Abstract
Background. Much variation is found in the development of hand function in children with unilateral cerebral palsy (CP). Objective. To explore how anatomic brain abnormalities can be used to predict the development of hand function. Methods. A total of 32 children with unilateral CP (16 boys and 16 girls) were evaluated at least once a year by the Assisting Hand Assessment (AHA). The data collection covered an age range from 18 months to 8 years (mean time in study, 4 years and 6 months). Computerized tomography or magnetic resonance imaging of the brain were assessed for patterns of brain damage, including the location of gray and extent of white-matter damage. The children were divided into groups according to lesion characteristics, and a series of univariate models were analyzed with a nonlinear mixed-effects model. The rate and maximum limit of development were calculated. Results. The highest predictive power of better development of hand function was the absence of a concurrent lesion to the basal ganglia and thalamus, independent of the basic type of brain lesion. This model predicted both the rate of increasing ability and hand function at age 8 years. Hand function was also predicted by the basic pattern of damage and by the extent of white-matter damage. The presence of unilateral or bilateral damage had no predictive value. Conclusions. Neuroradiological findings can be used to make a crude prediction of the future development of the use of the affected hand in young children with unilateral CP.
Keywords
Introduction
Early predictors of motor development in children who are at risk for cerebral palsy (CP) as a result of a perinatal brain lesion are sought by parents and professionals. They wish to know the risk for long-term impairments and whether to initiate early interventions to improve motor function. No such predictors are available. Impaired function in 1 hand is a major limitation in children with unilateral CP. We recently reported that use of the affected hand at 18 months of age according to the Assisting Hand Assessment (AHA) correlated with later hand development and could therefore be used to predict both the rate of development and the ability level at later age. 1 Children who spontaneously used their hand for grasping at age 18 months had a more rapid development and reached a higher ability level than those who did not use the hand or who at best could only hold on to objects. Children in the “higher ability group” reached 90% of their ability limit at 3 years of age, whereas children in the “lower ability group” developed more slowly and reached 90% of their limit at 7 years of age. 1
From a clinical perspective, it is relatively late to make predictions on future functioning at 18 months of age. One possible solution is whether neuroimaging information about the location and extent of a perinatal lesion can predict later hand motor function.2-6 Previous research has shown that the type of brain lesion is associated with overall motor function.2-3,7-11 However, description of hand function has not been the focus of these studies, and when it has been included, the developmental perspective has not been considered. A recent cross-sectional study demonstrated that better hand function (measured with the Melbourne Assessment of Unilateral Upper Limb Function) was found in children with periventricular white-matter lesions than in those with gray-matter lesions. Children with a pure white-matter lesion performed better than those with mixed lesions, and children with middle cerebral artery (MCA) distribution infarcts and lesions in the basal ganglia and thalamus had the lowest performance. 12 The large impact of basal ganglia damage on hand function has been shown in other cross-sectional studies.13,14 Because there is a large variation in the type, extent, and localization of brain abnormalities in CP, important predictive information can probably be obtained if the brain abnormality is considered in relation to the progress of the early development of hand function. The aim of the current study, therefore, was to explore how different characteristics of brain abnormalities can be used to predict the development of hand function in children with unilateral CP.
Methods
Participants
We recruited 30 children with unilateral CP for this study. They had been included in a previous study of the longitudinal development of hand function, 1 and clinical magnetic resonance (MR) images or computer tomography (CT) scans of the brain were available that were of sufficient quality to allow analysis of the brain lesions. We also included 2 more children for whom data had been collected according to the protocol of the previous study. In all, 32 children, 16 girls and 16 boys, participated. Their first assessment of hand function was conducted between 18 months and 5 years of age (mean, 2 years and 8 months; standard deviation, 13 months). According to the Manual Ability Classification System, 15 there were 4 children at Level I, 20 children at level II, and 8 children at level III (Table 1). The study was approved by the Research Ethics Board at the Karolinska Hospital in Stockholm, Sweden. Informed written consent was obtained from guardians.
