Abstract
Background. Neuroimmune activation in the spinal dorsal horn plays an important role in the pathogenesis of chronic pain after peripheral nerve injury. Objective. The aim of this study was to examine the role of neuroimmune activation in below-level neuropathic pain after traumatic spinal cord injury (SCI). Methods. Right hemilateral SCI was created in male Sprague–Dawley rats by controlled blunt impact through a T12 laminectomy. Pain-related behaviors were assessed using both evoked reflex responses and an operant conflict-avoidance test. Neuroimmune activation was blocked by the anti-inflammatory cytokine interleukin-10 (IL-10) delivered by a nonreplicating herpes simplex virus (HSV)–based gene transfer vector (vIL10). Markers of neuroimmune activation were assessed using immunohistochemistry and Western blot. Results. One week after SCI, injured animals demonstrated mechanical allodynia, thermal hyperalgesia, and mechanical hyperalgesia in the hind limbs below the level of injury. Animals inoculated with vIL10 had a statistically significant reduction in all of these measures compared to injured rats or injured rats inoculated with control vector. Conflict-avoidance behavior of injured rats inoculated with vIL10 was consistent with significantly reduced pain compared with injured rats injected with control vector. These behavioral results correlated with a significant decrease in spinal tumor necrosis factor α (mTNFα) expression assessed by Western blot and astrocyte activation assessed by glial fibrillary acidic protein immunohistochemistry. Conclusion. Below-level pain after SCI is characterized by neuroimmune activation (increase mTNFα and astrocyte activation). Blunting of the neuroimmune response by HSV-mediated delivery of IL-10 reduced pain-related behaviors, and may represent a potential novel therapeutic agent.
Introduction
Spinal cord injury (SCI) results in paralysis, loss of sensation, and dysregulation of autonomic function.1,2 Paradoxically, despite the loss of transmitted sensation, many patients with SCI suffer from central neuropathic pain, 3 a maladaptive state characterized by spontaneous pain and by evoked allodynia and/or hyperalgesia, in which normally innocuous or mildly painful stimuli in regions that are hypesthetic or numb elicit pain below the level of injury. Below-level pain is a clinically important form of central neuropathic pain that develops in a delayed fashion and is often refractory to medical treatment.4,5
There is substantial evidence for neuroimmune activation in the pathogenesis of post-SCI central neuropathic pain. 6 SCI results in increased transcription and translation of inflammatory mediators, including interleukin-1beta (IL-1β), interleukin-6 (IL-6), prostaglandin E2,7,8 and tumor necrosis factor alpha (TNFα), 9 and activation of glial cells10-13 below the level of injury. Drugs that inhibit or block this inflammatory process reduce peripherally induced neuropathic pain. 14 In a previous study, we found that interfering with spinal TNFα signaling below the level of the lesion can reduce pain-related behaviors in a hemisection model of SCI. 9
Gene transfer to dorsal root ganglion (DRG) neurons using nonreplicating herpes simplex virus (HSV) gene transfer vectors is an effective means to express and release neuroactive peptides, and subcutaneous inoculation can be used to transduce primary afferents in vivo to achieve peptide-mediated effects.15-17 Previous studies have demonstrated that interleukin-10 (IL-10) can reduce pain-related behaviors and improve outcome in several different rodent models of SCI.18-20 In this study, we tested a nonreplicating HSV vector expressing the anti-inflammatory cytokine IL-10 in a model of blunt trauma to the spinal cord and used both evoked (reflex) measures of pain behavior as well as a conflict-avoidance operant test to determine whether HSV-mediated expression of IL-10 reduced below-level pain in these animals.
Methods and Materials
Experimental Animals and Surgical Procedures
All experimental procedures were conducted in accordance with the guidelines outlined in the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals (1996) and were approved by the Institutional Animal Care and Use Committee at the University of Michigan and Ann Arbor Veterans Affairs Healthcare System. Male Sprague–Dawley rats weighing 250 to 300 g were used in these experiments. The animals were housed according to NIH guidelines and maintained for approximately 1 week of adjustment and training prior to surgery, given free access to food and water, and maintained on a 12:12 hour light:dark schedule at 21°C and 60% humidity. Following hemi-laminectomy, SCI was induced by a blunt compressive force 21 applied to the T12–L1 spinal cord using a microprocessor-controlled device (Infinite Horizon, Precision Systems & Instrumentation, Lexington, Kentucky) with a 2.5-mm steel impactor to apply a force of 125 kdyn at a maximum speed of 135 mm/s and dwell time of 0. 22 A blunt force model, rather than a hemisection, was chosen to more closely mimic human SCI. Our model was based on the unilateral contusion model and has been characterized in cervical spinal cord injury. Sham-operated animals underwent identical surgical dissection and exposure but did not undergo injury to the spinal cord. Rats with bilateral paralysis after SCI were excluded from the study. Postoperative care included 10 cm3/d of sterile saline and ampicillin 50 mg/kg/d for 3 days injected subcutaneously. All animals exhibited normal weight gain, cleanliness, bladder function, and level of activity. One week after injury, the 12 rats received a single inoculation subcutaneously in the plantar surface of the right hind limb with 30 µL containing 2 × 108 plaque forming units/mL of the IL-10 expressing vector (vIL10) or a control vector expressing LacZ (vZ). The construction and characterization of this vector (referred to in previous publications as “QHIL10”) have been reported.23,24 Four cohorts of 6 animals were tested: (a) sham-operated, (b) noninjected injured, (c) vIL10-injected injured, and (d) vZ-injected injured.
