Abstract
Introduction
The pattern and timing of innate cyclic behaviors such as walking are generated by neuronal networks, or central pattern generators (CPGs), in the spinal cord. In nonhuman primates 1 and other vertebrates2,3 CPGs produce well-coordinated movements that are highly reproducible. Humans are thought to have similar organization of spinal circuitry.4-9 In intact animals, CPGs are thought to be activated by supraspinal centers. Spinal cord injury interrupts the pathways linking the supraspinal and spinal centers, thereby disrupting activation of the spinal locomotor CPG and contributing to the impairment or loss of walking function. In individuals with spinal cord injuries (SCIs), interventions targeting the spinal CPG have been met with some success. Epidural electrical stimulation over the lumbosacral spinal cord elicits cyclic stepping patterns in individuals with motor-complete SCIs (MCSCIs).9,10 This input appears to increase the excitability of spinal motor circuits, thereby increasing the effectiveness of descending commands, allowing some voluntary movement in those with MCSCIs 10 and augmenting walking function in those with motor-incomplete SCIs (MISCIs). 11
In animal models, afferent input has a powerful influence on the timing and robustness of CPG output.2,12,13 Afferent input appears to influence locomotor pattern-generating circuitry in humans as well.4,14 Vibration applied to the belly of the thigh muscles elicits involuntary, unilateral, step-like movements in nondisabled (ND) individuals in a side-lying position. 14 Vibration to the contralateral thigh muscles elicits similar movements. When ND people are suspended in an upright position, muscle vibration applied to both legs elicits bilateral step-like movements. 15 Furthermore, continuous vibration applied to the thigh muscles increases walking speed in ND individuals, with effects dependent on direction of progression and vibration placement. 15 Further evidence supporting the utility of vibration in influencing stepping behavior comes from studies demonstrating that participation in an intervention that includes whole-body vibration is associated with increased walking speed in individuals with Parkinson disease 16 and MISCIs 17 and in elderly ND individuals. 18 This evidence suggests that vibration excites central structures, creating conditions that activate locomotor-related circuits. 14
We undertook this study to determine whether step-like responses are (1) evoked with muscle vibration in those with SCI, (2) similar among individuals with MCSCIs and MISCIs and ND individuals, (3) influenced by site of vibration, (4) influenced by locomotor training, and (5) stable over time. Because afferent and CPG-related circuits remain intact after SCI, we hypothesized that vibration would elicit step-like behaviors from those with SCI as has been shown to be the case in those with ND.14,15 However, based on evidence that the spinal circuits are strengthened through repeated activation,19-24 we hypothesized that responses would be associated with the extent to which the locomotor CPG was used and, therefore, that ND individuals would have the most consistent/robust responses, whereas those with MCSCIs would have the least consistent/robust responses. Likewise, we hypothesized that participation in locomotor training would result in increased responsiveness. We further hypothesized that vibration to muscles that are reciprocally activated during walking (ie, quadriceps or hamstring muscles) would elicit more consistent and robust responses compared with vibration of a muscle that functions primarily to stabilize the pelvis (ie, tensor fascia latae [TFL]).
Methods
Participants
A total of 19 individuals with SCI and 8 ND individuals (11 women, 16 men; ages 19-65 years) participated in this study. All participants gave written and verbal informed consent according to the protocol of a study approved by the Human Studies Research Office at the University of Miami Miller School of Medicine. For those with SCI, the inclusion criterion was chronic (≥1 year duration) SCI at or above the level of T10. American Spinal Injury Association (ASIA) motor and sensory scores 25 and ASIA Impairment Scale classification 26 were evaluated by a physical therapist not otherwise involved in the study.
Participants were categorized according to lower-extremity motor function status. The ND group (n = 8) included those with no known neurological impairment. The MCSCI group (n = 6) included those who were unable to voluntarily activate any lower-extremity muscle. The MISCI group (n = 13) included those who were able to voluntarily activate some lower-extremity muscles. Participants with MISCIs were further categorized into 2 subgroups. One MISCI subgroup (n = 6) included those who were scheduled to participate in body-weight-supported locomotor training 5 d/wk for 10 to 12 weeks. Details related to the locomotor training protocol and outcomes are available elsewhere. 27 The other MISCI subgroup included individuals who did not participate in the locomotor training intervention (n = 7). Participant demographic data are given in Table 1.
