Abstract
Background. Arm paresis remains a major impairment after stroke despite the best conventional rehabilitation. Randomized, controlled trials of intensive exercise programs have demonstrated improvements in arm function for patients with chronic stroke. However, the gains achieved have been relatively modest for the large investments in patient and therapist time. Objective. To evaluate the safety, acceptance, adherence, and effectiveness of a community-based exercise program for upper limb paresis in patients with chronic stroke and the effects of telerehabilitation monitoring in kiosks distributed through the community. Methods. Longitudinal cohort with geographic control group. The experimental group received devices needed for a home exercise program based on the Carr and Shepherd “Motor Learning Program” and were instructed to practice the exercises at least twice a week at the kiosk and at least 3 more days a week at home. The control group received usual care. Results. Compared with the control group, patients in the experimental group demonstrated significant gains in arm function as measured by the Wolf Motor Function Test, 9-Hole Peg Test, Motricity Index, and Nottingham Extended Activities of Daily Living Questionnaire. The intervention received high satisfaction ratings and produced no adverse events. Only 30% of the subjects attended kiosks regularly. Outcomes for this group did not differ significantly from those who only practiced at home. Conclusions. Home- and community-based exercise for arm paresis is safe and effective. Telerehabilitation interventions will need additional enhancements to improve effectiveness. The optimal upper extremity exercise prescription poststroke remains to be established.
Introduction
Less than one third of hemiparetic stroke patients recover full function of their paretic upper extremities, and a substantial proportion remain significantly impaired. 1 This clinical outcome does not appear to have changed substantially over the past decade.2,3 However, a number of studies have now demonstrated that upper extremity function can be improved in chronic stroke patients who have some residual motor control in their paretic extremities. The rationale and precise content of the rehabilitation programs have differed, from constraint-induced movement4,5 to bilateral arm exercise, 6 to robotic interventions, 7 to virtual reality8,9 among others. The gains demonstrated are modest but persistent, are comparable across different programs, and can occur more than 1 year after stroke.10-15 The resources necessary to implement these programs are substantial, and widespread adoption into routine clinical practice has not occurred.
Telerehabilitation can, in principle, deliver therapeutic interventions in home and community settings safely, for a longer duration and more cost-effectively than they can be provided in the inpatient rehabilitation setting. Telemonitoring has decreased recurrent hospitalizations in heart failure patients 16 and been effective in promoting exercise adherence in home-based aerobic exercise programs for cardiac rehabilitation.17,18 Piron et al 19 demonstrated modest gains equivalent to traditional therapy in a small-scale randomized controlled trial of telerehabilitation for chronic mild to moderate upper extremity paresis due to stroke. A small randomized controlled trial of a comprehensive telerehabilitation intervention for chronic lower extremity paresis 20 also suggests that such interventions can produce sustained improvements in function.
In this context, the Habilis system was developed under a grant from the European Union (ICT-PSP224985 Programme, coordinated by Signo Motus s.r.l.) for the CLEAR project “Clinical Leading Environment for the Assessment and Validation of Rehabilitation Protocols for Home Care,” to deploy telerehabilitation services delivered in home and community settings through a general purpose, low-cost, Web-based platform. 21 Habilis was tested in 4 European countries with interventions and related clinical protocols for patients with neurological, orthopedic, or pulmonary conditions or chronic pain. 22
As one intervention in the CLEAR project, we tested the safety and effectiveness of a home-based exercise program for chronic upper extremity paresis secondary to stroke, supplemented by the Habilis telerehabilitation system in Empoli and in Siena, Tuscany. This trial with a geographic no-treatment control was initiated after a pilot program with 15 subjects demonstrated patient acceptance, no adverse events, and suggested possible efficacy. 23 Our primary hypothesis was that this home-based exercise intervention would be safe and effective, demonstrating superior clinical and quality of life outcomes for the intervention group when compared with the control group. We further hypothesized that use of the Habilis telerehabilitation system would reinforce adherence to the exercise program, and that highly adherent subjects would have significantly greater improvements in clinical outcomes than others.
Methods
This is an effectiveness study with a geographic control group. Subjects with arm paresis caused by stroke were recruited by the rehabilitation services team in Local Health Authorities of Empoli and Siena, Tuscany. Subjects received detailed information from the physicians and physiotherapists coordinating the study as part of the process of obtaining informed consent. The study was powered on the preliminary data collected in the pilot study for the Motricity Index and the Wolf Motor Function Test (WMFT). Using these data, we determined that to achieve 90% power on the Motricity Index and 80% power on the WMFT required 40 subjects in the control group and 140 subjects in the experimental group. For logistical and policy reasons, this experimental design was preferable to equal-sized groups.
Inclusion Criteria
Participants were included in the study if their stroke onset was at least 3 months prior, they were at least 40 years old, maintained at least minimal residual function of the paretic limb, that is, ≥3 as measured using the Enjalbert Scale, 24 and had permission from their primary care provider. Subjects were excluded if they presented conditions that prevented understanding and/or execution of rehabilitation tasks, suffered from symptomatic congestive heart failure, unstable angina, pulmonary disease that required oxygen therapy, had recent myocardial infarction or hospitalization, had pain which interfered with exercise, or had poorly controlled blood pressure.
