Abstract
Background. While treatments for the behavioral deficits associated with traumatic brain injury (TBI) are currently limited, animal models suggest that zinc supplementation may increase resilience to TBI. Objective. This work tests the hypothesis that zinc supplementation after TBI can be used as treatment to improve behavioral outcomes such as anxiety, depression, and learning and memory. Methods. TBI was induced by controlled cortical impact to the medial frontal cortex. After TBI, rats were fed either a zinc adequate (ZA, 30 ppm) or zinc supplemented (ZS, 180 ppm) diet. Additional rats in each dietary group (ZA or ZS) were given a single intraperitoneal (ip) injection of zinc (30 mg/kg) 1 hour following injury. Results. Brain injury resulted in significant increases in anxiety-like and depression-like behaviors as well as impairments in learning and memory. None of the zinc treatments (dietary or ip zinc) improved TBI-induced anxiety. The 2-bottle saccharin preference test for anhedonia revealed that dietary ZS also did not improve depression-like behaviors. However, dietary ZS combined with an early ip zinc injection significantly reduced anhedonia (P < .001). Dietary supplementation after injury, but not zinc injection, significantly improved (P < .05) cognitive behavior as measured by the time spent finding the hidden platform in the Morris water maze test compared with injured rats fed a ZA diet. Conclusions. These data suggest that zinc supplementation may be an effective treatment option for improving behavioral deficits such as cognitive impairment and depression following TBI.
Introduction
Despite aggressive treatment and rehabilitation, many traumatic brain injury (TBI) patients experience a number of long-term cognitive, social, and psychiatric complications such as memory loss, anxiety, depression, aggression, and poor social functioning. 1 With more than 1.5 million Americans sustaining a TBI every year, effective treatment options are needed not only to reduce mortality rates but to also improve the quality of life of TBI survivors. 2
An early clinical study limited to patients with moderate to severe brain injury suggested that zinc administration after TBI may be beneficial. 3 Intravenous administration of zinc within 72 hours of injury followed by enteral zinc supplementation significantly improved protein synthesis and metabolism as evidenced by increased retinol-binding protein and pre-albumin levels. They also showed improved mean Glasgow Coma Scores, suggesting that zinc can improve neurological outcomes after TBI. More recent work using zinc supplementation to improve zinc status in stroke patients reported improvements in the National Institutes of Health stroke scale scores 30 days after hospital admission. 4 However, neither study examined other behavioral outcomes associated with brain injury such as depression, anxiety, or other cognitive measures. The use of zinc to treat mood disorders is also supported by clinical trials showing that oral zinc improves the effects of antidepressant drug therapy in patients with unipolar depression. 5 Moreover, patients previously refractory to antidepressants drugs showed improvements in depression scores when medication was supplemented with zinc. 6
Animal models of TBI provide the opportunity to examine the extent to which zinc supplementation can be used to improve behaviors after injury. In fact, recent work has shown that zinc supplementation prior to TBI reduced depression-like behaviors, markers of stress-related anxiety, and improved learning and memory. 7 While this work suggested that zinc supplementation can improve resilience to TBI, the work was not designed to test the use of zinc as a treatment after TBI. In fact, no work to date has examined the extent to which zinc can be used after injury to improve depression, anxiety, and memory. Thus, the current work used dietary zinc supplementation following injury to test the hypothesis that zinc supplementation after TBI will improve behavioral outcomes. Furthermore, because of reductions in food intake that typically occur in rodents and humans immediately after TBI, we explored the efficacy of early zinc injections (1 hour after TBI) alone and coupled with dietary supplementation.
Methods
Animal Care and Zinc Treatments
Young adult male Sprague-Dawley rats (Charles Rivers Laboratories, Wilmington, Massachusetts) were individually housed in temperature-controlled rooms with a 12-hour light–dark cycle. All procedures were approved by the Florida State University Animal Care and Use Committee. Before TBI, all rats were fed a commercially prepared semipurified zinc adequate diet (ZA, 30 ppm) for 5 days using zinc carbonate as the zinc source (Research Diets Inc, New Brunswick, New Jersey). Following TBI surgery, rats were divided into a total of 4 groups (n = 8 per group) as described below. First, rats were placed into a dietary treatment group in which they were fed either the ZA or a zinc supplemented diet (ZS, 180 ppm). These 2 groups were further divided into groups that received diet alone (ZA or ZS) or a single intraperitoneal (ip) zinc injection (30 mg/kg) 1 hour following injury (TBI ZA + ipZn or TBI ZS + ipZn). Rats continued on their assigned diets throughout the remainder of the experiment.