Participant Demographics
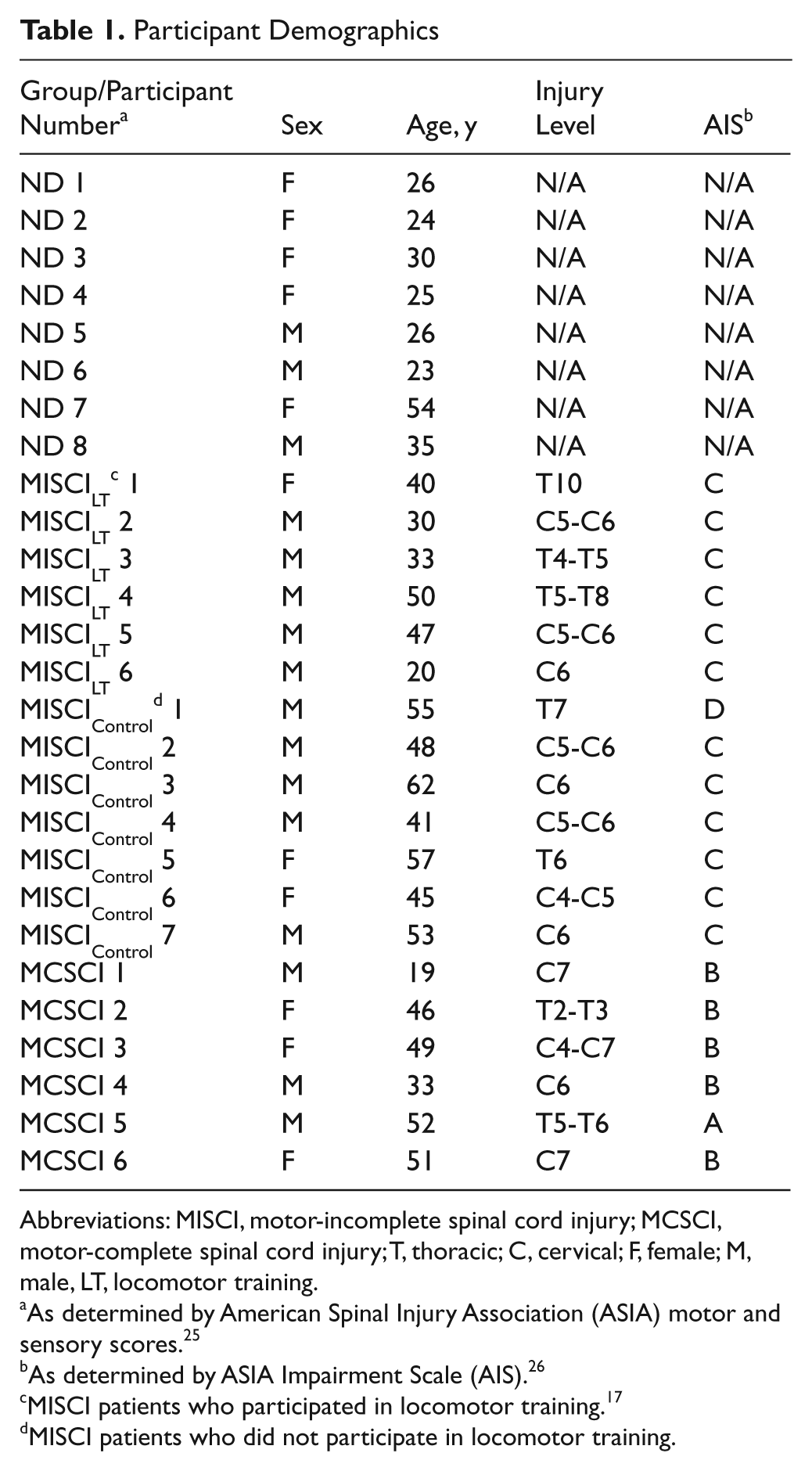
Abbreviations: MISCI, motor-incomplete spinal cord injury; MCSCI, motor-complete spinal cord injury; T, thoracic; C, cervical; F, female; M, male, LT, locomotor training.
As determined by American Spinal Injury Association (ASIA) motor and sensory scores. 25
As determined by ASIA Impairment Scale (AIS). 26
MISCI patients who participated in locomotor training. 17
MISCI patients who did not participate in locomotor training.
Testing Procedure
For collection of kinematic data, reflective markers were placed at the following points: C7, T10, the sacrum, on the test leg at the anterior superior iliac spine, greater trochanter, lateral knee joint line, lateral malleolus, lateral heel, and lateral fifth metatarsal head. Data were collected at 60 Hz using an 8-camera, 3D motion capture system (Peak Motus Software, Centennial, Colorado) in a calibrated test space. The weaker leg was used as the test leg in those with MISCIs who had asymmetrical lower-extremity motor scores, whereas the nondominant leg (“the leg you would use/would have used to stand on when you kick a ball”) was used as the test leg in ND individuals and those with MCSCI and MISCI who had symmetrical motor scores.