Exercise Intervention
Subjects in the intervention group received a task-oriented, individually tailored, intensive, 3-month rehabilitation program which integrated hospital outpatient rehabilitation with home and “kiosk” practice. The treatment program was based on the Carr and Shepherd “Motor Learning Program” 25 because of its emphasis on patient practice at home with therapist “coaches.” Before initiating the home- and community-based program, hospital physiotherapists instructed the subjects on how to practice the rehabilitation exercises and use the Habilis platform. This generally required three 2-hour sessions. When able to perform the home tasks and use the Habilis platform adequately, subjects were instructed to repeat the learned tasks at least twice a week in the kiosk and 3 more days at home for a minimum of 5 times per week. The exercises used very simple rehabilitation devices, low cost and easily available in the community (different shape and size objects to manipulate, puzzles, printed paths to follow with pens of different shape and size, etc). The appropriate training material for home practice was given to each subject in a “training suitcase” by the hospital physiotherapist. A caregiver was also instructed, if necessary, to assist the subject in his or her home exercise sessions.
For each subject, the hospital physiotherapists individualized the exercise prescription for exercise type, intensity (number of repetitions) and frequency (number of sessions per week) based on characteristics of the impairments observed in baseline and follow-up assessments. At home, subjects were expected to practice their exercise program using the devices provided in the rehabilitation suitcase in conjunction with a Home Program Sheet that specified their individualized exercises prescription. Each practice session was recorded by the subject/caregiver in a logbook.
During the course of the study, subjects were progressed through exercises that encompassed the essential components of reaching, grasping, holding and manipulation with increasing level of difficulty. The level of difficulty was chosen so as to make the tasks “challenging, but not impossible.” 25 This was done at a monthly clinic visit if subjects did not attend the kiosk. Telephone calls were used to handle complaints and questions, but not to review and adjust the exercise prescription.
Telerehabilitation
Since the availability of affordable broadband connections was quite low, the kiosks were intended to provide patients an environment located near home to be easily accessible, where subjects could perform continued intensive practice and receive the necessary supervision and assessment. Eight kiosks were located in social centers or voluntary associations in local municipalities. Each kiosk was equipped with at least 2 client stations, including computers with touch screens and webcams. The stations had the same training material used at home, as well as other opportunities for computer based exercises implemented on the Habilis platform. Each subject had to log on by using his or her personal username and password to perform their individually tailored exercise program.
In the kiosks, the exercises were assigned by the physiotherapist from the station located in the hospital via the Habilis platform and sent through the web. To make it easier for the patient to understand the assigned training program, all exercises were video-recorded by the physiotherapist. From the hospital station, the physiotherapist could observe patient performance by videoconference, give advice for improving practice at regular intervals (once a week), and reassess the patients. Alternatively, patients could record his or her practice to be later reviewed by the physiotherapist. Finally, patients could request immediate advice from the physiotherapist via the Habilis platform. If the treatment plan needed to be revised or if adverse clinical events were present, the physiotherapist asked patients to return to the hospital to review the treatment program and/or a visit a physician. Kiosks were supervised by volunteers who ensured opening/closing of the kiosk, cleaning the environment, and maintaining the equipment in proper order. A caregiver could assist the patient in kiosk activities if needed.
Control Treatment
Patients with stroke and residual upper limb impairments who were residents in municipalities not served by kiosks were recruited to the control group. Subjects were included in the control group if the inclusion/exclusion criteria were met and they gave informed consent. Usual care consisted of visits to their general practitioners as needed, and sessions of outpatient physical therapy delivered on demand. Two control subjects received 10 sessions each of outpatient rehabilitation during the study period. The other control subjects received no therapies during the time of the study.
Measurement
Safety was measured by adverse events. All adverse events reported spontaneously by the subject, or observed by the investigator or staff, were reported to the local ethical committee. For reporting purposes, adverse events were defined as any undesirable experience occurring to a subject during the clinical trial, whether or not considered related to the intervention protocol. However, in assessing safety of the intervention, only study-related adverse events were considered.
All tests were conducted by the same trained physiotherapists. To assess baseline comparability, a variety of characteristics known to influence clinical outcomes were measured (see Table 1).
