Abstract
In well-recovered stroke patients with preserved hand movement, motor dysfunction relates to interhemispheric and intracortical inhibition in affected hand muscles. In less fully recovered patients unable to move their hand, the neural substrates of recovered arm movements, crucial for performance of daily living tasks, are not well understood. Here, we evaluated interhemispheric and intracortical inhibition in paretic arm muscles of patients with no recovery of hand movement (n = 16, upper extremity Fugl-Meyer Assessment = 27.0 ± 8.6). We recorded silent periods (contralateral and ipsilateral) induced by transcranial magnetic stimulation during voluntary isometric contraction of the paretic biceps and triceps brachii muscles (correlates of intracortical and interhemispheric inhibition, respectively) and investigated links between the silent periods and motor recovery, an issue that has not been previously explored. We report that interhemispheric inhibition, stronger in the paretic triceps than biceps brachii muscles, significantly correlated with the magnitude of residual impairment (lower Fugl-Meyer scores). In contrast, intracortical inhibition in the paretic biceps brachii, but not in the triceps, correlated positively with motor recovery (Fugl-Meyer scores) and negatively with spasticity (lower Modified Ashworth scores). Our results suggest that interhemispheric inhibition and intracortical inhibition of paretic upper arm muscles relate to motor recovery in different ways. While interhemispheric inhibition may contribute to poorer recovery, muscle-specific intracortical inhibition may relate to successful motor recovery and lesser spasticity.
Keywords
Introduction
Over the past nearly 2 decades, there has been a great deal of investigation into mechanisms of impairment and recovery of hand movement after human stroke. This work has demonstrated that limitations in recovery of functional hand movements poststroke are often linked to abnormalities in intracortical and interhemispheric inhibition. These findings have provided insight into the mechanisms of behavioral rehabilitation approaches, such as constraint-induced movement therapy,1-5 and have informed the development of cortical stimulation paradigms to improve hand recovery.6-9
Previous studies have used transcranial magnetic stimulation (TMS) to investigate intracortical inhibition of primary motor cortex (M1) hand representations in well-recovered stroke patients with at least partial recovery of hand function. Paired-pulse measurements of short-interval intracortical inhibition (SICI), 10 associated with GABAA-mediated intracortical inhibition, 11 have shown abnormally decreased levels of intracortical inhibition targeting the paretic hand.1,2,12-15 In contrast, intracortical inhibition reflected by the contralateral silent period (cSP), associated with GABAB receptor–mediated inhibition, 11 is reported to be abnormally increased in the paretic hand1,15-19 and to decrease with recovery. 16 Thus, it appears that SICI, reflecting GABAA-mediated intracortical inhibition, is abnormally decreased while cSP, reflecting GABAB receptor–mediated inhibition, is abnormally increased in the paretic hand post-stroke.
In addition to intracortical inhibition, interhemispheric inhibition between M1 hand representations in stroke patients with hand recovery has also been widely studied, and like intracortical inhibition, it has been studied using both paired-pulse and silent period TMS techniques. Paired-pulse measurements have shown that interhemispheric inhibition targeting the affected hemisphere (ie, paretic hand) is stronger than that targeting the unaffected hemisphere20-22 and abnormally persistent during paretic finger movement preparation,23,24 particularly in those with poorer hand recovery. Ipsilateral silent period measurements have provided further support for the notion that interhemispheric inhibition targeting the paretic hand is stronger than that targeting the nonparetic hand15,25 and that measured in controls. 26
Mechanisms of upper arm motor recovery in stroke patients unable to use their hands, however, are not well understood. To examine interhemispheric and intracortical inhibition in paretic elbow flexors and extensors, we evaluated silent periods during voluntary isometric contractions of paretic arm biceps (flexor) or triceps (extensor) brachii and measured the correlation between these measures and clinical and behavioral tests of motor ability, reaching performance, and spasticity. Recognizing that specific electrophysiological measurements, such as silent periods, reflect only a portion of the larger processes of intracortical and interhemispheric inhibition, we emphasize that when we refer to intracortical and interhemispheric inhibition we are referring only to that reflected by the contralateral and ipsilateral silent periods, respectively.
Given that many patients have particular difficulty deactivating elbow flexors, we postulated that inhibition targeting an elbow flexor muscle (biceps brachii) would be less than that targeting an elbow extensor (triceps brachii) and that biceps inhibition would correlate negatively with motor impairment. We report that interhemispheric inhibition and intracortical inhibition of these paretic upper arm muscles relate to paretic arm motor recovery differently in this population.
Materials and Methods
Participants
Sixteen individuals with chronic hemispheric stroke participated in the study (8 females; age: 59.0 ± 9.0 years; time poststroke: 5.0 ± 3.8 years; Table 1) after providing written informed consent according to a protocol approved by the local ethical review boards. Testing was conducted at 1 of 2 sites, the National Institutes of Health Clinical Center (Bethesda, MD) or MedStar National Rehabilitation Hospital (Washington, DC), using identical equipment and methods. Inclusion criteria included being at least 6 months poststroke and having the ability to reach forward at least 5 cm without compensatory trunk movement. Potential participants were excluded if they were less than 18 years of age, pregnant, had cerebellar or brainstem lesions, or any contraindications to TMS (eg, metal objects inside eyes or skull, history of seizures). We also excluded patients with voluntary wrist and finger movement on the paretic side because we wished to specifically target patients with less complete recovery (ie, more residual motor dysfunction) than those who have been typically studied in the past. Thus, all participants had relatively severe arm impairment and lacked voluntary finger and wrist movement. On average, they scored 27.0 ± 8.6 on the Upper Extremity Fugl-Meyer Assessment. All participants had subcortical lesions, some of which extended into the cortex, but all of which spared the primary motor cortex (M1). In general, the lesions resulted from large ischemic or hemorrhagic infarcts of the middle cerebral artery.
