Abstract
Background. In stroke rehabilitation, considerable emphasis is placed on improving muscle strength with less focus on the speed of movement. Muscle power (product of force and velocity) is essential for balance and mobility but velocity of movement is impaired after stroke. Objective. The purpose of this efficacy study is to determine if a single session of fast functional movements can increase muscle activation and the speed of movement in participants with a subacute stroke. Methods. In total, 32 individuals poststroke and 32 age- and sex-matched controls performed a single session of 50 fast squats and steps. Electromyographic (EMG) activity was measured bilaterally in the rectus femoris (RF), biceps femoris (BF), tibialis anterior (TA), and soleus muscles. The average EMG area and the movement speed were calculated over 10 trials. The effect of exercise was determined as the change from the second set (Start) to the last set (End) of 10 trials. Results. The stroke group had significant increases in EMG area of the TA, BF, and RF during the squatting exercise. There was an increase in EMG area of the RF and BF when the paretic leg was stepping. Improvements in EMG area of the soleus and RF when the paretic leg was in stance accompanied increases in EMG area when the nonparetic leg was stepping. There was a trend for improved movement speed for both exercises. Conclusion. A single session of exercises emphasizing speed of movement can be used to improve muscle activation in persons with mild to moderately severe strokes.
Introduction
Progressive resistance exercises are used commonly to improve muscle strength after stroke.1-3 These studies do not distinguish between the neural and muscle hypertrophy components of the strength gains; neural changes that can occur very quickly whereas hypertrophy can take several weeks. 4 The assumption that strength gains in the paretic muscle will improve performance of functional activities, such as dynamic balance and mobility, has been challenged in recent reviews.5,6 For instance, a poor correlation has been found between muscle strength and dynamic balance after stroke, 7 whereas in older adults, dynamic balance was associated with velocity of movement. 8
Muscle power, the product of force and velocity, has been shown to be essential for balance and mobility 9 and is reduced after stroke. 10 Indeed, velocity of movement may be a more important component of power than force for functional movement. 11 Deficits in velocity are well documented after stroke.12-14 Thus the retraining of power through speed of movement and not only strength may be important for the rehabilitation of balance and mobility after stroke.
The use of fast movements as an intervention has received little attention as a rehabilitation strategy after stroke. Strength training did not translate to improved muscle power poststroke 3 but muscle power has been shown to increase in a group of older adults employing fast velocity movements when compared with a strength training group.15,16 Marigold et al 17 included agility exercises in an exercise regimen in chronic stroke patients and found improvements in reaction time and muscle activation in response to platform perturbations in the agility group.
Impairments in muscle activation during balance 18 and gait 19 are well known following stroke. Specific to fast functional movement, deficits in the muscle activation during fast squatting movements have been reported previously. 20 Whole body movements in standing, such as squatting and stepping, combine limb movement with the necessary postural responses to maintain stable stance. We have shown that, during the squat, the temporal coupling between the timing of the knee movement and the postural responses elicited by the movement was impaired. 20 Thus, the retraining of fast functional movements, such as squats, may be important for the coupling of balance with movement after stroke.
It is not known if the speed of movement and muscle activation can be improved with fast functional movement exercise in persons after stroke. Therefore, the aim of this efficacy study was to determine if a single session of exercises emphasizing speed of movement could improve muscle activation patterns and the speed of performance of the exercises. We hypothesized that a single session of fast functional exercises would increase the electromyographic (EMG) burst area and increase the speed of movement performance in persons after stroke.
Methods
Subjects
Subjects were included if they experienced hemiparesis as a result of a stroke, were recently discharged from an inpatient stroke rehabilitation unit, and were able to stand independently for one minute. Participants were excluded if they had other neurological, cardiac, musculoskeletal, or respiratory conditions that would interfere with the study protocol. A total of 32 sex- and age-matched controls were recruited through the University of Western Ontario and its Retirement Research Association. The study was approved by the Review Board for Health Sciences Research Involving Human Subjects at the University of Western Ontario. All participants gave written informed consent.
Clinical Measurements
To describe the level of sensorimotor recovery in persons after stroke, standing balance, motor recovery, and sensation were assessed; standing balance was also assessed in the control group. Standing balance was examined with the Berg Balance Scale. 21 The Berg Balance Scale has been identified as a tool for predicting recurrent falls in persons poststroke after completing a rehabilitation program, with less than 49 out of 56 being at risk. 22 The Chedoke McMaster Stroke Assessment Impairment Inventory was used to assess the motor recovery of the stroke group of the leg and foot. 23 The scores of the leg and the foot were combined for a maximal score of 14. Cutaneous sensation of the plantar aspect of the foot and ankle proprioception were evaluated according to standard clinical procedures. 24
Physiological Measurements
The EMG activity was recorded using single differential sensors (DE-2.3), 1.0 × 0.1 cm strips with a fixed interelectrode distance of 1 cm equipped with a preamplifier (gain, 1000 V/V; bandwidth, 20-450 Hz; Delsys Inc, Boston, MA). The sensors were placed bilaterally over the muscle bellies of the rectus femoris (RF), biceps femoris (BF), tibialis anterior (TA), and soleus (SOL) muscle. A ground electrode was positioned over the lateral malleolus.
