Abstract
The relationship of poststroke spasticity and motor recovery can be confusing. “True” motor recovery refers to return of motor behaviors to prestroke state with the same end-effectors and temporo-spatial pattern. This requires neural recovery and repair, and presumably occurs mainly in the acute and subacute stages. However, according to the International Classification of Functioning, Disability and Health, motor recovery after stroke is also defined as “improvement in performance of functional tasks,” i.e., functional recovery, which is mainly mediated by compensatory mechanisms. Therefore, stroke survivors can execute motor tasks in spite of disordered motor control and the presence of spasticity. Spasticity interferes with execution of normal motor behaviors (“true” motor recovery), throughout the evolution of stroke from acute to chronic stages. Spasticity reduction does not affect functional recovery in the acute and subacute stages; however, appropriate management of spasticity could lead to improvement of motor function, that is, functional recovery, during the chronic stage of stroke. We assert that spasticity results from upregulation of medial cortico-reticulo-spinal pathways that are disinhibited due to damage of the motor cortex or corticobulbar pathways. Spasticity emerges as a manifestation of maladaptive plasticity in the early stages of recovery and can persist into the chronic stage. It coexists and shares similar pathophysiological processes with related motor impairments, such as abnormal force control, muscle coactivation and motor synergies, and diffuse interlimb muscle activation. Accordingly, we propose a new definition of spasticity to better account for its pathophysiology and the complex nuances of different definitions of motor recovery.
Introduction
Spastic hemiparesis (spasticity and weakness on one side of the body) is the hallmark motor impairment after a stroke. Other features include, but are not limited to, associated reactions and disordered motor control. Weakness occurs immediately after stroke, but spasticity typically emerges later. Whereas strength may improve, spasticity gradually worsens over time. Motor recovery at 3 months or 6 months poststroke can be predicted from the initial degree of impairment and is largely caused by spontaneous repair and restoration of the stroke lesion, in spite of standard inpatient and outpatient therapies.1-3 It is also widely recognized that spasticity and weakness interact, leading to contractures, abnormal joint postures, and limited movement, further amplifying difficulty in performing activities of daily living. 4 However, recent studies showed that management of poststroke spasticity with botulinum toxin can effectively reduce poststroke spasticity but does not routinely improve volitional movement and function.5-7 Alternatively, machine-assisted intensive therapy can improve voluntary motor function but has no effect on spasticity. 8 This leads to the question: does spasticity truly interfere with motor recovery after stroke?
Motor recovery has different meanings for clinicians, stroke survivors, public health officials, and scientists. 9 Spasticity is also commonly used as an umbrella term to describe a clinical phenomenon of increased resistance to passive stretch. There has been considerable progress in understanding the underlying pathophysiology of poststroke spasticity in recent years. In this article, we will focus on the following topics to better understand the relations between poststroke spasticity and motor recovery:
understand different definitions of motor recovery;
review the pathophysiology of poststroke spasticity;
propose a new definition of poststroke spasticity;
enumerate how spasticity can interfere with motor recovery from acute to chronic stages of stroke; and
list rehabilitation approaches to manage poststroke spasticity to improve motor function.
Understanding Different Definitions of Motor Recovery
Motor recovery after stroke can be defined in various ways. To neurorehabilitation scientists, motor recovery means “true recovery” of motor function, characterized by a return of some or all motor behaviors to their normal, prestroke state.9-11 Frequently, such recovery refers to successful execution of a motor task using the same limbs or end effectors in the same manner as it was performed before stroke via restitution or repair of damaged neural tissues. 10 Because of the redundancy of the human motor system, 12 stroke survivors are often able to use alternative muscle combinations or limb movements to accomplish the same task using different motor patterns—that is, compensation.9-11 For example, instead of extending the elbow joint to grab a bottle of water in a reaching task, a stroke survivor may accomplish the task by leaning his/her trunk forward because of the reduced range of limb motion, which is routinely limited by shoulder and elbow flexor spasticity. From a functional standpoint, the task is completed successfully, although it is accomplished through compensatory movements. To improve the ability to perform such a functional task—that is, functional recovery—is one of the primary goals of stroke rehabilitation.