Basso, Beattie, and Bresnahan Locomotor Scale
All the analyses were performed by an investigator blinded to the treatment group. The Basso, Beattie, and Bresnahan (BBB) locomotor scale 25 was used to exclude animals with confounding weakness. Rats were placed, one at a time, in a flat open field within a molded plastic box, allowed to acclimate to the field, and examined for 4 to 5 minutes. Animals with a BBB score of less than 12 (unable to produce frequent to consistent supported plantar steps with occasional forelimb–hindlimb coordination) were excluded from the study.
Behavioral testing
Evoked pain responses ipsilateral to the lesion were examined once per week for 5 weeks following SCI or sham surgery. Behavioral testing at week 1 was performed before the inoculation of the vectors to avoid potential confounding pain from the injection. An operant pain test was performed once at the conclusion of the study.
Mechanical allodynia
Rats were randomly placed in nontransparent plastic cubicles on a mesh floor for an acclimatization period of at least 30 minutes. Mechanical allodynia was determined by assessing paw withdrawal to von Frey hairs of graded tensile strength. A series of calibrated von Frey filaments (0.4, 0.7, 1.2, 1.5, 2.0, 3.6, 5.5, 8.5, 11.8, and 15.1 g) were presented serially to the hind paw in ascending order of strength starting at 0.4 g, with each filament applied for 6 seconds with sufficient force to cause slight bending against the paw. The filament was applied to the volar surface between the pads and was repeated at the same spot each time with at least a 2-minute interstimulus interval. A positive response was defined as rapid withdrawal and/or licking of the paw immediately on application of the stimulus, which was then followed by application of the next lower von Frey filament. After a negative response, the next higher von Frey filament was applied. Animals that did not respond to a pressure of 15.1 g were assigned to this cutoff value. The tactile stimulus producing a 50% likelihood of withdrawal was determined using the up–down method.26,27
Thermal hyperalgesia
The latency to hind paw withdrawal from a thermal stimulus was determined by exposing the plantar surface of the hind paw to radiant heat using a modified Hargreaves thermal testing device. 28 Briefly, rats were placed in individual enclosures on a glass plate maintained at 30°C, and a radiant thermal stimulus was positioned underneath the glass plate directly under the hind paw. Activation of the bulb simultaneously activated a timer, and both were immediately turned off by paw withdrawal or terminated at 20 seconds to avoid tissue damage. Urine and feces were immediately cleaned and removed when present.
Mechanical hyperalgesia
Nociceptive threshold to mechanical stimulation was measured using the Randall–Sellito method. 29 With a clean towel, animals were lightly restrained by hand with their heads covered and the right hind paw placed in an analgesiameter (Ugo Basile, Comeiro-Varese, Italy) that applied a linearly increasing pressure (76.8 g/s) by means of a blunt Plexiglas cone to the palmer region. The same point in which the pressure was applied was used each time and the interstimulus interval was at least 5 minutes. The pressure (g) at which the rat withdrew its paw was recorded as the threshold for withdrawal from a mechanical stimulus.
Operant pain test
Operant pain testing was conducted using a Mechanical Conflict-Avoidance System (Coy Labs, Grass Lake, Michigan). The apparatus consisted of a rectangular track connecting a brightly lit box on one end and an unlit dark box at the other. Rats placed in the light box will spontaneously cross the track to access the dark box. For 3 days of training, each rat was given 60 seconds to exit the light box and was given another 60 seconds to cross the track to enter the dark box. On the fourth day, after a final training run, the floor of the track was replaced with sharp metal spikes (0.4-mm tip diameter, spaced 10 mm apart). The amount of time spent crossing the track was recorded. Rats that did not exit the light box within the 60-second period were assigned a time of 0 seconds. Rats were first tested with a spike height of 2 mm, followed by testing with a spike height of 3 mm. This test presents a conflict in that rats must either chose to (a) remain in an aversive environment (brightly lit box) and avoid noxious mechanical stimulation (spikes) or (b) escape the adverse environment by subjecting themselves noxious stimulation by crossing a track of spikes to gain access to a preferred dark environment. Only vZ-injected and vIL10-injected animals underwent operant testing.
