Abstract
Background
Somatosensory deficits are prevalent after stroke, but effective interventions are limited. Brain stimulation of the contralesional primary somatosensory cortex (S1) is a promising adjunct to peripherally administered rehabilitation therapies.
Objective
To assess short-term effects of repetitive transcranial magnetic stimulation (rTMS) targeting contralesional (S1) of the upper extremity.
Methods
Using a single-session randomized crossover design, stroke survivors with upper extremity somatosensory loss participated in 3 rTMS treatments targeting contralesional S1: Sham, 5 Hz, and 1 Hz. rTMS was delivered concurrently with peripheral of sensory electrical stimulation and vibration of the affected hand. Outcomes included 2-point discrimination (2PD), proprioception, vibration perception threshold, monofilament threshold (size), and somatosensory evoked potential (SEP). Measures were collected before, immediately after treatment, and 1 hour after treatment. Mixed models were fit to analyze the effects of the 3 interventions.
Results
Subjects were 59.8 ± 8.1 years old and 45 ± 39 months poststroke. There was improvement in 2PD after 5-Hz rTMS for the stroke-affected (F(2, 76.163) = 3.5, P = .035) and unaffected arm (F(2, 192.786) = 10.6, P < .0001). Peak-to-peak SEP amplitudes were greater after 5-Hz rTMS for N33-P45 (F(2, 133.027) = 3.518, P = .032) and N45-P60 (F(2, 67.353) = 3.212, P = .047). Latencies shortened after 5-Hz rTMS for N20 (F(2, 69.64) = 3.37, P = .04), N60 (F(2, 47.343) = 4.375, P = .018), and P100 (F(2, 37.608) = 3.537, P = .039) peaks. There were no differences between changes immediately after the intervention and an hour later.
Conclusions
Short-term application of facilitatory high-frequency rTMS (5Hz) to contralesional S1 combined with peripheral somatosensory stimulation may promote somatosensory function. This intervention may serve as a useful adjunct in somatosensory rehabilitation after stroke.
Keywords
Introduction
Loss of somatosensation is present in upwards of 90% of stroke survivors.1-3 Impairment of proprioception, perception of touch, or pain limits an individual’s ability to avoid injury and impacts how they experience the world around them. Furthermore, somatosensory deficits significantly impair motor control, impede functional performance2,4-7 and negatively impact quality of life. 8 Though somatosensation has an important role in the functioning of the upper limb 7 and is routinely assessed clinically, 7 interventions that address somatosensory deficits are lacking.
Peripherally directed therapies9,10 (such as electrical stimulation, vibration, and others) can produce some improvement in upper limb somatosensory function, but restoration is typically not realized. One limitation of these methods is that they target the periphery (eg, upper limbs)11,12 rather than the brain, which is the source of the deficit and the key area for targeting therapies to promote upper limb functional recovery and reorganization. 13
Recovery after stroke is driven by functional and structural brain reorganization.14-16 Several possible neuroplastic patterns have been observed during rehabilitation, for example, functional re-mapping of surviving pathways or shift of function to homologous and functionally related regions.13,17 It may be possible to promote changes in motor performance by stimulating the brain with electrical currents.18,19 Noninvasive methods using repetitive TMS (rTMS) have been shown to induce brain changes and enhance functional recovery by directly modulating brain activity. 20 Most noninvasive brain stimulation studies have focused on movement-related rehabilitation interventions targeting the primary motor area (M1). After stroke, there is functional interhemispheric imbalance between the right and left motor regions. A diminished activity of the ipsilesional M1 by stroke is further suppressed by overactivity of the contralesional M1. 21 rTMS has been used to correct this imbalance. A common approach involves facilitating activity of ipsilesional M1 and/or inhibiting activity of contralesional M1 (believed to have strong interhemispheric inhibitory effects). 20 Although facilitatory (5 Hz) rTMS of the ipsilesional S1 paired with motor therapy showed improvement of both motor abilities and somatosensory discrimination abilities, 22 the role of rTMS in poststroke somatosensory recovery has not been fully tested. Promisingly, it has been shown that rTMS can affect sensory perception in healthy subjects. 23
Targeting primary somatosensory cortex (S1) to restore somatosensory function may be a logical corollary of the approaches adopted in the area of motor recovery. Contralesional S1 may be a more reasonable target than its ipsilesional counterpart for many reasons. First, modulating structurally intact brain may produce a more reliable and consistent effect across patients given the lack of confounding influence of differing lesion sizes, locations, and geometry. 24 Second, although the majority of sensory processing is in the contralateral hemisphere, it is believed that there is a bihemispheric involvement in sensory processing as demonstrated by functional magnetic resonance imaging (fMRI)25,26 and electroencephalography (EEG) studies 27 and because unilateral infarct may result in bilateral somatosensory deficits.1,28
The objective of this proof-of-principle study was to explore the effect of rTMS targeting contralesional S1. Using a single-session randomized crossover design, participants received high-frequency rTMS (5 Hz, H-rTMS), low-frequency rTMS (1 Hz, L-rTMS), and sham rTMS (S-rTMS) targeting contralesional S1. High-frequency rTMS increases cortical excitability and thus is facilitatory, while low-frequency rTMS decreases cortical excitability and is inhibitory. 29 Both high and low rTMS frequencies were tested because it is unclear whether facilitating or inhibiting contralesional S1 is beneficial for somatosensory function, 30 and because effects of inhibiting contralesional M1 have been equivocal in studies of poststroke motor recovery.20,31 Somatosensory processing of the various somatosensory modalities take different paths in the brain 32 and the response of different somatosensory modalities to rehabilitation intervention can vary.2,33 Therefore, we included a range of somatosensory outcomes probing both primary somatosensation and discriminatory abilities as well as testing both posterior column and the spinothalamic pathways. Immediate and delayed effects of rTMS on clinical somatosensory measures and neurophysiologic indices characterizing somatosensory cortical processing, that is, somatosensory evoked potentials (SEPs) were studied.
Methods
Participants
Subjects were recruited by word of mouth and through use of flyers from the local population. Sixteen patients with upper extremity somatosensory deficits following first-ever stroke (>6 months post) were enrolled. Subjects were screened for study entry and deemed eligible to enroll if they had a detectable difference between the affected and unaffected upper limbs in ≥1 study measure of somatosensation. The inclusion criteria were intentionally broad because of the nature of the study and therefore some individuals with the most severe deficits did not perceive all the sensory modalities. Exclusion criteria were contraindications for TMS such as metal implants, history of seizures, or use of substances that lower seizure threshold. 34 A description of limbs in the text is as follows: affected side—contralateral to the stroke lesions; and unaffected side—ipsilateral to the stroke lesions.
Overview of Study Design
In a randomized, crossover experiment, participants underwent single sessions of 3 different types of rTMS at intervals ≥1 week: (1) facilitatory H-rTMS, (2) inhibitory L-rTMS, and (3) sham. H-rTMS involved application of 5-Hz rTMS while L-rTMS involved 1-Hz rTMS to contralesional S1. Sham rTMS was delivered using a placebo coil. In every session, rTMS was delivered concurrently while participants received peripheral sensory stimulation of the affected hand. Measurements were performed before (Pre), immediately after each session (Post 1), and 1 hour after each session (Post 2). Figure 1 depicts an overview of the study design. The rationale for the timing of data collection was based on prior reports of rTMS effects lasting up to 1 hour. 35 Participants underwent a baseline session to familiarize them with the testing protocol. A randomized-block design using the 6 order permutations within the block was used to randomize the order of the different rTMS sessions. The rTMS protocols were delivered ≥1 week apart to allow for a washout period. 36 The study enrolled patients from 2 hospitals in Cleveland, Ohio: the Louis Stokes Cleveland Veterans Affairs Medical Center (LSCVAMC) and the Cleveland Clinic (CC). The protocol at both sites was identical unless otherwise noted. Local institutional review boards of each hospital approved the study. Participants provided written informed consent before participating.