Baseline Characteristics and Scores. a
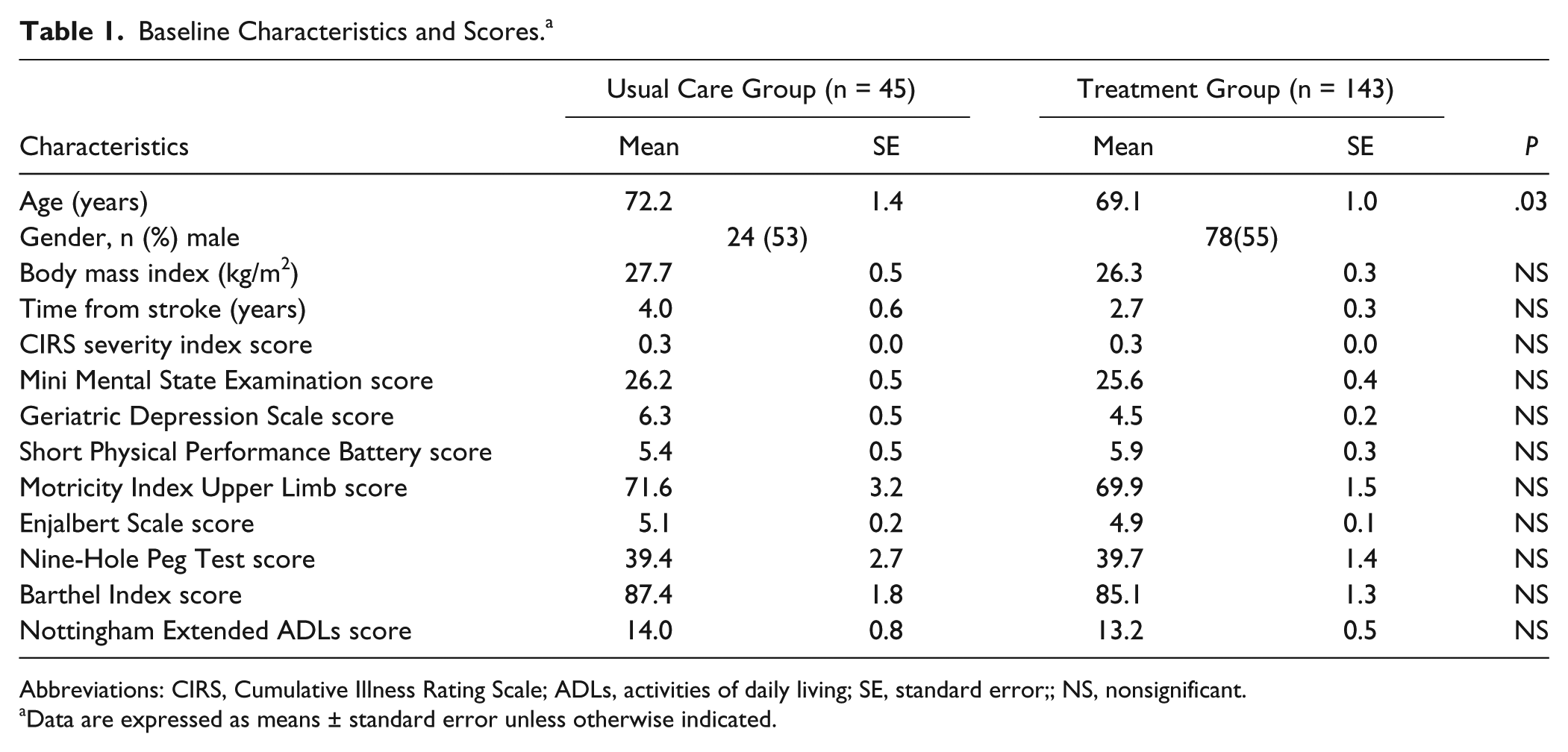
Abbreviations: CIRS, Cumulative Illness Rating Scale; ADLs, activities of daily living; SE, standard error;; NS, nonsignificant.
Data are expressed as means ± standard error unless otherwise indicated.
Outcome measures were obtained immediately before and after the 3-month treatment period. The primary outcomes measures were those of paretic upper limb impairment (Motricity Index, 26 WMFT, 27 and 9-Hole Peg Test 28 ). These measurements have good reliability and are responsive to change in patients with stroke. 29 Secondary outcomes included widely used measures of disability (the Barthel Index, 30 Nottingham Extended Activities of Daily Living Questionnaire [NEADL], 31 Short Physical Performance Battery [SPPB], 32 and quality of life as measured by the Stroke Impact Scale [SIS] 33 ). The SIS was scored according to the SIS manual. The Ashworth 34 scale was used to measure spasticity, and each subject was evaluated by a trained physiotherapist.
Patient perception of treatment was assessed using the Habilis evaluation questionnaire augmented by several dimensions from the Unified Theory of User Acceptance and Use of Technology (UTAUT). 35 Structured interviews using Likert-type scales were administered to subjects and caregivers. For each dimension, subjects were asked to score 4 to 6 items on a 7-point Likert-type scale. The data were then categorized as: low expectations/not satisfied (score 1-2), average scores (score 3-5), and high expectations/satisfied (scores 6-7). Subjects were also asked whether they had used a computer prior to their stroke.
We measured adherence at the end of the study by examining the subject log books for home exercise and computer-generated logs from the kiosks to determine the actual frequency of exercise at each site. To assess the relationship between adherence to the study protocol and improvement, we subdivided subjects into 3 categories: low adherence, average adherence, and high adherence. High adherence subjects practiced at home at least twice per week and at kiosks at least twice per week. Average adherence subjects practiced at least twice per week in either home or kiosk. Low adherence subjects practiced less than twice per week in either location.
Statistical Analysis
The SPSS (version 11.5 for Windows) statistical package was used. Means and standard errors for ordinal variables and frequency distribution for nominal variables were used for descriptive analyses. To compare outcomes between groups, we calculated change scores on each variable for each subject (measure at 3 months − baseline). Change scores for the treatment and control groups were compared with both parametric (t test) and nonparametric (Mann–Whitney U test) statistics. We have presented the more conservative nonparametric results, although the results for both analyses were similar. The Motricity Index and WMFT were the primary outcomes measures for the study. To adjust for multiple comparisons, we set the false discovery rate 36 to .05. This procedure 37 is more appropriate than the Bonferroni correction to adjust highly correlated outcomes measures, and for the data in Table 2 adjusts the P value required for significance to .037.
Means ± Standard Error for 3-Month Clinical Outcomes.