All 19 individuals with SCI and the 8 ND individuals participated in 2 test sessions separated by 10 to 12 weeks; this time period corresponded to testing sessions prior to (initial) and after (final) 10 to 12 weeks of training for the MISCI individuals in the locomotor training group. Participants were positioned in a side-lying, gravity-minimized position on a padded table similar to previous studies.14,28 All participants were secured to the table by a strap to ensure safety and to minimize movement of the trunk during leg movements. The test leg was suspended by 2 straps, 1 placed above the ankle joint and the other placed above the knee joint. Strap lengths were carefully adjusted to maintain the test leg in neutral internal/external rotation with the feet shoulder-width apart. This positioning permitted cyclic motion of the leg under conditions that reduced the influence of gravity. Participants were told only that the investigation was to assess the effects of the vibration.
A hand-held vibrator (Pro-Massager USJ-301, US Jaclean, Inc, Gardena, California) was used to deliver vibration to the test leg muscle. The vibrator had a frequency of 60 Hz and unloaded amplitude of approximately 1 mm. Each testing session consisted of 5 trials. In 2 trials, participants performed, or attempted to perform, voluntary air-stepping in the forward direction (to simulate forward walking) and voluntary air-stepping in the backward direction (to simulate backward walking). All participants, including those with MCSCIs, were asked to attempt voluntary air-stepping. In those who had voluntary lower-extremity movement, these voluntary air-stepping trials were used to ensure that the range of motion of the hip and knee joints was unrestricted in the test position. In those with MCSCIs, these trials were used to confirm the absence of voluntary lower-extremity movement. Following the voluntary air-stepping trials, 3 trials of involuntary, vibration-elicited air-stepping were recorded during vibration to each of 3 muscles: the quadriceps (Qvib), the hamstrings (Hvib), and the tensor fasciae latae (TFLvib). During the involuntary air-stepping trials, participants were instructed to relax and not to intervene if they perceived movement. The sequence of the vibration trials was randomized by muscle to counter any possible order effects. During vibration-elicited air-stepping trials, vibration was applied to the muscle belly according to previously published methods. 14 The target muscle was vibrated for 5 minutes prior to the start of kinematic data collection to ensure a steady state of the elicited response, and vibration was continued throughout the duration of the ensuing 30-s kinematic data capture period.
Data Analysis
Air-stepping parameters were calculated from measures defined by the joint markers. The hip angle was defined by the trunk and thigh segments, and the knee angle was defined by the thigh and shank segments; kinematic joint angle data were filtered using a Butterworth filter (6-Hz cutoff frequency). Hip and knee angle relative motion data from each trial were used to quantify the responses. These data have previously been used to illustrate features that distinguish intralimb coordination between normal and pathological gait 29 and to assess cycle-to-cycle consistency of intralimb coupling relationships in cyclic behaviors such as locomotion.30-33 Analysis of the consistency over multiple cycles has been suggested to offer insights into the control mechanisms underlying coordination of cyclic behavior.30,32 Individual air-step cycles were identified, with the onset of a cycle defined as the frame of maximum hip flexion and the end of the cycle defined by the frame preceding the subsequent maximum hip flexion frame. The total number of cycles was determined for each trial and used to calculate the mean number of kinematic frames per cycle within each trial. A spline interpolation was used to normalize all cycles to the mean number of frames per cycle.
The consistency and robustness of each trial were calculated. Consistency of the step-like responses was defined as repeatability of the hip-angle-to-knee-angle coupling relationship over multiple cycles, which represents the strength of the intralimb coordination. Consistency was quantified using a vector-based approach, wherein the angular component of the coefficient of correspondence30,32 (ACC; 0 =
Robustness was defined as the area circumscribed by the step cycle, which represents the excursion of the hip–knee movement. To quantify the robustness of the step-like responses, the mean hip angle and knee angle values of corresponding frames over all cycles was calculated resulting in a mean air-step cycle (air-stepavg) for each trial. The air-stepavg was used to quantify the excursion of the hip (hipROM) and knee (kneeROM). The net area (AREA, in degrees2) of the air-stepavg was computed using Green theorem. This theorem has 2 primary requirements: (1) a 2-dimensional shape and (2) a closed loop (the beginning frame must equal the end frame). For cycles wherein the air-stepavg did not meet the latter requirement, the loop was closed by joining the first and last frame with a line of least length, and the AREA was then computed. A larger value of AREA was interpreted as a more robust air-step.
Statistical Analysis
The Statistical Package for the Social Sciences 15.0 (SPSS Inc, Chicago, Illinois) was used for all statistical analyses. We first established the validity of the AREA value as a measure of robustness of the step-like responses using a Pearson correlation to assess the strength of the relationship between AREA and hipROM and between AREA and kneeROM. High correlation between these measures would indicate that the AREA value accurately represents, within a single value, the excursion of both the hip and the knee during the air-stepping cycles.