Participants
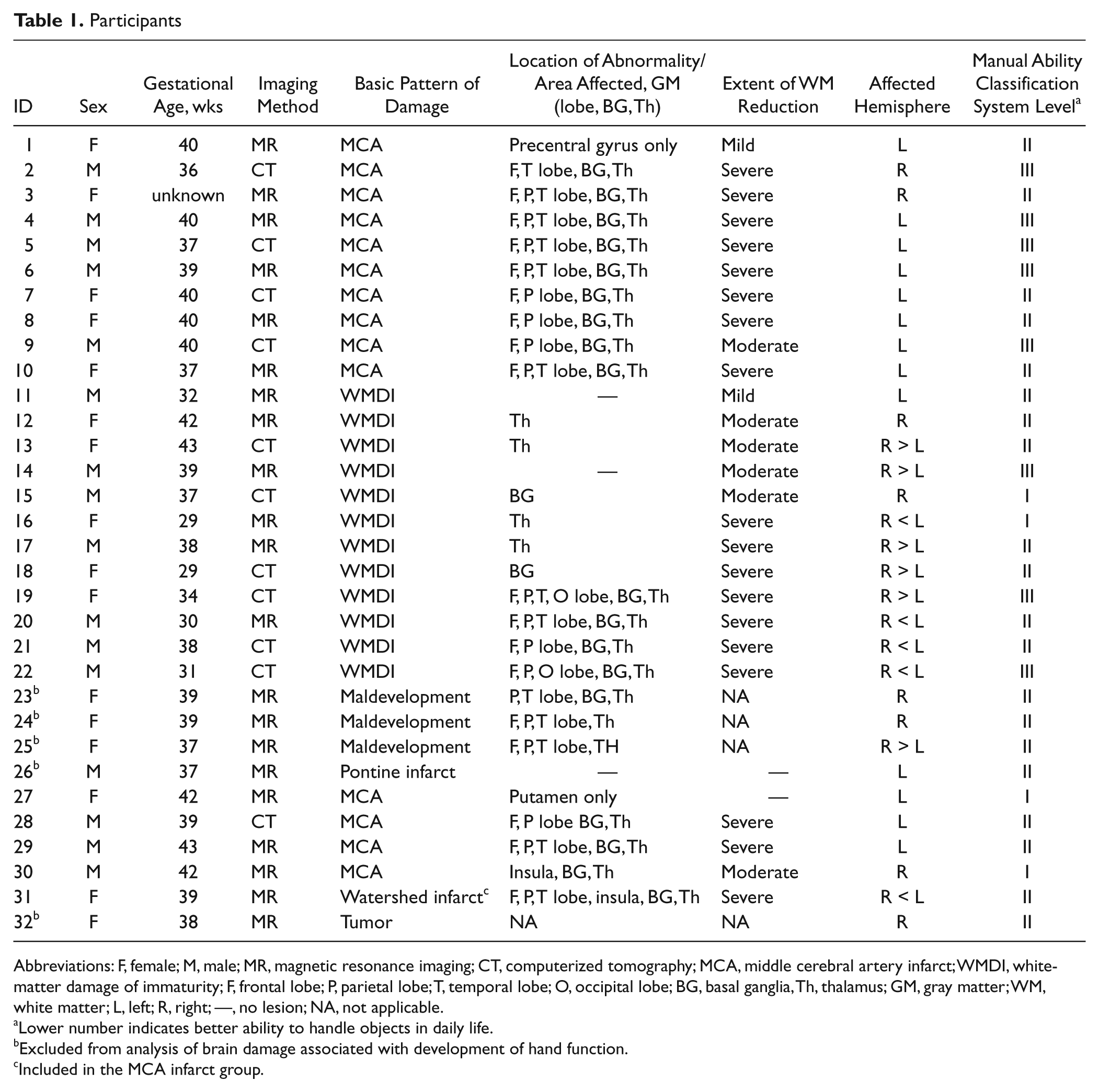
Abbreviations: F, female; M, male; MR, magnetic resonance imaging; CT, computerized tomography; MCA, middle cerebral artery infarct; WMDI, white-matter damage of immaturity; F, frontal lobe; P, parietal lobe; T, temporal lobe; O, occipital lobe; BG, basal ganglia, Th, thalamus; GM, gray matter; WM, white matter; L, left; R, right; —, no lesion; NA, not applicable.