Exercise Protocol
All subjects were fitted into a safety harness that did not provide any body weight support during the exercises. The exercise protocol comprised 50 fast squats and 50 steps. Rest breaks were given as needed. For the squats, subjects stood with each leg on 2 separate adjacent AMTI OR6-6-1000 force platforms (Advanced Mechanical Technology, Watertown, MA). Subjects performed a squat of approximately 30° of knee flexion following the instruction “unlock your knees and stop the downward movement as quickly as possible.” There was a 5-second gap between squats. For the steps, subjects stood behind the force platforms and were instructed to step onto one of the force platforms. The location of the starting position varied according to the length of step of the individual. Subjects were instructed to “lean forward until you feel you are losing your balance and take a single step.” Subjects returned to the start position by pushing back from the force platform with the stepping leg. Subjects completed 5 steps on one leg before alternating to the opposite leg and this was continued until 50 steps with each leg were completed.
Data Acquisition
The EMG signals were recorded using Myomonitor IV Wireless Transmission & Datalogging System with EMGWorks software (Delsys Inc, Boston, MA) at a 2000 Hz sampling rate. A linear uniaxial accelerometer (Colbourn Instruments T45-10) was attached to each knee superior to the patella since this is the best configuration to detect the initial knee unlocking 25 and has been used successfully to detect gait phases in healthy subjects and individuals with hemiparesis with few errors. 26 Simultaneous recording of force and moment signals from the force platforms (6 from each platform) and the 2 accelerometer signals was conducted using a separate 16-bit acquisition system (Power 1401 with Spike2 software, Cambridge Electronic Design, Cambridge, UK) at a sampling rate of 500 Hz (force platform) and 1000 Hz (accelerometer). The recordings on both acquisition systems were synchronized by a Delsys trigger module that provided a TTL signal at the beginning of the EMG recording. The data were analyzed offline with Spike2.
Data Analyses
The onset of knee movement was detected when the slope of the acceleration curve was greater than 0. The speed of the squat was obtained by combining the vertical ground reaction forces (Fz) of the 2 force platforms and the resultant signal was used to calculate the vertical acceleration (a) of the center of mass with the following formula, a = 9.81 × (Fz − W)/W, where W is the subject’s body weight. 27 The vertical velocity of the center of mass was obtained by the time integration of the acceleration curve. The speed of the steps was determined as the peak positive amplitude from the knee accelerometer; this measured the initial forward acceleration of the stepping leg.
To examine if the temporal coupling between knee movement and the postural responses elicited by the squatting movement is adaptable with exercise, the difference in the timing of the peak knee acceleration and timing of the peak center of pressure (COP) excursion in the anterior/posterior (AP) direction (acceleration-COP) was calculated according to Gray et al. 20 The arrows in Figure 1 illustrate the temporal coupling in the timing of the peak knee acceleration with the peak posterior COP excursion (acceleration phase) and the timing of the peak knee deceleration with the peak anterior COP excursion (deceleration phase).