Because data did not meet the requirements of homogeneity of variance needed for use of parametric tests, comparisons were made using nonparametric statistical tests. Mann-Whitney
No significant between-group differences were identified among the 3 groups of SCI participants in initial ACC or AREA values (see the Results section); therefore, data from all SCI participants were pooled for comparisons on initial ACC and initial AREA values between vibration sites (Qvib, Hvib, or TFLvib). This testing identified the TFL vibration to be the most favorable site of vibration (see the Results section); therefore, all further comparisons were made based on the ACC and AREA values obtained from the TFLvib.
To quantify differences between participants with SCI and ND individuals in consistency or robustness of the step-like responses elicited with TFLvib, values of the initial ACC and initial AREA values were compared. To determine whether consistency and robustness of the responses were influenced by locomotor training, between-group comparisons were made of the initial and final ACC and AREA values of individuals with MISCI who participated in locomotor training versus individuals with SCI who did not undergo locomotor training. To assess the within-group stability of the step-like responses over time, we compared the ACC and AREA values from the initial test with those from the final test for all individuals with SCI and ND individuals using 1-tailed, paired
Results
All individuals with SCI demonstrated step-like responses to vibration, as did ND participants. Relative motion plots of the hip and knee angle and air-stepavg for a single individual from each of the ND, MISCI, and MCSCI groups are illustrated in Figure 1, and representative videos of kinematic data for each of the participant groups are available as supplemental digital content on the journal Web site (see videos, supplemental digital content). There was an excellent
34
correlation between the AREA values and the respective measures of initial hipROM (Pearson
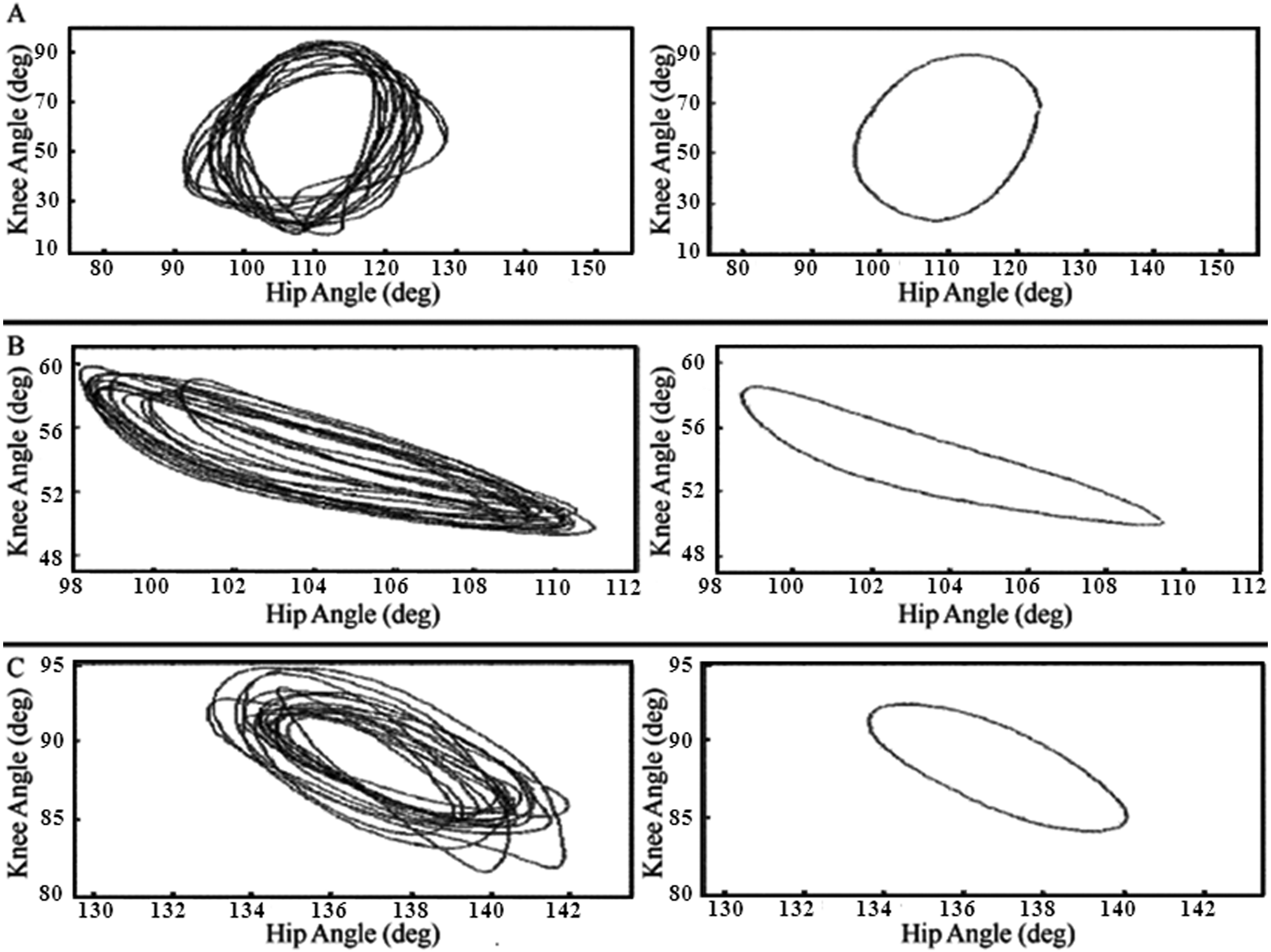
Example hip–knee relative motion and air-stepavg plots: intralimb relative motion (left panel) and air-stepavg (right panel) plots are shown for a (A) nondisabled individuals, (B) those with motor-incomplete spinal cord injury (SCI), and (C) those with motor-complete SCI (MCSCI).
