Abstract
Background. Electrical and magnetic brain stimulation can improve motor function following stroke in humans, rats, and nonhuman primates, especially when paired with rehabilitative training (RT). Previously, we found in rodent stroke models that epidural electrical cortical stimulation (CS) of the ipsilesional motor cortex (MC) combined with motor RT enhances motor function and motor cortical plasticity. It was unknown whether CS following experimental traumatic brain injury (TBI) would have similar effects. Objective. To test the effects of CS combined with motor training after moderate/severe TBI on behavioral outcome and motor cortical organization. Methods. Following unilateral controlled cortical impact (CCI) over the caudal forelimb area of the MC in adult male rats, forelimb reach training was administered daily for 9 weeks concurrently with subthreshold, 100-Hz monopolar CS or no-stimulation control procedures. The rate and magnitude of behavioral improvements and changes in forelimb movement representations in the injured MC as revealed by intracortical microstimulation were measured. Results. CCI resulted in severe motor impairments persisting throughout the 9 weeks of training in both groups, but CS-treated animals had significantly greater behavioral improvements. CS also increased wrist motor cortical representation, one of the main movements used in the training task, when compared with RT alone. However, the overall recovery level was modest, leaving animals still extremely impaired. Conclusions. These data suggest that CS may be useful for improving rehabilitation efficacy after TBI but also raise the possibility that the CS parameters that are highly effective following stroke are suboptimal after moderate/severe TBI.
There are few well-established treatments that improve motor outcomes after traumatic brain injury (TBI). In contrast, the stroke literature strongly supports that task-specific motor practice (rehabilitative training [RT]) can greatly improve motor function. Adjunctive therapies, such as cortical stimulation (CS), can further enhance the efficacy of RT. In monkeys 1 and rats,2-5 CS of the peri-infarct motor cortex (MC) coupled with impaired-limb RT greatly enhances forelimb functional recovery and increases wrist representation area 3 and neuronal structural plasticity 6 in the perilesion cortex.
Although it seems reasonable to assume that treatments that reduce motor impairments following stroke would produce similar results following TBI, our data do not support this assumption. We and others have found that rehabilitative treatments, such as reach training, alone are insufficient to drive functional recovery following a controlled cortical impact (CCI) to the MC,7,8 and this may be, in part, a result of more limited neural plasticity following CCI. 4 Thus, it was unknown if CS combined with RT would enhance behavioral function following CCI as it has following ischemia.
We investigated whether CS combined with RT would enhance the efficacy of RT and enhance the integrity of motor functioning after CCI. A total of 17 male Long-Evans rats (~5 months old) received a CCI centered over the caudal forelimb area (CFA) and then either received CS during RT (CS+RT; n = 9) or RT alone (n = 8) on a tray-reaching task. We used CS parameters previously found to be most effective following experimental stroke.2,6,9 We also investigated whether CS+RT compared with RT alone increased cortical forelimb movement representation area, as previously found in stroke models.2,3
As previously described, 9 rats were trained prior to surgery to criterion on the single-pellet-reaching task with their preferred limb, then received a CCI of the CFA opposite the preferred reaching limb and epidural electrodes were implanted over remaining MC (see Supplemental Methods online).4,7
Tray reaching was used as RT, which required that animals reach for ~200 pellets placed on an inclined tray or for 20 minutes, whichever was first.7,9 All rats were attached to stimulator cables and placed into the reaching chamber. Only CS+RT animals received continuous stimulation delivered at 50% of that week’s movement threshold, defined as the minimal current necessary to produce visible movements of the forelimb, head, or neck. No stimulation was ever delivered to the RT-alone group, and movement threshold was never assessed. Impaired forelimb function was probed with the single-pellet-reaching task6,9 on days 8 and 9 post-CCI and then over 2 consecutive days after each week’s RT. Data were analyzed as the mean per week of the percentage success—Pellet placed in mouth/Total reaches—using repeated-measures ANOVA. All probe tests were performed without CS.
After 9 weeks of RT, standard intracortical microstimulation (ICMS) mapping2,3,10 was used to reveal the organization of movement representations in the remaining MC of the injured hemisphere. The rostral forelimb area (RFA) and CFA were exhaustively mapped. Animals were then transcardially perfused with phosphate-buffered saline and 4% paraformaldehyde. Eight 50-µm coronal sections, 600 µm apart, through the cerebrum were Nissl stained for lesion volume analysis, calculated as follows: (Area of intact cortex − Area of injured cortex) × Distance between sections.