Patient Characteristics.
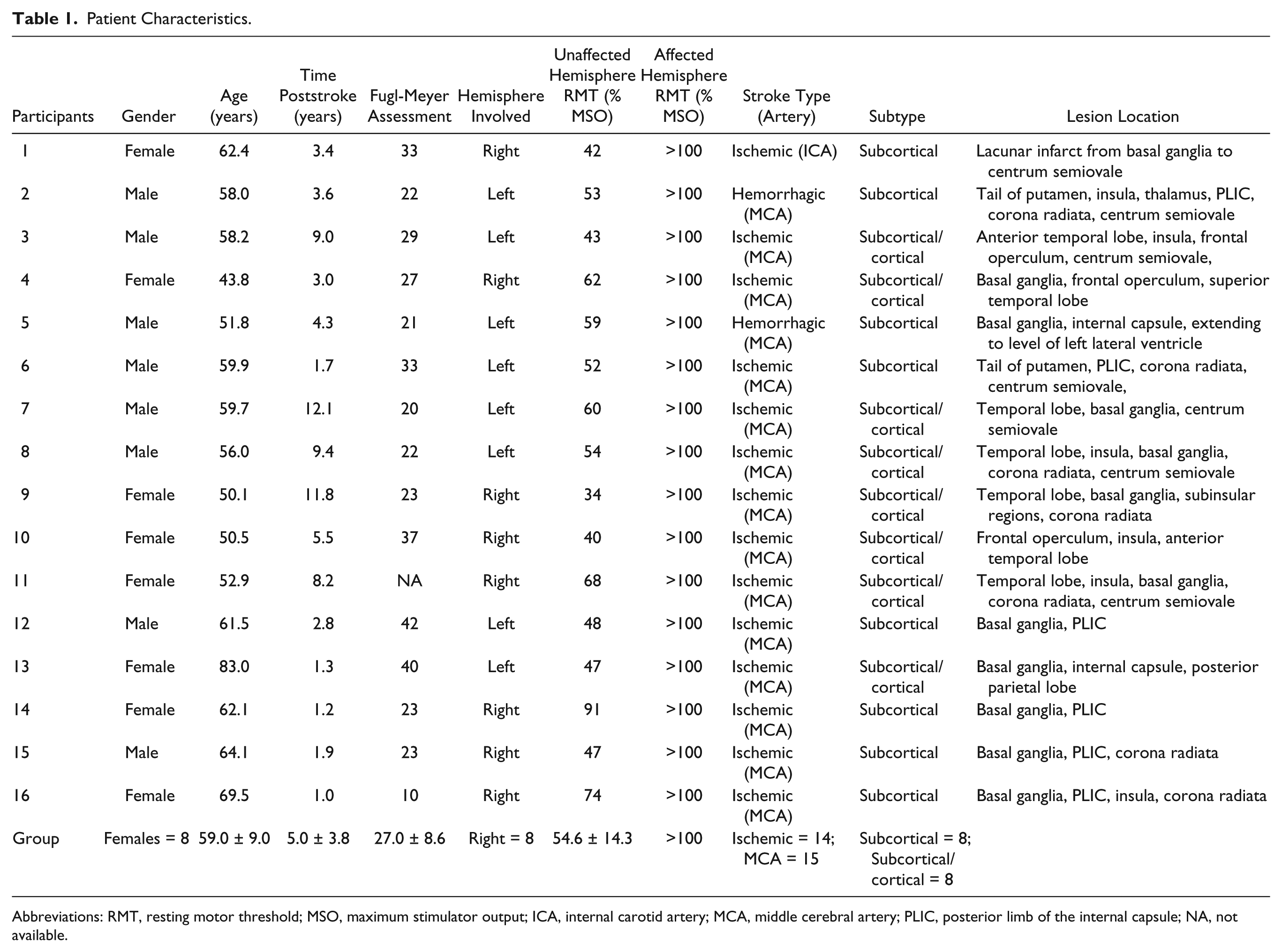
Abbreviations: RMT, resting motor threshold; MSO, maximum stimulator output; ICA, internal carotid artery; MCA, middle cerebral artery; PLIC, posterior limb of the internal capsule; NA, not available.
Procedures
Participants were seated in an adjustable high-backed chair with arm rests. Participants’ shoulders were positioned at 0° flexion and elbows at 90° flexion, with forearms supported by cushioned arm rests. Electromyography (EMG) signals were recorded using active differential surface EMG electrodes (B&L Engineering, Santa Ana, CA) placed on the biceps brachii (short head) and triceps brachii (long head) of both arms. Standard skin preparation and muscle identification procedures were used to ensure the reliability of EMG signals. The electrodes had a pre-amplified gain of 330 and input impedance greater than 100 MΩ, providing good electromagnetic artifact suppression when used with TMS. EMG signals were digitized at 10 kHz, 16-bit precision (Cambridge Electronic Design Ltd, Cambridge, England) and subsequently high-pass filtered at 10 Hz (second-order Butterworth) in MATLAB (MathWorks, Natick, MA) to reduce direct current offsets and movement artifacts.