When comparing differences among the sites of vibration, whereas mean values for consistency of the TFLvib responses (ACC = 0.84 ± 0.05) were slightly higher, these responses were not significantly different compared with the Qvib (ACC = 0.76 ± 0.04) or the Hvib (ACC = 0.77 ± 0.06) responses. However, the robustness of the TFLvib response (AREA = 26.60 ± 11.04 degrees2) was significantly greater compared with both the Qvib (AREA = 6.81 ± 2.68 degrees2;
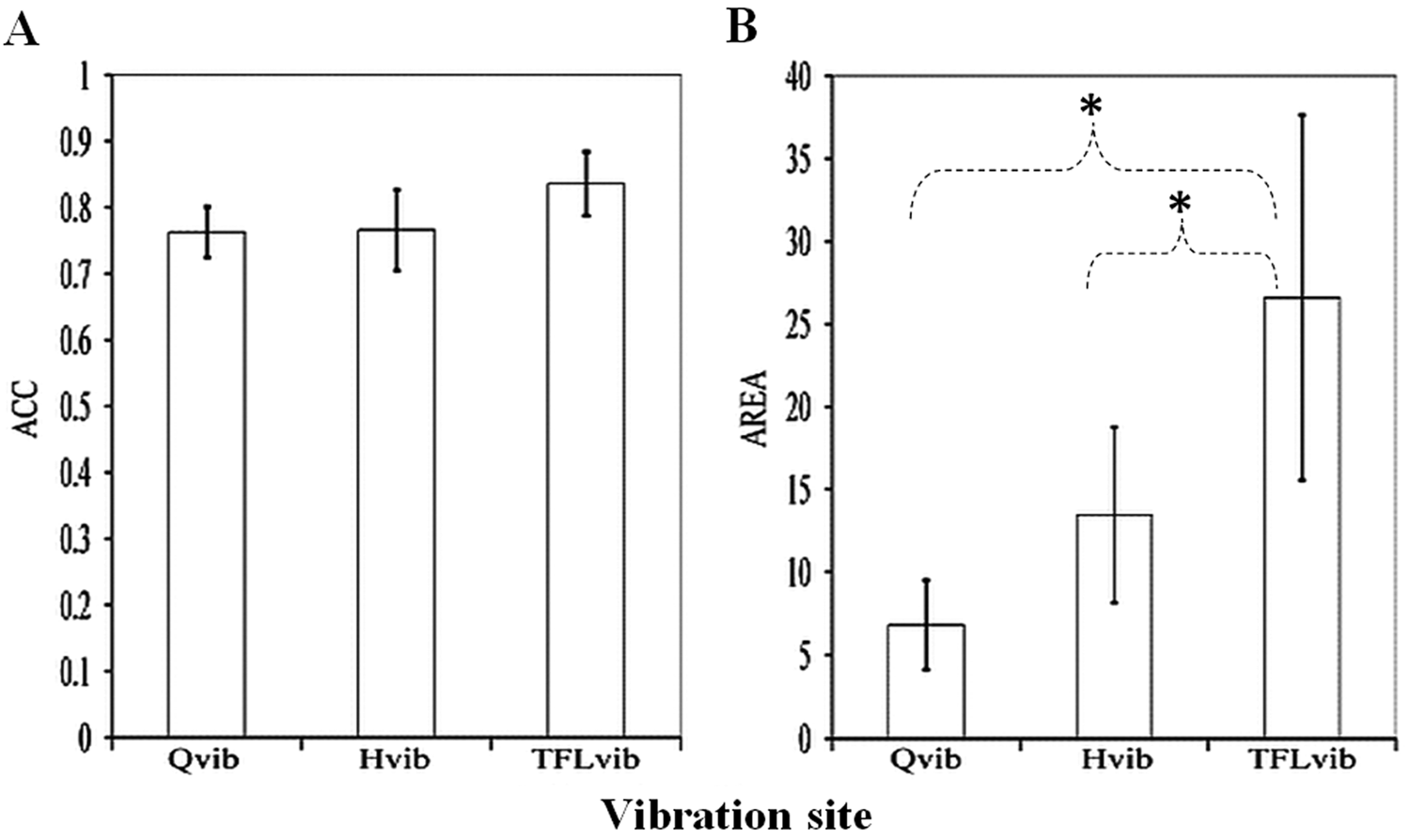
Effect of vibration site on vibration-elicited step-like responses in individuals with spinal cord injury: tested vibration sites included the muscle belly of quadriceps (Qvib), hamstrings (Hvib), and tensor fascia latae (TFLvib). Illustrated are response parameters (mean values ± standard error): (A) consistency (angular component of the coefficient of correspondence [ACC]) and (B) robustness (net area [AREA]). *Significant differences in AREA were found between TFLvib and Qvib (
There was no significant between-group difference identified in the comparison of the vibration-elicited step-like responses between the MISCI and MCSCI groups for the initial ACC values for TFLvib (
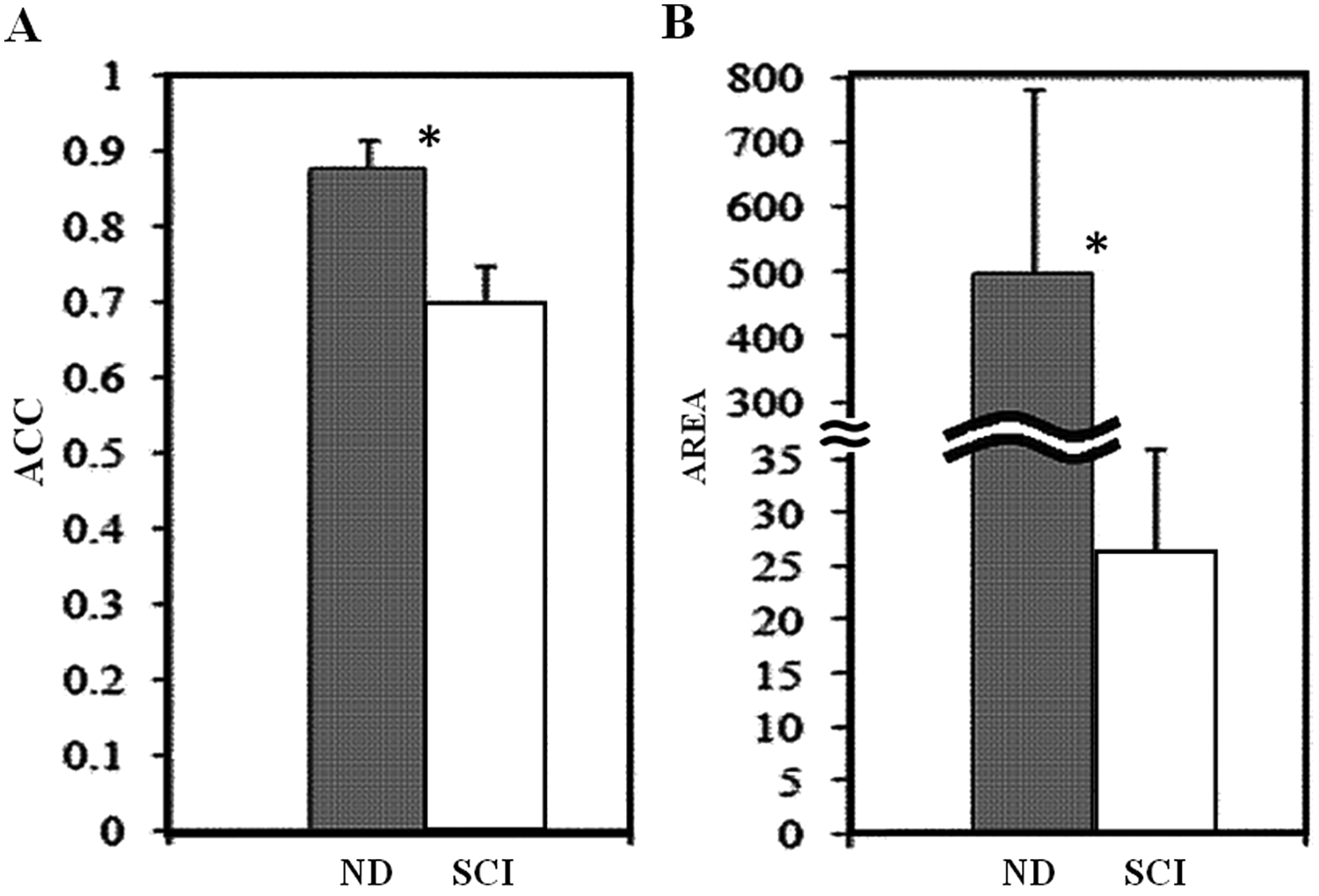
Comparison of initial vibration-elicited step-like responses in ND and SCI participants: Responses (mean values ± standard error) are shown for ND individuals (gray bar) and pooled participants with SCI (white bar). The
Comparison of the initial and final test responses for those with MISCI who participated versus those who did not participate in locomotor training identified no significant between-group differences in ACC (
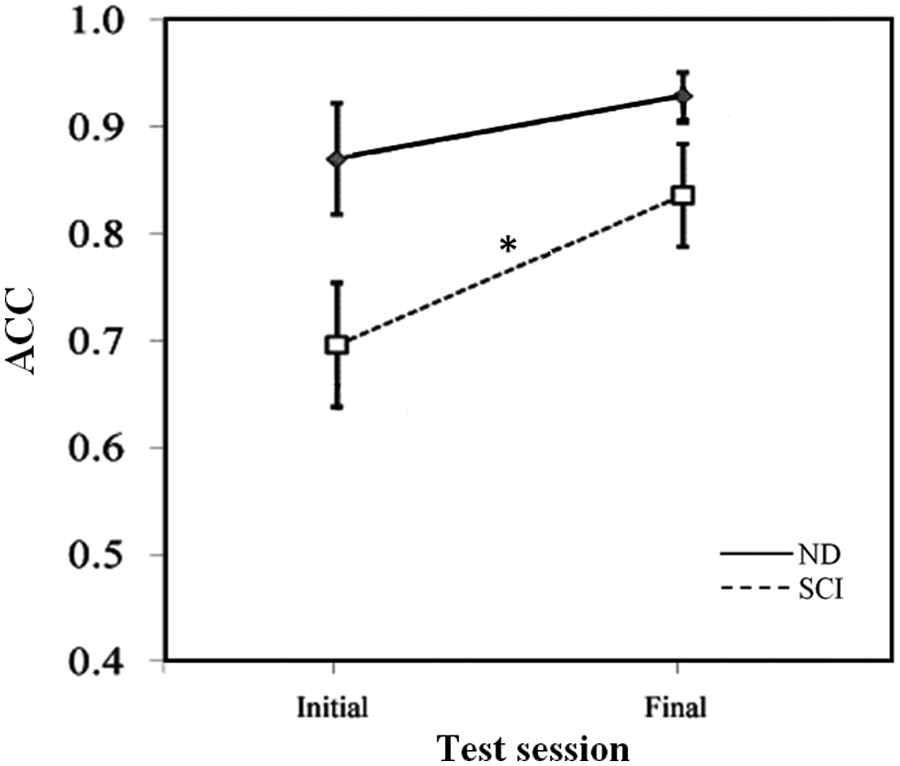