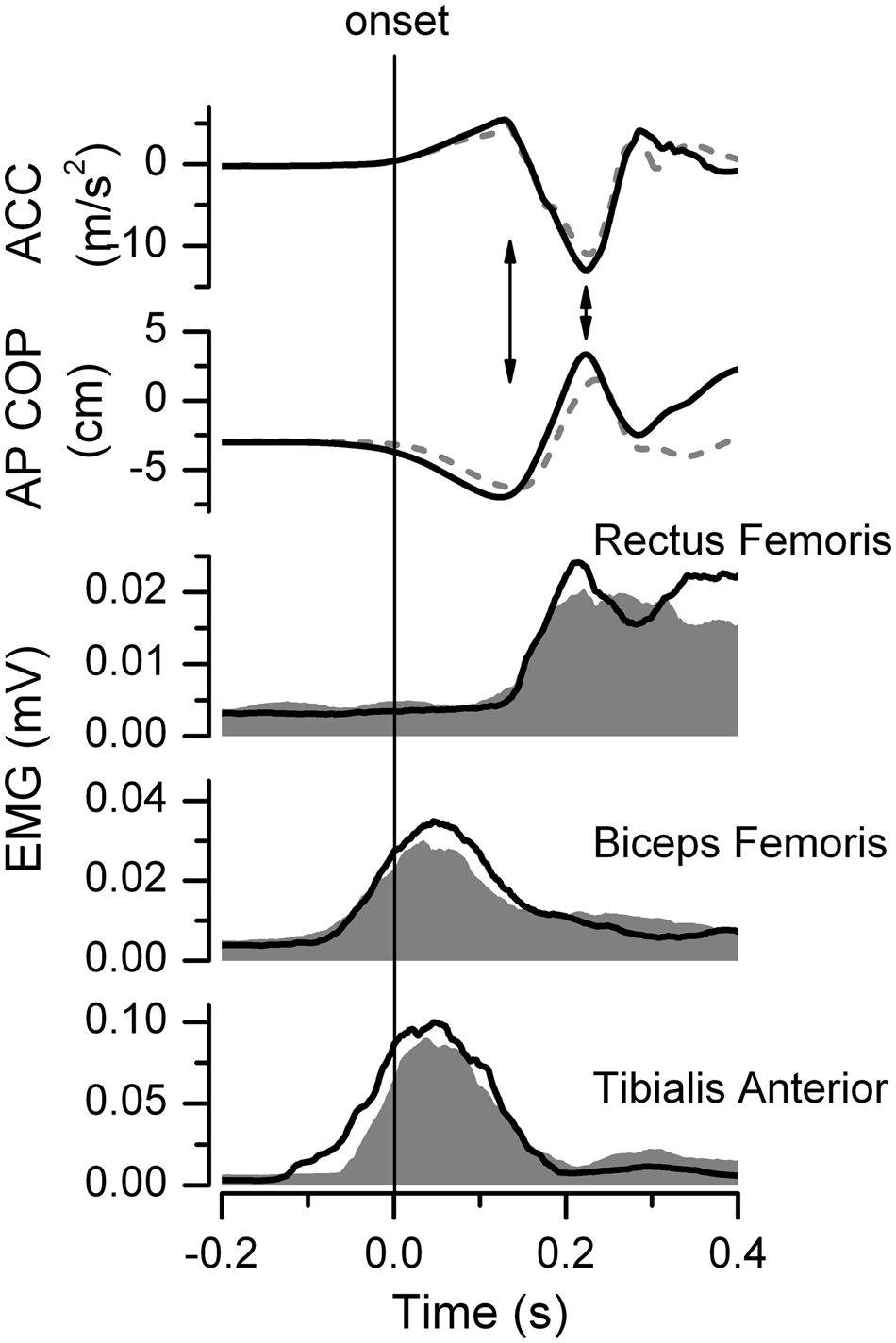
Squat: Knee accelerations (ACC), center of pressure (COP) in anterior/posterior (AP) direction, and electromyographic (EMG) signals (rectus femoris, biceps femoris, tibialis anterior) for representative subject from the stroke group
All EMG signals were rectified before averaging. For the squats, the EMG for each trial was aligned to the onset of knee movement and the 50 trials (5 sets of 10 trials) were averaged for 2 seconds starting 1 second before the onset of movement. Because of the high intersubject variability in the duration of the step, the steps were time normalized 28 with the onset of knee movement of the stepping leg being 0% and the foot contact detected by the initial deflection of Fz being 100%. The stepping and stance legs were averaged separately in 5 sets of 10 trials. The second set of 10 trials for the steps and squats, which will be referred to as “Start” and the last set, referred to as the “End,” were used for data analysis. The first 10 trials were considered as practice for both groups for both the squats and the steps.
A muscle burst was identified when the EMG activity was greater than the mean + 2 standard deviations of the baseline activity, which was measured for 100 ms or 10% of step duration during quiet stance prior to the squat or step, respectively. The EMG burst area in the squats was defined as the area 75 ms before and after the peak EMG burst amplitude. The EMG burst area of the step was defined as the area 15% of step duration before and after the peak EMG burst amplitude. The EMG area was normalized by dividing by the baseline area for the same duration during quiet stance preceding the squat or the step.
Statistical Analyses
For all statistical analyses, the level of significance was set at an alpha value of 0.05. Statistical analysis was performed using SPSS for Windows v17.0 (SPSS, an IBM Company, Chicago, IL). Nonparametric statistics were used because the data did not fulfill the criterion of normality. No differences between legs were found in any measured variables in the control group so the right and left legs were averaged. The between-leg effects (paretic, nonparetic, control) were compared using a Kruskal–Wallis test for both the squats and steps at the Start for the EMG area, and the temporal coupling in acceleration and deceleration phases for the squats, and the peak knee acceleration amplitude for the steps. A Mann–Whitney U was used to compare the center of mass velocity between the control and stroke groups in the squat exercises. When a significant between-leg effect occurred, a Mann–Whitney U was used to determine the leg difference. The effect of exercise (Start, End) was examined for each leg separately using the Wilcoxon signed-rank test. Bonferroni corrections were performed to correct for multiple comparisons with an adjusted P value (P ≤ .025). All data are reported as mean ± standard deviation. Percentages were calculated as either [(stroke value/control value) − 1] × 100% or (End − Start)/Start × 100%.