Western Blot
Four rats of each group that underwent behavioral testing were sacrificed for Western blot analysis. Under deep anesthesia with chloral hydrate, the lumbar enlargement of the spinal cord (L4, L5, and L6 in rats) and DRG ipsilateral to the SCI were removed, immediately frozen on dry ice and stored at −80°C. The tissue of the dorsal horn ipsilateral to SCI was isolated and homogenized in extraction buffer (50 mM Tris, pH 8.0 containing 0.5% Triton X-100, 150 mM NaCl, 1 mM EDTA, and protease inhibitor cocktail from Sigma), protein concentration was determined with a BCA Protein Assay Kit (Pierce Biotechnology, Rockford, Illinois), and 50 µg of protein per sample separated on sodium dodecyl sulfate–polyacrylamide gel electrophoresis gel (4% to 12% gradient gel; Bio-Rad, Hercules, California) and transferred to polyvinylidene difluoride membranes (Millipore Corp, Bedford, Massachusetts) that were blocked with 5% milk in phosphate-buffered saline with 0.1% Tween 20 for 1 hour at room temperature, and incubated overnight at 4°C with anti-TNF antibody (rabbit, 1:200; Millipore Corp). The blots were then incubated for 2 hours at room temperature with horseradish peroxidase–conjugated secondary antibody (1:2000; Santa Cruz Biotechnology, Santa Cruz, California), developed in chemiluminescence solution (Pierce Biotechnology, Rockford, Illinois) for 5 minutes, and immunoreactive bands were determined directly using a BioRad chemiluminesence imager and BioRad software. mTNFα levels were normalized using the amount of β-actin (anti-mouse, 1:2000; Sigma-Aldrich, St Louis, Missouri) as a standard.
Immunohistochemistry
The animals that were not sacrificed for Western analysis were deeply anesthetized with chloral hydrate at 300 mg/kg, perfused with 0.9% saline followed by Zamboni solution (2% paraformaldehyde, 0.15% picric acid, 0.1 mol/L phosphate buffer, pH 7.4). 30 The lumbar enlargement of spinal cord (L4, L5, and L6) was removed, cut transversely, and post fixed for 24 hours in Zamboni solution. After cryoprotection with 30% sucrose in phosphate buffer at 4°C, 10-µm sections mounted on glass slides were incubated overnight at 4°C with an anti-GFAP (anti–glial fibrillary acidic protein) antibody (mouse, 1:4000; Sigma-Aldrich) followed by fluorescent complementary secondary anti-mouse Alexa 488 antibody (rabbit, 1:1000; Santa Cruz Biotechnology). Images of the lumbar (L4, L5, and L6) dorsal horn ipsilateral to side of SCI were photographed using an Olympus BX-DSU confocal microscope, and the intensity of staining determined by visual inspection.
Data Analysis
The statistical significance of differences between groups was evaluated by analysis of variance and single comparisons were performed with Student t test with the use of SAS 9.2 (SAS Institute, Cary, North Carolina). For repeated measures of behavioral function, the general linear model for repeated measures was used to identify significant effects of treatment condition on the behavioral measure of neuropathic pain (SPSS18, IBM SPSS Inc, Chicago, Illinois). P values less than .05 were counted as significant. All data are presented as mean ± standard error of the mean (mean ± SEM).