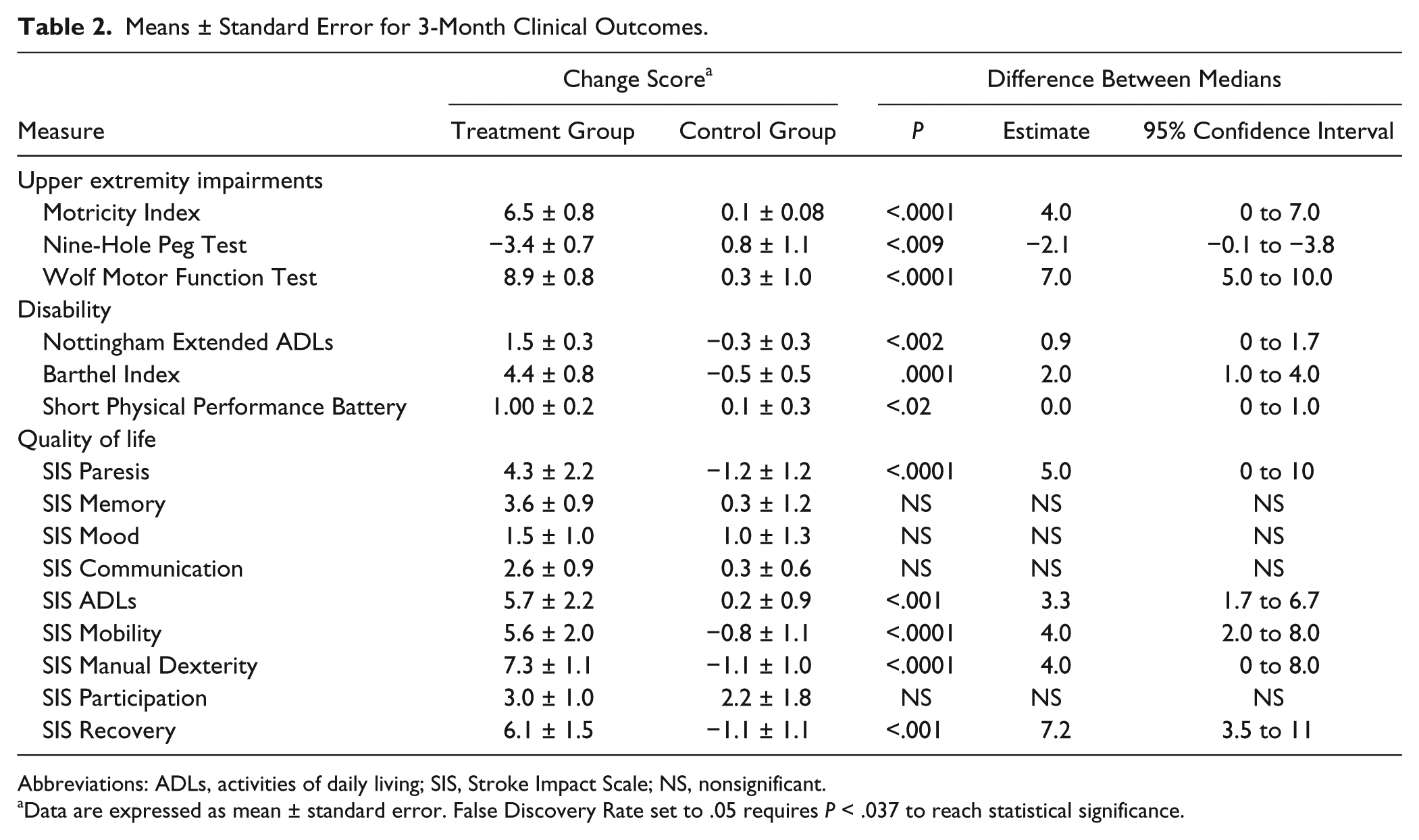
Abbreviations: ADLs, activities of daily living; SIS, Stroke Impact Scale; NS, nonsignificant.
Data are expressed as mean ± standard error. False Discovery Rate set to .05 requires P < .037 to reach statistical significance.
Changes were also evaluated at the individual level (within-group). For the Motricity Index, subjects were identified as clinically improved if their 3-month score improved by at least 10% of their rehabilitation potential (rehabilitation potential = 100 − score at baseline) as compared with baseline; clinically unchanged if the improvement was between 0% and 9% of potential; and worsened if the 3-month score was worse than baseline. For the 9-Hole Peg Test, subjects were identified as clinically improved when their 3-month time improved by at least 10% as compared with baseline; clinically unchanged if the improvement was between 0% and 9% of baseline; and worsened if the 3-month score was worse than baseline.
Logistic Regressions
Logistic regressions were used to estimate which factors were most predictive of arm function improvement (as defined above on the Motricity Index). For each subject, a series of independent variables were examined.
As a first step in identifying factors that were predictive of meaningful arm improvement, we conducted univariate analyses for all of the potential predictor variables, that is, the full data set. Independent sample t tests were used to establish group differences for normally distributed continuous variables, Mann–Whitney U tests for those not normally distributed, and χ2 tests for categorical variables. We then performed multivariate logistic regression analyses to determine the combined effects of the appropriate predictor variables on our primary outcomes. Potential predictor variables with a significant univariate association with the outcome (P < .05) were retained as potential predictor variables in regression models. In the final multivariate logistic regression models for improved arm function, the α level was set at .05.