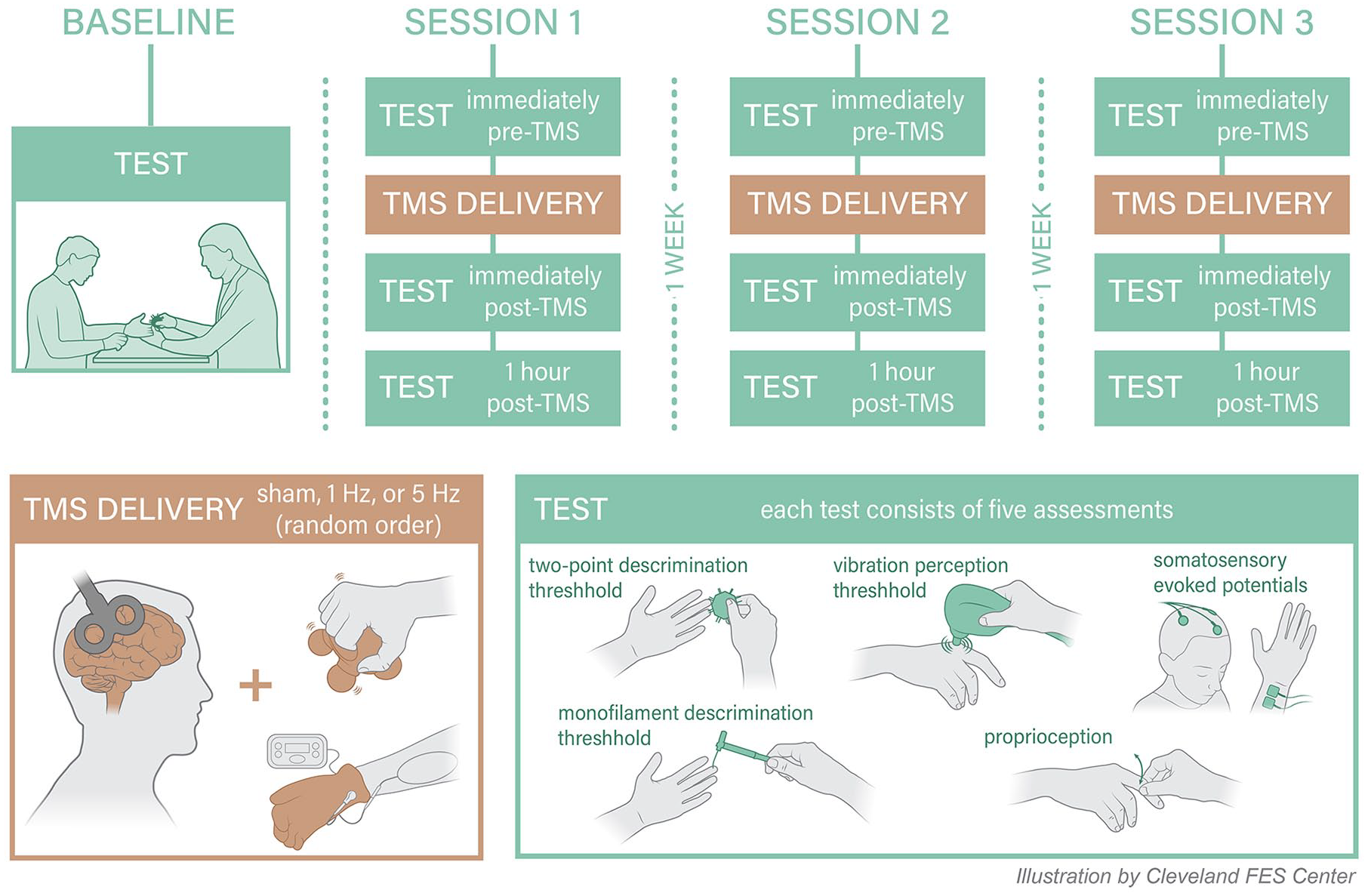
Overview of the study design.
Intervention
rTMS
Patients were seated in a chair with a headrest and arms resting on a table. rTMS was applied using a biphasic stimulator (Magstim Super Rapid 200 2 magnetic stimulator [Magstim Company Ltd] at LSCDVAMC site and MagPro R30 [Dantec] at CC site]. Coil targeting was guided by frameless stereotactic navigation (Brainsight2, Rogue Research, Inc). At the beginning of each session, evoked response to single pulses of TMS, called motor-evoked potentials (MEPs), were collected from contralateral first dorsal interosseus muscle (FDI). Surface electromyography was acquired via silver-silver chloride 8 mm bipolar electrodes (Powerlab 4/25T [AD Instruments Inc]) with a reference electrode over the lateral epicondyle (CC) or with Brainsight2 EMG pod via 10-mm surface gel adhesive electrodes with reference electrode over the lateral forearm (LSCDVAMC). Hotspot for FDI was identified as the site that evoked ≥50 µV MEPs at the lowest TMS intensity (motor threshold [MT]) reliably in 6 out of 10 trials. 37 Motor hotspot for FDI served as a guide to identify the anatomic location of S1 and MT intensity served as a means to establish rTMS intensities.
The site for S1 stimulation was identified 2 cm posterior to the FDI hotspot in M1.23,30 The intensity of rTMS application was subthreshold, applied at 90% of the resting MT required to elicit FDI responses in accordance with safety guidelines. 34 The H-rTMS protocol consisted of 5-Hz rTMS at 90% resting MT for a total of 1250 pulses as follows: 5 blocks of 250 pulses with an interblock interval of 60 seconds; each block consisted of five 50-pulse trains with an intertrain interval of 2 seconds.23,38 This protocol showed effect on sensory function in healthy controls.23,38 The L-rTMS protocol involved continuous 1-Hz rTMS of 1200 pulses, which is the most commonly used inhibitory rTMS paradigm. S-rTMS was provided using a sham coil and 5-Hz protocol. At LSCDVAMC, the active coil was a flat 70 mm figure-of-eight magnetic air-cooled coil with a maximum magnetic field strength of 1.5 T (Tesla) and an average inductance of 15.5 µH; the sham coil was identical in appearance to the active coil but had a maximum magnetic field of 0.2 T and an average inductance of 2.8 µH. At CC, both sham and active rTMS were provided with the dual-sided active/sham 70 mm figure-of-eight MagVenture A/P coil that has an inductance of 11.3 to 12.0 mH (for both active and sham) and a maximum magnetic field of 1.4 T on the active side and 0.07 T on the sham side. The outputs of both sham coils are well below stimulus threshold while producing similar tactile and auditory effects.
Peripheral Sensory Stimulation
During rTMS, peripheral sensory stimulation consisting of 5 minutes of sensory-level electrical stimulation and 5 minutes of vibratory stimulation to the affected hand was administered. Sensory-level electrical stimulation was delivered by an EMPI 300 PV Neuromuscular Stimulator at a frequency of 50 Hz at sub-motor intensity with an Electro-Mesh glove (Prizm Medical); 1 second ramp on/off; 20 seconds on and 5 seconds off. For vibratory stimulation, the subject’s hand was positioned with the palmar surface in contact with the handle of a mini massager (Homedics) and fingers gently curled over the handle (Figure 1). Subjects were asked to not actively grasp the massager rather to rest their hand in this position.