Change in consistency of vibration-elicited step-like responses between initial and final tests: ACC of the vibration-elicited step-like responses are displayed by group and test session (mean values ± standard error). The ND group (long dashed line) and pooled SCI group (short dashed line) were tested during the initial and final test sessions. Abbreviations: ND, nondisabled; SCI, spinal cord injury; ACC, angular component of the coefficient of correspondence. *Indicates significant within-group differences between the initial and final test sessions for the pooled SCI group (
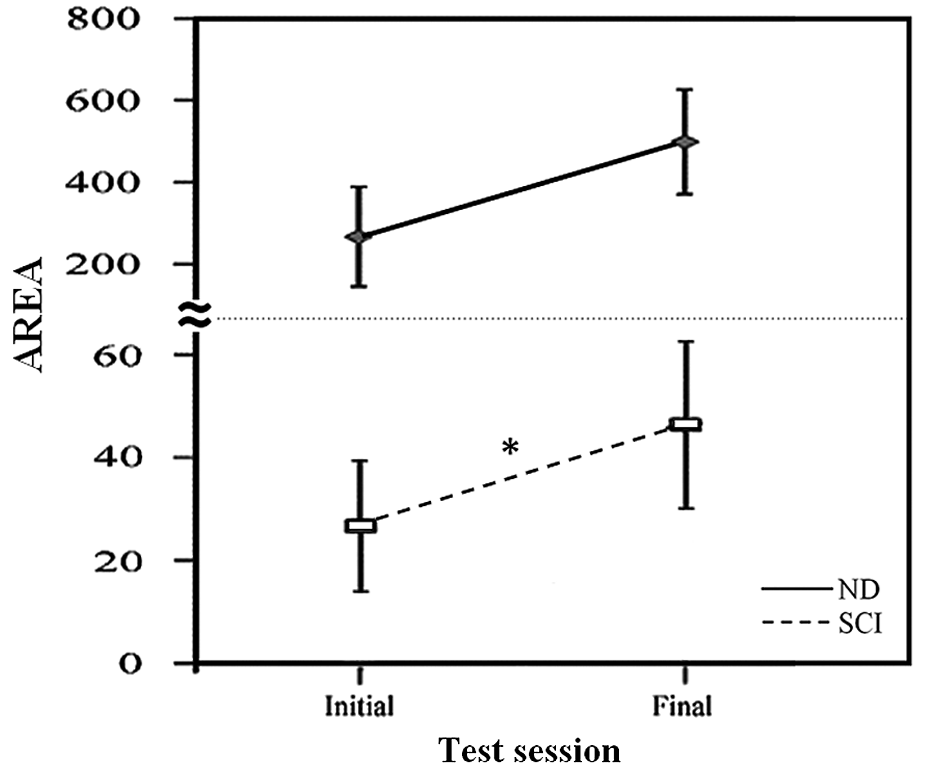
Change in robustness of vibration-elicited step-like responses between initial and final tests. AREA of the vibration-elicited step-like responses are displayed by group and test session (mean values ± standard error). The ND group (long dashed line) and pooled SCI group (short dashed line) were tested during the initial and final test sessions. The
Discussion
Our results indicate that vibration applied to muscles of the thigh elicits step-like responses in individuals with chronic SCI, regardless of degree of completeness. This is consistent with prior reports of involuntary stepping movements elicited with muscle vibration in ND individuals 14 and with epidural electrical stimulation in individuals with SCI.9,28 Although the responses elicited in individuals with SCI were not as consistent or robust as those from ND participants, contrary to our hypothesis, there were no differences in responses between those with MISCI and those with MCSCI. The similarity in step-like responses in those with MCSCI and those with MISCI are interesting in light of the fact that there were clear differences between these groups in terms of the extent of remaining voluntary drive to the lower extremities. When asked to attempt to perform voluntary air-stepping, none of the 6 