Lower number indicates better ability to handle objects in daily life.
Excluded from analysis of brain damage associated with development of hand function.
Included in the MCA infarct group.
Hand Function Measure
The main outcome was the AHA, which measures and describes how effectively children with unilateral disability use their affected hand in bimanual tasks.16,17 The children were tested repeatedly with the AHA over an average time of 4 years and 6 months (range, 2 years and 2 months to 7 years and 2 months), covering an age range from 18 months to 8 years. Each child was tested at least once a year, rendering 3 to 11 assessments (mean, 6.7) per child, for a total of 215 AHA assessments. The AHA raw scores were converted to interval-level data by means of a Rasch analysis, and linearly rescaled Rasch measures in the range, 0 to 100 AHA units were obtained (1 AHA unit = 0.19 Rasch measurement logits), in which a higher number indicates a higher ability. The smallest detectable difference for the AHA is 0.97 logits, 17 which corresponds to 5 AHA units.
Neuroimaging
In all, 21 children had MR scans, and 11 children had CT scans of the brain, obtained for clinical purposes. The age at imaging ranged from 9 months to 9 years. Conventional structural MR or CT scans of the brain had been acquired using various imaging protocols and equipment. CT scans were only used if the quality was good enough to make a detailed description in the analysis. In the case of MR scans, 1.5 T MRI systems were used. All images were visually reassessed specifically for this study by 2 experienced neuroradiologists (OF, AK), both of whom were unaware of the clinical diagnosis and functional outcome. Images were assessed for the basic pattern of damage and for the location and extent of white-matter and gray-matter damage (Table 1). The basic pattern of damage was classified as white-matter damage of immaturity (WMDI), infarcts of the MCA, or maldevelopment. In children with bilateral lesions, only the most affected hemisphere was reported and analyzed. In each case, the most affected hemisphere was contralateral to the hemiparetic side. The extent of white-matter damage was visually classified as mild, moderate, or severe. The gray-matter damage was located in the cerebral lobes, basal ganglia, and thalamus. Basal ganglia and thalamus damage was defined as reduced volume, which in some cases were associated with signal changes on T2-weighted MR images.
Statistical Analysis
A nonlinear mixed effects model was used to create developmental trajectories of each child. In the nonlinear model, the developmental trajectories were assumed to take the shape of a negative exponential function 18 : AHA = limit − (limit − start) × exp (−rate × age). In the model, the AHA score was assumed to start at zero at birth and increase at a fast rate in early ages. The rate of increase slows at later ages and eventually levels out to reach a maximum limit of development, which in this study is equal to the AHA measure at age 8 years. These assumptions were confirmed in the previous study, 1 which had been based on almost the same cohort of children. The participants were divided into 2 groups depending on their AHA measure at 18 months as had been done also in the previous study. 1
To investigate associations between the development of hand function and the type of brain lesion, 5 different hypothetical models were constructed. The models were based on earlier literature investigating how brain damage influences motor function.2,3,12-14 Because of the small sample size, each model could only include 2 groups of children. In the first model, we hypothesized that the basic pattern of damage could predict the development of hand function, and the children were divided into 2 groups: WMDI and MCA infarct. In the second and third models, we hypothesized that damage in the basal ganglia and thalamus could predict development independently of the basic pattern of damage. In model 2, children were divided depending on whether the basal ganglia were damaged or not. In the third model, children who had concurrent involvement of both basal ganglia and the thalamus were compared with children with only one or none of these structures involved. In the fourth model, we hypothesized that there would be a difference in development between children with different extents of white–matter reduction independently of the basic pattern of damage. In the fifth model, we compared the development of children with unilateral damage with those with bilateral damage. For each model, that was analyzed, the differences between mean rate and limit of development between the groups and the 95% confidence interval of the difference were calculated. The P value of the differences between groups and Akaike information criterion (AIC) were reported for each model. The AIC is a measure that can be used to compare model fit between different estimated models. In Table 2, the AIC is interpreted as “smaller is better.” P values less than .05 were considered to be a statistically significant difference. Plots of standardized residuals and Q-Q plots were inspected to validate model assumptions.