A figure-of-eight double 70 mm TMS coil attached to a Magstim 200 stimulator (Magstim Company Ltd, Wales, UK) was used to deliver the stimuli. When stimulating the unaffected hemisphere M1, coil position (oriented to induce posterior-to-anterior current flow in the underlying cortex) was determined by identifying the location on the scalp where MEPs in biceps and triceps of the nonparetic arm could be optimally elicited by single-pulse TMS with the muscles at rest, the so-called “hotspot.” Resting motor threshold (RMT) for each muscle was determined by identifying the lowest stimulation intensity to elicit MEPs with peak-to-peak amplitude larger than 50 µV in 5 out of 10 trials. Since RMTs for nonparetic arm biceps and triceps were similar (54.6 ± 14.3% of maximum stimulator output for biceps, 53.90 ± 13.2% for triceps), the average of RMTs for both muscles was used to determine subsequent stimulation intensities for each subject.
Unable to elicit MEPs in the paretic arm at rest even using high stimulation intensities (Table 1), we instead asked participants to produce isometric contractions of the target muscle by either pulling up against a fixed strap placed just proximal to the wrist (elbow flexion) or pushing down into the chair’s arm rest (elbow extension) to activate biceps or triceps, respectively. Real-time visual feedback of the EMG activity, a low target level (around 30 µV), and frequent rest periods were used to ensure that background activation remained constant during coil localization and subsequent testing. Background activation of the target muscle was constantly monitored by the investigator throughout the experiment. Any trial in which there was little or no EMG activation in the target muscle was immediately discarded, and after a rest period, an additional trial was collected to replace it. Participants were relatively accurate in maintaining the target activation level (mean prestimulus EMG was 25.9 ± 3.7 µV for biceps and 23.6 ± 3.0 µV for triceps).
We used the highest stimulation intensity tolerated by the participant to identify the location that, at least intermittently, produced a facilitatory EMG response or, as was more frequently the case, a period of suppression of the ongoing EMG activation. In addition, a stereotactic neuro-navigation system (Brainsight, Rogue Research Inc, Montreal, Quebec, Canada) was used to confirm that the hotspot locations for both hemispheres were located on the precentral gyrus, the affected hemisphere hotspot was approximately in the mirror location of that for the unaffected hemisphere, and to ensure location accuracy of each TMS delivery throughout the experiment. Even with background muscle activation and high stimulation intensities, facilitatory responses (ie, MEPs) were rarely elicited and thus it was not possible to determine active motor threshold.
Unaffected Hemisphere Stimulation
During sustained muscle activation, stimulation of the hemisphere ipsilateral to the active muscle produces a transcallosal volley that elicits a period of inhibition of the ongoing EMG activity known as an “ipsilateral silent period” (iSP).27-29 To measure the iSP, EMG was recorded from the paretic arm during biceps or triceps isometric muscle contraction while TMS was delivered to the unaffected (ipsilateral) hemisphere M1 hotspot at 110%, 130%, and 150% of the nonparetic RMT (20 trials at each intensity for each muscle; Figure 1A).
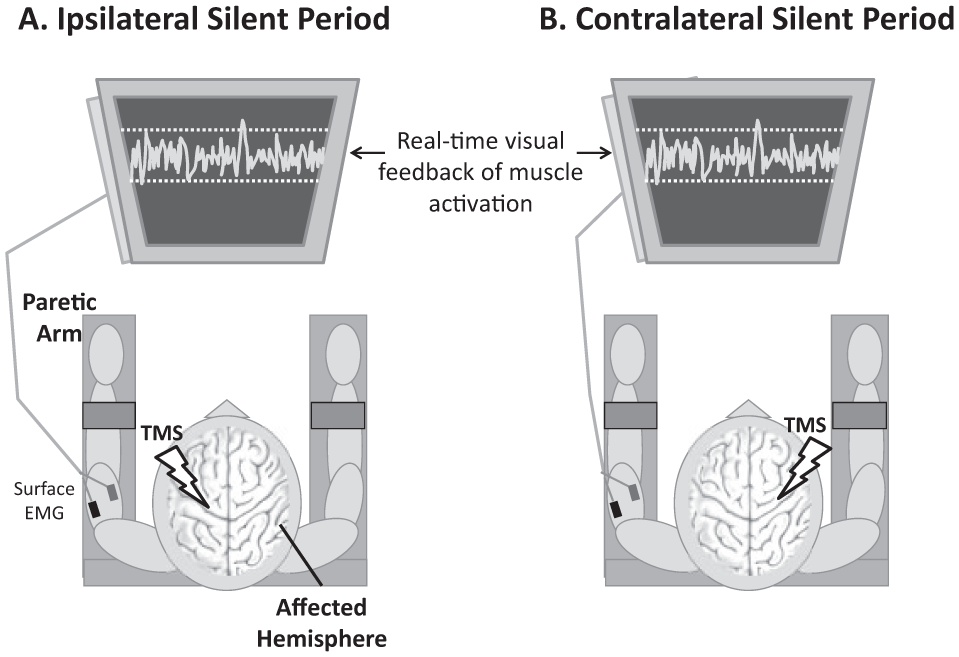
To elicit an ipsilateral silent period (A), patients activated their paretic arm by isometrically contracting the target muscle (biceps or triceps) while TMS was applied to the unaffected hemisphere primary motor cortex. The resulting period of decreased EMG activity reflects interhemispheric inhibition from the unaffected to the affected hemisphere. To elicit a contralateral silent period (B), the paretic arm was activated while TMS was applied to the affected hemisphere primary motor cortex, eliciting a period of decreased EMG activity that reflects intracortical inhibition.