Outcome Measures
Clinical Measures
Somatosensory modalities included 2-point, proprioception, monofilament size, and vibratory discrimination (Figure 1). Subjects were blindfolded during testing and skin temperature was maintained at a constant level. Both the clinical assessor and the study participant were blinded to the order of rTMS protocol delivery and the same assessor tested a given subject for each of the 3 rTMS sessions.
Two-point discrimination was measured with Disk-Criminator disks by determining the subjects’ ability to perceive 2 points on the disk as 2 separate points rather than as a single point. 39 The distances between the 2 points ranged between 2 and 15 mm. One and 2 sensory points were presented in a pseudo-random order to subjects’ fourth digit volar fingertip surface. A threshold is determined when 70% accuracy is exhibited for identifying the difference between single versus double point stimulation. 39 Accuracy of threshold was confirmed by retesting the level above and below the determined threshold level. A score of 16 mm was assigned when the subject could not accurately differentiate 1 versus 2 points at the maximum distance (15 mm). Mean average for healthy controls has been reported to be 3.2 (±0.9) for the fourth digit of the dominant hand. 40 The volar surface of the fourth digit was assessed due to the high incidence of median nerve entrapment common in this population which could confound the findings. 41
For proprioception, an examiner held the subjects’ second digit at the medial and lateral surfaces of the proximal interphalangeal joint (forearm pronated) and flexed or extended the subject’s second metacarpophalangeal joint in a random sequence of approximately 20° flexion and extension. Subjects were asked whether the joint was moved upwards (extension) or downwards (flexion). Proprioception accuracy was expressed as a percent of correct responses 42 and a chance score for the testing as was conducted was 50%.
The monofilament test assessed the threshold for perceiving tactile stimulation at the fourth digit volar fingertip with Tactile Semmes-Weinstein Monofilaments. 43 Monofilaments of varying sizes (2.83, 3.61, 4.31, 4.56, 6.65) were presented in random time intervals achieving filament bent into “C” or blanched skin underneath for 6.65. We report the smallest filament reliably detected using standard clinical testing procedures. For individuals who could not sense the thickest filament, a score of 7.6 was recorded. Filaments were presented in descending order thickest to finest. Normal monofilament score is ≤2.83. 43
Vibration perception threshold was determined using the Biothesiometer (Bio-Medical Instrument Co). The Biothesiometer pestle, which vibrates at a frequency of 120 Hz, was placed at the second metacarpophalangeal joint. 44 Vibration amplitude was gradually increased. Test score corresponded to device units (vibration amplitude) at which subjects detected vibration averaged across 3 trials. 44 A normal score is <7 V. 45
Somatosensory Evoked Potentials
SEPs were recorded with a Cadwell Sierra Wave (Cadwell; LSCDVAMC) or with Powerlab 4/25T (AD Instruments Inc) and a Grass Stimulator (Natus Neurology; CC). 46 The recording electrodes (1 cm diameter, gold cup electrodes filled with conductive paste) were placed 2 cm posterior to C3 and C4 (10-20 international system of EEG electrode placement) and the reference electrode at Fz (Figure 1). Electrode impedance was kept below 5 kΩ. Stimulus was applied to the median nerve at the wrist (anode at the wrist crease and cathode 2 cm proximal). Ground electrodes were placed at the lateral epicondyle of the stimulated arm. Square wave stimulus of 200 µs pulse width and frequency of 5.1 Hz was applied with sufficient amplitude to cause 1 to 2 cm of thumb movement. The evoked response from 500 stimuli were recorded and averaged for a single trial. Three SEP trials were recorded then analyzed. Latencies (in milliseconds) were determined for N20, P25, N33, P45, N60, P100, and N120. Peak-to-peak amplitudes (in µV) were extracted for N20-P25, P25-N33, N33-P45, P45-N60, N60-P100, P100-N120. 46
Statistical Analysis
Descriptive statistics were applied to determine the shape of data distributions. Baseline scores (Pre) were compared using nonparametric Friedman test for repeated measurements and Wilcoxon signed rank test for paired comparisons. Linear mixed models were fit to analyze the response of both clinical and SEP outcome measures to the 3 intervention protocols. We included subject-level random intercept, as well as compound symmetry repeated covariance type structure, to reflect within-subject and serial correlation. The outcome (dependent) variables in the mixed models were the difference in respective outcome measure score from Pre to Post 1 and Pre to Post 2 (eg, change in 2-point discrimination at Post 1 and at Post 2). The 2 explanatory variables were the following: (1) a categorical variable denoted as rTMS session type: either S-rTMS, L-rTMS or H-rTMS; and (2) a categorical time variable, indicating whether the outcome value is a difference from Pre to Post 1 or Pre to Post 2. Note that statistically significant treatment variable association indicates that the change over time differs by rTMS type. We also conducted pairwise analysis of the treatment effects, to ascertain directionality. Pairwise comparison based on estimated marginal means included Sidak’s method for correction for multiple comparisons. The reported P values were adjusted for multiple comparisons.
Results
Subject characteristics are provided in Table 1. Mean differences between the stroke-affected and unaffected hand were statistically significant for 2-point discrimination, monofilament threshold, proprioception, and vibration detection (P < .05; Table 2). The unaffected hand had diminished somatosensation with frequency of 94% compared with the normative values (Table 2).
Subjects’ Characteristics.
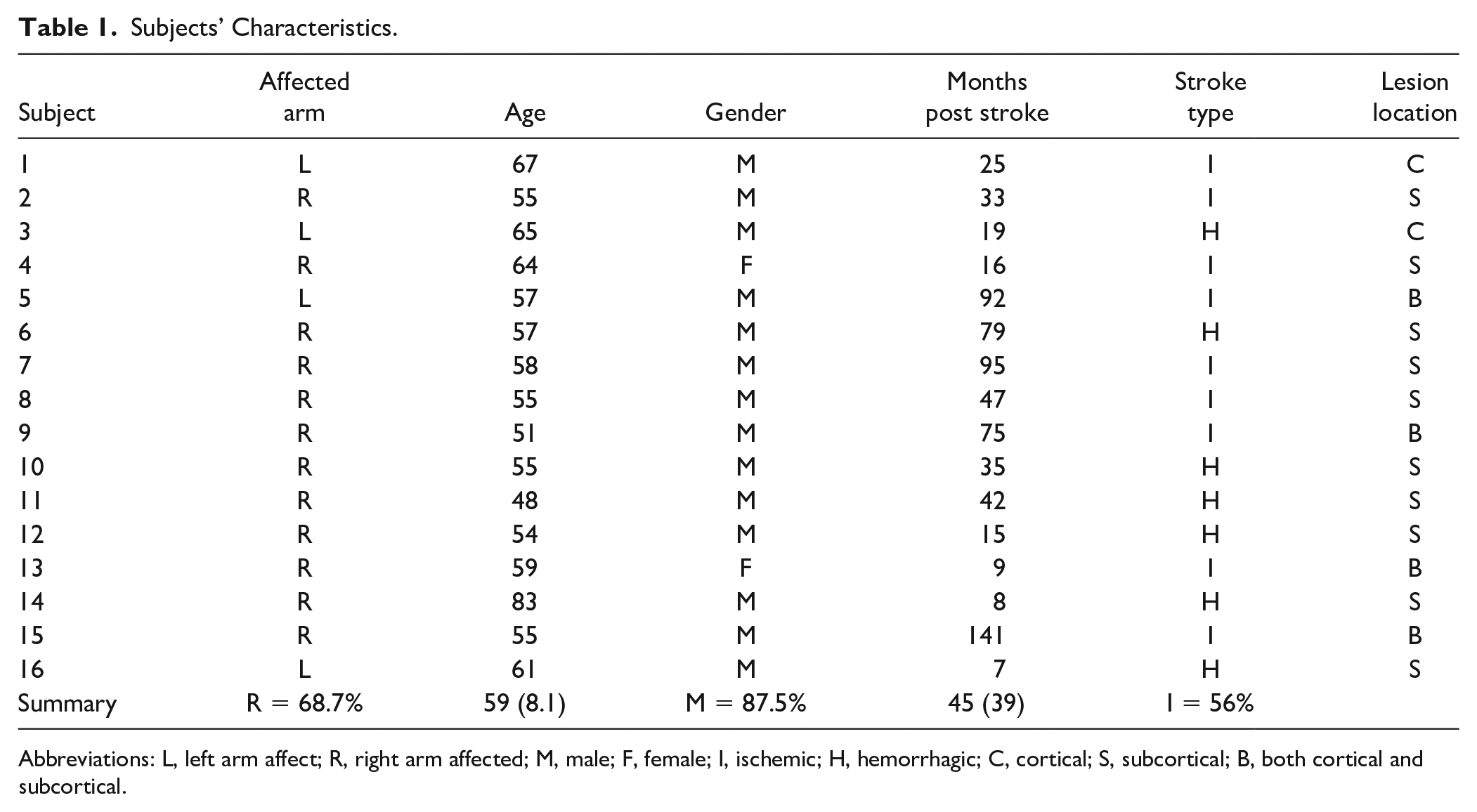
Abbreviations: L, left arm affect; R, right arm affected; M, male; F, female; I, ischemic; H, hemorrhagic; C, cortical; S, subcortical; B, both cortical and subcortical.