Traumatic Brain Injury
TBI was induced by a controlled cortical impact to the medial frontal cortex. Prior to stereotaxic placement, rats (n = 8 per group) were anesthetized using isoflurane gas. Aseptic procedures were used throughout the surgical procedures, and body temperature was maintained using a homeothermic blanket. After a midline incision, a 6 mm diameter mid-sagittal bilateral craniotomy 3 mm rostral to bregma was performed. A 5 mm diameter pneumatic cortical contusion device (MyNeuroLab, Inc, Richmond, Illinois) was used to produce a 3.0 mm deep contusion using an impact velocity of 2.25 m/s and an impact time of 500 ms. 7 Following TBI, the incision was immediately sutured. Additional sham-operated controls (n = 8) fed the ZA diet were anesthetized and received the midline incision, but not the craniotomy or impact. Behavioral testing began 1 week after TBI or sham surgery. Body weights were measured daily for 10 days following TBI.
Light–Dark Box
Anxiety-like behaviors were measured using standard protocols for light–dark box in rodents. 8 Each rodent was placed in the dark chamber of the light–dark box and allowed to explore the box for a period of 5 minutes. The number of crosses into the light compartment and the amount of time spent in light was recorded for each animal. Both the light and dark chambers were cleaned with 70% ethanol in between animals.
Saccharin Preference
As a measure of depression-like behavior, a standard 2-bottle choice paradigm for saccharin preference was used to examine the effect of injury and zinc treatment on the development of anhedonia. 7 Eight days following injury, rats were given a choice between saccharin and deionized water for a 4-day period; 2 days at 0.025% saccharin followed by 2 days at 0.05% saccharin. Intake of water and saccharin was measured daily. To avoid preferences associated with bottle placement, the position of the bottles were changed daily.
Morris Water Maze
Spatial learning and memory was examined using the Morris water maze (MWM) test. 9 The water maze apparatus consisted of a large circular tank filled with water and rendered opaque by the addition of nontoxic water-based white paint. Additionally, a platform was submerged and hidden 1 cm under the surface of the water in the southwest quadrant (target quadrant). The water maze was surrounded by curtains to eliminate any spatial cues from inside the testing room. Visual cues were placed on the north, south, east, and west quadrants of the pool. Spatial acquisition consisted of 2 trials per day, with an intertrial interval of 5 minutes for 10 days in which the rat was placed in 1 of the 3 semirandom start locations (excluding the southwest quadrant containing the hidden platform). If the animal was unable to locate the hidden platform during the 90-second trial period, it was then physically guided to the platform and placed on it for a period of 20 seconds. Following completion of the second trial each day, the rodent was dried and returned to its home cage. Time to find the hidden platform (seconds) was recorded for each animal. Twenty-four hours following the spatial acquisition trials, animals were given a single probe trial in which the hidden platform was removed from the southwest quadrant. Animals were allowed to explore the maze for 60 seconds. Both the time spent in the southwest quadrant and the number of crosses over where the platform was located were recorded. Visible platform trials (cued learning) were conducted 24 hours following the probe trial. In this control trial, all spatial cues were removed, the platform was raised 1 cm above the water, and an orange flag was attached to the platform. Rodents were given four 90-second trials to swim to the visible platform. The platform was moved to a semirandom location for each trial and the time to the platform was recorded. Animals unable to swim to a visible platform were removed from all data sets before analysis.
Statistical Analysis
Body weight and MWM acquisition trials were analyzed using a 2-way analysis of variance (ANOVA) repeated measures with a Bonferroni post hoc test. Anhedonia, anxiety, and MWM probe trials were analyzed using a 1-way ANOVA with a Bonferroni post hoc test. All data sets were expressed as the mean ± SEM and statistical significance was set at P < .05 with 95% confidence (Prism; GraphPad, San Diego, California).
Results
Body Weight
Before TBI or sham surgery, there were no significant differences in mean body weights between animal groups (194.6 ± 1.3; Figure 1). While TBI resulted in an approximately 8% decrease (P < .05) in mean body weight, body weights did not differ between TBI treatment groups. TBI significantly decreased body weight for 8 days following injury.