Results
A Nonreplicating HSV-Based Vector Expressing IL10 Injected Into the Skin Infects DRG Neurons to Release IL10 In Vivo
Two vector constructs were employed in this study. vIL10 contains the full-length rat IL10 gene tagged with hemagglutinin (HA) under the control of the human cytomegalovirus immediate-early promoter (HCMC IEp). Control vector vZ was identical to vIL10 but contained the lacZ gene in place of IL10-HA. The construction of vIL10 has been described previously (designated in that report as QHIL10. 23 In previous studies, we demonstrated that vIL10 produces IL10 from primary DRG neurons infected in vitro and in the spinal cord in vivo. 24
Injection of the HSV Vector Expressing IL-10 After SCI Reduces Evoked Pain-Related Behaviors
By 1 week after contusion injury, the animals showed considerable improvement in motor function. The mean actual force applied to induce SCI was 138.3 kdyn with a mean speed of 133.7 mm/s. One animal was removed from further study because of bilateral hindlimb paralysis and was not replaced with another animal leaving the LacZ group with 5 animals total. The remaining animals all exhibited BBB scores greater than 12 (data not shown) consistent with weight support sufficient to allow full behavioral testing of somatosensory function. At 1 week after injury, animals with SCI also demonstrated significant increases in pain-related behaviors, including: mechanical hyperalgesia (withdrawal threshold 90.0 ± 5.86 g for sham vs 52.3 ± 7.51 g for injured; P < .01), thermal hyperalgesia (withdrawal latency 13.0 ± 1.42 seconds for sham vs 7.6 ± 0.28 seconds for injured; P < .01), and mechanical allodynia (threshold 11.7 ± 2.78 g in sham vs 1.9 ± 0.28 g in injured; P < .01; Figure 1). Subcutaneous inoculation of vIL10 1 week after SCI reduced mechanical hyperalgesia (withdrawal threshold from 61.1 ± 3.75 g to 92.2 ± 5.44 g); thermal hyperalgesia (withdrawal latency from 7.58 ± 0.58 seconds to 12.5 ± 2.39 seconds); and mechanical allodynia (threshold from 3.86 ± 0.71 g to 11.0 ± 3.85 g) by 2 weeks after injury (Figure 2). All 3 pain behavior measures in SCI rats inoculated with vIL10 paralleled those of sham rats from week 2 to 5. Rats injected with control vector vZ did not show any significant differences from the noninjected injured group; vZ-injected rats continued to demonstrate SCI-induced neuropathic pain behaviors. The differences between vIL10-injected animals and injured noninjected or injured vZ-injected animals was statistically significant (P < .01, repeated-measures analysis of variance) for all 3 pain behavioral measures throughout the 4-week period beginning 1 week after injection. These results suggest that expression of IL10 from the HSV vector reduced neuropathic pain as measured by evoked responses.
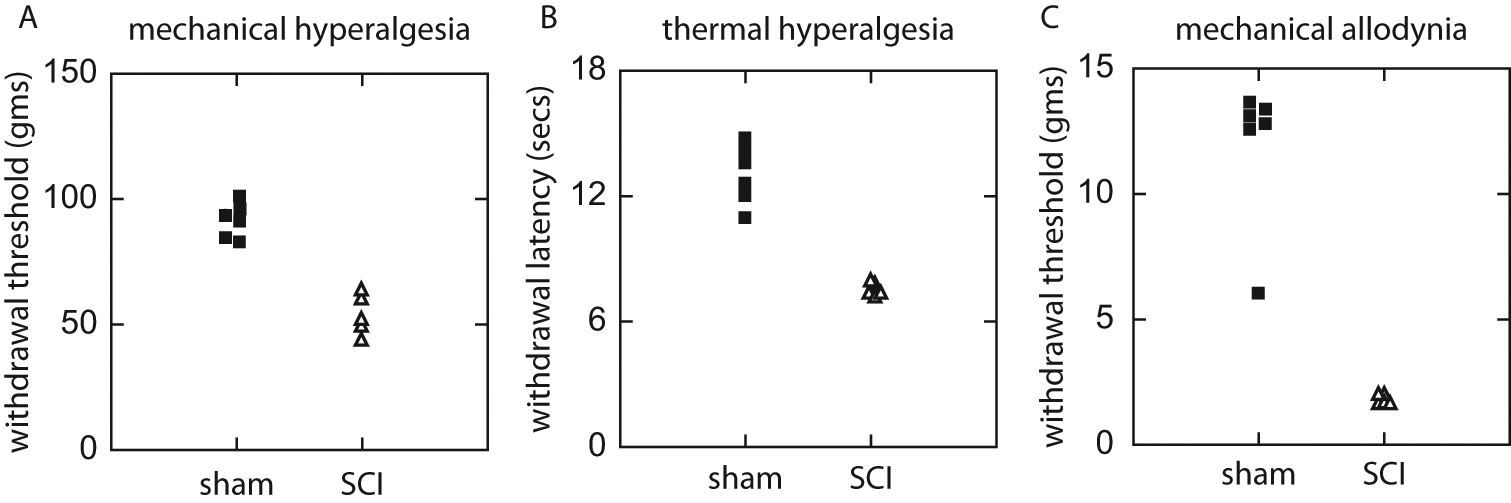
Pain-related behaviors 1 week after spinal cord injury (SCI). Blunt trauma to the lumbar spinal cord resulted in pain-related behaviors in the ipsilateral hindlimb below the level of the lesion. Filled squares represent sham (uninjured animals) and open triangles are injured animals without treatment. Animals with SCI demonstrated (A) mechanical hyperalgesia measured by the Randall–Selitto method, (B) thermal hyperalgesia measured by a modified Hargreaves test, and (C) mechanical allodynia using von Frey filaments.