Baseline Testing Values Averaged Across the 3 rTMS Sessions for Both Stroke-Affected and Unaffected Arms (Mean (SD)).
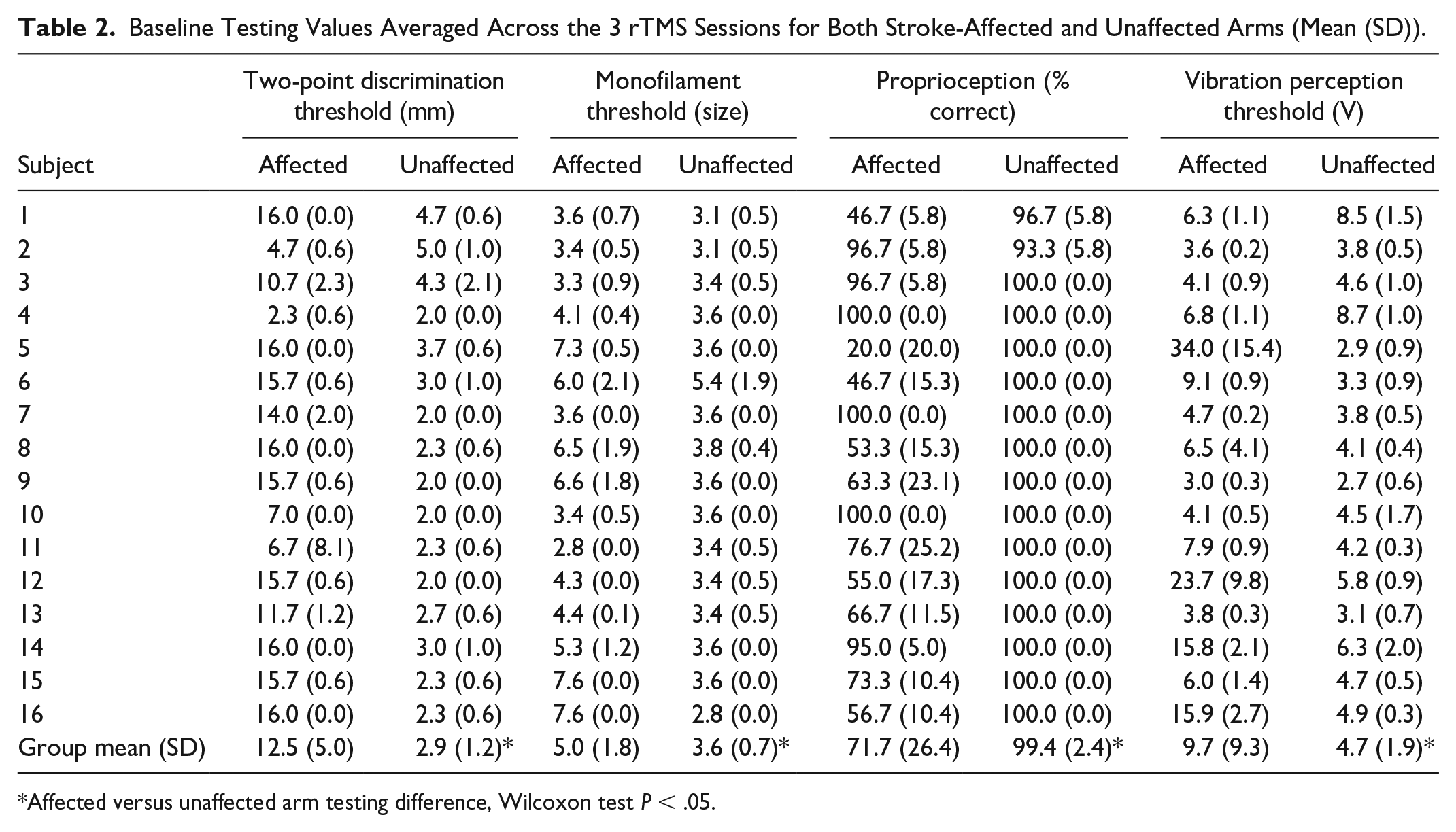
Affected versus unaffected arm testing difference, Wilcoxon test P < .05.
Baseline Stability of Measures
For the stroke affected arm, pre-intervention somatosensory measurements were similar at all 3 rTMS sessions for 2-point discrimination, monofilament threshold and vibration threshold (Table 3). However, for proprioception, there was a slight decrease in proprioception accuracy over the 3 sessions (Friedman test P = .03). For the unaffected arm, there were no differences among pre-intervention somatosensory test scores. There were no differences in baseline values for any SEP components (Table 3). The average duration between sessions 1 and 2 was 16.7 (10.4) (mean [SD]) days and between sessions 2 and 3, 16.4 (18.5) days.
Pre-Intervention (Baseline) Measurements Across the 3 Consecutive Intervention Sessions a .