Comparison of Limits and Rates Between Brain Lesion Variables
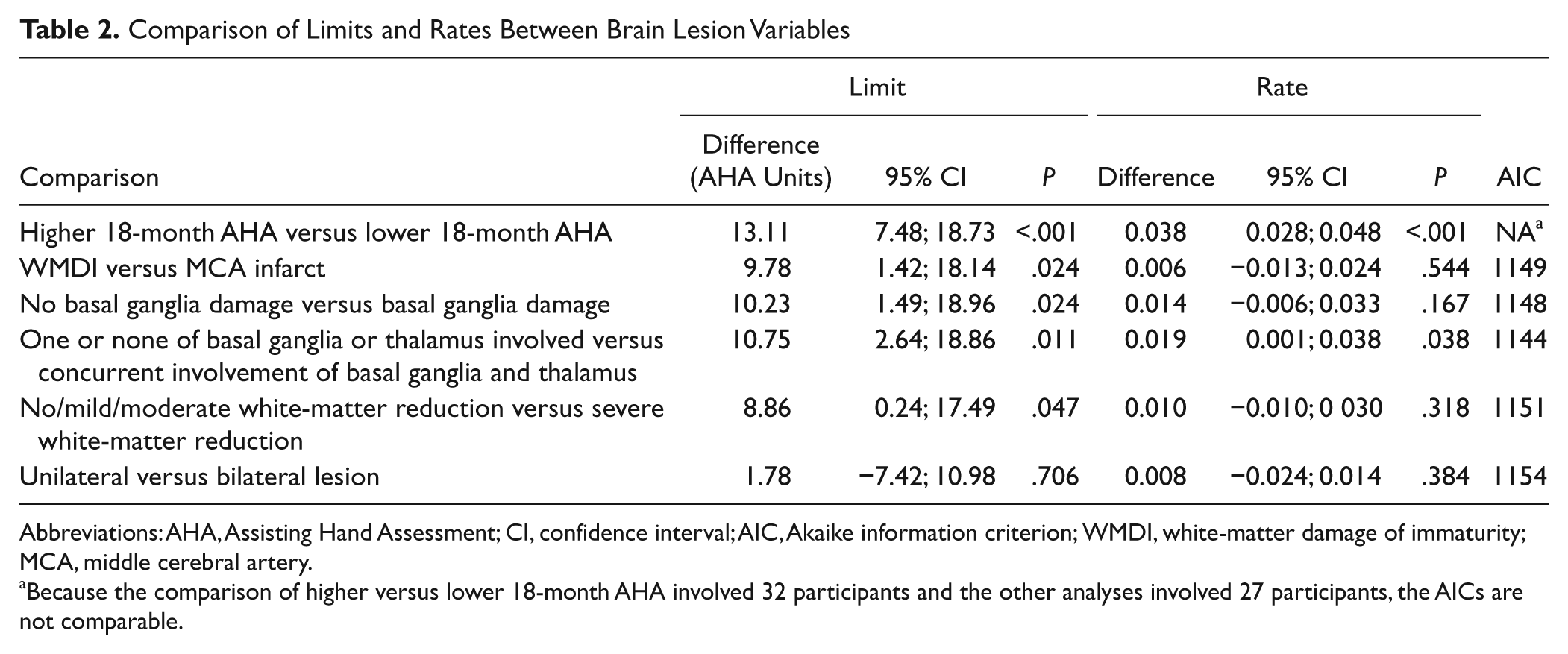
Abbreviations: AHA, Assisting Hand Assessment; CI, confidence interval; AIC, Akaike information criterion; WMDI, white-matter damage of immaturity; MCA, middle cerebral artery.
Because the comparison of higher versus lower 18-month AHA involved 32 participants and the other analyses involved 27 participants, the AICs are not comparable.
