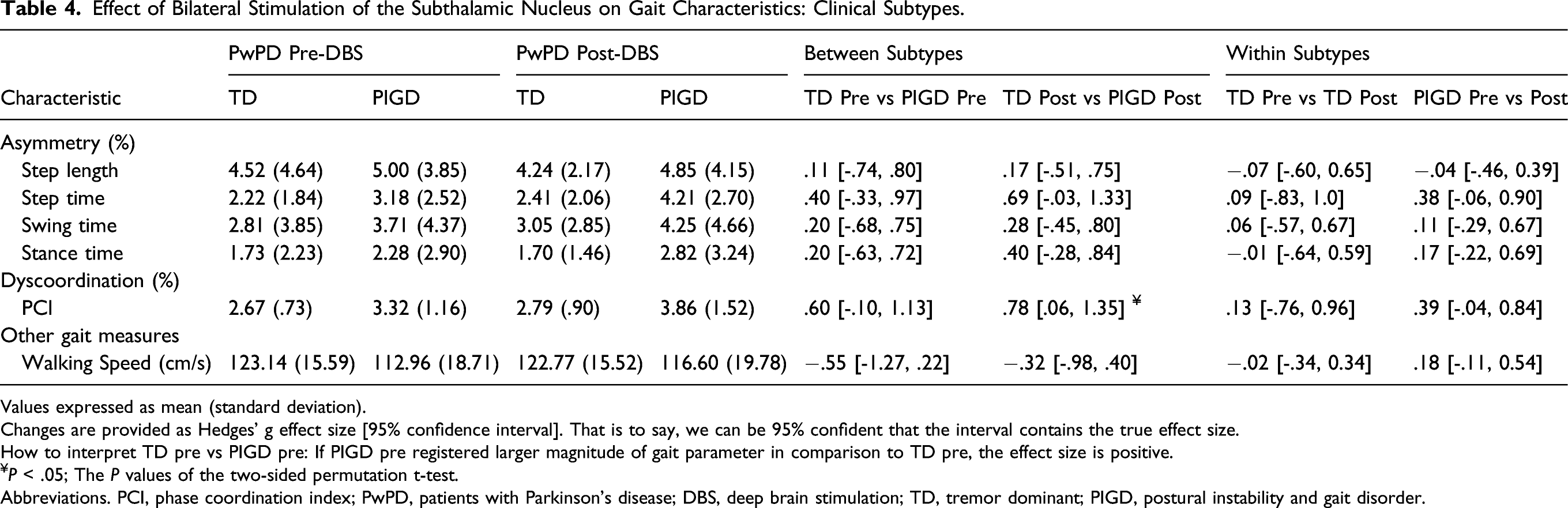
Values expressed as mean (standard deviation).
Changes are provided as Hedges’ g effect size [95% confidence interval]. That is to say, we can be 95% confident that the interval contains the true effect size.
How to interpret TD pre vs PIGD pre: If PIGD pre registered larger magnitude of gait parameter in comparison to TD pre, the effect size is positive.
¥
Abbreviations. PCI, phase coordination index; PwPD, patients with Parkinson’s disease; DBS, deep brain stimulation; TD, tremor dominant; PIGD, postural instability and gait disorder.
Within subtypes
Step time asymmetry (.38 [-.06, .90]) and PCI (.39 [-.04, .84]) increased in the PIGD group following surgery. Differences in all other gait characteristics within subtypes, between pre- and post-surgery were negligible (Table 4).
Discussion
Our exploratory study was designed to investigate the therapeutic effects of STN‐DBS on gait asymmetry, dyscoordination, and walking speed 6 months post-surgery in PwPD. Our results revealed the following: (1) As anticipated, PwPD showed significant clinical motor and non-motor improvement (MDS-UPDRS I through IV) after surgery, together with a significant reduction in dopaminergic medication. (2) STN-DBS induced increase in step time asymmetry and dyscoordination at follow-up. (3) Group differences in baseline (before surgery) gait characteristics are critical to discretize treatment planning, as indicated by higher level of asymmetry, dyscoordination, and slower walking speed in PIGD patients compared to the TD group. Six-month increase in step time asymmetry and dyscoordination after surgery were higher in the PIGD group than the TD group. Collectively, our study documents novel aspects of functional differences in DBS treatment response guided by different motor subtypes with an eye toward their relevance as objective biomarkers for use in clinical settings.