As seen in Figure 1, CCI over the CFA greatly impaired performance on the single-pellet-reaching task in both groups compared with preinjury [t(1, 16) = 8.37; P < .001]. All animals significantly improved in reaching success after 9 weeks of RT. However, the CS+RT group had a significantly greater magnitude of improvement over time compared with the RT-alone group [Day × Group: F(10, 150) = 2.39; P = .012]. There was a significant effect of Day [F(10, 150) = 18.37; P < .001)] but no significant Group effect [F(1, 15) = 1.93, P > .05]. Post hoc analysis indicated that there were significant differences between groups on weeks 4 and 5: P < .05. Movement thresholds significantly decreased from week 1 (3.57 ± 0.61) compared with week 9 (2.06 ± 0.18) in the CS+RT group [t(1, 8) = 2.622; P = .013] and fell within our previously reported range following stroke.2,6,9
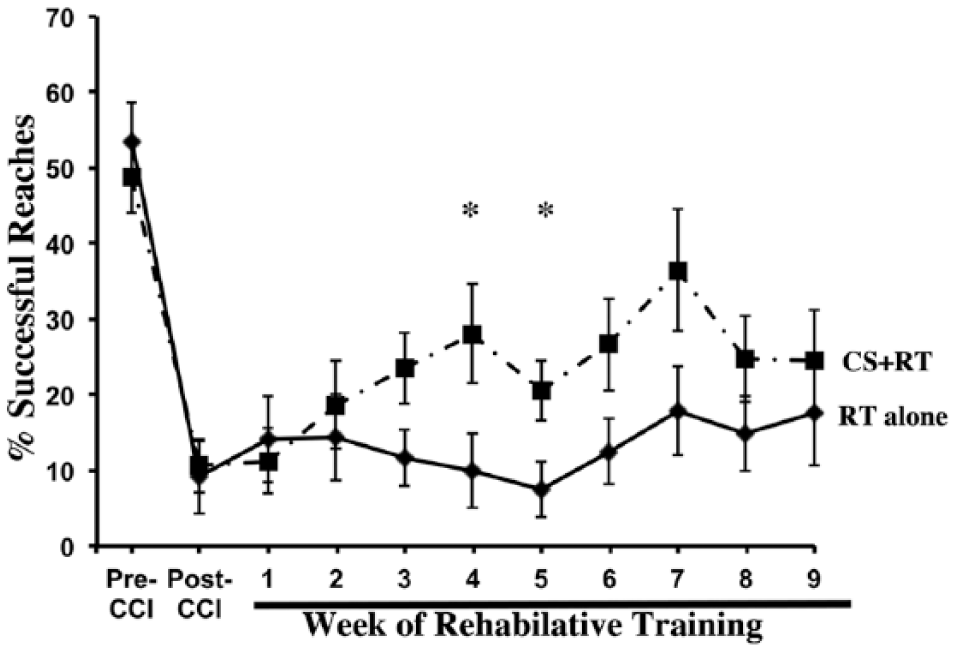
Following a unilateral CCI centered over the CFA, reaching performance with the impaired forelimb drastically declined. All animals improved after 9 weeks of RT, although the improvement was far from preinjury levels. CS+RT significantly increased reaching performance compared with RT alone. Data are means ± SEM. *P ≤ .05.
As seen in Figure 2, CS significantly increased the area of the remaining MC (CFA + RFA) from which wrist [F(1, 15) = 7.48; P = .015], but not elbow [F(1, 16) = 0.424; P > .05], movements were evoked at ≤100 µA compared with RT alone. However, the mean wrist ICMS-evoked movement thresholds were not significantly different (P > .05) between CS+RT (61.1 ± 4.22) and RT alone (63.0 ± 4.0). The RT-alone group had no CFA and only sparse RFA wrist representation even at current levels above the standard ICMS upper boundary of 60 µA, further suggestive of profoundly disrupted MC function. Only in animals that received CS+RT were we able to elicit wrist movements below 60 µA, and these thresholds were relatively high compared with our stroke CS+RT study. 2 There were no significant differences in injury volume between CS+RT (19.8 ± 3.85) and RT alone (22.5 ± 4.22): F(1, 13) = 0.23; P = .64.