Among the important goals of stroke rehabilitation are to improve body function and to maximize functional independence, participation, and social reintegration via coordinated delivery of therapies and interventions in an interdisciplinary approach. As defined by the International Classification of Functioning, Disability and Health, in the motor domain, common impairments of body functions after stroke and resultant limitations in activities are listed in Figure 1. According to this model, improvement in performance of any activity, for example, walking, as a result of therapy or intervention (Figure 2), is considered “recovery” of a particular body function or activity, although a patient still walks in an abnormal pattern with compensatory mechanisms. From a functioning point of view, this definition of motor recovery—that is, functional recovery, characterized by compensation without neural restoration—is shared by payers, clinicians, stroke survivors, and their family members and caregivers. 13
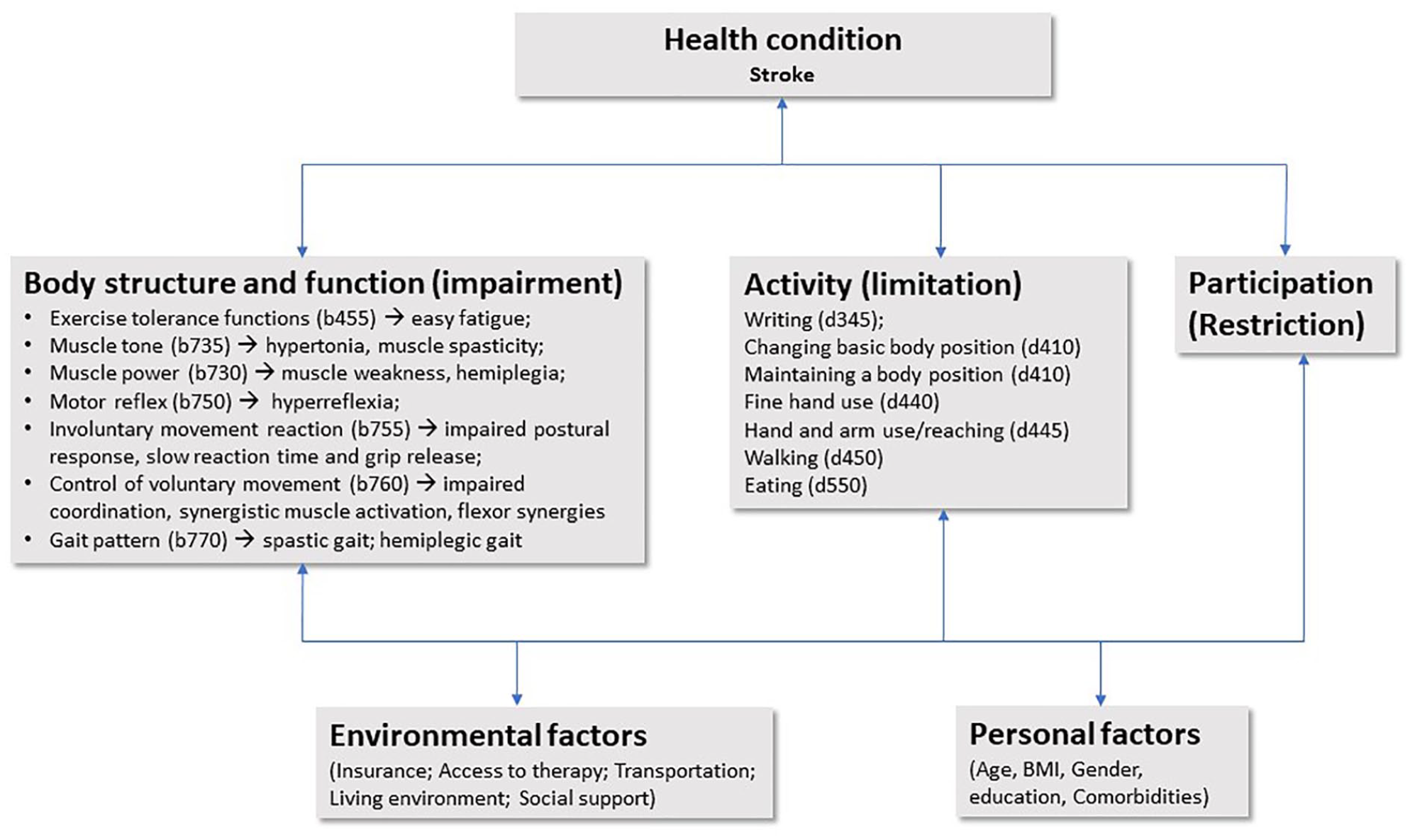
Examples of motor impairments and function limitations after stroke within the International Classification of Functioning, Disability and Health (ICF) model.