We then performed logistic regressions in the same manner to determine the factors associated with adherence to the study protocol, including level of disability, kiosk accessibility, comorbidity, and other potential barriers to home exercise or accessing the kiosk.
Results
We entered 256 subjects into the effectiveness study. Of these, 203 subjects entered the treatment group. Fifteen subjects were excluded because they did not meet inclusion criteria; 43 subjects dropped out and 2 subjects died during the study, leaving 143 subjects in the treatment group with outcomes measures for analysis (Figure 1). We entered 53 subjects into the usual care group. Five of these subjects were found not to have met inclusion criteria, and 3 dropped out, leaving 45 subjects for analysis. After completion of the effectiveness study, all control subjects were offered the option of treatment and 22 accepted. After completion of the intervention, the results from this group were combined with the initial intervention group for the analysis of patient satisfaction and adherence (Figure 1). At entry into the study, the only significant difference between the usual care and experimental groups was that the usual care group had a mean age of 72.2 years while the experimental group had a mean age of 69.1 years (P < .03). For all other variables of interest, including stroke severity, time from stroke onset, performance on 9-Hole Peg Test, Motricity Index, Barthel Index, SIS, NEADL, Mini-Mental State Examination, 38 Geriatric Depression Scale (GDS), 39 Cumulative Illness Rating Scale (CIRS), 40 and visual analogue scale (VAS) for pain, 41 there were no significant differences between the groups (Table 1).
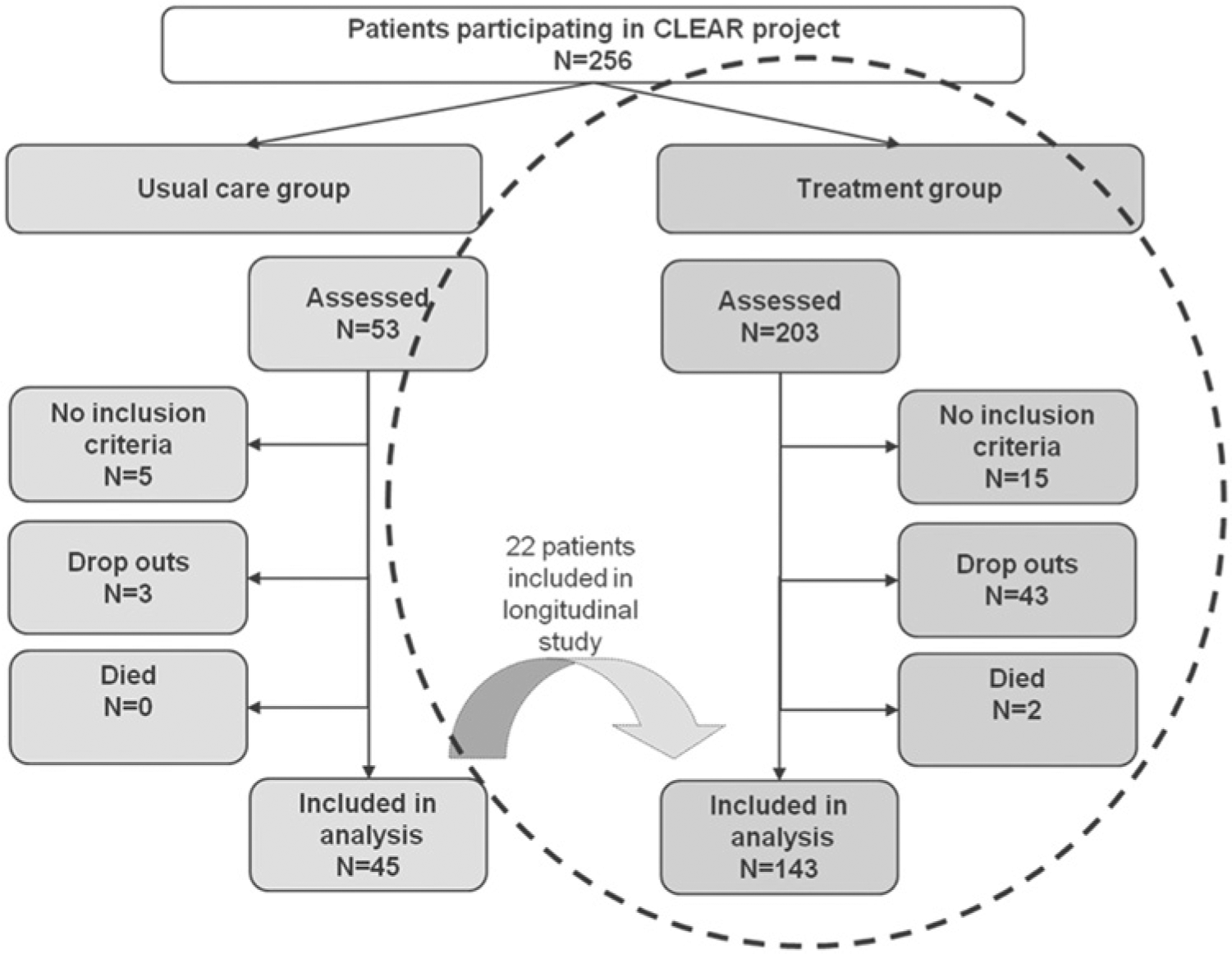
Flow of participants through the study.