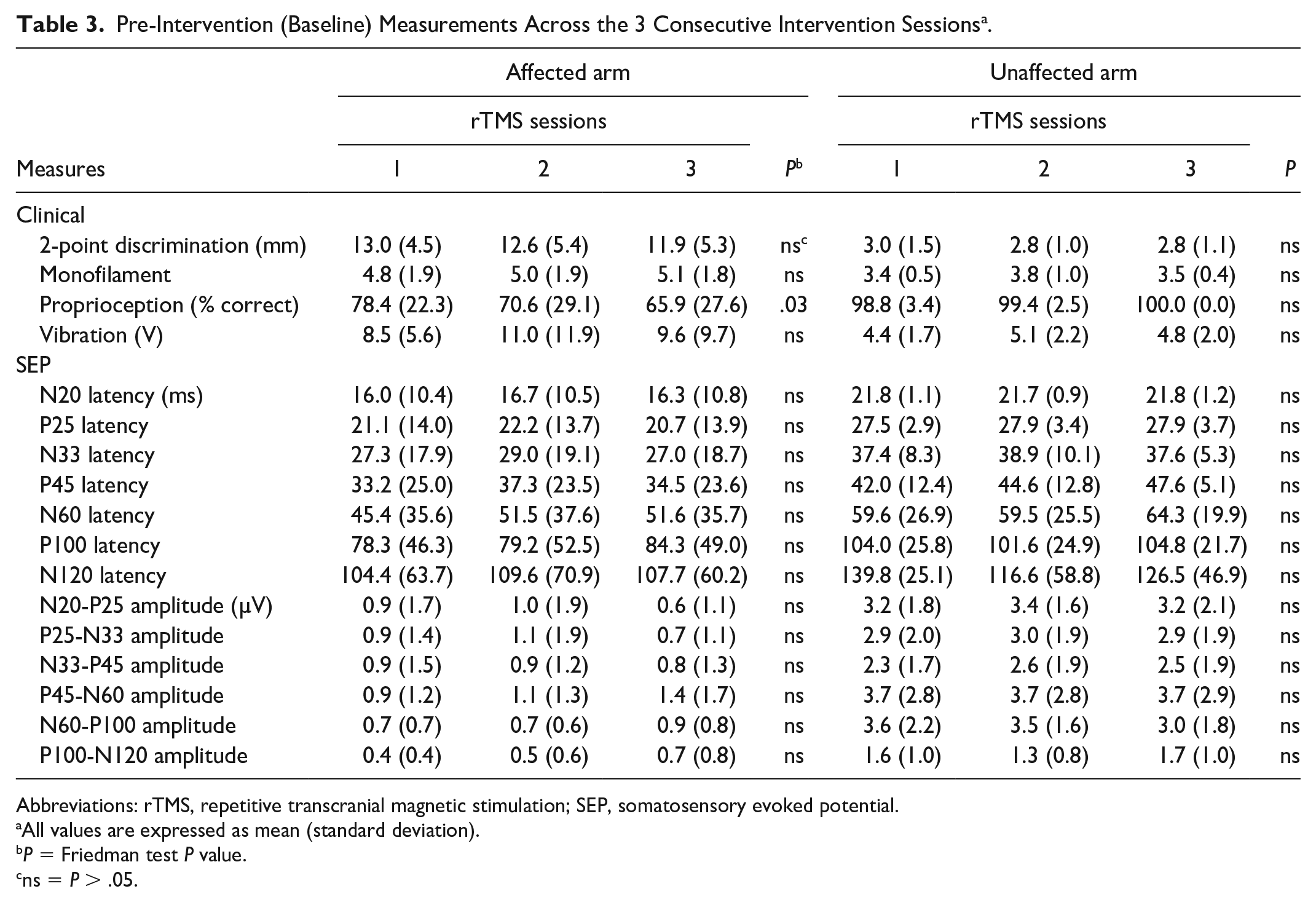
Abbreviations: rTMS, repetitive transcranial magnetic stimulation; SEP, somatosensory evoked potential.
All values are expressed as mean (standard deviation).
P = Friedman test P value.
ns = P > .05.
Effects of rTMS on Clinical Somatosensory Function
Two-Point Discrimination
Affected arm
Based on the mixed model analysis, we found for the primary outcome measure, 2-point discrimination, a statistically significant treatment effect for session type (F(2, 76.163) = 3.5, P = .035). The pairwise analysis demonstrated an improvement of 2-point discrimination of the affected arm in response to H-rTMS compared to S-rTMS (estimated mean difference of −1.187 mm, SE = 0.459; P = .011). There was a trend toward greater improvement after H-rTMS compared with L-rTMS responses (estimated mean difference = −0.773 mm, SE = 0.459, P = .096) but no difference between L-rTMS and S-rTMS. Figure 2A and Table 4 depict a change in 2-point discrimination ability in response to different rTMS interventions.
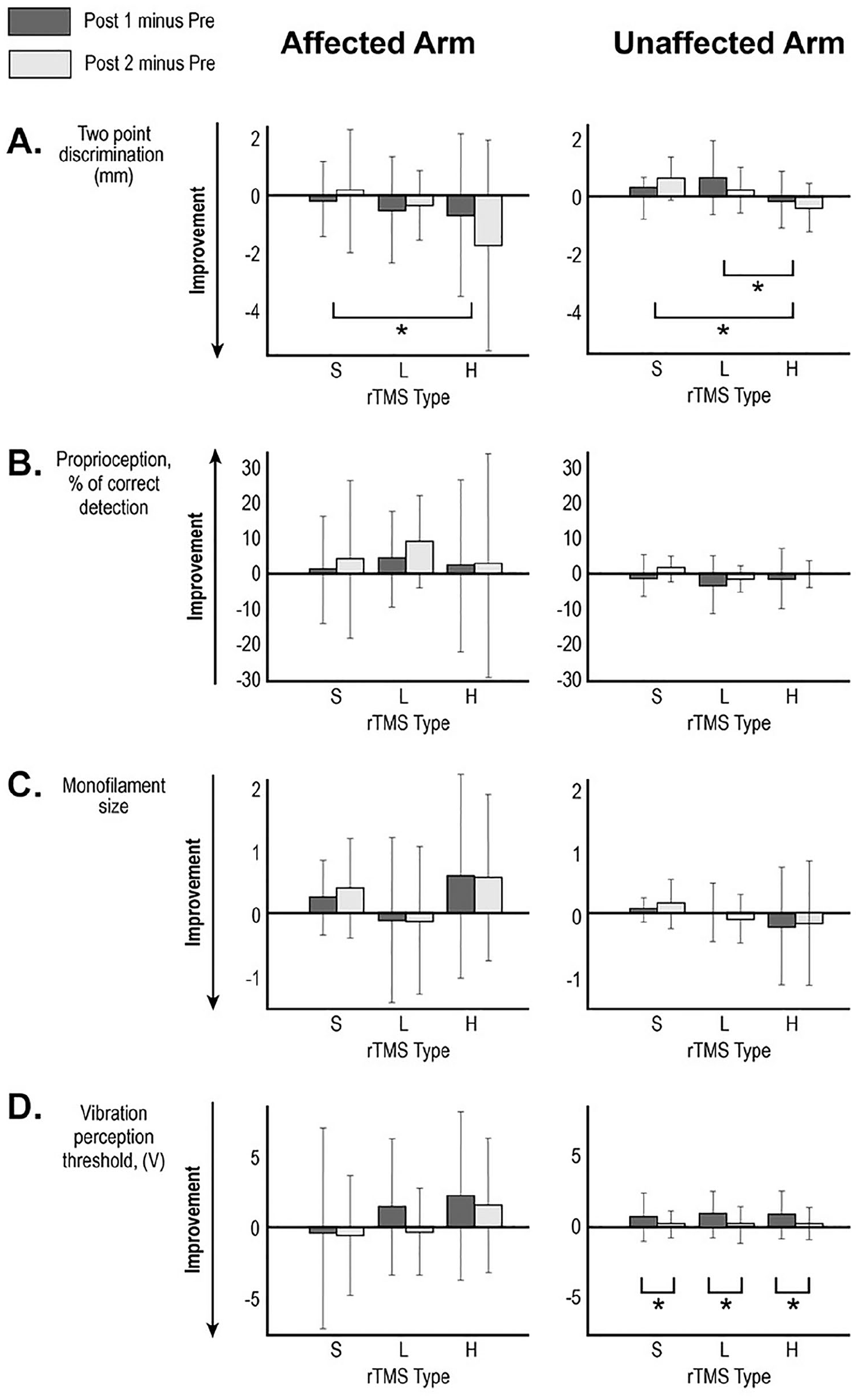
Changes in clinical test scores following the 3 interventions performed ≥1 week apart. Mean changes for (A) 2-point discrimination, (B) proprioception, (C) monofilament size, and (D) vibration perception threshold at Post 1 (immediately after rTMS; dark bar) and Post 2 (1 hour after rTMS; light bar) are shown with error bar representing standard deviation. Stroke-affected arm data is in the left-sided graphs and stroke-unaffected arm is the right-sided graphs. *Brackets indicate statistically significant post hoc paired comparisons in the mixed model analysis based on estimated marginal means. S, sham rTMS; L, low-frequency rTMS; H, high-frequency rTMS.