Results
A total of 21 men and 11 women with hemiparesis participated in the study; 4 had a hemorrhagic stroke and 28 had an ischemic stroke. In the stroke group, 10 subjects had right hemiparesis and 22 had left hemiparesis. The mean age was 55.6 ± 13.5 years and the mean time poststroke was 11.3 ± 4.1 weeks, with a hospital mean length of stay of 52.4 ± 20.7 days (range = 18-89 days). The mean Berg Balance Scale score was 52.1 ± 4.5 (range = 40-56) out of 56 and the Chedoke-McMaster Stroke Assessment was 10.2 ± 3.1 (range = 6-14) out of 14. The sensation was intact in all individuals in the stroke group. The healthy group had a mean age of 56.1 ± 14.0 years and a mean Berg Balance Scale score of 55.9 ± 0.2.
Squats
The center of mass velocity was 35.0% lower in the stroke group (Start: 37.1 ± 10.4 cm/s) compared with the control group (Start: 57.1 ± 10.9 cm/s; P < .001). Over the course of the exercise, the center of mass velocity in the control group did not change (End: 55.8 ± 12.2 cm/s, P = .51), but in the stroke group there was a tendency for an increase of 7.0% (End: 39.6 ± 14.5 cm/s, P = .12).
At the Start, the EMG burst area of the paretic TA was 21.9% lower than in controls (P = .002, Table 1). The soleus data are not presented for the squats because a SOL burst was observed in less than one-third of all subjects (10 control subjects and 7 stroke subjects). Figure 1 depicts the knee acceleration, COP, and EMG bursts from the paretic leg in a representative subject in the stroke group. Comparing the Start with the End, there were increases in EMG area in the paretic TA (28.0%, P = .005), the paretic BF (27.3%, P = .001), and the paretic RF (23.6%, P = .002). There were no significant changes in the EMG area during the squats in the control group or the nonparetic leg.
Electromyography Area: Squats a
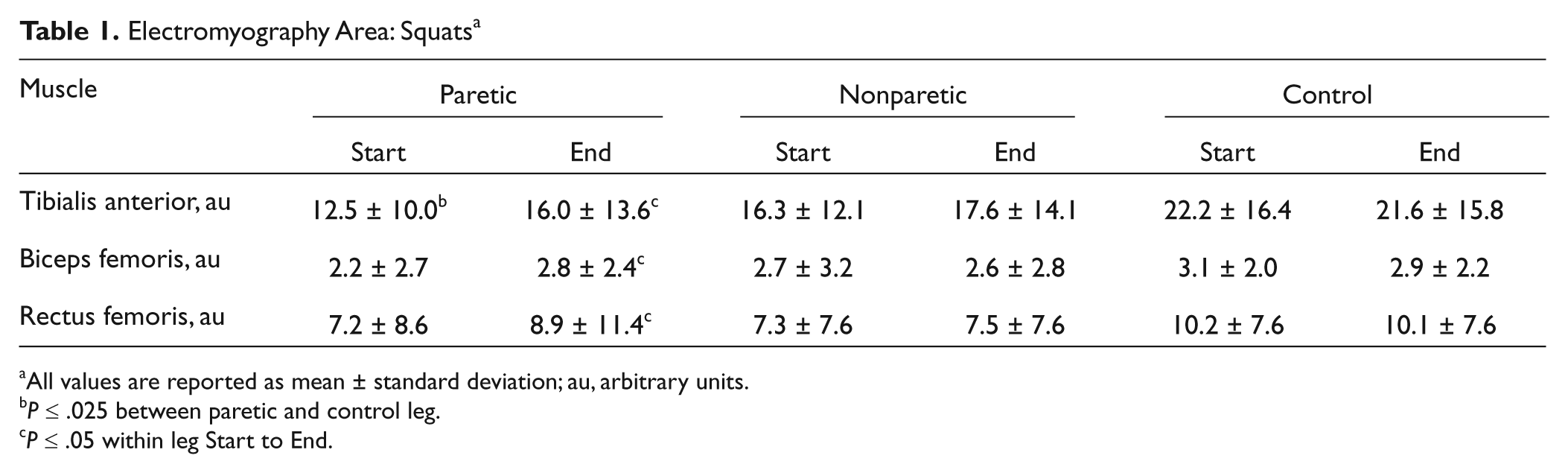
All values are reported as mean ± standard deviation; au, arbitrary units.
P ≤ .025 between paretic and control leg.
P ≤ .05 within leg Start to End.