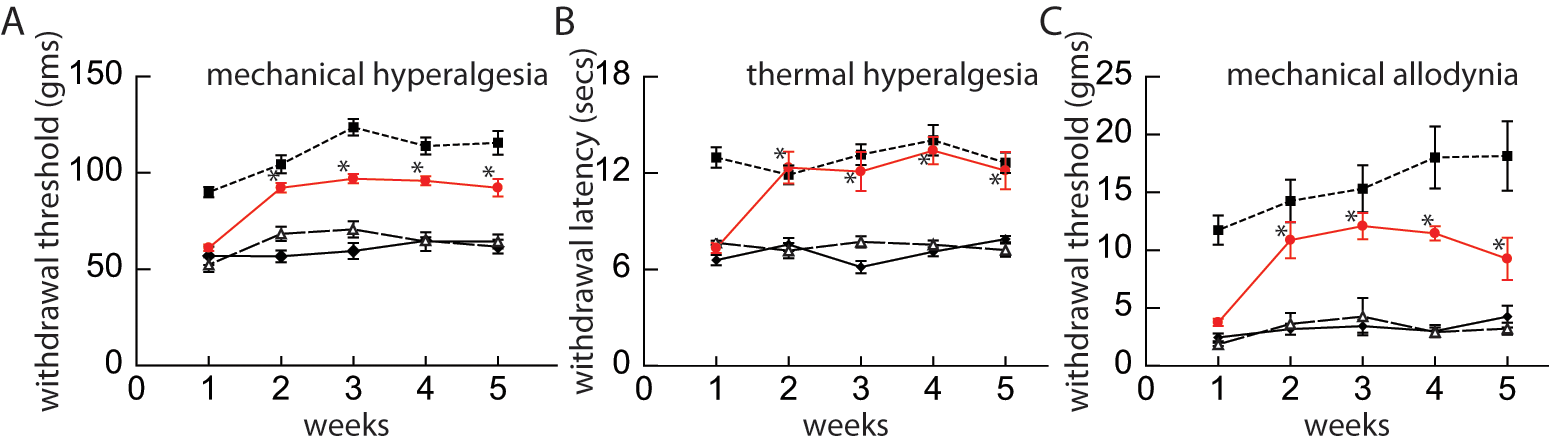
Pain-related behavior after injection of vIL10 vector. Injection of the herpes simplex virus (HSV) vector expressing IL10 after spinal cord injury (SCI) reduces evoked pain-related behaviors. Injured rats injected with vIL10 (red circles) 1 week after injury showed a significant reduction in (A) mechanical hyperalgesia (Randall Selitto), (B) thermal hyperalgesia (Hargreaves), and (C) mechanical allodynia (von Frey), compared with injured rats with no treatment (open triangle) and injured rats injected with vZ (filled diamonds) throughout the 5-week period. Filled squares represent the behavior of sham-operated animals. The difference between vIL10-injected animals and injured noninjected or injured vZ-injected animals was statistically significant from the 2-week through the 5-week time points (*P < .01).
Injection of the HSV Vector Expressing IL10 After SCI Reduces Pain Measured by a Conflict-Avoidance Operant Test
Pain is a complex experience with sensory-discriminative, affective-motivational, and cognitive components that may not be fully assessed by spinally mediated reflex tests.31,32 To complement the results of the evoked behaviors, we employed an operant mechanical conflict-avoidance test33-35 to compare the vIL10- and vZ-inoculated animals. Following training, with a smooth surface (no spikes), all rats in both the vIL10- and the vZ-inoculated cohorts readily crossed the track from the light box into the dark box without hesitation as shown in Figure 3. When escape to the dark box was impeded by 2-mm spikes, vIL10-injected rats spent significantly more time on the track (26.8 ± 3.93 seconds) as compared with vZ-injected rats (18.6 ± 7.45 seconds; P = .04). This difference became more pronounced when spike height was increased to 3 mm (vIL10-injected rats 16.9 ± 5.70 seconds vs vZ-injected rats 6.7 ± 7.54 seconds; P = .02). This difference in part reflects the fact that at a spike height of 3 mm, 50.0% of vZ-injected rats chose not to cross (stayed in the light box) whereas all (100.0%) the vIL10-injected rats crossed the track with the spikes to reach the dark box. These results are consistent with those obtained using evoked reflex tests and provide additional confirmation that vector inoculation with vIL10 reduces SCI-induced neuropathic pain.