Findings confirmed our primary hypothesis, that the intervention would be safe and effective. The intervention was judged safe, as there were no study-related adverse events reported. After 3 months of intervention, there were significant differences between the experimental and control groups on impairment as documented in Table 2. The mean change scores (score at 3 months − score at baseline) were significantly different between treatment and control groups for the primary outcomes measures, Motricity Index, 9-Hole Peg Test, and WMFT with P < .0001, .009, and .0001, respectively. The 95% confidence limits for median differences between groups were 4.0 (0.0 to 7.0) for Motricity Scores, −2.1 (−0.1 to −3.8) for the 9-Hole Peg Test, and 7.0 (5.0 to 10.0) for the WMFT. The change in the WMFT exceeds the minimum clinically significant change previously reported for stroke recovery. 42
Measures of disability demonstrated a similar pattern of change between treatment and control groups (Table 2). The NEADL and Barthel Index change scores were significantly larger for the treatment group (P < .002 and P < .0001, respectively). The 95% confidence limits for the median differences in these variables were 0 to 1.7 and 1.0 to 4.0, respectively. Although the change in the SPPB was significant using both parametric and nonparametric tests, the magnitude of this change was quite small (probably reflecting the large component of lower extremity function measured in the SPPB). Quality-of-life measures (Table 2) also demonstrated a significant difference between mean change in the treatment and control groups, with SIS variables relevant to the intervention exhibiting significant changes: paresis, ADLs, mobility, manual dexterity all of which demonstrated differences significant at P < .0001. The perception of recovery was also significantly better in the treatment group (P < .001).
Results of the evaluation of change at the individual level (within-group) are illustrated in Figure 2. After 3 months, only 13% of the control group were classified as clinically improved on the Motricity Index, while 80% were unchanged and 7% worsened. In contrast, 49% of the treatment group improved, 48% were unchanged, and 3% worsened (χ2 P < .0001). On the 9-Hole Peg Test (Figure 2), 24% of the control group improved, 36% were unchanged, and 40% worsened. In the treatment group, 38% improved, 41% were unchanged, and 21% worsened (χ2 P < .02).
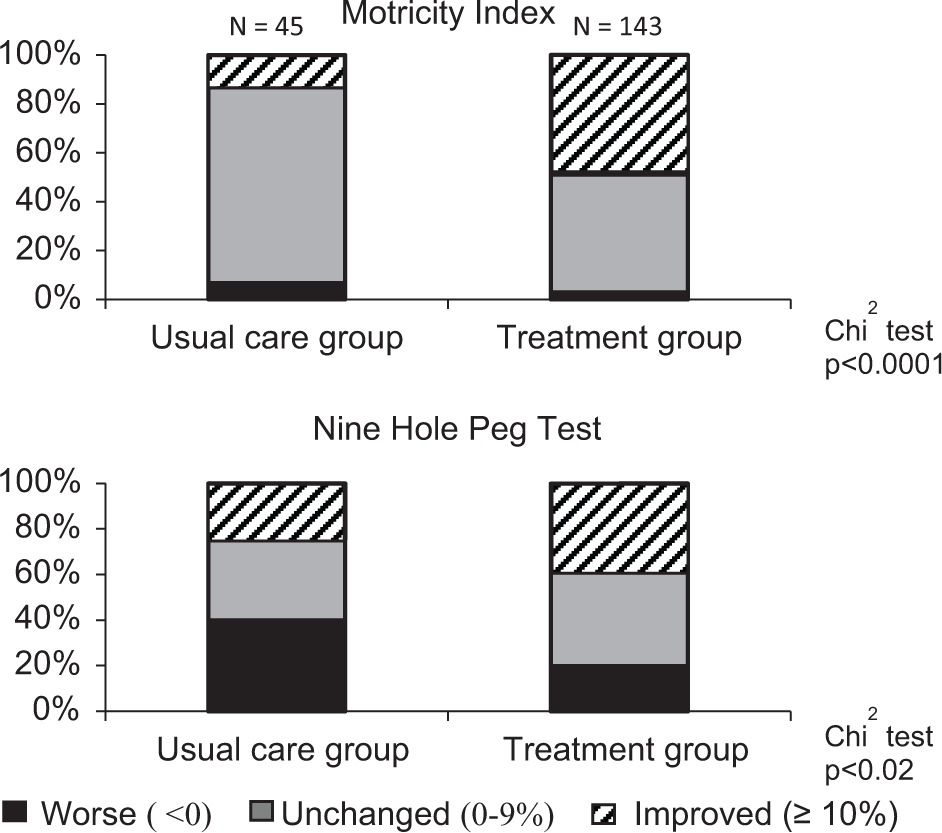
Clinically significant change in Motricity Index (MI) and 9-Hole Peg Test. Graphs indicate the proportion of subjects in treatment and control groups by level of change between baseline and posttreatment testing. Minimally significant clinical change for MI is defined as greater than 10% of potential improvement. The changes in the treatment versus the control group are significantly different as determined by χ2.