The temporal coupling was impaired in the paretic leg, with larger differences in the timing of knee movement with COP excursion in both the acceleration phase (P < .001) and the deceleration phase (P = .01) than the control group (Figure 2). Improvements in the temporal coupling in the acceleration phase occurred at the End of the squats, with a decrease of 36.3 ms (P < .001) in the paretic leg, which brought the value into the normal range. The temporal coupling in the deceleration phase improved by 7.8 ms (P = .025) in the paretic leg, whereas no significant changes were found in nonparetic leg or controls.
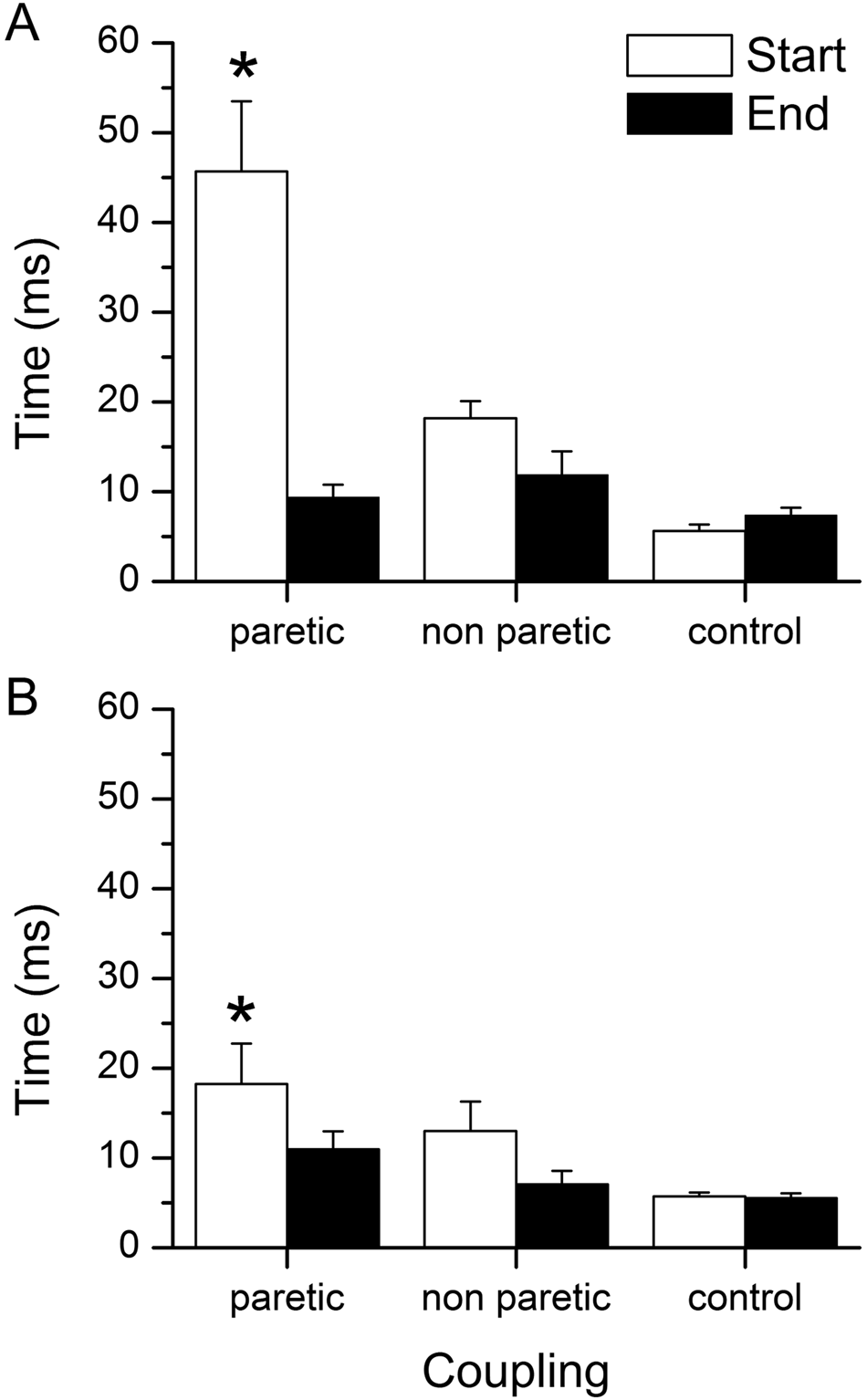
The temporal coupling of peak knee movement and center of pressure (COP) excursion in acceleration (panel A) and deceleration (panel B) for the paretic, nonparetic, and control legs at the Start of the squats (white bar) and the End of the squats (black bar)
Steps: Stepping Leg
At the Start, the peak knee acceleration amplitude was lower in both the paretic leg, 27.9% (P = .01), and the nonparetic leg, 26.2% (P = .02), than the control group. With exercise, the peak acceleration amplitude increased by 13.6% (P = .11) in the paretic leg and 13.3% (P = .02) in the nonparetic leg, although only the increase in the nonparetic leg was statistically significant.