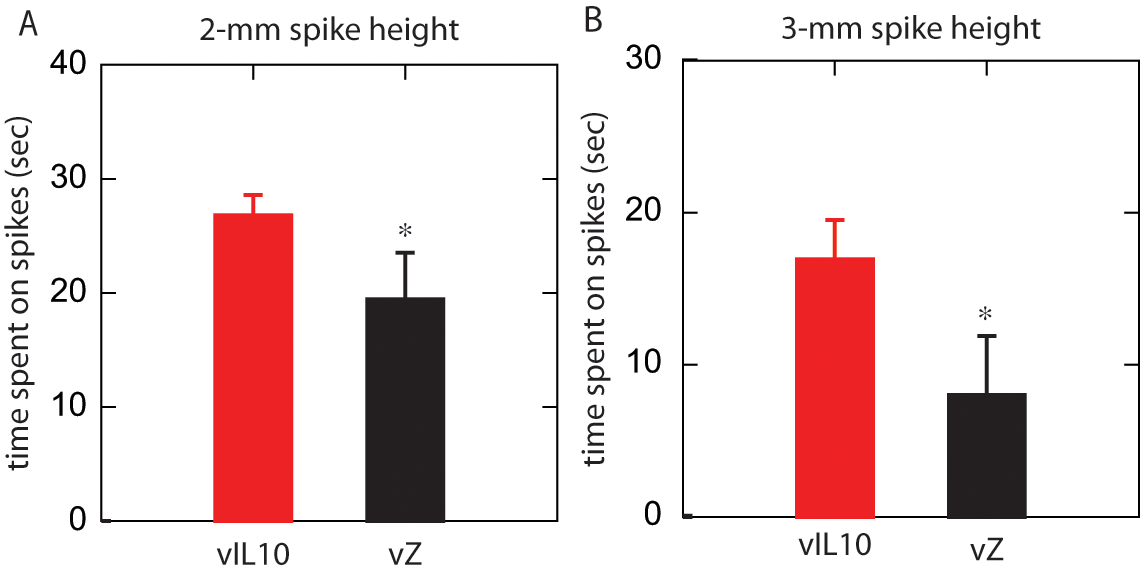
Injection of the herpes simplex virus (HSV) vector expressing IL10 after spinal cord injury (SCI) reduces pain measured by an operant mechanical conflict-avoidance test. Whether the rat decided to cross and the length of time it took for the rats to cross a track of metal spikes were recorded. (A) At a spike height of 2 mm, injured rats injected with the control vector, vZ, spent significantly less time on the spikes compared with injured rats injected with vIL10 (P = .04). (B) At a spike height of 3 mm, this difference was more profound (P = .02), and 50.0% of vZ-injected rats chose not to cross whereas all (100.0%) the vIL10 injected rats crossed.
Injection of the HSV Vector Expressing IL10 After SCI Reduces Immune Activation in the Spinal Cord
Neuroimmune activation in the spinal cord below the level of injury was assessed by Western blot for TNFα. At 5 weeks after SCI, noninjected injured and vZ-injected injured animals demonstrated an approximately sevenfold increase in mTNFα in the dorsal horn at the level of the lumbar enlargement (below the level of the injury) compared with sham-operated animals (Figure 4). Injured animals inoculated with the vIL10 had mTNFα levels that were similar to the sham-operated animals and were significantly lower than noninjected injured animals and vZ-injected injured animals (P < .01).