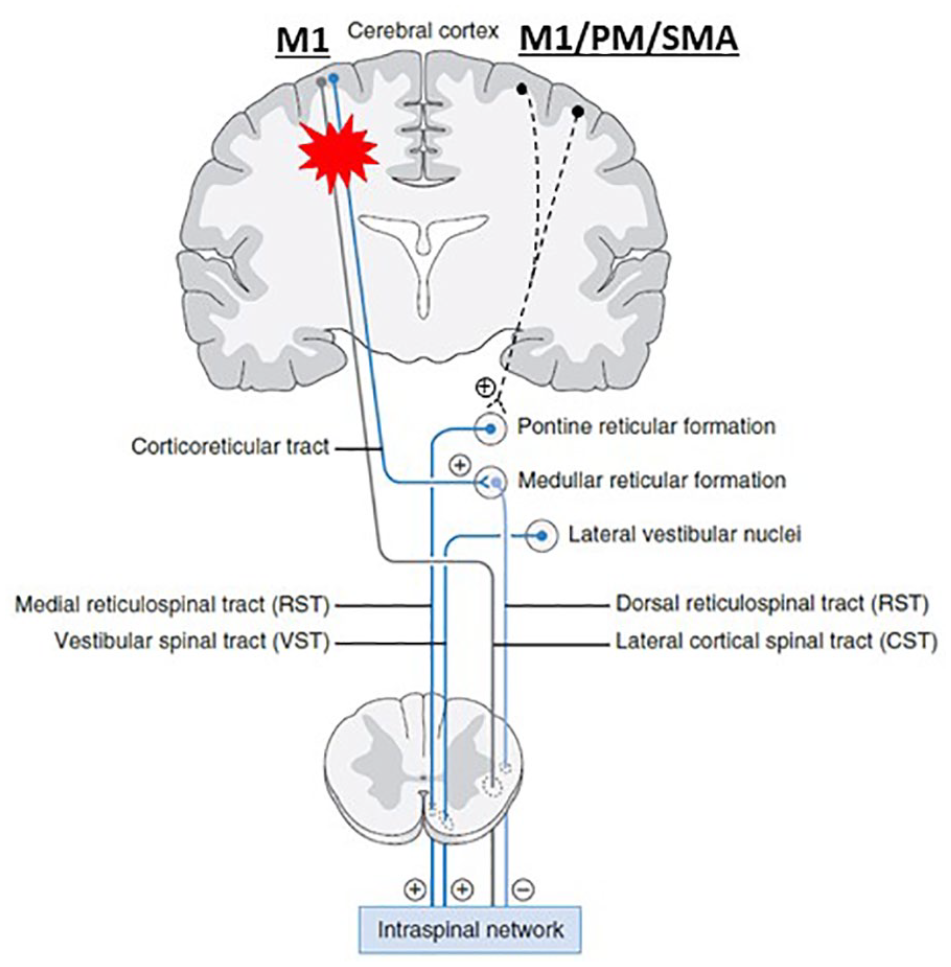
Modified from Li et al 24 on the unifying hypothesis. The pontomedullary reticular formation (PMRF) in the pons receives diffuse inputs primarily from the ipsilateral motor cortex, including the premotor (PM) and supplementary motor area (SMA) and primary motor cortex (M1), and descends ipsilaterally. This medial cortico-reticulo-spinal tract (cortico-RST) provides excitatory descending inputs to spinal motor neurons. The PMRF in the medulla receives inputs primarily from the contralateral motor cortex. This dorsolateral cortico-RST provides inhibitory descending inputs to the spinal motor circuitry. When damages occur to the motor cortex and its descending CST and cortico-RST after stroke on one hemisphere (red asterisk), their output signals diminish. Subsequently, the medial cortico-RST excitability from the contralesional hemisphere becomes unopposed, gradually upregulated, and hyperexcitable. Eventually, spinal motor neurons and the stretch reflex circuitry are hyperexcitable or may be spontaneously firing. (+): Excitatory; (−): inhibitory. Note: other descending pathways are not illustrated. They are considered either insignificant or connected with RST.