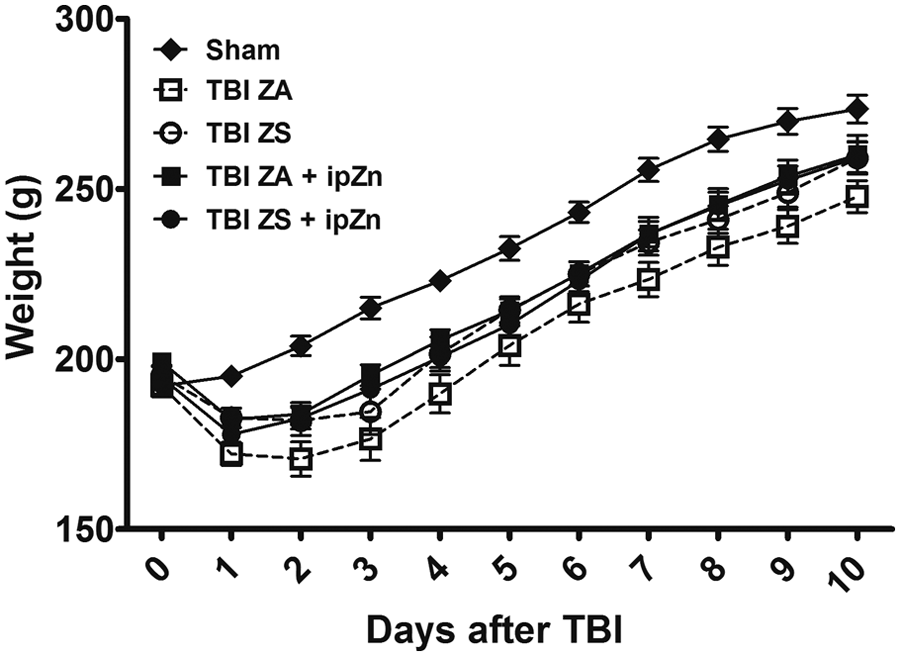
Effect of injury and zinc treatments on mean body weight. Rats (n = 8 per group) were subject to a traumatic brain injury (TBI) or sham surgery (Sham) and fed either a zinc adequate (ZA, 30 ppm) or zinc supplemented (ZS, 180 ppm) diet. Shams were fed the ZA diet. Additional ZA or ZS rats were given a single intraperitoneal (ip) zinc injection (30 mg/kg; TBI ZA + ipZn or TBI ZS + ipZn) 1 hour after TBI. Body weight (g) was measured daily for 10 days following TBI. Bars indicate mean ± SEM. TBI rats groups are significantly different than Sham rats at days 2 to 8 at P < .05.
Anxiety
TBI induced anxiety-like behavior. In the light–dark box test of exploratory behavior, rats subjected to TBI spent 50% less time in the light compartment (P < .01) and had 38% fewer crosses into the light chamber (P < .05) compared with sham-operated controls. Sham-operated controls spent 100 ± 10 seconds in the light chamber compared with TBI ZA rats, which spent only 50 ± 13 seconds in light. None of the supplementation treatments (diet, injection, or combination of diet and injection) improved these TBI-induced increases in anxiety-like behavior.
Anhedonia
Examination of the data from the 2-bottle test for the depression-like behavior anhedonia revealed that ZA rats showed a significantly decreased preference for saccharin after injury (Figure 2, P < .0001). While neither dietary supplementation nor a single ip zinc injection improved this behavior, rats given the zinc injection and fed the ZS diet (TBI ZS + ipZn) showed significantly fewer depression-like behaviors following TBI (P < .0001).