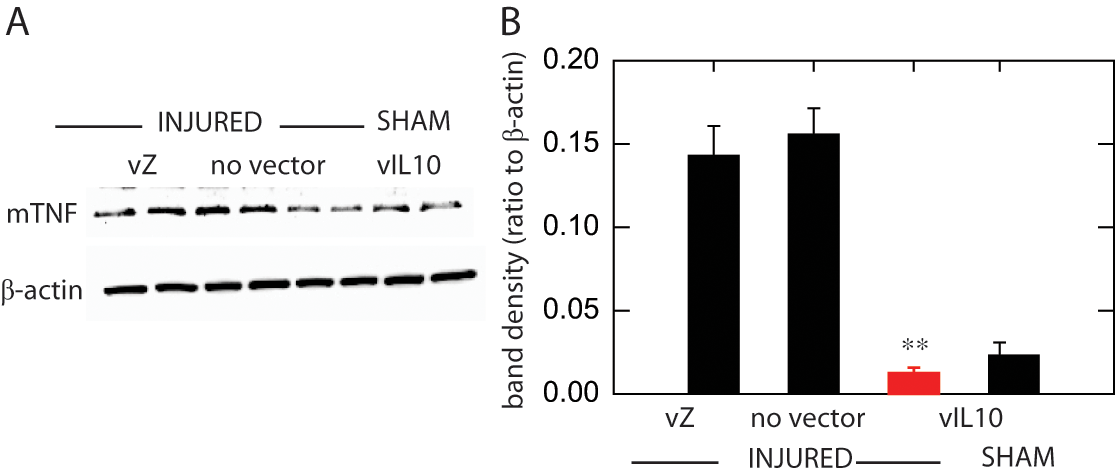
Western blot of tumor necrosis factor alpha (TNFα). The dorsal horn in lumbar enlargement of the spinal cord was collected and processed for Western blot analysis with antibodies against TNFα. Injection of the herpes simplex virus (HSV) vector expressing IL10 after spinal cord injury (SCI) reduces immune activation in the spinal cord. (A) Western blot. (B) Quantitation of Western blot, calculated as a ratio to β-actin control (**P < .01).
Suppression of inflammation is further supported by the decrease in immunohistochemical marker of glial activation GFAP in injured rats inoculated with vIL10. At 5 weeks after injury, noninjected injured animals and vZ-injected injured animals showed substantial increases in GFAP immunoreactivity 36 that was much greater than that seen in sham-operated animals or in vIL10-injected injured animals (Figure 5).
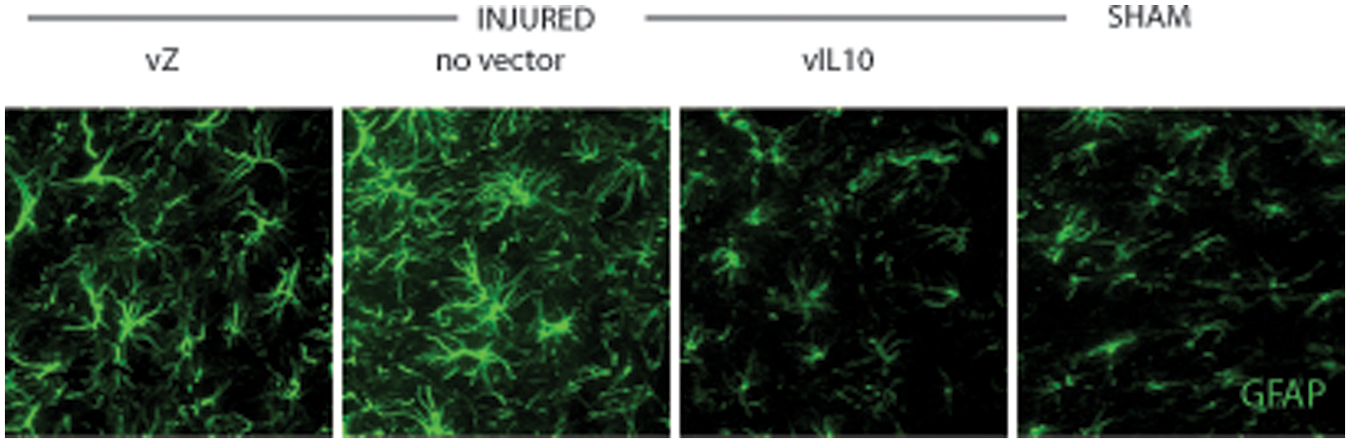
Glial fibrillary acidic protein (GFAP) immunocytochemistry. There was increased GFAP immunoreactivity in the dorsal horn of lumbar spinal cord in injured rats that was reduced by inoculation of vIL10 but not by inoculation of control vector vZ.
Discussion
Inoculation of the HSV vector expressing the IL-10 gene significantly reduced mechanical allodynia, thermal hyperalgesia, and mechanical hyperalgesia below the level of injury after blunt trauma to the spinal cord. These findings are supported by a reduction in supraspinally mediated pain behaviors as measured by an operant conflict-avoidance paradigm. The reduction in pain-related behaviors correlated with a significant decrease in levels of mTNFα and a reduction in astrocyte activation in the dorsal horn of the spinal cord. Taken together, these findings are consistent with previous studies demonstrating that below-level neuropathic pain associated with SCI is characterized by neuroimmune activation below the level of the lesion and show that reducing neuroimmune activation below the level of the lesion reduces pain.