Affected Hemisphere Stimulation
During voluntary muscle activation, stimulation of the contralateral M1 produces a period of reduced EMG activity known as the contralateral silent period (cSP), 30 which reflects GABAB-mediated intracortical inhibition. 11 To measure the cSP, EMG was recorded from the affected arm during biceps or triceps isometric muscle contraction while TMS was delivered to the affected (contralateral) hemisphere M1 hotspot at 80%, 90%, and 100% of maximum stimulator output (10 trials at each intensity for each muscle; Figure 1B).
Measurements of Motor Recovery
To characterize motor recovery we collected metrics of overall arm impairment, spasticity at the elbow, and performance of a proximal arm reaching task. The upper extremity portion of the Fugl-Meyer Assessment 31 and the Modified Ashworth Scale, 32 administered by a licensed physical therapist, served as measures of motor impairment/ability 33 and spasticity, respectively. To specifically measure proximal arm motor performance, patients performed a forward reaching task that required shoulder flexion with elbow extension, but no finger or wrist movement. Participants were seated at a table in a high-backed chair with crossed nonelastic straps across the torso to prevent compensatory movements of the trunk. In response to a randomly-timed visual “Go” cue, participants reached forward as quickly as possible to contact a large circular button on the table (the reaching “target”). The target was placed at 80% of each participant’s maximum forward reaching distance (21.4 ± 7.7 cm from the front edge of the table). After 10 familiarization trials, participants performed 20 test trials. Reaching response time was defined as the time elapsed from the appearance of the “Go” cue to button contact and was quantified as the median of the 20 trials for each individual. Note that this measure does not differentiate between reaction time and movement time and therefore represents the time required for not only movement execution but movement preparation as well.
Data Analysis
Physiological data were analyzed off-line using custom software programmed in MATLAB. Prestimulus mean EMG was calculated from the averaged rectified waveform and was defined as the mean value during a 100-ms time window (from 150 ms to 50 ms prior to TMS onset). For iSP, the onset of the silent period was defined as the point at which the averaged rectified EMG dropped and stayed below the prestimulus level for at least 5 ms. For cSP, the onset was defined as the time of stimulus delivery since, though they were rare in this population, any MEPs preceding the silent period could obscure cSP onset. Trials in which onsets were obscured by prolonged TMS artifact were discarded. Silent period offsets were defined as the point after the onset at which the averaged rectified EMG returned to and stayed at or above the prestimulus level for at least 5 ms. Silent period (iSP and cSP) measurements included percent inhibition, 27 calculated from the rectified and ensemble averaged waveform and expressed as a percentage of prestimulus EMG according to the following equation:
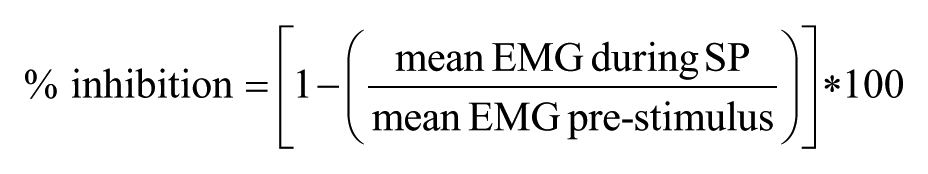
Calculated in this manner, a higher value indicates stronger inhibition. We also measured silent period duration34-36 and rate of occurrence, a unitless value between 0 and 1 defined as the number of trials in which a silent period was successfully elicited divided by the total number of trials (0 = silent period was not elicited in any of the trials; 1 = silent period elicited in all trials). To measure the rate of occurrence, each trial was analyzed individually to determine the presence or absence of a silent period. A trial was defined as positive for a silent period when it contained a poststimulus period lasting at least 25 ms during which the mean rectified EMG values remained below the mean rectified prestimulus EMG level. Each trial was also visually inspected to validate the result of the algorithm.
Dependent variables were analyzed using a 2 (Muscles) × 3 (Stimulation Intensities) ANOVA with repeated measures. In the case of significant interaction effects, paired
Results
We observed clear distinctions between inhibition targeting paretic arm flexor versus extensor muscles (biceps and triceps, respectively), as well as muscle-specific correlations between inhibition and paretic arm motor recovery.