The Pathophysiology of Poststroke Spasticity
Muscular hypertonia is an umbrella term for a common clinical phenomenon of increased joint resistance to passive stretch. Hypertonia can have different neural and nonneural components, whereas spasticity is primarily of neural origin.4,14 A still widely used definition is the following:
With research evidence and progress, understanding of spasticity has progressively evolved. This earlier definition has been challenged and alternative definitions have been proposed.16,17 For example, Pandyan et al
16
proposed this definition:
This definition expands spasticity as a disorder of sensorimotor control, rather than simply a phenomenon of increased joint or limb resistance (muscle tone) or a motor disorder. Furthermore, it describes its clinical presentation of intermittent or sustained muscle overactivity, rather than attributing the underlying mechanism solely to stretch reflex hyperexcitability.
Our understanding of the pathophysiology of poststroke spasticity has advanced significantly over the past 3 to 4 decades.18-24 The damage to the motor cortex and to its descending corticospinal tract (CST) causes muscle weakness (usually hemiparesis) immediately after stroke and may subsequently lead to incoordination and joint immobilization. Neuroplasticity occurs after stroke as well. Spontaneous neuronal recovery and further neural repair in the lesioned hemisphere contribute to motor recovery in both the acute and subacute phases—that is, true motor recovery. 9 On the other hand, lesions of corticobulbar pathways to the medullary bulge and medullary reticular inhibitory centers are usually associated with lesions of motor cortices and/or the descending CST because of their anatomical proximity. As a result, bulbospinal pathways, particularly the medial reticulospinal (RS) pathways, gradually becomes hyperexcitable as a result of loss of supraspinal inhibition (Figure 3). This is mainly a phenomenon of disinhibition or unmasking effects. RS hyperexcitability provides unopposed excitatory descending inputs to spinal stretch reflex circuits, resulting in elevated excitability of spinal motor neurons and of stretch reflex circuits and hence the common observation of velocity-dependent and length-dependent increase in resistance to passive stretch.18,22,25 Recent animal studies have shown that medial RS pathways receive ipsilateral cortical inputs from the contralesional cortex.26-28 These findings have led us to propose a unifying hypothesis to better understand the relations among weakness (hemiparesis), spasticity, and related motor impairments (Figure 3). 