Results
Development of Hand Function
The increase of AHA score was similar to that described in a previous study, 1 in spite of the minor alteration of the present cohort (Table 2).
Brain Abnormalities
The analysis of MR and CT scans showed that 12 children had WMDI, 14 children had MCA infarcts, 1 had watershed-type of infarct, 3 children had maldevelopment, 1 had a pontine infarct, and 1 had a tumor (Table 1). Because of the small numbers, the children with maldevelopment, pontine infarct, and tumor were excluded from further analysis. Thus, only children with WMDI (Figures 1C and 1D), MCA infarct (Figures 1A and 1B), or watershed-type of infarct (n = 27) were included in the subsequent analysis (the child with the watershed-type of infarct was analyzed together with those with MCA infarct; Table 1).
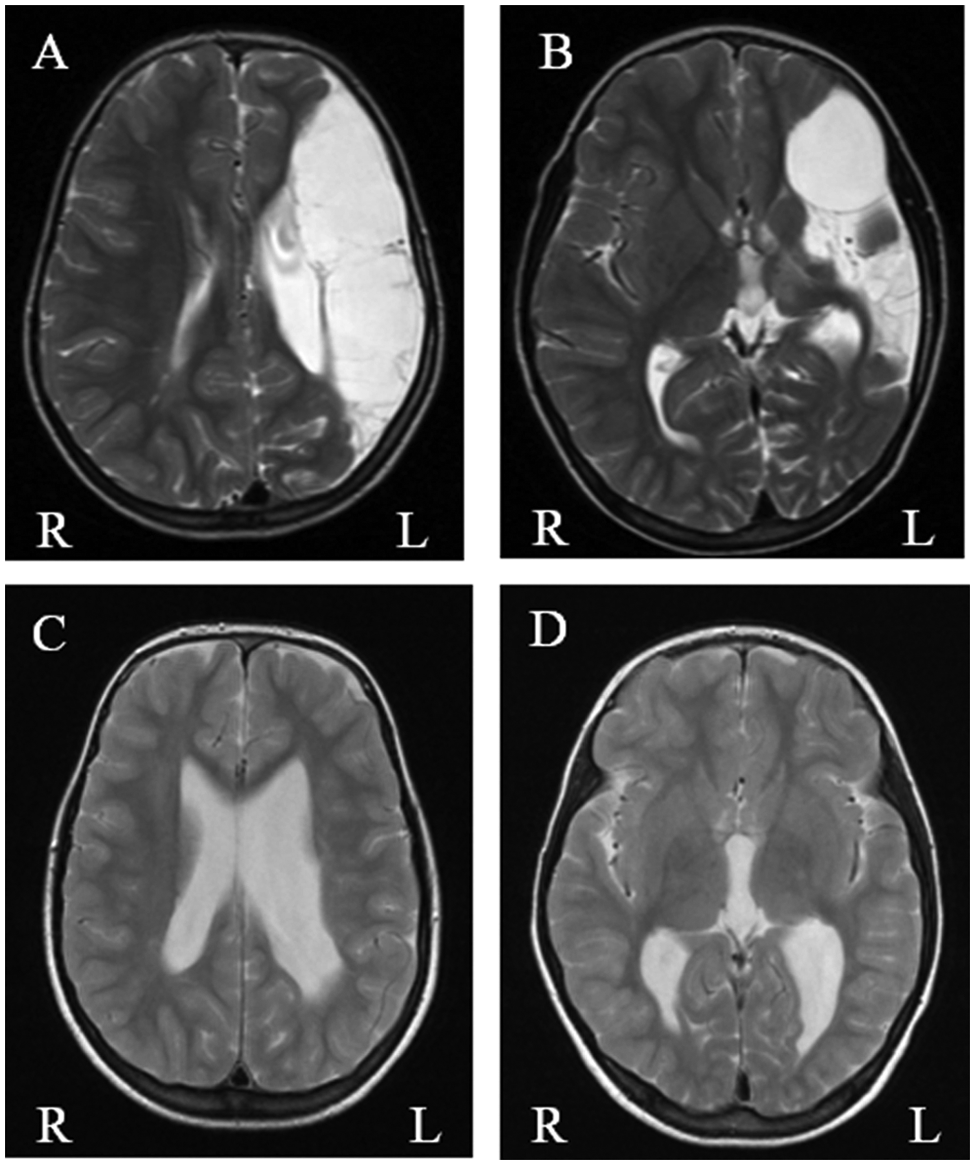
A, B. (ID no. 4 in Table 1) Infarct of middle cerebral artery on the left side with defect in cortical gray matter and subcortical white matter in the frontal, parietal, and temporal lobe. There is also atrophy of the left cerebral hemisphere (A, B). Deep gray matter is affected with volume reduction and signal changes in the basal ganglia and thalamus (B). C, D. (ID no. 16 in Table 1) Bilateral white-matter damage of immaturity with severe reduction of periventricular white matter on the most affected left side (C), associated bilateral ventricular enlargement and volume reduction of left thalamus (D). Basal ganglia are normal.