Ipsilateral Silent Period (iSP)
iSPs were readily elicited in both muscles with unaffected hemisphere stimulation (ipsilateral to pre-activated paretic arm muscles). They were present in 71 ± 5% and 75 ± 5% of trials for biceps and triceps, respectively. However, these muscles showed marked differences in iSP % inhibition (Figure 2). There was a significant main effect of Muscle (
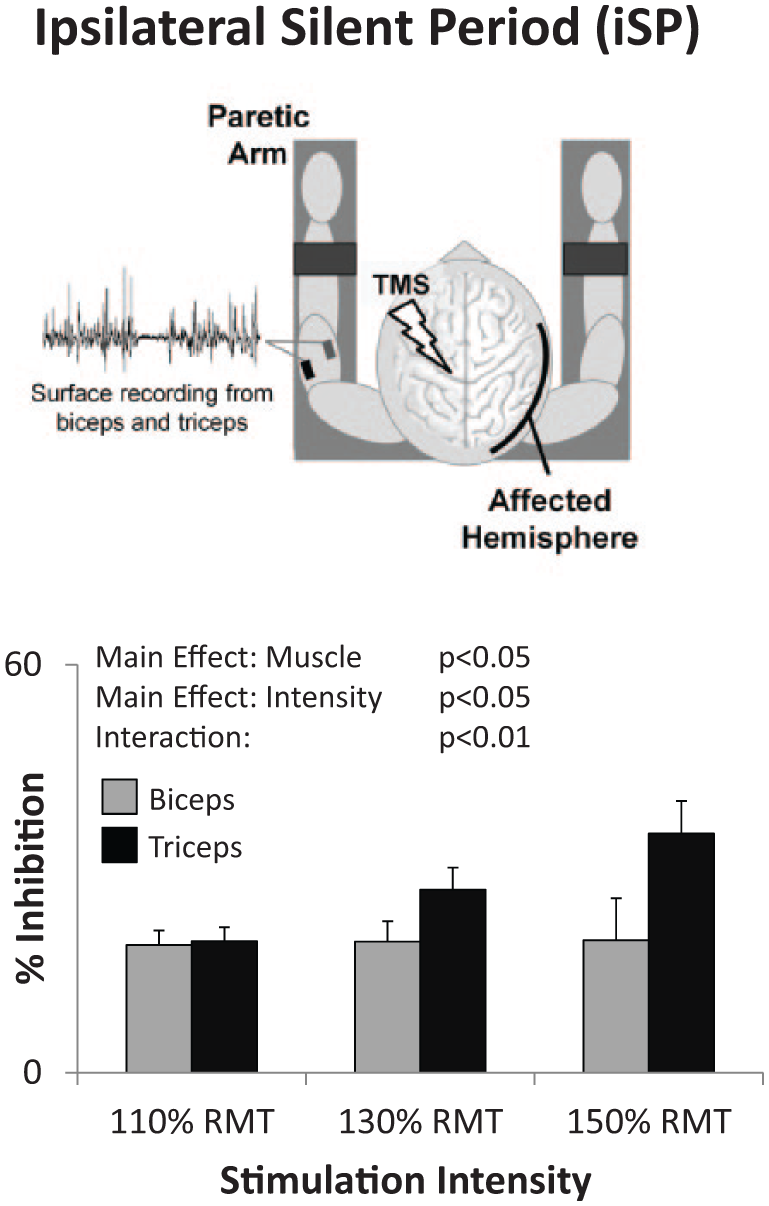
With stimulation of the unaffected hemisphere, the ipsilateral silent period (iSP) elicited in the paretic arm triceps muscle was greater than that in biceps (Main Effect of Muscle), increased with increasing stimulation intensity (Main Effect of Intensity), and increased more in triceps than biceps (Interaction Effect).
iSPs correlated negatively with Fugl-Meyer scores (ie, larger iSPs correlated with more severe impairment; biceps:
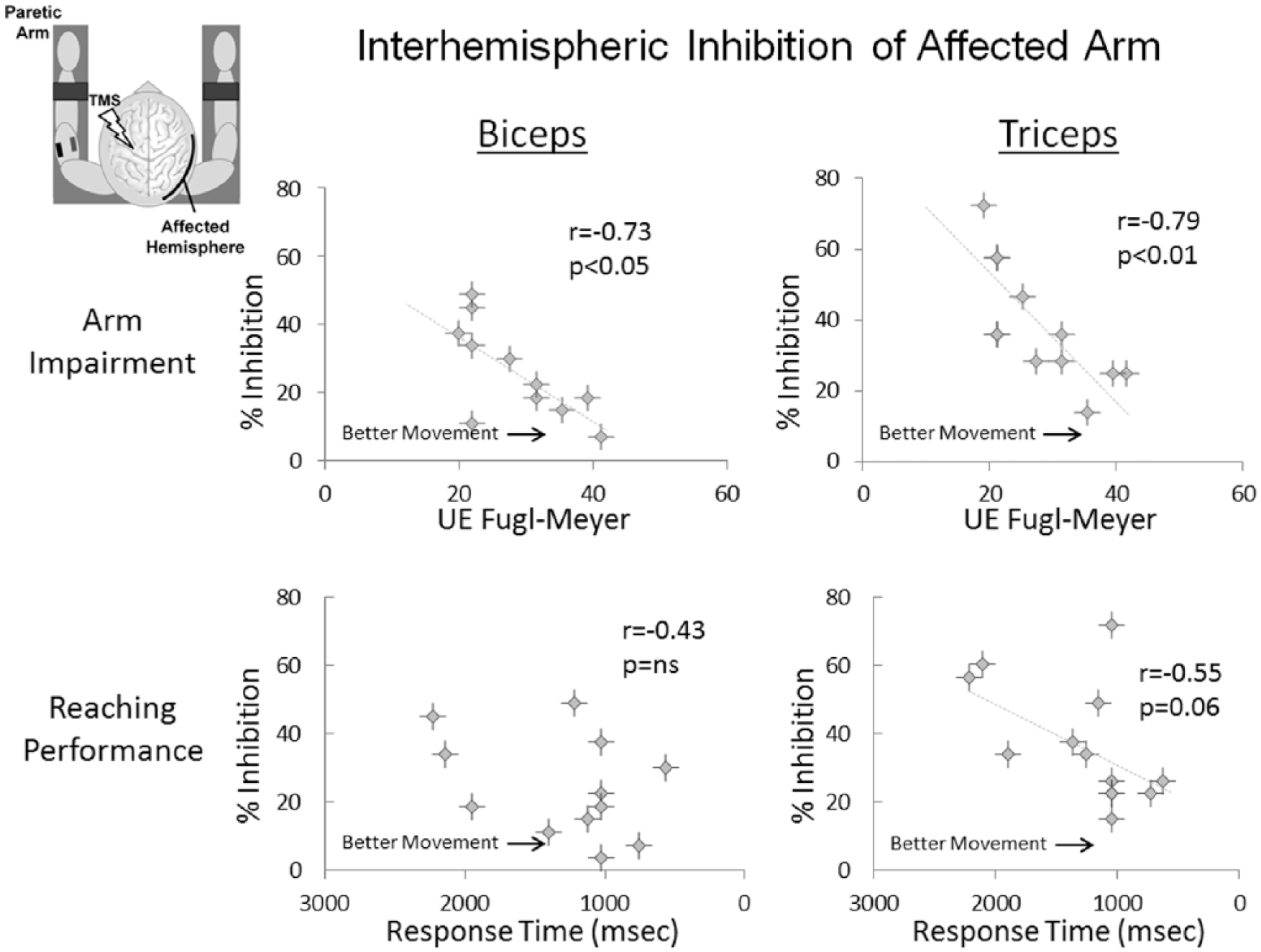