Findings were mixed regarding our second hypothesis, that use of the Habilis telerehabilitation system would reinforce adherence and that highly adherent subjects would have greater improvements in outcomes than others. Only 50 of the 165 (143 treatment group + 22 from control group accepting treatment after the effectiveness study completed) were highly adherent to the study protocol, 85 demonstrated average adherence, and 30 low adherence. Nearly all of the subjects in the average adherence group performed only home exercise and did not attend the kiosks with any regularity. Highly adherent subjects exercised both at home and at kiosks. The difference in arm function outcome (Motricity Index) between low adherence and average adherence subjects is significant at P < .02 (χ2 test), but the difference in outcome between average and highly adherent subjects is not significant (Figure 3). We reviewed changes in other measures related to upper extremity impairment, that is, WMFT, 9-Hole Peg Test, and NEADL, and found no significant differences between subjects with average and high adherence. We examined more closely the relationship between these measures of improvement and the total amount of exercise that subjects reported. The mean total hours of exercise overall was 3.3 hours per week; 16% of subjects exercised 1 hour or less per week; 60% of subjects exercised 3 or more hours, and 28% exercised 5 or more hours per week. Linear regression demonstrated a significant (P < .05) relationship, but only 2% of the total variance in MI improvement was explained by subject total exercise hours, and improvement on the 9-Hole Peg Test and WMFT were not linearly related to hours of exercise. Even the low adherence group reported a high level of satisfaction with the exercise program (5 out of 7 on the Likert-type scale) and there was a significant difference in overall patient satisfaction with each increment of adherence (P < .05, analysis of variance). Not surprisingly, highly adherent subjects rated the kiosk component of the training program more highly than other subjects.
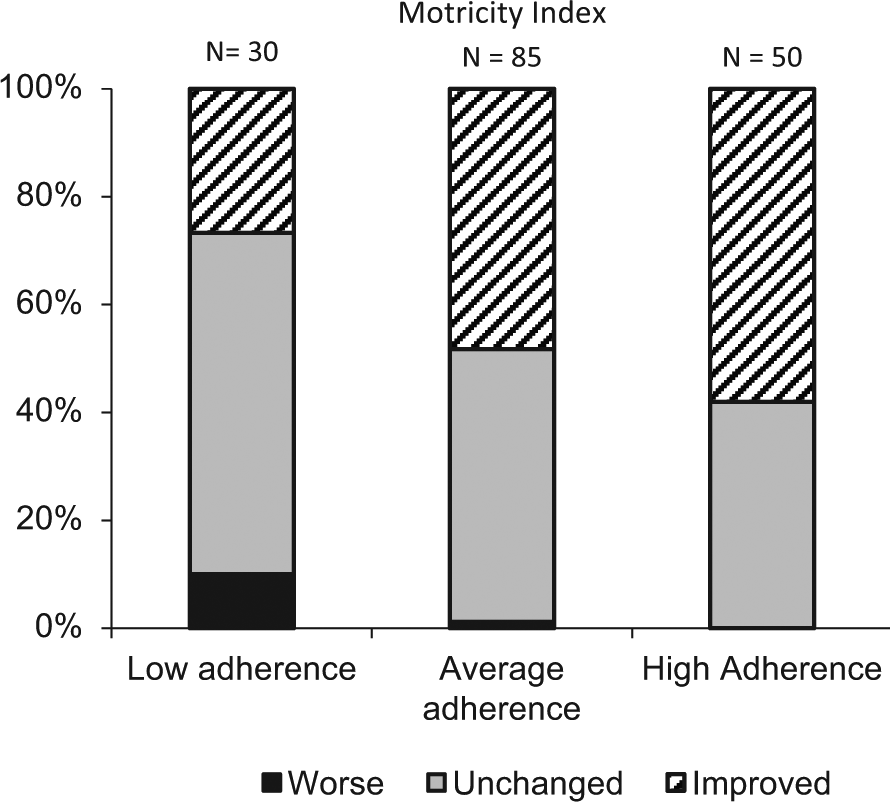
Significant improvement in Motricity Index scores stratified by adherence.
Results of the final logistic regressions to identify which factors were most predictive of motor improvement are reported in Table 3. The 165 patients who completed the intervention were included in these regressions. An odds ratio of 1 indicates no effect, a ratio less than 1 indicates a negative effect, and a ratio more than 1 indicates a positive effect. Female patients had a 2-fold probability of improvement over male patients. Severity of spasticity negatively influenced outcome. There was a very small negative effect associated with higher baseline Motricity Index scores. Adherence to the protocol was the strongest predictor, with average and highly adherent subjects having a 2-fold higher probability of recovery of arm function as compared with low adherence subjects. Table 3 also includes the results of logistic regressions to examine the factors predicting adherence to the treatment protocol. The distance from home to kiosk negatively affected the probability of adherence to the treatment protocol, but the ability of subjects to travel independently to the kiosk was a much stronger predictor. No other variables were significant predictors of adherence to the treatment protocol.
Logistic Regression Predictors of Improvement and Adherence.