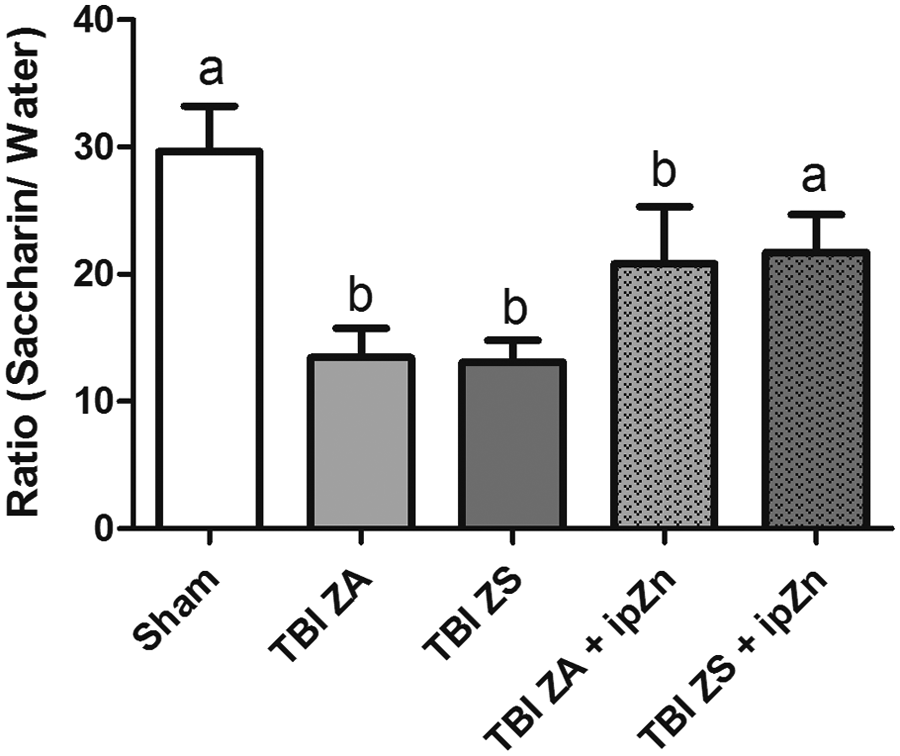
Effect of injury and zinc treatments on depression-like behavior. Rats were subject to a traumatic brain injury (TBI) or sham surgery and fed either a zinc adequate (ZA) or zinc supplemented (ZS) diet. Shams were fed the ZA diet. Additional ZA or ZS rats were given a single intraperitoneal (ip) zinc injection (30 mg/kg; TBI ZA + ipZn or TBI ZS + ipZn) 1 hour after TBI. Bars represent intake ratios of saccharin to water over a 4-day period (mean ± SEM). Bars with different letters (a or b) are significantly different from each other at P < .001.
Spatial Learning and Memory
Analysis of MWM performance data showed statistically significant effects of treatment (P < .0001), time after injury (P < .0001), as well as in interaction between these 2 main effects (P = .0017). Post hoc analysis revealed that injury resulted in impairments in spatial learning and memory such that on day 1 of MWM testing sham-operated controls were able to find the hidden platform within 49 ± 8 seconds while TBI ZA rats took 86 ± 4 seconds to complete the same task (P < .0001, Figure 3). These significant differences were maintained until day 9 of testing. Rats fed the ZS diet following TBI showed significant improvements in spatial learning on days 4 to 7 compared with rats fed the ZA diet (P < .05). Furthermore, there was no difference between sham-operated controls and the TBI ZS rats on days 3 to 10 of MWM testing.
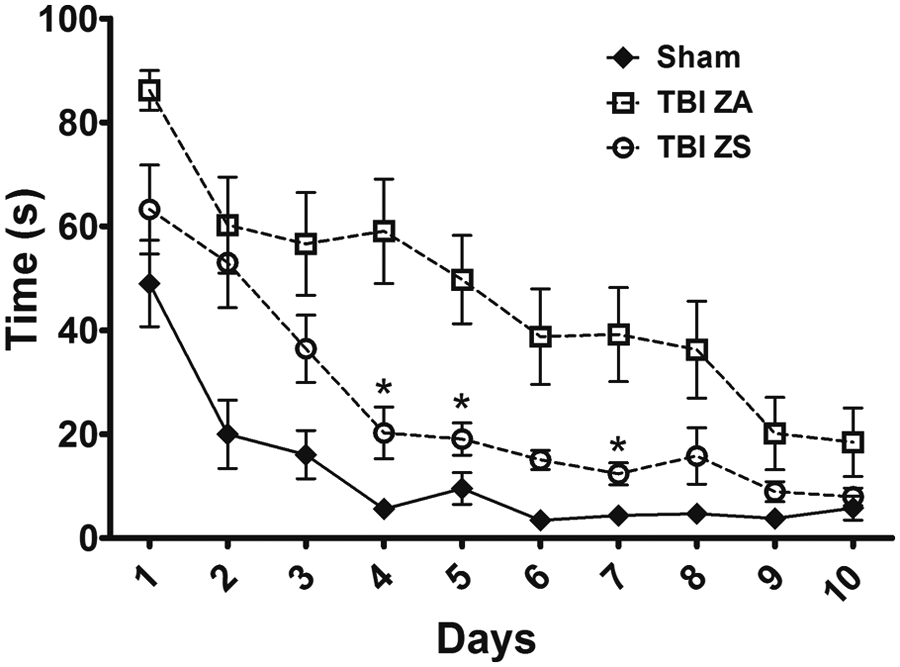
Effect of injury and dietary zinc treatment on spatial learning and memory. Rats were subject to a traumatic brain injury (TBI) or sham surgery and fed either a zinc adequate (ZA) or zinc supplemented (ZS) diet. Shams were fed the ZA diet. Morris water maze (MWM) testing began 8 days after injury. Individual points are time (seconds) to find the hidden platform (mean ± SEM). TBI ZA rats were significantly different from Sham rats on days 1 to 8 of MWM testing. *Significantly different than TBI ZA at P < .05.