In the stepping leg, two bursts were observed in the BF, the first BF burst (1st BF) started before the initial movement of the knee and the second burst occurred around foot contact (2nd BF). At the Start, the EMG area was less than the control leg in the paretic SOL (35.5%, P = .01), paretic 1st BF (56.9%, P < .001), paretic 2nd BF (58.0%, P < .001), paretic RF (59.6%, P = .01), and nonparetic 2nd BF (55.7%, P < .001; Table 2).
Electromyography Area and Knee Acceleration in Stepping Leg a
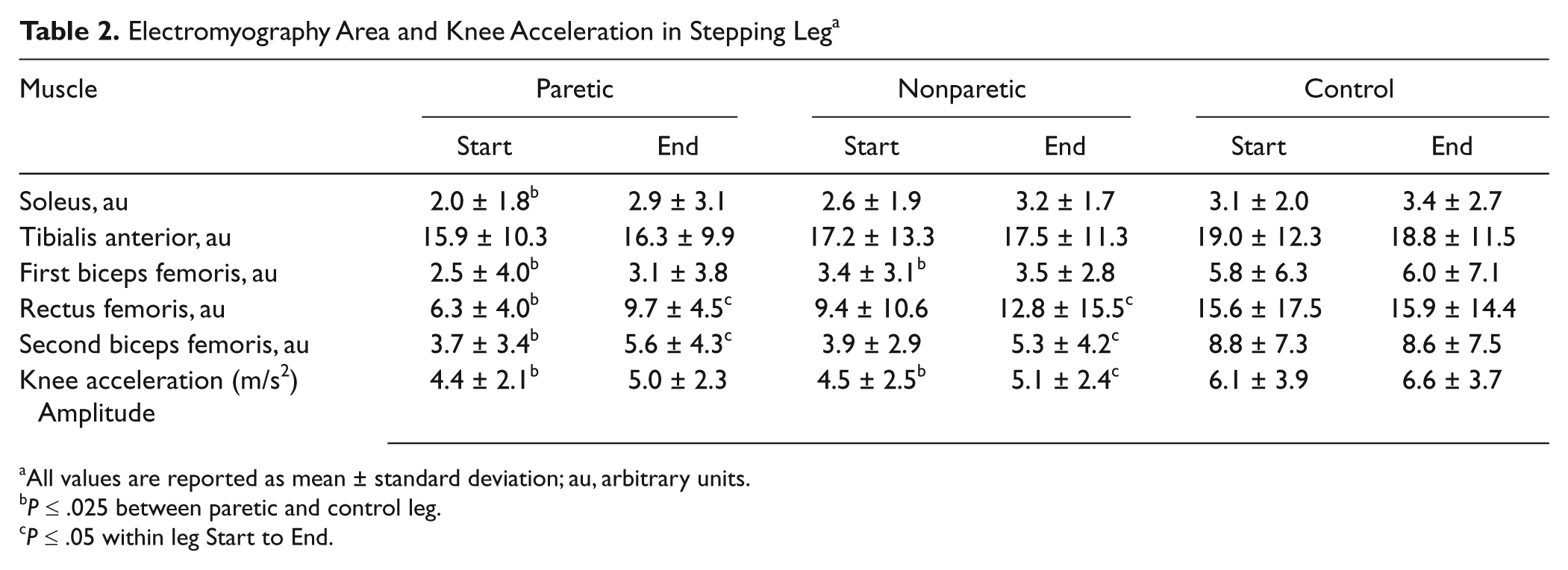
All values are reported as mean ± standard deviation; au, arbitrary units.
P ≤ .025 between paretic and control leg.
P ≤ .05 within leg Start to End.
With exercise, an increase in the EMG area was evident in the paretic 2nd BF (51.4%, P = .001), paretic RF (54.0%, P < .001), nonparetic RF (36.2%, P = .02), and nonparetic 2nd BF (35.9%, P = .006) at the End compared with the Start (Figure 3A and C). There were no significant changes in the EMG area or knee acceleration in the stepping leg in the control group.