Association Between Brain Abnormalities and Development of Hand Function
In the first model investigated (ie, basic pattern of damage; see Methods section), the children with WMDI (n = 12) had significantly higher limits of development (difference, 9.8 AHA units) than children with MCA infarct (n = 15). Thus, the basic pattern of damage could predict the limit but not the rate of development of affected hand use (Table 2, Figure 2A).
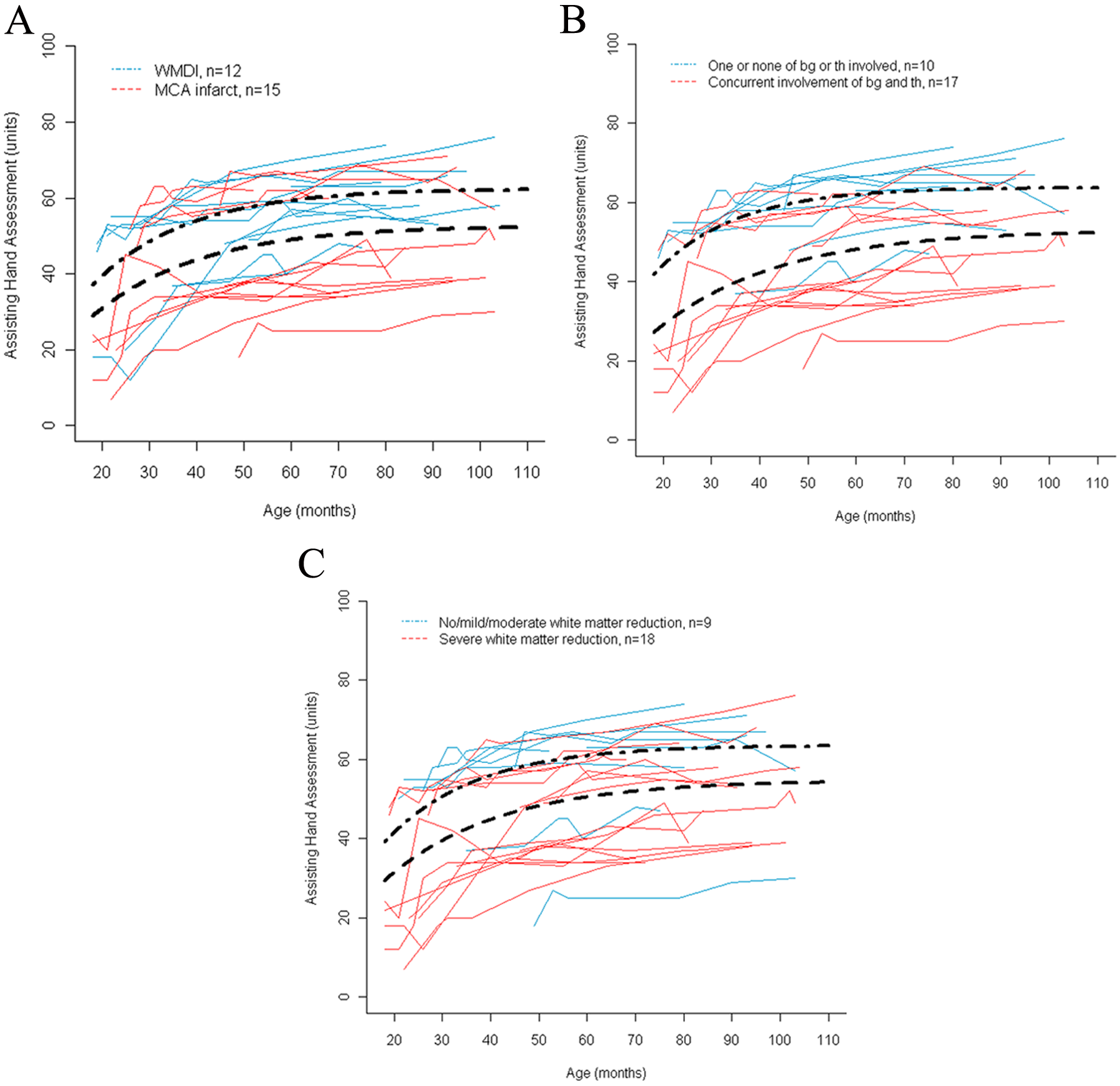
Development of use of the affected hand measured with the Assisting Hand Assessment. A thin line represents the development of 1 child. Bold lines represent mean development. Rate of development is indicated by the steepness in the curve and limit is seen where the curve levels out at higher age. A significant difference between groups in both rate and limit is seen in model 3 (Figure 2B). A. Children were grouped according to their basic pattern of damage, white-matter damage of immaturity (WMDI), or middle cerebral artery (MCA) infarct (model 1). B. Children were grouped according to lesions to basal ganglia and the thalamus; children with one or both of these structures intact are compared with those with concurrent involvement of basal ganglia (bg) and thalamus (th) (model 3). C. Children were grouped according to extent of white-matter reduction: no, mild, or moderate white-matter reduction versus severe white-matter reduction (model 4).
In the second model, children with no basal ganglia damage (n = 8) had significantly higher limits of development of the affected hand (a difference of 10.2 AHA units) than children with basal ganglia damage (n = 19), whereas the rate of development was not significantly different between the groups (Table 2). In the third model, the group of children that had damage to only one or none of the basal ganglia and thalamus (n = 10) had both significantly higher limits (a difference of 10.8 AHA units) and faster rates of development than children with concurrent lesions of both the basal ganglia and thalamus (n = 17; Table 2, Figure 2B). Thus, coexisting damage of both basal ganglia and the thalamus was a strong predictor of the development of hand function in this sample, regardless of whether the basic pattern of damage was WMDI or an MCA infarct. This model had the lowest AIC and thus explained most of the variance between children.