24 Specifically, weakness occurs immediately after stroke as a result of the lesioned motor cortex and its descending CST, whereas spasticity and related motor impairments emerge and evolve when hyperexcitability of ipsilateral cortico-reticulo-spinal pathways from the contralesional cortex develop secondary to abnormal neuroplasticity. Spasticity and related neuromuscular impairments are parallel manifestations of this common pathophysiological process. Yet an alternative view was hypothesized a few years ago that damage to the pyramidal tracts is necessary and sufficient for the production of spasticity. 29
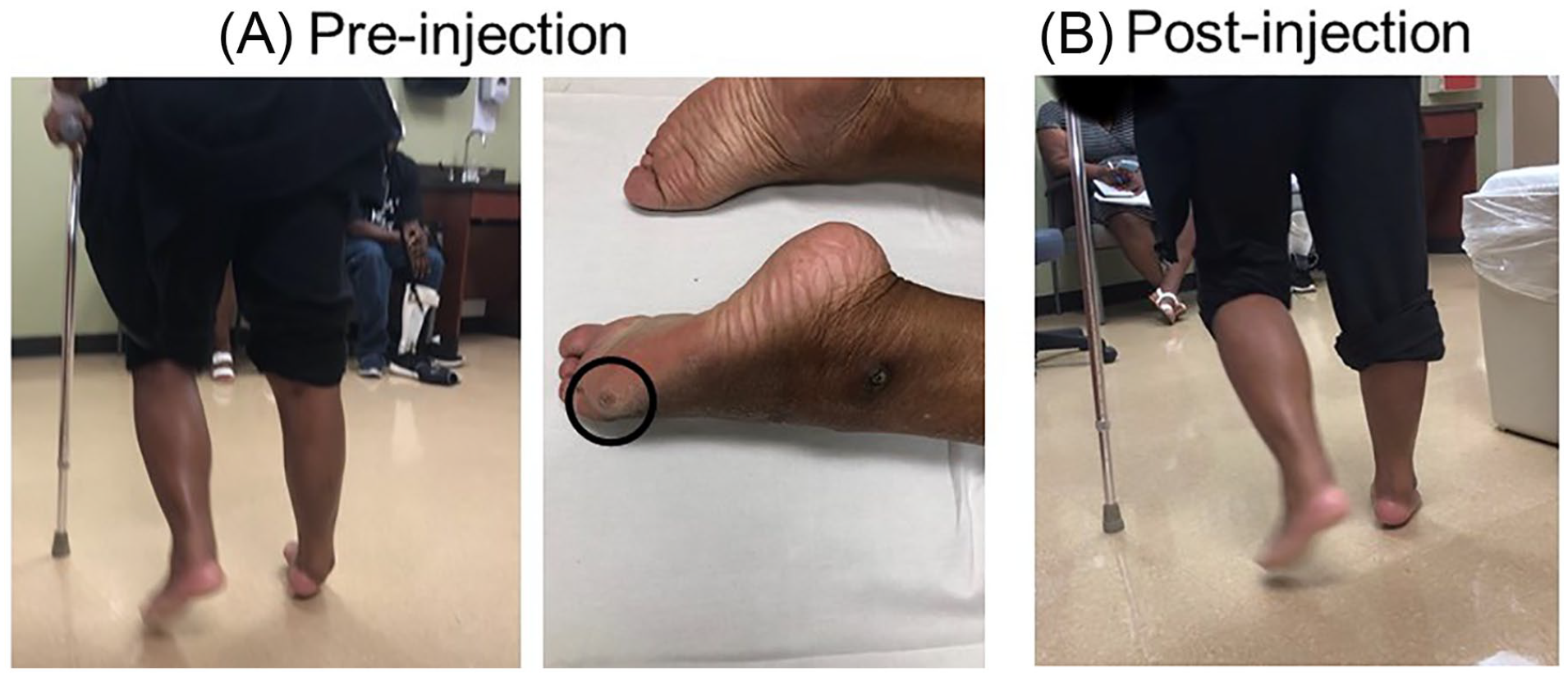
A stroke survivor with right spastic hemiparesis and right ankle equinovarus. She received phenol blocks to tibial nerve motor branches to tibialis posterior muscles and calf muscles and to peroneal nerve motor branches to tibialis anterior muscles. The black circle indicates a callus
A New Definition of Poststroke Spasticity
To reflect these advances, and to better understand the underlying pathophysiology of poststroke spasticity and its relations with other parallel neuromuscular impairments, we propose a new definition of poststroke spasticity:
We should also point out that spasticity is manifested only in resting muscles, whereas the other related motor impairments arise during activation.
Spasticity and Its Interference With “True” Motor Recovery
This new definition advances our understanding of the pathophysiological basis of spasticity and of other associated motor impairments and their interactions. In essence, both spasticity and other motor impairments are clinical presentations and readouts of the common process of abnormal neuroplasticity. This new definition helps elaborate on spasticity and its interference with “true” motor recovery throughout the course of recovery. The interference with true recovery in the early stages is better understood from a longitudinal view of poststroke spasticity. In the subacute and chronic stages, the interference is manifested in different aspects of motor behavior, including abnormal force control, abnormal muscle coactivation, synergistic muscle activation, interlimb coupling, and overall neuromechanical consequences.