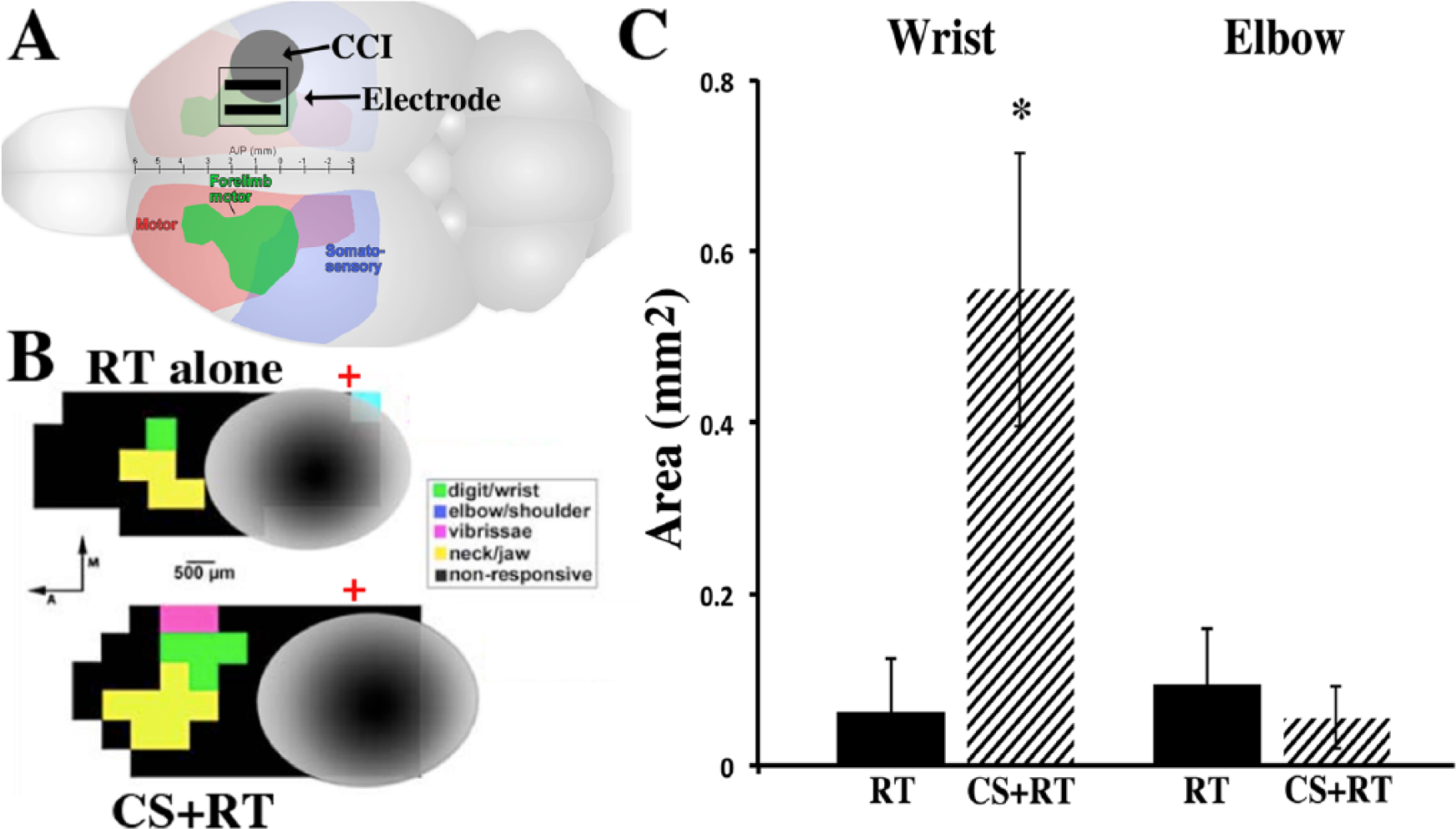
A. As seen in the rat brain schematic, CCI (gray circle) was induced over the forelimb overlap area of the sensory motor cortex (arrow). Epidural electrode contacts (parallel black bars) were placed rostral and medial to the injury over the remaining forelimb area (CFA and RFA, in green). B. Representative ICMS-derived motor maps surrounding injured tissue (gray) following 9 weeks of RT alone (top) and CS+RT (bottom). Green squares are wrist, yellow are jaw, pink are whisker, and black are nonresponsive sites. Red crosses denote bregma. C. CS significantly increased the total area of wrist movement representation compared with RT alone, but not the elbow movement representation area. Data are means ± SEM. *P < .05.
The organization and size of ICMS-derived motor maps are thought to reflect intracortical synaptic connectivity contributing to the movement. 10 CCI profoundly diminished forelimb representations in the remaining forelimb territory surrounding the contusion as compared with noninjured animals in other TBI studies and following CFA-focused ischemic lesions and CS+RT. 2 Although we do not have direct evidence in this study, presumably the loss of wrist representation reflects loss and dysfunction of forelimb movement circuits. 10 This is consistent with the lack of robust improvements over 9 weeks of RT with or without CS. Following experimental stroke of the CFA, robust improvements occur following 10 days to 3 weeks of CS+RT compared with no training or RT only.2,3,6,9 However, the greater wrist representation area in CS+RT compared with RT alone likely reflects greater functional integrity of the surviving circuits and/or better interactions within these circuits contributing to forelimb movements,2,3,10 consistent with its association with motor improvements.
Although these findings do support the hypothesis that CS following TBI may be a beneficial adjunctive treatment, and CS was able to induce movement thresholds at the levels seen after stroke, CS+RT left considerable room for further improvement. The lack of stronger effects may be a result of injury severity, because we have previously shown that CS effectiveness is reduced in severely impaired rats with ischemic CFA infarcts. 6 We have also previously found that more robust rehabilitation combinations (compared with stroke) are needed to drive motor improvements after similar CCIs.4,7 The CS parameters used in this study were optimized in stroke models, and it is quite possible that a different frequency and/or intensity of CS and its combination with more intense rehabilitation would drive better motor outcomes and motor cortical reorganization.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This material is the result of work supported by NINDS NS065866 (DLA, TAJ, DAK).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