Baseline Values and Changes of Clinical and SEP Measures in Response to Different rTMS Interventions (Affected Arm) a .
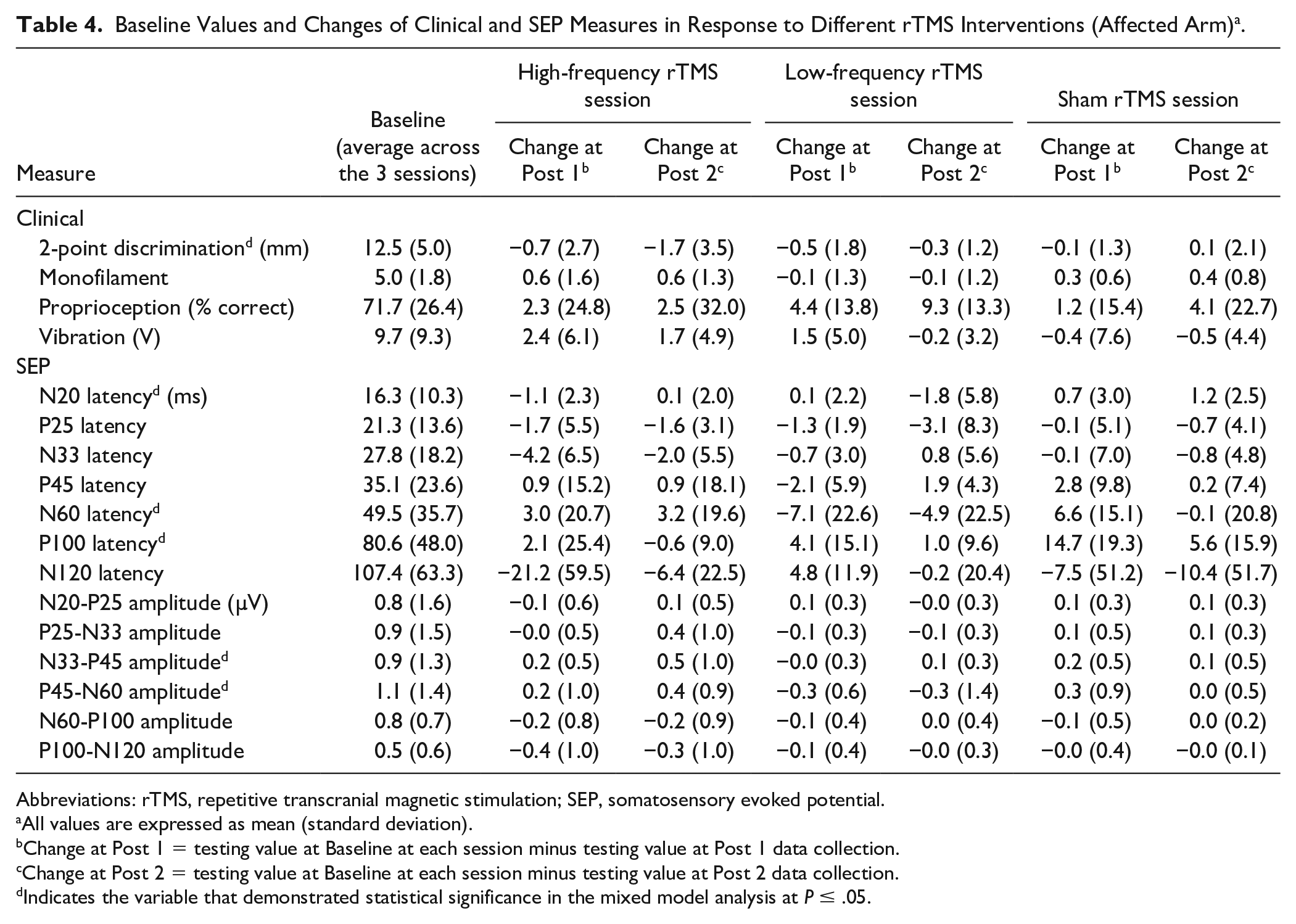
Abbreviations: rTMS, repetitive transcranial magnetic stimulation; SEP, somatosensory evoked potential.
All values are expressed as mean (standard deviation).
Change at Post 1 = testing value at Baseline at each session minus testing value at Post 1 data collection.
Change at Post 2 = testing value at Baseline at each session minus testing value at Post 2 data collection.
Indicates the variable that demonstrated statistical significance in the mixed model analysis at P ≤ .05.
Unaffected arm
Mixed model analysis showed a statistically significant improvement of 2-point discrimination of the unaffected arm following H-rTMS intervention (F(2, 192.786) = 10.6, P < .0001). Based on the pairwise analysis, H-rTMS produced greater improvement compared with S-rTMS (estimated mean difference = −0.710 mm, SE = 0.173; P < .0001) and greater improvement compared with L-rTMS (estimated mean difference = −0.663 mm, SE = 0.173; P < .0001). Table 5 and Figure 2A depict a change in 2-point discrimination ability in response to different rTMS.
Baseline Values and Changes of Clinical and SEP Measures in Response to Different rTMS Interventions (Unaffected Arm) a .
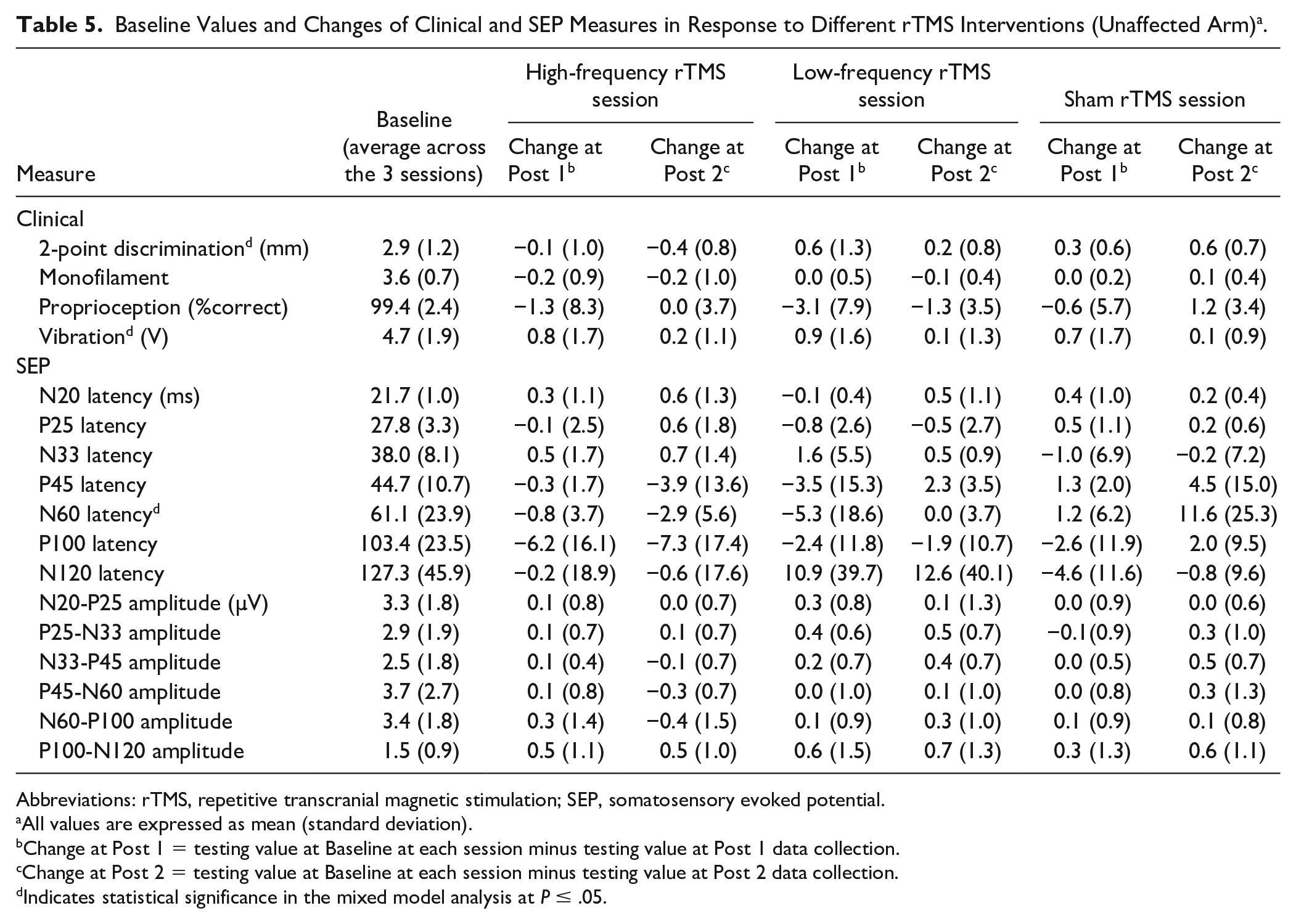
Abbreviations: rTMS, repetitive transcranial magnetic stimulation; SEP, somatosensory evoked potential.