A Longitudinal View of Poststroke Spasticity
Weakness for voluntary movement occurs at stroke onset, yet true motor recovery commences almost immediately because the central nervous system undergoes repair in the acute and subacute phases. The severity of the initial motor impairment can be used to predict the amount of recovery that could be expected at 3 months after stroke—that is, proportional recovery.1-3 As abnormal neuroplasticity develops, spasticity starts to emerge. The median time to detect spasticity, as measured by the modified Ashworth Scale ≥1, is 34 days after stroke onset. 30 But it is only seen in a small portion (19%) of stroke survivors with moderate to severe motor impairments by 3 months poststroke. 31 However, the presence of spasticity in the early phase (<4 weeks), even very mild, is an important predictor of severe spasticity and its consequences at 6 months 32 or after 12 months. 33 It is important to note that spontaneous spasticity reduction rarely occurs. It rather evolves and becomes more severe over time. 33 By 12 months after stroke, spasticity is present in 43.2% of stroke survivors. 34 In chronic stroke survivors with moderate and severe motor impairments, the prevalence of spasticity is up to 97%. 35 In the course of emergence and development, spasticity interacts with weakness, and it can amplify the problem of weakness and results in disordered motor control.19,36
Along with neuronal repair and true motor recovery in the acute and subacute stages, 9 spasticity starts to emerge and evolve as abnormal neuroplasticity develops. Even if spasticity is suppressed or treated aggressively during these early stages, no correlated functional improvement in a later stage has been systematically reported.37,38 Therefore, in these acute and subacute stages, spasticity does not appear to interfere with functional recovery. In these early stages, stroke survivors with incomplete motor recovery and spasticity execute motor tasks, such as writing, reaching, and walking, involving different end effectors (muscles) and abnormal movement patterns. According to this definition of “true” motor recovery, which often refers to execution of motor tasks using the same limbs or end effectors in the same manner as before stroke,9-11 spasticity does interfere with “true” motor recovery, even in the early stages.
Abnormal Force Control
Spasticity often presents clinically as sustained or intermittent muscle overactivity at rest and during voluntary movement, in addition to velocity-dependent increase in resistance at rest.16,39-41 Rymer and colleagues have demonstrated that spinal motor neurons are hyperexcitable or spontaneously firing in stroke survivors with spastic paresis. Spontaneous firing increases with the level of voluntary muscle contraction,39,40 and it is sustained after voluntary contraction ends. 41 These findings suggest that spasticity-related muscle overactivity and underlying spontaneous motor neuron firing are not under voluntary control and could be viewed as a reflection of “motor noise.” Although these involuntary activities are reflected in electromyographic (EMG) recordings, they cannot contribute effectively to torque production, 42 thus resulting in central activation deficits. 43 As such, they may contribute to abnormal EMG-force relations.44-46 Furthermore, they may make it difficult for stroke survivors to initiate and terminate a hand grip on the spastic paretic side.47,48 It is also difficult for them to maintain a constant force output on the spastic-paretic side as compared with the contralateral side.41,49-51 The weaker the spastic muscle, the greater the force variability that was observed. 41 Spastic muscles are weak muscles. Stroke survivors need to exert more effort to execute the same tasks during activities of daily living. They report less endurance with easy fatigue. 52
Abnormal Coactivation
Spasticity and associated involuntary activation alter the normal activation patterns between agonists and antagonists of a joint and lead to inappropriate coactivation. 53 During intentional finger extension, for example, involuntary activation of spastic finger flexors often overcomes weak finger extensor strength, resulting in paradoxical finger flexion. 54 Inappropriate coactivation occurs during isometric contraction of elbow and shoulder muscles 55 and during passive stretch of elbow flexors. 56
This abnormal coactivation potentially further impairs motor learning. In a recent study, 57 participants were instructed to make rapid horizontal elbow extension movements with various loads. Accuracy of reaching movements was significantly decreased when the spasticity zone (ie, triggering reflex responses in biceps muscles) was included; however, it was similar in stroke individuals when the spasticity zone was not included as compared with performance in healthy individuals. The results suggest that spasticity may obscure motor learning ability after stroke.