In the fourth model, the children were divided into 4 groups according to the extent of the white–matter lesion (see Table 1). Because of the small group sizes, the groups with no, mild, or moderate reduction in white matter were collapsed into 1 group (n = 9) and compared with the group with severe white–matter reduction (n = 18). The children with no, mild, or moderate white–matter reduction had a significantly higher limit of development (a difference of 8.9 AHA units) than the children with severe white–matter reduction (Table 2, Figure 2C). The fifth model showed that there was no difference in either the limit or the rate of development between children with unilateral (n = 17) and those with bilateral damage (n = 10) of the brain (Table 2).
Discussion
This study suggests that imaging of the brain can be used to make a crude prediction of the future development of the use of the affected hand. There were associations between the type, location, and size of the perinatal brain lesions in children with unilateral CP and their developmental trajectories of hand function. In agreement with a number of cross-sectional studies, children with WMDI lesions reached higher developmental limits of hand function than children with MCA infarcts.10,12,19,20 However, the highest predictive power of better developmental trajectory was the absence of combined involvement of both basal ganglia and thalamus, independently of the basic type of brain lesion. This model was the only one that could predict both the rate and the limit of development of hand function and resulted in the largest difference in the limit of development—that is, development was faster and better for those with no or limited involvement of the basal ganglia and thalamus.