Associations between interhemispheric inhibition, measured by the ipsilateral silent period (iSP), and overall motor impairment of the arm (measured by the Upper Extremity Fugl-Meyer Assessment) and proximal arm motor performance (measured by reaching response time). Note that response time values are displayed longest to shortest (left to right), since longer response time indicates slower task completion. For both biceps and triceps, higher iSPs correlated with more severe motor impairment of the arm (top row). For triceps, higher iSP values also tended to be associated with longer (ie, slower) reaching response times (bottom right).
Contralateral Silent Period (cSP)
Like iSPs, cSPs were readily elicited in both biceps and triceps (83 ± 3% and 91 ± 3% of trials, respectively). There was a significant Main Effect of Muscle (
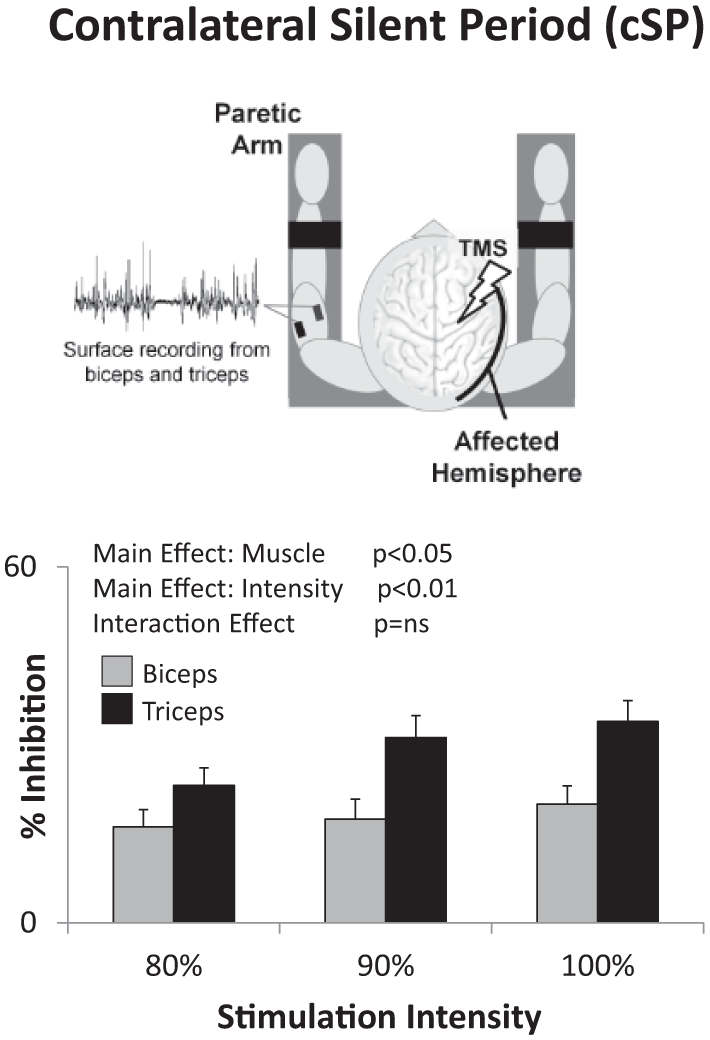
With transcranial magnetic stimulation (TMS) of the stroke-affected hemisphere, the contralateral silent period (cSP) was greater in triceps than biceps (Main Effect of Muscle) and increased with increasing stimulation intensity (Main Effect of Intensity).