participants in the MCSCI group were able to perform the voluntary movements, whereas all participants in the MISCI group were able to perform at least minimal voluntary air-stepping. Therefore, it seems that factors other than the extent of available voluntary drive must account for the lack of difference in vibration-elicited responses between the MCSCI and MISCI participants.
We expected that there would be an activity-related increase in the vibration-elicited step-like behaviors in those who participated in locomotor training. Use- or activation-related differences have been demonstrated in studies of spinal reflex circuits, wherein those with MISCI who walk on a regular basis exhibit spinal reflex activity similar to that of ND individuals but those with MISCI who do not walk have spinal reflex activity that more closely resembles reflexes of individuals with MCSCI. 35 However, there were no differences in consistency or robustness of the responses in individuals with MISCI who participated in locomotor training compared with those who did not. Therefore, it seems that factors other than the extent to which the locomotor CPG is used account for differences in responsiveness of the vibration-elicited step-like behavior.
Our results seem to suggest that there may be an influence of vibration on the step-like responses that is retained over time. When comparing initial with final test results, we found that the responses increased in consistency and robustness in those with SCI, with a trend toward increases in robustness in ND participants. It is possible that this finding is explained by evidence from prior studies that have shown that a single session of afferent stimulus may be sufficient to induce neuroplastic changes in the spinal cord 36 and cortex. 37 Research has shown that these changes may persist following removal of the stimulus for periods as long as 1 month. 38