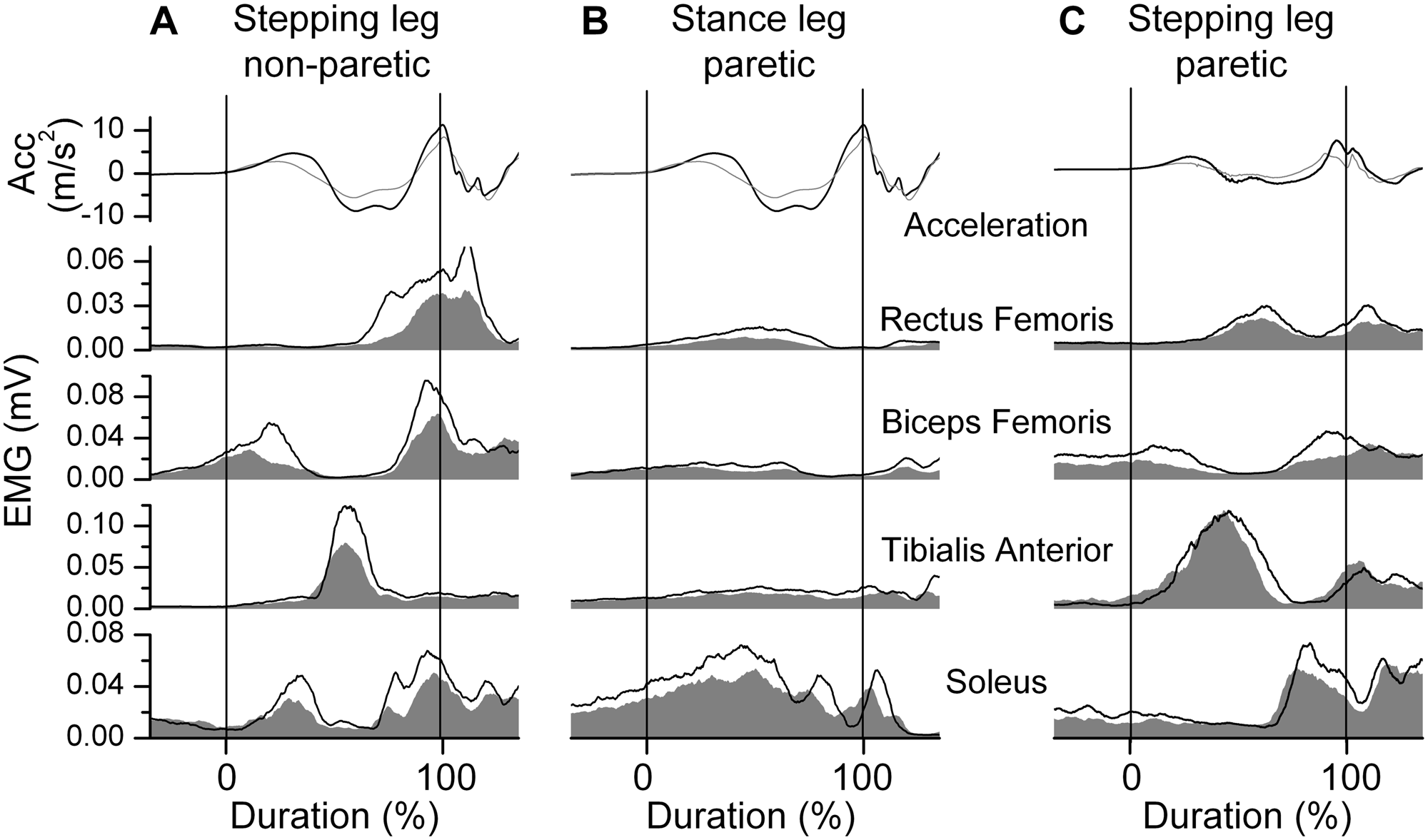
Stepping: Knee acceleration (ACC) and electromyographic (EMG) signals for 2 representative subjects from the stroke group
Steps: Stance Leg
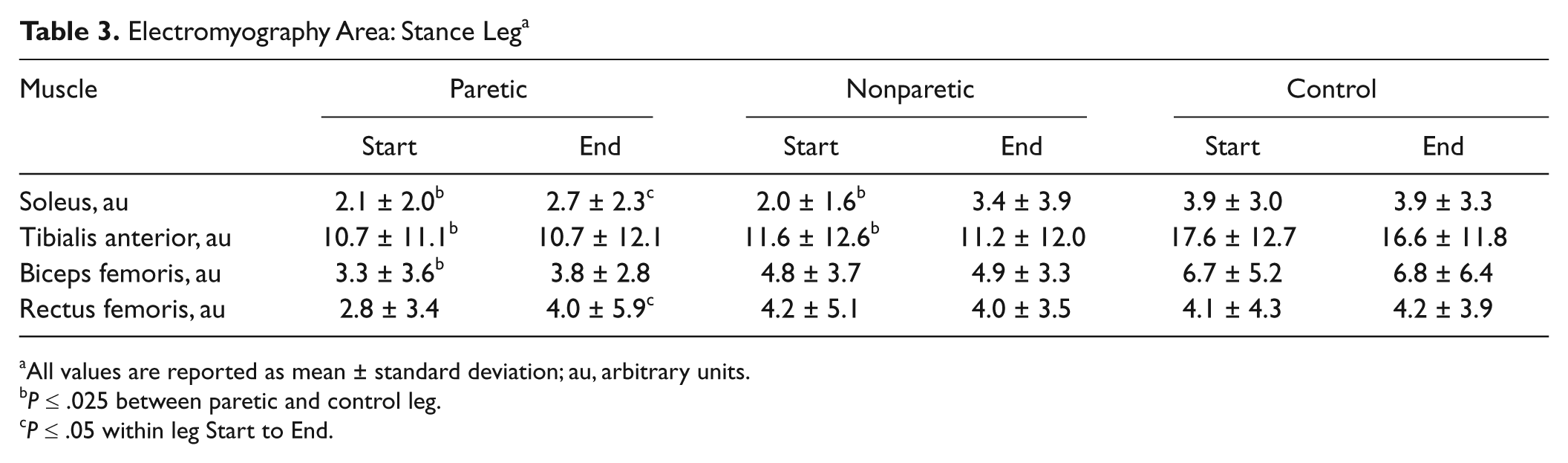
At the Start, the stroke group had lower EMG areas in the paretic SOL (46.1%, P = .008), paretic TA (39.2%, P = .008), paretic BF (50.7%, P < .001), nonparetic SOL (48.7%, P = .008), and nonparetic TA (34.1%, P = .02) compared with the control leg (Table 3).