In contrast to the negative correlation observed between iSP and motor ability (Figure 3), in the paretic biceps, cSPs correlated positively with Fugl-Meyer scores (ie, larger cSPs were associated with higher levels of motor ability;
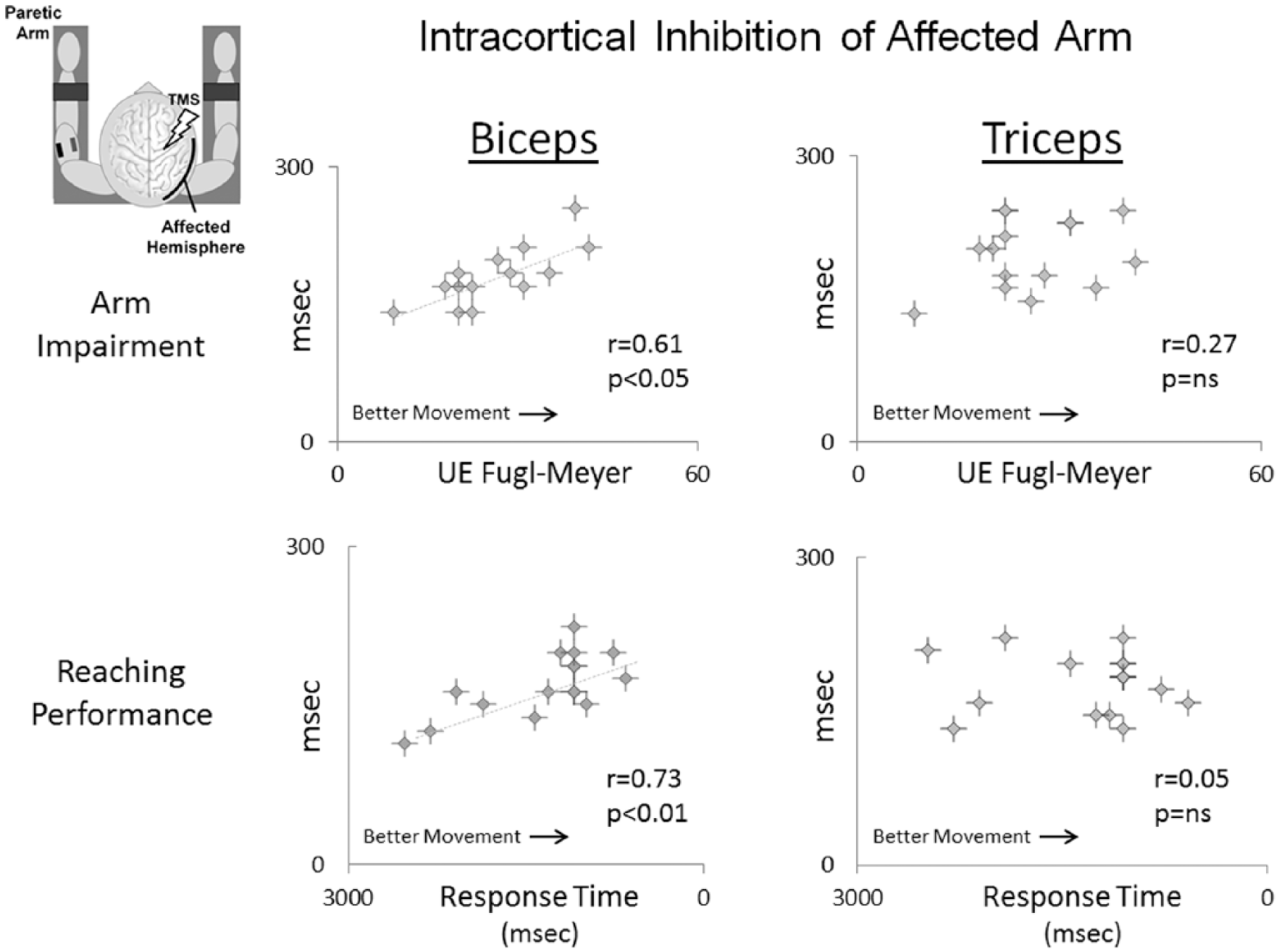
Associations between intracortical inhibition, measured by the contralateral silent period (cSP), and overall motor impairment of the arm (measured by the Upper Extremity Fugl-Meyer Assessment) and proximal arm motor performance (measured by reaching response time). Note that biceps cSP (left) was positively associated with Fugl-Meyer score and reaching response time, while triceps cSP (right) showed no correlation with either measure.
Discussion
In summary, iSP was stronger in the paretic triceps than biceps brachii and correlated negatively with motor recovery. Additionally, cSP correlated positively with motor recovery only in the paretic biceps but not triceps. Thus, in patients with severe stroke-related arm dysfunction, metrics of interhemispheric and intracortical inhibition differed across paretic upper arm muscles and in their relationship with motor recovery.