Mild asymmetry in the walking patterns of asymptomatic healthy individuals is quite common and plausibly functional. 38 It is likely that such asymmetrical patterns during walking persist as a result of the natural hemispheric functional specificities in the relative contribution of lower limbs to propulsion (facilitate forward progression) and braking (facilitate postural stabilization).38,39 In PwPD, asymmetry markedly increases and has been directly related to poor walking ability40,41 and adverse episodes such as FOG and falls.16,42,43 The underlying pathophysiology leading to these deficits are multifactorial and have not yet been completely understood. We may conceivably (albeit inconclusively) attribute these deficits to a bilateral but asymmetric neurodegenerative process that is characteristic of PD (asymmetrical motor function may persist over the years despite bilateral disease progression 44 ). Patients with PD (PwPD) also appear to have reduced corpus callosum function, which interferes with the normal bilateral coordination of limb movements during walking. 45 Although, there is only a marginal agreement on the underpinnings of asymmetrical motor and gait symptoms in PwPD, such features are starting to provide essential movement biomarkers to guide diagnosis and treatment.19,21
DBS treatment seems like an ideal approach for improving asymmetry and dyscoordination of walking as it provides the opportunity to modify stimulation parameters for each hemisphere independently. However, we observed that STN-DBS increased gait asymmetry and dyscoordination, when comparing pre vs post-surgery. Similar to our findings, one recent study 11 also reported increased gait asymmetry 6-month post-DBS compared to preoperative assessment. These results differ from a previous work that demonstrated a significant improvement in gait asymmetry in response to STN-DBS treatment. 16 The difference in results can be attributed primarily to the study design (Johnsen and colleagues only presented postoperative DBS OFF vs ON gait outcomes in OFF medication state). Postoperative microlesion effects (e.g., some permanent lesion related to electrode insertion) may introduce bias 46 and does not allow us to equate and compare the pre-DBS state (of our study) to OFF DBS state (in Johnsen et al 2009). Also relevant, however, is the definition of gait asymmetry (Johnsen and colleagues looked at spatial distance between heel to projected center of mass 16 ). The increase in asymmetry and dyscoordination that we are reporting is calculated based on temporal metrics (step time asymmetry and PCI).
With specific interest to the observation of increased step time asymmetry (and PCI) and negligible effects on step length asymmetry, our study plausibly suggests a differential effect of STN-DBS on the temporal deficit or a possible adaptation of temporal characteristics to allow regulation of spatial characteristics. This coincides with other studies showing that STN-DBS increases walking speed in patients by spatial (increasing step length) rather than temporal changes (increasing cadence or step time).2,17 There is surmounting evidence indicating that cortical centers are responsible for movement initiation, while “ongoing” movements are steadily regulated via subcortical regions within the basal ganglia and the brain stem.47,48 These regions are responsible for providing internal cues to cortical and subcortical regions, for example, the pre-motor and supplementary motor area, accounting for the regulation of spatial parameters. 49 In this regard, it is possible that the regulation of temporal parameters is not under the influence of cortico-thalamo-basal ganglia circuitry. 50 However, further research is necessary to substantiate the notion that these parameters (cadence and step length, but also asymmetry in step time and length) may reflect distinct regulation but allow mutual interactions. 51
Gait impairments in PwPD, from the symptom onset to their advanced stages, including response to treatments (levodopa 52 and DBS 53 ), demonstrate patient as well as group (TD vs PIGD) specific traits. In our study, the TD group had comparatively less impaired gait to the overall group of patients both before and after surgery. On the other hand, PIGD patients demonstrated a more pronounced increase (more than the longitudinal changes due to disease progression reported previously in PwPD 54 ) in asymmetry and dyscoordination compared to the TD group due to DBS, consistent with a previous clinical investigation. 53 Natural prognosis (PIGD may naturally progress more quickly in disease course 55 ) and inadequate targeting of the STN sub territories responsible for the manifestation of the PIGD subtype 56 likely underlie differences in treatment outcomes, but future studies may shed light on these hypotheses.