All values are expressed as mean (standard deviation).
Change at Post 1 = testing value at Baseline at each session minus testing value at Post 1 data collection.
Change at Post 2 = testing value at Baseline at each session minus testing value at Post 2 data collection.
Indicates statistical significance in the mixed model analysis at P ≤ .05.
Proprioception and Monofilament Threshold
For both affected and unaffected arms, mixed model analysis did not show a statistically significant difference between responses to the 3 types of rTMS for proprioception (Figure 2B; Tables 4 and 5) or monofilament threshold (Figure 2C; Tables 4 and 5). However results for monofilament threshold were trending toward significance—affected: F(2, 75.303) = 2.769, P = .069; unaffected: F(2, 74.34) = 2.737, P = .071.
Vibration
Affected Arm
There were no statistically significant mixed model results for vibration for the affected arm (F(2, 75.545) = 2.026, P = .139; Table 4 and Figure 2D).
Unaffected Arm
Mixed model analysis demonstrated a statistically significant reduction in vibration perception at Post 1 compared with Post 2 (F(2, 153.350) = 7.843, P = .006). There was no effect of session type (P = .928). These transient changes in vibration perception were similar following all treatment types (Table 5, Figure 2D).
Effect of rTMS on Somatosensory Evoked Potential
Affected Arm SEP
We used mixed model analysis to evaluate changes in SEP component measures in response to the 3 types of rTMS. Table 4 shows data for SEP at baseline and changes observed at Post 1 and Post 2. There were statistically significant findings for session type in models where independent variables were peak to peak amplitude changes for N33-P45 (F(2, 133.027) = 3.518, P = .032) and P45-N60 (F(2, 67.353) = 3.212, P = .047) as well as changes in N20 latency (F(2, 69.64) = 3.37, P = .04), N60 latency (F(2, 47.343) = 4.375, P = .018), and P100 latency (F(2, 37.608) = 3.537, P = .039). For N33-P45 amplitude change, margin of mean test demonstrated that an amplitude change following H-rTMS was greater compared to L-rTMS (estimated marginal mean difference = 0.291 µV, SE = 0.113 P = .032). For P45-N60, there was a trend toward H-rTMS producing greater response compared to L-rTMS (estimated marginal mean difference = 0.611 µV, SE = 0.253, P = .054). For N20 latency change, H-rTMS resulted in a shorter latency for N20 compared to change following S-rTMS (estimated marginal means difference = −2.049 ms, SE = 0.8, P = .037). For N60 latency change, H-rTMS also resulted in a shorter latency for N60 compared to change following S-rTMS (estimated marginal means difference = −10.288 ms, SE = 3.7, P = .025). Similarly, for P100 latency change, H-rTMS resulted in a shorter latency compared to change following S-rTMS (estimated marginal means difference = −16.215 ms, SE = 6.1, P = .035).
Unaffected Arm SEP
Table 5 demonstrates both baseline SEP components values and changes in these measures in response to each of the rTMS interventions. Based on mixed model analysis, N60 latency showed a statistically significant result for session type (F(2, 64.76) = 3.875, P = .026). Marginal mean estimate tests showed that following H-rTMS, there was a shortening of latency for N60 peak compared with S-rTMS (estimated marginal means = −3.56 µV, SE = 1.29, P = .022).
Discussion
This proof-of-principle study evaluates for the first time whether facilitation or inhibition of contralesional S1 using rTMS paired with peripheral sensory stimulation can elicit improvement in somatosensory function in chronic stroke. Our results suggest that facilitatory H-rTMS (5 Hz) plus simultaneous peripheral sensory stimulation to the affected arm improves 2-point discrimination. In addition, our exploratory analysis of SEP indicates enhancement of somatosensory evoked responses following H-rTMS. Both clinical and physiological effects lasted at least an hour as suggested by the lack of differences between the changes found immediately after and one hour following the interventions. There were no differences among the pre-intervention testing for all sessions.
Somatosensory Modality-Specific Response
A statistically significant response to facilitatory H-rTMS (5 Hz) plus simultaneous peripheral sensory stimulation of the affected arm was observed for 2-point discrimination ability on the stroke-affected side. Other somatosensory modalities (proprioception, vibration, and monofilament) did not demonstrate a statistically significant change. This modality-specific response maybe due to differences in somatosensory signal processing, that is, proprioceptive versus tactile, primary somatosensory perception (monofilament) versus associative (2-point discrimination). It is possible that discriminative somatosensory function is more likely to be impacted by an ipsilateral intervention due to a bilateral processing of somatosensory discrimination compared to the processing of primary somatosensory input. In other words, tactile sensing is involved in monofilament test versus 2-point discrimination. Others have also reported a modality-specific response to brain stimulation following 10-Hz rTMS of the primary motor region where facilitatory stimulation reduced painful perception of thermal stimuli but did not alter ability to sense nonpainful tactile stimulation. 47 Another factor that may be influencing the modality specific response is the choice of the peripheral stimulus paired with brain stimulation. For example, a modality-specific response in healthy controls receiving rTMS over M1 paired with passive joint movement resulted in a significant change of corticospinal excitability while stimulation with rTMS alone did not. 48 In our study, we used sensory-level electrical stimulation and vibration. Perhaps, other combinations of peripheral somatosensory therapy and brain treatment protocols need to be evaluated to determine if they have a specific effect on other somatosensory modalities. Somatosensation is complex, and thus an array of measures was employed to assess various aspects of somatosensation. At baseline, 2-point discrimination had the largest difference between affected and unaffected hand and thus the tool may have been sensitive enough to detect this change. It may be that the other measurement tools were not sensitive enough to detect change in the other somatosensory modalities assessed. Indeed, available somatosensory testing has many limitations that include a lack of sensitivity, poor responsiveness, decreased validity, and a lack of objectiveness 49 and this may have contributed to having only 1 out of 4 sensory measures demonstrating a statistically significant response to the intervention. Additionally, the study employed impairment-level measures and no measurement of function was directly assessed. Therefore, the impact of this observed change in 2-point discrimination on overall function was not studied. However, the fact that we found an effect on 2-point discrimination in a small sample using standard clinical tests is intriguing and suggests future study is warranted with measures that more finely assess somatosensory function.