Figure 4 shows the effect of zinc injections 1 hour post-injury. Figure 4A shows that a single ip zinc injection (TBI ZA + ipZn) did not improve performance in MWM compared with TBI ZA rats. Indeed, mean time to find the hidden platform was not different at any time point during this test. Rats given both the dietary supplementation and the zinc injection (TBI ZS + ipZn) did not show further improvements in time spent finding the hidden platform compared with rats given only the dietary zinc supplementation (TBI ZS, Figure 4B). These groups were not significantly different at any time point during this 10-day test.
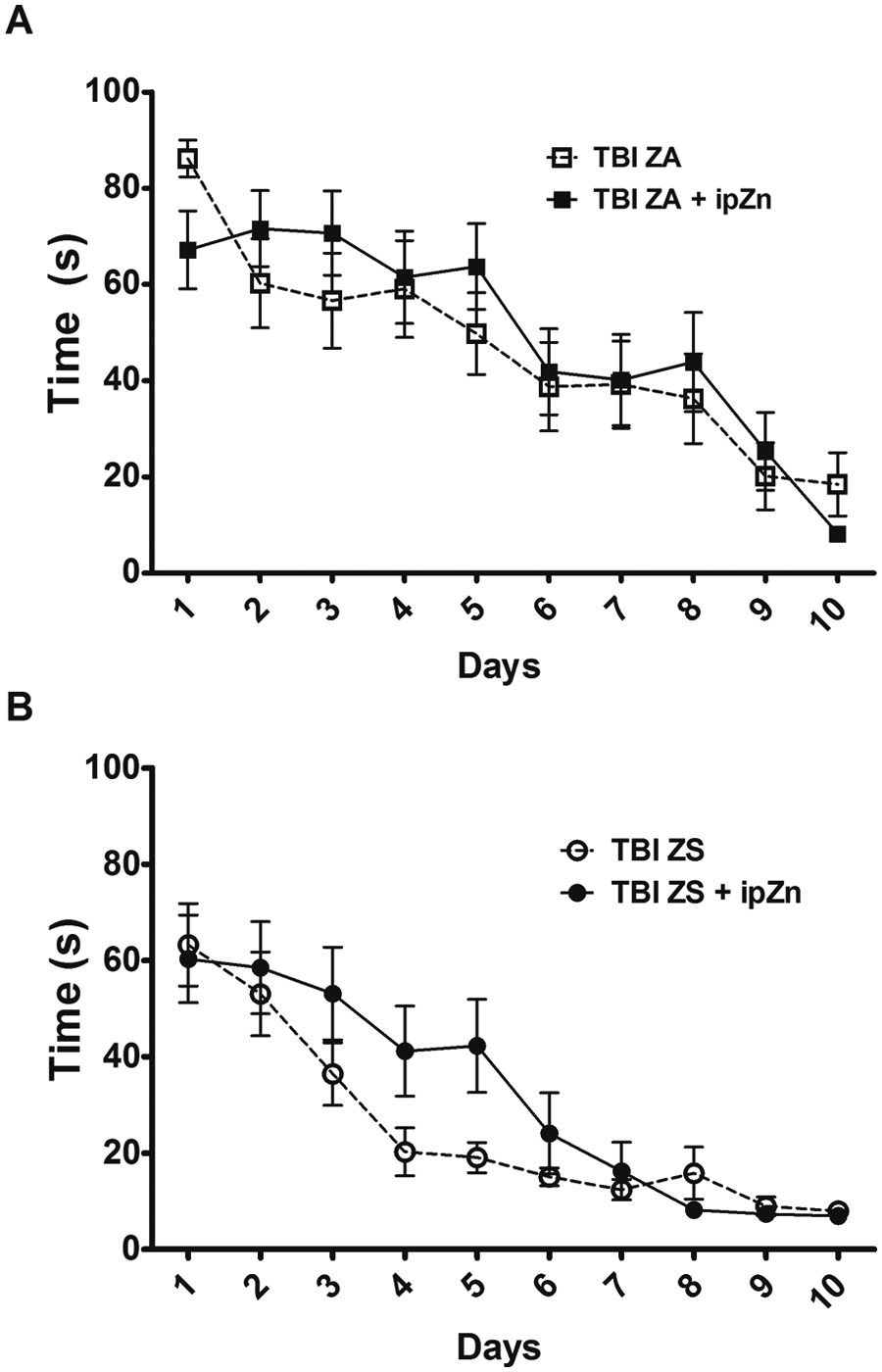
Effect of zinc treatments (ip zinc injection) on spatial learning and memory after TBI. Morris water maze testing began 8 days after injury. Individual points are time (seconds) to find the hidden platform (mean ± SEM). All rats were subject to a traumatic brain injury (TBI). (A) After TBI, rats were fed a zinc adequate (ZA) diet. Additional ZA rats were given a single intraperitoneal (ip) zinc injection (30 mg/kg, TBI ZA + ipZn) 1 hour following TBI. (B) After TBI, rats were fed a zinc supplemented (ZS) diet. Additional rats were given a single ip zinc injection (30 mg/kg, TBI ZS + ipZn) 1 hour following TBI.
A single probe trial conducted at the end of the behavioral assessments to assess spatial memory revealed that all rats learned the task such that rats spent approximately 34% time (20.3 ± 1.3 seconds) in the quadrant where the platform was located during acquisition trials. There was no difference in either time spent or crosses into this quadrant between treatment groups or sham controls.
Discussion
Several lines of evidence led us to hypothesize that zinc treatments could be used to improve a variety of outcomes after TBI and compare the efficacy of dietary and early injected zinc administration. First, our previous work showed that administration of dietary zinc starting 4 weeks prior to injury and continued throughout the testing period resulted in significant improvements in depression, anxiety, and learning and memory. 