The expression of IL10 by vIL10 vector–transduced cells has been reported previously. 23 Infection of primary DRG neurons in vitro with this vector at a multiplicity of infection of 1 for 48 hours results in the release of 1500 pg/mL of IL10. Transduction of DRG neurons in vivo achieved by subcutaneous inoculation of the vector in the foot resulted in production of transgene-coded IL-10 in DRG neurons and transport of the gene product to terminals in the spinal cord. The amount of IL-10 released in vivo after vector transduction was not measured, but the behavioral and biochemical effects observed previously 23 and in the current study indicate that a biologically significant amount of IL-10 was released from transduced cells in vivo. We did not examine the time course of expression in the current study, but in previous experiments with other HSV vectors in which transgene expression was driven by the HCMV IEp, we have consistently observed initial high levels of expression that tapers off over the course of several weeks in vivo.37,38
Several lines of evidence suggest that neuroimmune activation of microglia and astrocytes in the dorsal horn plays an important role in the pathogenesis of neuropathic pain.39-42 Glial activation occurs after peripheral nerve damage,43,44 and administration of drugs that block the effects cytokines45,46 or that block glial activation47,48 can be used to prevent or reverse neuropathic pain in rodent models. In addition, pain-related behaviors similar to those seen in neuropathic pain can be reproduced by activation of spinal cord glia 49 or by direct intrathecal administration of proinflammatory cytokines 50 in the absence of nerve injury.
Similar observations have been made in models of SCI pain. 12 After thoracic cord contusion, extensive glial activation is observed at the level of the lesion as well as extending significant distances above and below the lesion, in areas of degenerating injured ascending and descending fibers. 51 Following excitotoxic 52 or contusive 53 SCI, phosphorylation of ERK as a marker of glial activation is found at both acute and chronic time points at the level of the lesion. Furthermore there is evidence of microglial activation below the level of the lesion13,54 and increased calcitonin gene related peptide immunoreactivity.55,56 Systemic administration of agents that block microglial activation reduce pain-related evoked behaviors in SCI models, 11 supporting a role for microglial activation in this process.
TNFα is one marker of immune activation, and plays a key role in the cascade of immune responses through induction of the expression and release of other proinflammatory cytokines. We have shown that administration of a nonreplicating HSV vector to release a soluble form of the truncated p55 TNFα receptor (p55 sTNFR) reduces evoked pain-related behaviors following SCI. 9
The results of the current study differ from a recent publication by Baastrup et al 57 who found that following a blunt traumatic injury to the thoracic spinal cord, reflex-evoked responses below the level of injury were increased, but using a place escape/avoidance paradigm they found no evidence for cerebrally mediated aversive reaction to applied stimuli. There are 2 important differences between our study and that of Baastrup et al. First, the lesion they created was more severe, with the impact delivered centrally to the cord, resulting in severe disruption of the substance of the cord involving almost 90% of the cross-sectional area, 57 whereas in our model of injury, we used less force and delivered the impact to one side of the cord, rather than on the midline. Second, the operant test employed by Baastrup et al used touch with von Frey filaments as the aversive stimulus, a model of mechanical allodynia. The device we employed used noxious spikes to measure mechanical hyperalgesia. Contextually, our rats inoculated with vIL10 made a decision to cross over and spend more time on the sharp spikes compared to vZ-injected animals, indicating lower levels of neuropathic pain. In contrast, half of the vZ-injected animals decided not to cross the spikes suggesting greater pain sensitivity. Decision making in such circumstances reflects supraspinal cognitive processing of nociceptive input, which in this study parallels the findings obtained using reflex measures.
IL-10 is an anti-inflammatory cytokine with a broad range of actions that may blunt the neuro-immune response not only through its effects on TNFα but also by decreasing levels of IL-1β. 58 In addition to its anti-inflammatory effects, IL-10 provides direct trophic support and protection of neurons after neuronal injury 59 and promotes neuronal survival after SCI. 59 The results of this study have implications for potential human therapy with the HSV vector–mediated expression of the IL-10 gene. We recently completed a successful phase 1 clinical trial of a similar HSV vector expressing enkephalin for the treatment of chronic cancer pain 60 and a phase 2 trial of this vector has been initiated. Blunting of the neuroimmune response by HSV-mediated delivery of IL-10 reduced pain-related behaviors and may represent another potential novel therapeutic agent for chronic pain.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Two authors (SEH, TJM) have a proprietary interest in the Mechanical Conflict-Avoidance System used in this research, including but not limited to, a patent, trademark, copyright, or licensing agreement, or the right to receive royalties from product commercialization.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Department of Veterans Affairs and the National Institutes of Health (DJF and MM) and a student fellowship from Alpha Omega Alpha (DL).