Unilateral Stimulation, Bilateral Effect
Importantly, our findings suggest that contralesional S1 is playing a role in processing of tactile discrimination in individuals in the chronic stage after stroke. This finding might contradict the classic functional sensory anatomy teaching stating that peripheral signals evoke contralateral activity and, therefore, are processed in the contralateral hemisphere. 50 Indeed, recent functional imaging studies describe bihemispheric activation following unilateral somatosensory stimulation as seen with both fMRI and somatosensory evoked potential tests.51,52 The pathways of sensory signal transfer to bilateral hemispheres may involve transcallosal connections and possibly direct ipsilateral uncrossed afferent pathways.27,53-55 Therefore, it is reasonable to expect that after stroke, these secondary contralesional somatosensory processing regions play a role in restoring the lost function.
The concept of focal rTMS influencing function of bilateral somatosensory networks and bilateral somatosensory abilities has been previously evaluated in healthy adults and in pain-related research. In healthy adults, Premji et al 56 demonstrated that unilateral facilitatory stimulation using an intermittent theta-burst stimulation (iTBS) protocol increased somatosensory evoked potential amplitude at the site of stimulation and also on the nonstimulated hemisphere. Another study of healthy adults showed that 5-pulse 10-Hz TMS burst targeting right parietal region when paired with same side peripheral median nerve stimulation increased fMRI activation. 57 In pain therapy, unilateral 10-Hz rTMS targeting primary motor area (M1) reduced thermal pain threshold in both hands. 47 Overall, our findings and those of others support contralesional somatosensory cortex modulation as a reasonable brain stimulation target to enhance poststroke somatosensory restoration.
Facilitatory, Not Inhibitory, rTMS Targeting Contralesional S1
Brain reorganization may take several paths to support recovery of somatosensory function after stroke. If we consider a paradigm of interhemispheric balance that is disturbed after stroke, one pattern of plasticity may be toward rebalance of interhemispheric interactions. 21 Indeed, in motor control studies using rTMS, inhibition of the contralesional hemisphere has been shown to enhance motor function of the stroke affected upper limb presumably by correcting imbalance. 21 Interhemispheric interactions have been demonstrated for healthy controls where inhibition of somatosensory pathways on one side resulted in enhanced excitability of SEP on the opposite side30,58 and improved somatosensory function.58-60 After stroke, anesthesia of the unaffected upper limb improved somatosensory perception on the affected side. 61 However, these were peripherally administered inhibitory interventions of the unaffected limb and we cannot expect the same response for modulation of the brain somatosensory networks. In fact, contralesional S1 may be supporting the lost function of the opposite hemisphere since somatosensory signal processing is bilateral. It is possible the mechanism of somatosensory improvement following contralesional S1 facilitation is tapping into pathways not related to transcallosal inhibition. These mechanisms may involve facilitation of alternative somatosensory processing centers. Therefore, it is quite reasonable to find that the facilitation of contralesional S1 and not its inhibition benefits recovery of somatosensory function after stroke.
Unaffected Upper Limb Response to Stimulation Supports Protocol Feasibility
We observed a statistically significant improvement of 2-point discrimination of the unaffected-by-stroke arm. This finding is an important confirmation that our protocol achieves modulation of somatosensory function. H-rTMS, which is known to enhance cortical activity, improved 2-point discrimination of the unaffected hand, contralaterally to stimulated cortex. The positive changes in response to H-rTMS were contrasted with both S-rTMS and L-rTMS. Others demonstrated in healthy adults that 2 types of facilitatory TMS (5-Hz rTMS and iTBS) can transiently increase 2-point discrimination.59,60 Notably, in our cohort of stroke survivors we observed changes in 2-point discrimination even though we did not use a more sensitive 2-point discrimination set up with interpoint distances less than 2 mm as was applied in studies with healthy controls. Perhaps, the response is more robust in individuals with stroke deficits compared with healthy controls.
A transient decrease in vibration sense was observed immediately after each intervention. This was not a lasting effect confirmed by the lack of differences among the testing at each session’s baseline. It may be that adaptation of the cutaneous mechanoreceptors during the vibratory stimulation of the hand was the cause for this temporary change. 62 Although vibration was applied to the stroke-affected hand, the unaffected hand may have inadvertently received vibratory stimulation while both arms were resting on a table where the affected hand was placed on a vibrating device.
SEP Changes in Response to rTMS
Our exploratory analysis suggests that H-rTMS may enhance the SEP signal induced by peripheral stimulation of the median nerve. Specifically, for the stroke affected side there was a reduction of N20, N60, P100 peak latencies and greater amplitudes of N25-P33 and N33-P45 peaks following H-rTMS. The stroke-unaffected SEP pathway demonstrated changes for the N60 peak latency following H-rTMS. Changes in both early and late SEP components for both stroke-affected and stroke-unaffected somatosensory tracts suggest unilateral modulation of contralesional S1 may have an impact on the bilateral somatosensory processing network. The earlier peaks (N20, P25, N33, P45) may reflect changes at the level of thalamocortical projections to S1 cortex areas 3b and area1. 63 Changes in the late SEP components (N60 and P100) suggest involvement of SII and other higher-order sensory27,64 and sensory-motor 65 integration regions. SEP changes provide additional support of potential therapeutic feasibility of our stimulation paradigm to evoke response in the function of the somatosensory network.
After-Effect Duration
There were no differences between Post 1 and Post 2 test results. Thus, when the changes were achieved, they lasted at least 60 minutes. This finding is expected and suggests the interventions induced an after-effect of up to 60 minutes (reviewed in Hernandez-Pavon and Harvey 24 ). Physiological mechanisms of the after-effect are unclear but are thought to involve long-term potentiation and long-term depression.24,66 In fact, analysis of SEP changes also demonstrated lasting after-effects in both increased amplitude and decreased latencies, and thus provide indirect evidence for functional brain changes remaining after stimulation for at least an hour.24,66 It is expected that a single rehabilitation session would not provide a permanent functional recovery and future multisession studies are needed to evaluate a long-lasting of this intervention.
Limitations
Three main limitations may have led to the limited response of different somatosensory modalities. First, that we only demonstrated a significant change on 2-point discrimination may be because for this measure there was the largest separation between the affected and unaffected sides. Therefore, it had the highest opportunity for improvement compared to the other measures. Second, the outcome measures may have not been sensitive enough to detect change. For instance, the proprioception measure may have been too subjective in nature to detect change. Third, different forms of peripheral stimulation paired with NIBS may yield better results. It should also be noted the study cohort was younger than average for the stroke population and this may limit the generalizing study results to the greater stroke population. Future research using more sensitive somatosensory measuring tools, 67 testing a population with a broader range of sensory deficits and multisession design of complimentary peripherally directed stimulation paired with NIBS should overcome these limitations.
Conclusion
Using a single-session randomized crossover design, we report preliminary evidence in support of the effect of facilitatory contralesional rTMS on 2-point discrimination in chronic stroke. This may be due to the higher incidence of 2-point discrimination deficit compared with other sensory modalities in our patient cohort. In motor rehabilitation literature, a single-session crossover intervention paradigm has been widely used as a first step. In these studies, different brain stimulation interventions were compared on the same patients with the goal of identifying a therapy for application in multisession clinical trials. Further studies are needed to explore the role of rTMS in rehabilitation of somatosensory function after stroke.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Award Number I01BX007080 from the Rehabilitation Research & Development Service of the VA Office of Research and Development; by the Case Western Reserve University/Cleveland Clinic CTSA Grant Number UL1 RR024989 from the National Center for Research Resources (NCRR), a component of the National Institutes of Health and NIH roadmap for Medical Research.