7 Because rats were fed the supplemented diets both before and after the injury, the study design did not permit us to determine the efficacy of dietary zinc supplementation after injury alone. This study filled that gap in knowledge by feeding rats zinc adequate diets prior to injury and supplemented or control diets after injury. The data suggest that supplementation prior to injury is more effective than supplementation after injury. While MWM performance was improved in both studies, anxiety-like and depression-like behaviors were not improved when the zinc supplemented diet was provided only after the TBI.
It should be noted that while injury resulted in decreased body weights, zinc treatments did not alter body weight after TBI. These data are consistent with previous reports showing that the zinc supplemented diet does not alter body weight or food intake in rats. 7 Thus, the behavioral changes were not due to body weight alterations. Moreover, previous data have shown that dietary zinc supplementation does not alter behavior of uninjured rats. 7
Additional support for the possible role of zinc in improving TBI outcomes came from the clinical trial in moderately and severely injured TBI patients suggesting improvements in protein balance, Glasgow Coma Scores, and mortality after zinc treatment. 3 This study used both parenterally administered zinc and enteral zinc. Intravenous zinc, at either adequate (2.5 mg/d) or supplemental (12 mg/d) levels, was administered within 72 hours of injury. The supplemented group continued on enteral zinc supplementation (22 mg/d) for the next 2 weeks, while the adequate group received a placebo. The design of the study did not permit the investigators to determine the efficacy of the enteral zinc alone or if the combination of the 2 modes of administration is needed for improved outcomes.