In general, the associations between lesions to different brain structures and hand function are as expected and in line with those reported in the literature. It is, however, interesting that the basal ganglia/thalamus model was the strongest predictor of development. We have previously described the significant impact of basal ganglia involvement on the sensorimotor control of the grasping of objects in children with CP, 13 and recently Feys et al 12 showed a correlation between basal ganglia/thalamus damage and clinical assessments of hand function. One reason for the more deleterious influence of basal ganglia/thalamus involvement might be the critical role of these structures in sensorimotor integration and the fact that this role cannot be compensated for by other brain structures. Other lesions seem to be compensated for by the substantial cortical plasticity that is known to be present after unilateral, perinatal lesions.14,19,21 The finding that children with severe white–matter reduction have a lower limit of development than those with no, mild, or moderate white–matter reduction is, of course, expected and in line with previous studies.11,12,14 The differences found in all these models rely on a small number of children and need to be confirmed in future studies. Several children in this study had bilateral brain damage despite a clear unilateral presentation of symptoms, which had resulted in the diagnosis of unilateral CP. Bilateral brain abnormalities in children with unilateral CP have also been reported elsewhere.12,20,22 However, the lack of impact of such bilateral abnormalities on the use of the hemiplegic hand is, to our knowledge, reported here for the first time.
In this study, we used longitudinal data on hand function covering the age range from 18 months to 8 years in contrast with earlier studies that have included cross-sectional data, mainly from children at older ages.2,3,12,20 By using the nonlinear model, we could create and compare individual developmental trajectories for each child. Despite the fact that the group of children was small, we have shown that the brain abnormalities have an impact on the developmental trajectories. The differences in mean limit of development between groups in models 1 through 4 ranged between approximately 9 and 13 AHA units, which is a considerable difference of clinical importance. This difference can be exemplified by describing the mean development of the 2 groups in model 3. The children with one or none of the basal ganglia or thalamus involved used both their hands in play already at the age of 18 months, using their affected hand to grasp but with some slowness or difficulty. They improved during the preschool years, and at age 8 years, bimanual performance was only mildly affected by their hemiplegia. The children with concurrent lesion to basal ganglia and thalamus on the other hand typically did not use the affected hand spontaneously at 18 months of age: at best, they kept hold of an object that the assessor had placed into their hand. At age 8 years, these children mostly performed bimanual tasks independently but with increased effort, slowness, or difficulty. There was a variation within each group described in the present study, indicating a more complex interaction between the different models and the possibility that other factors explain differences in development, but this was not possible to explore with the current sample size.
Another limitation of this study is the lack of children in the upper range of the AHA scale—that is, children with mild unilateral CP. 16 Most of the children in this study had a moderate to severe type, whereas the general population of children with unilateral CP covers the whole scale range of 0 to 100 AHA units. 16 With a larger sample and a representative proportion of children, it would have been possible to carry out multivariate analysis, enabling a direct analysis and comparison of the impact of each variable. Such an analysis might also have shown that more of the models could predict the rate of development.
Implications
Although the brain imaging was performed at different ages, we think that with modern imaging technologies, there is an increasing possibility to make detailed descriptions of perinatal brain lesions as early as the neonatal period (first year). By focusing on the imaging findings that have been shown to be associated with the rates and limits of the developmental trajectories, it is possible to make a preliminary prediction of hand development at an early age. However, the knowledge from imaging findings must be combined with the child’s functional ability because it is well known that not all brain lesions, such as neonatal stroke or lesions in the prenatal period, lead to CP.5,23 Therefore, the combined information about the brain lesion and functional performance will be of clinical importance in discussions about prognosis with families and when deciding whether to initiate early intervention programs to maximize upper-extremity function.
Footnotes
Acknowledgements
We express our deepest gratitude to the children and families who participated in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Promobilia Foundation, the Sunnerdahl Foundation, the Swedish Research Council (K2009-63X14534-07-3), the Centre for Health Care Sciences at Karolinska Institutet, and the Strategic Research Programme in Care Sciences and Vinnova.