Limitations
We note the following limitations in our study. First, the demographic characteristics (age, sex distribution, height, and weight) of the PwPD group were significantly different from asymptomatic controls group, but we did not factor this in the estimation of effect sizes in the present study. A better approach would be to use a statistical test that includes them as covariates; however, this may require large sample sizes which remain to be explored in future analyses. Second, the lack of information about fall history of the participants limited our ability to answer the question whether asymmetric and dyscoordinated gait is associated with increased fall risk after STN-DBS in PwPD. Third, there were also limitations in the design of the study (gait being a secondary outcome) that did not allow us to replicate novel unilateral stimulation protocols suggested toward improving gait asymmetry and dyscoordination in patients with Parkinson’s disease. 23 Fourth, the non-randomized and unblinded design might be another limitation of our study. Last, the relatively small sample of female PwPD may have limited our ability to confirm previous observations57,58 of gender-related factors for STN-DBS outcomes in PwPD.
Conclusion
The present findings show the efficacy of DBS of the STN in reducing motor symptoms and related clinical outcomes in PwPD at 6 months after surgery. Despite clinical improvement, STN-DBS treatment altered temporal asymmetry and dyscoordination of walking. In particular, PIGD patients demonstrated a more pronounced decline in these gait characteristics compared to the TD group.
STN is a complex subcortical motor network and regulate a myriad of motor features required in daily life of PwPD. 59 Current approaches to STN-DBS therapy are based on an inadequate assessment of such features and thus hinder providing any definitive recommendations about the therapy. Our study provides preliminary insights into the mechanisms possibly regulating DBS outcomes for subtle, yet important gait features like symmetry and coordination. However, our findings ought to be validated in future randomized controlled trials prior to translating into clinical rehabilitation settings.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683211041309 – Supplemental Material for Does Subthalamic Deep Brain Stimulation Impact Asymmetry and Dyscoordination of Gait in Parkinson’s Disease?
Supplemental Material, sj-pdf-1-nnr-10.1177_15459683211041309 for Does Subthalamic Deep Brain Stimulation Impact Asymmetry and Dyscoordination of Gait in Parkinson’s Disease? by Deepak K. Ravi, Christian R. Baumann, Elena Bernasconi, Michelle Gwerder, Niklas K. Ignasiak, Mechtild Uhl, Lennart Stieglitz, William R. Taylor and Navrag B. Singh in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgments
The authors would like to thank all the subjects for participating in the study and their families for the support provided.
Author Contributions
NKI, DKR, NBS, CRB, and WRT conceived and designed the study. CRB, LS, and MU coordinated the recruitment, pre-surgery, and post-surgery of the participants. DKR, MG, EB, and NKI coordinated the gait measurements. DKR, MG, and EB analyzed the data. DKR, NBS, and WRT drafted the manuscript. CRB provided clinical content expertise while drafting and revising the manuscript. LS provided critical opinion and revision of the manuscript as a subject expert. All the authors reviewed and approved the manuscript for submission. WRT is the guarantor.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: DKR was supported by a PhD scholarship from
Data Availability
Please contact the corresponding author for requests regarding data sharing and collaboration.
Ethics Approval
The study was approved (approval no: 2015-00141) by Zurich Cantonal Ethics Commission and carried out in accordance with the Declaration of Helsinki, the guidelines of Good Clinical Practice, and the Swiss regulatory authority’s requirements. The subjects all provided written, informed consent prior to participation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