The current work sought to fill this gap in our understanding. An examination of the anhedonia data shows that the use of enteral (dietary) zinc supplementation following TBI did not improve this measure of depression. However, it should be noted that, although not statistically significant, there was a clear trend toward reduced depression-like behavior in rats receiving only the zinc injection. This is an interesting finding because zinc injections have been shown to be efficacious in other models of depression. In the rodent depression models of bulbectomy and chronic unpredictable stress, a single zinc injection significantly reduced depression-like behaviors in the forced swim test.10,11 TBI-induced depression may be a novel model of mood disorders and is certainly clinically relevant because major depression is the most common long-term complication of brain injury affecting as much as 40% of TBI patients. 12 Furthermore, classical antidepressants such as selective serotonin reuptake inhibitors may not be an effective treatment for TBI patients suffering from depression. 13
Despite the fact that neither diet nor injections significantly improved depression-like symptoms, the combined use of dietary zinc and a zinc injection reduced anhedonia in injured animals. Together these data suggest that both routes of zinc administration are needed for efficacy. However, it should be noted that it is possible the therapy worked, not because it included an injection per se but because it delivered zinc quickly after the injury. Clearly, this raises the question of proposed mechanisms by which zinc may be acting to improve outcomes after TBI. First, we do not fully understand the mechanisms responsible for zinc entry and uptake into the brain. While there appear to be specific zinc transport sites on the luminal surface of brain capillary endothelial cells, the role of specific transporters has not been fully elucidated. 14 Second, the mechanisms responsible for the action of zinc injections after TBI are not known. The current work suggests that bypassing intestinal absorption may facilitate behavioral improvements. This is consistent with recent work showing that intestinal zinc transporters are downregulated in response to dietary treatment. 15 Thus, future work will be needed to determine if downregulation of gut zinc transporters are responsible for the reduced efficacy of zinc for the treatment of depression in the diet-only groups. However, it should be noted that diet alone significantly improved MWM performance, suggesting that the mechanisms responsible for the effects of zinc in depression and memory are different.
Other possible mechanisms that could be contributing to the current outcomes include the action of free zinc at the synapse, particularly in the hippocampus where free zinc is colocalized with the neurotransmitter glutamate and participates in neuronal transmission. While zinc is efficacious in the current work, previous data have suggested that free zinc may be neurotoxic and play a role in neuronal death after traumatic or ischemic brain injury.16,17 We have previously measured cell death after cortical brain injury in rats fed zinc supplemented diets for up to 4 weeks and found no increases in cell death. 18 Furthermore, published reports show that zinc chelation did not result in improved spatial learning in injured rats. 19
Finally, the possible role of zinc in the processes of neuronal precursor proliferation in the hippocampus after TBI should be considered as previous work has shown that zinc deficiency impairs stem cell proliferation in this region of the brain. 20 Hippocampal stem cells have been repeatedly shown to play a role in depression and antidepressant action.21,22 Future work will be needed to explore the role of zinc supplementation, both dietary and injected, on stem cell proliferation and neurogenesis.
The issue of early zinc intervention, either parenterally or enterally, is important. First, it appears that there is a window of opportunity for virtually all treatments following TBI. The window of opportunity for zinc treatment is not known, but the current work suggests that administration of zinc within the first several hours after injury should be investigated. The issue of early zinc administration is also important when permissive underfeeding is used in patients with TBI. Permissive underfeeding (the reduction in total caloric intake by 60% to 70% of estimated needs) has been shown to reduce mortality. 23 The current data suggest that zinc levels should not be reduced when caloric intake is low after TBI.
Conclusions
The current data show that dietary zinc treatment after TBI results in significant improvements in spatial learning and memory. While early zinc treatment may be important for treating TBI-associated depression, future work is needed to not only determine the window of opportunity for this treatment but to also determine the extent to which a chronic dietary zinc treatment following TBI may improve depression. It should also be noted that treatment prior to injury appears to be the most effective approach with clearly robust improvements in stress-related anxiety, depression, and spatial memory. 7 The current work suggests that the efficacy of zinc is reduced when used solely as a treatment for TBI-associated depression and anxiety compared with zinc supplementation prior to injury. Based on the current and previous works chronic zinc supplementation is recommended for populations at risk for TBI. Zinc, along with other dietary supplements, environmental enrichment, and rehabilitation strategies24,25 may also be tested in combinations to lessen behavioral deficits associated with brain injury.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the US Army Medical Research and Material Command.