Synergistic Activation
Certain patterns of involuntary activation of spastic muscles across multiple joints are observed in stroke survivors with spastic hemiparesis. Most commonly seen patterns are flexor synergy in the upper extremity and extensor synergy in the lower extremity. 58 In the flexion synergy, increase in shoulder adductor activation is associated with involuntary activity in elbow, wrist, and finger flexors. Dewald et al 55 provide evidence that the level of synergistic flexion in the arm is related to task-dependent involvement of the contralesional cortex and ipsilesional RS pathways.59-63 The multijoint synergistic activation makes daily tasks difficult, such as reaching. As compared with healthy controls, stroke survivors take more time to reach out to the target within their workspace but with increased dispersion and segmentation on their path and with shorter amplitude. Severity of spasticity was also found to correlate with reaching performance. 64 Similarly, multijoint synergistic activation patterns are observed in the lower extremity during walking. Normally, leg muscles are coordinated and organized into functional groups, or modules, according to their biomechanical functions during different types of locomotor activities. These modules can be recruited in a variety of combinations to meet different locomotion and balance needs.65-67 In stroke survivors with spastic hemiparesis, these modules are merged into fewer motor synergies, leading to less flexibility and slower speed during walking 68 and poststroke hemiplegic gait. 69
Interlimb Coupling
Synergistic activation is attributed to diffuse projections of RS pathways to multiple muscles. Diffuse projections and associated activations are also seen in muscles between limbs. As compared with the supine position, elbow, wrist, and finger flexor spasticity increases significantly during standing. 70 Wrist and flinger flexor spasticity significantly increases during walking than standing. The between-limb coupling is reflected by the result that voluntary finger flexion produces significant activation of the rectus femoris muscle. 71 The interlimb coupling is also observed between 2 arms. When a stroke survivor voluntarily flexes the elbow and makes a fist on the contralateral nonimpaired arm, the spastic paretic arm goes into elbow flexion and finger flexion as well. 72 This phenomenon of motor overflow is minimum during unilateral elbow flexion on the spastic-paretic side. This is potentially attributable to activation of cortico-reticulo-spinal pathways ipsilaterally from the contralesional cortex during activation of the nonimpaired side (see Figure 3). 24
Neuromechanical Consequences
Spasticity can worsen the consequences of weakness.19,36 Weakness often leads to immobility. Spasticity-related involuntary muscle overactivity and synergistic activation tend to immobilize joints in abnormal positions. In turn, this potentiates development of tendon shortening and muscle contracture over time, thus leading to abnormal posture—for example, stereotypical presentations of clenched fist and ankle equinovarus deformity.
The abnormal posture of one joint can have mechanical effects on other joints through the kinetic chain, particularly in the leg during walking. The ankle joint in plantarflexion during the stance phase relocates the ground reaction force further anterior to the knee joint and the hip joint, subsequently causing knee and hip extension to keep the ground reaction force close to the center of hip and knee joints. In turn, this kinetic chain position potentiates and worsens spasticity of knee and hip extensors. To clear the foot with extended hip and knee joints, the person with spastic hemiparesis has to circumduct the spastic-paretic leg during the swing phase. This example shows the mechanical consequence of muscle spasticity during “typical” hemiplegic gait. 69 However, clinical presentation of poststroke gait varies depending on severity of spasticity, involvement, and distribution of spastic muscles.69,73,74 Another example is shown in Figure 2A. Spastic equinovarus of the ankle and foot localizes weight-bearing on the lateral foot, undoubtedly causing calluses and pressure sores over time. In the upper extremity, the scapula could be restricted in a more laterally rotated position because of spasticity of rhomboid muscles. The acromion and glenoid cause a mechanical stop for the humeral head during shoulder forward flexion. 75 This scapular dyskinesia makes common daily activity difficult—for example, feeding.
Rehabilitation Strategies
To reiterate, “true” motor recovery is associated with spontaneous neural repair, whereas spasticity and related neuromuscular impairments emerge and evolve from the same pathophysiological process of abnormal neuroplasticity in the early stages. This process plateaus in the chronic stage. To date, there is no effective intervention to correct this process of abnormal plasticity. On the other hand, spasticity and concomitant disordered motor control and weakness can continue to interact in a vicious cycle, resulting in abnormal joint positions and other mechanical consequences. Functional recovery could be achieved if spasticity is adequately managed and the cycle is interrupted (Figure 2). Therefore, motor rehabilitation strategies include the following:
focusing on neural repair in the early stages to facilitate “true” motor recovery;
strengthening weak muscles throughout the course of recovery; and
adequately and appropriately managing spasticity to break the vicious cycle, thus improving motor function.