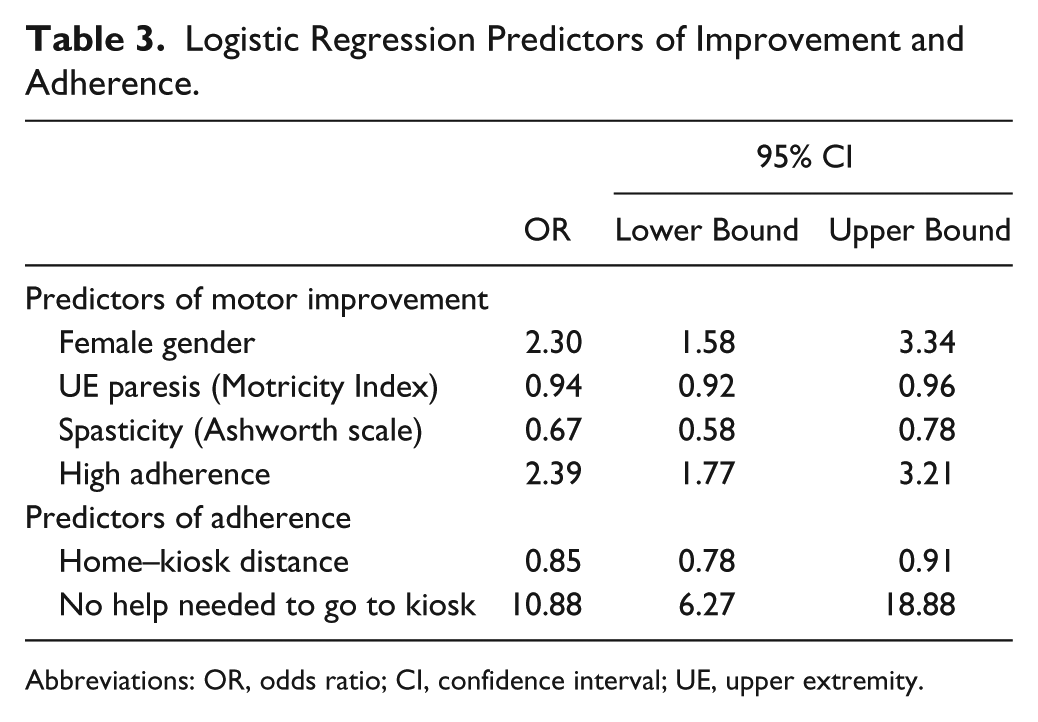
Abbreviations: OR, odds ratio; CI, confidence interval; UE, upper extremity.
Discussion
Findings confirm our primary hypothesis that the intervention would be safe and effective. There were no serious study related adverse events, and compared with the control group and their own baseline, the group receiving the home-based exercise intervention improved significantly in measures of upper arm function. Comparison with results from recent large randomized trials, 5 featuring more intensive and expensive interventions, 7 is difficult because of differences in study populations and design, but the results from this study appear to be of the same order of magnitude. Perhaps more important, the intervention appeared to have measurable impact on subjects’ performance in everyday activities, as measured by the change on the NEADL, and on their quality of life, as measured by the SIS. These findings parallel the results observed in a cross-country comparison of upper extremity rehabilitation in acute stroke, 43 which found that patients receiving outpatient rehabilitation achieved gains in function comparable to those receiving inpatient rehabilitation. The findings in this study are significantly more positive than those in a recent meta-analysis on home-based therapy programs for upper limb functional recovery after stroke 44 ; however, most of the 166 subjects included in the meta-analysis were in the subacute period poststroke, whereas subjects for our study were not limited to subacute.
Not surprisingly, subjects who had average or high adherence had a higher probability of improvement, and had greater satisfaction with the exercise intervention than those with low adherence. Female subjects had much better outcomes than males in this study. We have no explanation for this finding, and indeed, previous studies suggest that female stroke patients have worse functional recoveries. 45 Adherence to exercise did not differ significantly by gender in the present study. The negative influence of spasticity on improvement is not surprising, while the very slight effect of initial arm function most likely represents a ceiling effect of the Motricity Index in our population.
We expected highly adherent subjects would have significantly greater improvements in clinical outcomes than those with average adherence. However, this was not the case. It appears that the combination of practicing these relatively simple, low-cost exercises at least twice a week, with monitoring and assessment at least monthly, effectively improves motor function and quality of life regardless of whether subjects used the kiosks or practiced exclusively at home. People who used the kiosks exercised more than those who did not, suggesting that telerehabilitation may provide a useful motivational strategy. Key issues for future research include evaluation of home-based telerehabilitation, the relationship between intensity of exercise and outcomes and the long-term effects of exercise. Telerehabilitation interventions will have to be more accessible to achieve high levels of patient adherence. Increased engagement may be possible through dynamic adaptation, 46 real-time feedback,19,47 and virtual reality. 48 An ongoing multisite randomized controlled trial of home stroke rehabilitation 49 should enhance our understanding in this important area.
Our study has significant limitations. Subjects were assigned to treatment arms based on their locations rather than by random assignment. While random assignment is preferable, the demographic and clinical variables describing the treatment and control subjects were remarkably similar. Patient assessors were not blinded to treatment assignment. The lack of blinding suggests caution regarding the significance of the changes on the NEADL, which may be more susceptible to bias as compared with the WMFT, 9-Hole Peg Test, and Motricity Index, 50 which provide more objective measures. Findings related to adherence should be interpreted with some caution, in view of the well-known observation that patients complying with any treatment are more likely to have positive outcomes 51 than noncompliant subjects. Additionally, frequency of home exercise is based on self-report and detail regarding progression was not collected.
Footnotes
Acknowledgements
We thank Dr Paul Albert, National Institute of Child Health and Human Development (NICHD), for statistical consultation.
Declaration of Conflicting Interests
The author(s) disclosed the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sandro Scattareggia Marchese is CEO of Signo Motus, producing the Habilis platform.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by grant ICT-PSP-224985 from the European Union.