Ipsilateral Silent Period
The iSP produced when stimulating the unaffected hemisphere during isometric activation of the paretic arm reflects the strength of interhemispheric inhibition from the unaffected to the affected hemisphere and is thought to be transcallosally mediated28,38,39 and to reflect the activity of networks similar to those measured using a more traditional paired-pulse TMS technique. 29 In this patient population, interhemispheric inhibition targeting the paretic arm triceps muscle was stronger than that targeting the biceps, in spite of which the relationship between interhemispheric inhibition and motor recovery was similar for both muscles: stronger inhibition from unaffected to affected hemisphere M1 was associated with poorer motor recovery. This relationship is similar to that previously reported in hand muscles of more mildly impaired patients 23 and replicates our previous report. 40
Cortical Silent Period
cSPs were elicited by stimulating affected hemisphere M1 during paretic arm muscle activation. The cSP primarily reflects intracortical inhibitory processes,34,36,41-46 to which GABAB receptors contribute.11,47,48
We report stronger intracortical inhibition in paretic triceps than biceps. However, the relationship between intracortical inhibition and paretic arm motor recovery differed from that observed between interhemispheric inhibition and motor recovery. First, stronger intracortical inhibition in biceps was associated with better motor recovery, that is, higher Fugl-Meyer scores, faster reaching response times, and lower Modified Ashworth scores (unlike interhemispheric inhibition, in which stronger inhibition was associated with poorer motor recovery). Second, the relationship between intracortical inhibition and motor recovery differed between biceps and triceps (unlike interhemispheric inhibition, in which the correlation with motor recovery was similar for biceps and triceps). Intracortical inhibition in the paretic biceps correlated with motor recovery, while intracortical inhibition in the paretic triceps did not (Figure 5).
In general, cSPs elicited from hand muscles in well-recovered stroke patients are reported to be abnormally increased and to decrease with recovery.1,16-18,49 Our finding of a positive relationship between biceps cSP and motor ability (higher Fugl-Meyer scores), proximal arm motor performance (faster reaching response times), and spasticity (lower Modified Ashworth scores) appears to differ with these previously reported findings. However, in regard to the latter, even in patients with some recovery of hand movement there have been reports of a relationship between stronger intracortical inhibition in the affected hemisphere and lower Modified Ashworth scores.50-53 Interestingly, this association found with cSPs has been also reported when studying intracortical inhibition with paired-pulse TMS (a measure thought to reflect inhibition mediated by a different GABA subtype 2 ). The reasons for this relationship are not known but studies from both human and nonhuman primates may provide some clues. It is possible that intracortical inhibition of corticoreticular excitation, either via the corticoreticular tract 54 or via reticular collaterals of the corticospinal tract, which are most abundant in corticospinal neurons projecting to proximal arm muscles, 55 contribute to cSP. Alternatively, medial brainstem pathways such as those in the pontomedullary reticular formation (PMRF) project to proximal upper limb muscles56-58 and, following an extensive lesion of the corticospinal tract, projections from PMRF to upper limb flexor muscles have been shown to be preferentially strengthened. 59 The possibility that intracortical inhibition could interact with the preferential activation of flexors that may occur at the brainstem level poststroke (and the relationship this may have with spasticity and other aspects of arm impairment) is an important topic for future investigation.
Limitations
There are difficulties inherent to the study of patients without hand function, thus the relative absence of information on this population in the literature. First, because MEPs could not be elicited at rest, it was not possible to determine resting motor thresholds in the affected hemisphere in distal or proximal arm muscles of these patients (Table 1). For this reason we were confined to comparing iSP and cSP across muscle groups and their relation to motor impairment. This limitation also made it difficult to compare our results in stroke patients with complete hand paralysis with less impaired individuals, in whom RMTs are more easily determined. Additionally, most of the information on intracortical and interhemispheric inhibition in the literature has been obtained by testing distal hand muscles, not feasible in our patients with hand paralysis.
The reaching response time results must be interpreted with caution in that this measurement does not differentiate between reaction time and movement time, and thus represents not only movement execution but movement preparation time as well. The response times recorded were quite long (most were between 1000 and 2000 ms), possibly due to the difficulty these patients had in completing the reaching task, but without the ability to differentiate reaction and movement time, it is impossible to know. It is critical for future studies to clarify this issue. Another important limitation to the present study is that we do not yet have information on how these measurements may differ between muscles in age-matched healthy volunteers. Thus, we cannot conclude that the between-muscle difference we observed was abnormal, per se, only that it was in some cases correlated with motor function. Finally, the average age of our sample (59 ± 9 years) is considerably younger than the average age of the overall stroke population (70-71 years). 60 Thus, the generalizability of these results to the larger stroke population is an important topic for future investigation.
Conclusions
The results of this study indicate differences in poststroke cortical physiology between upper arm muscles in stroke patients with severe arm dysfunction and their relationship to the level of motor recovery attained. While interhemispheric inhibition relates to motor dysfunction, muscle-specific intracortical inhibition may contribute to motor recovery and lesser spasticity.
Footnotes
Acknowledgements
We are grateful to all of the study participants for volunteering their time. We also thank Drs Barbara Bregman, Stuart Baker, and Theresa Jones for helpful discussions on earlier versions of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NIH K01HD060886 (MHL); NIH/NINDS Competitive Intramural Postdoctoral Fellowship (MHL); National Capital Area Rehabilitation Research Network (NCARRN) pilot award to MHL via NICHD/NINDS HD050845 (Barbara S. Bregman, PI).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