It has been recently shown that successful management of spasticity in the very early stage after stroke does not facilitate functional recovery. 38 Therefore, we will focus on rehabilitation strategies to manage spasticity for motor recovery in the chronic stage.
There are many different goals to reduce spasticity—for example, to preserve range of motion and prevent muscle contracture,76,77 reduce pain,78-80 improve hygiene,78,79 and improve consciousness in patients with disorders of consciousness. 81 There are a variety of treatment options, including physical modalities, oral medications, botulinum toxin injection and phenol neurolysis, and surgery. 58 It is important to highlight that spasticity reduction itself does not equate to reduction of motor impairments or improvement of motor function. 7 However, appropriate management of spasticity and its associated neuromuscular impairments could improve motor function in a subgroup of patients, as discussed in the next sessions.
Restoration of Mechanical Alignment
As described above, spasticity can have neuromechanical consequences. There are systematic approaches to correcting mechanical malalignment, particularly for the lower extremity. 73 Restoration of mechanical alignment is usually accompanied with functional improvement, such as improved walking pattern and speed.69,73 Figure 2 is an example. Severe spasticity of ankle invertors (tibialis anterior muscles, tibialis posterior muscles, and calf muscles) causes spastic equinovarus of the right ankle and foot joint. This abnormal ankle joint posture shifts weight-bearing to the lateral foot, and a callus developed as a result (black circle in Figure 2). Immediately after phenol neurolysis of motor branches of the tibial and fibular nerves, her right ankle equinovarus was corrected, and her gait pattern was much improved. In addition to focal management with nerve blocks or botulinum toxin injection, spasticity reduction and restoration of mechanical alignment to improve gait and ambulation can also be achieved through intrathecal baclofen pump management at the regional level.82,83
Neuromuscular Reeducation
It is important to understand and differentiate that weakness and spasticity are mediated by different mechanisms. 24 Inappropriate muscle coactivation and imbalanced strength between overactive spastic muscles and weak antagonists could result in disordered motor control and impaired function. Focal spasticity management could provide spasticity reduction and neuromuscular reeducation to improve motor control and function. Difficulty in opening and closing the hand is likely a result of weak finger extensors and spastic and weak finger flexors. Coactivation of spastic finger flexors often results in paradoxical finger flexion during finger extension attempts. 54 When finger flexor spasticity is reduced through oral medication, hand opening and closing become smoother and more efficient. 84 Similar results are seen when botulinum toxin is injected to finger flexors for spasticity reduction. 47 A combination of finger flexor spasticity reduction via botulinum toxin injection and finger extensor strengthening via electrical stimulation provides better neuromuscular retraining and thus yields hand function improvement. 85 When elbow, wrist, and finger flexor spasticity are reduced, stroke patients are able to perform reaching tasks better. 86 Similarly, focal spasticity management of overactive muscles for the shoulder joint also improves muscle activation patterns and results in better shoulder joint position and increased active range of motion.75,87
Concluding Remarks
In summary, the relations between motor recovery and spasticity after stroke depend on the definition of motor recovery. Spasticity and related motor impairments coexist and are different manifestations of similar pathophysiological origins. The disorder in threshold control is one way to describe such pathophysiological processes.88-90 As a result, stroke survivors execute motor tasks with disordered motor control in the presence of spasticity. Therefore, spasticity interferes with true motor recovery from acute to chronic stages. Although spasticity reduction does not affect functional recovery in the acute and subacute stages, appropriate management of spasticity could restore mechanical alignment and/or correct neuromuscular activation patterns, thus allowing the opportunity to improve motor function—that is, functional recovery—in the chronic stage of stroke. The potential interference of spasticity with true and functional motor recovery should be recognized and addressed in future stroke recovery research.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: W. Zev Rymer is a member of the American Society for Neurorehabilitation.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