Electromyography Area: Stance Leg a
All values are reported as mean ± standard deviation; au, arbitrary units.
P ≤ .025 between paretic and control leg.
P ≤ .05 within leg Start to End.
With exercise, no changes in EMG parameters in the stance leg were found in the control group. However, the EMG area increased in the paretic SOL (28.6%, P = .02) and the paretic RF (42.9%, P = .025; Figure 3B).
Discussion
The main finding of this study is that exercise comprising fast functional movements improves the muscle activation after stroke. The improvement in muscle activation was accompanied by an improved temporal coupling of the movement with the postural responses during a squat and a tendency for increased speed of performance of the squats and steps.
The functional movements were performed more slowly in the stroke group than in the control group. There was a tendency for the exercise to elicit an increase in the speed of movement but a significant change in performance may require more than a single session of exercise. Importantly, the current study demonstrated the ability for the EMG area to increase with a single session of exercise.
Deficits in EMG area were not observed in all paretic muscles. Previous research has shown that the type of contraction influences the degree of deficits demonstrated in the paretic muscles, with greater preservation of force during eccentric contractions and greater deficits during concentric contractions.14,29 This may explain why deficits in the paretic TA were observed in the squats because the TA acts concentrically to initiate the squat. RF did not differ from control group, possibly because RF controls the end position of the squat eccentrically.
In the squats, the paretic EMG areas reached values similar to the control group at the End except for the TA muscle. In addition, the temporal coupling of the paretic leg movements with the COP excursion approached control values. The improved temporal coupling may have resulted from the larger EMG activation, which advanced the timing of the peak movement acceleration. It is possible that the bilateral nature of the squat was a factor mediating the improvement because bilateral tasks, albeit examined in the upper extremity, demonstrated an increase in peak velocity and improved symmetry compared with unilateral training.30,31
During the steps, similar improvements were observed in both the paretic and nonparetic leg, with increases in the EMG area of the RF and second BF bursts suggesting better control of the knee and hip for weight acceptance. Importantly, the increased speed and EMG area of the nonparetic stepping leg was accompanied by an increase in the SOL and the RF EMG areas in the paretic stance leg, thereby providing an increase in the extensor muscle activation of the paretic stance leg to support the nonparetic leg when stepping. Increased speed of the nonparetic step has great functional relevance as persons after stroke rely on stepping strategies to maintain balance after standing perturbations. 32
Although speculative, the mechanisms underlying the changes in EMG area and temporal coupling may be attributed to either an increased excitability at the alpha motor neuron level 33 or an increase in cortical excitability. 34 Perez et al 34 examined healthy individuals performing a motor learning task comprising voluntary ankle movements for 32 minutes and observed an increased cortical excitability in the motor cortex area of the TA. The improvements in temporal coupling may be attributed to changes at the cortical level because the coordination of the timing of the COP displacement with knee movement is unlikely to occur at the level of the motor neuron.
This feasibility study demonstrated that a single session of exercise comprising fast functional movements can evoke changes in muscle activation in the subacute phase in persons with mild to moderately severe strokes. These changes were accompanied by improved temporal coupling of the movement and postural control elements of the squat and an increased speed of stepping in the nonparetic leg. It is not known if comparable changes can be evoked in persons with more severe strokes. Exercise strategies other than squats, such as fast seated bilateral leg exercise as in cycling or functional strength training of the legs, may also be feasible ways to evoke greater muscle activation in persons poststroke.35,36 Although the appropriate dosage of a fast functional exercise program to instill retention remains to be determined, the results of this study bode well for future randomized controlled trials to examine the effectiveness of training speed of movement on balance and mobility following mild to moderately severe stroke.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Heart and Stroke Foundation of Ontario grant awarded to SJG.