Contrary to our hypothesis, the most favorable site for eliciting responses to vibration was over the TFL, a pelvic stabilizer, rather than the quadriceps or hamstring muscles that are more directly involved in the generation of stepping. Hip muscle afferents are known to provide a powerful modulatory influence on the locomotor CPG,4,39,40 and previous research has shown that the effects of vibration can spread to muscles other than the one being vibrated. 41 The TFL is a thinner muscle, and this may have contributed to a more effective transmission of the vibratory stimulus in this muscle. Because the TFL is situated midway between the hip flexor and extensor muscles, and all receive afferent innervation from the lumbosacral spinal cord, it is possible that vibration applied at this location produces widespread excitation to the spinal circuitry influencing both the hip flexors and extensors. Similarly, research using electrical epidural stimulation over the lumbosacral enlargement has been shown to elicit movements in individuals with MCSCI9,10,28 and to facilitate walking in individuals with MISCI. 11 The theoretical rationale for the effect of epidural stimulation is that it provides an excitatory input to the locomotor CPG. Likewise, it has been suggested that vibration may excite circuits at several segments of the spinal cord and trigger locomotor-like movements. 14 Vibration is known to have a broad influence on afferent activation and spinal circuitry (for a review see Lance 42 ), and stimuli of this nature may represent the type of excitatory drive that Graham Brown 43 and others44,45 theorized would produce rhythmic oscillations of the flexor and extensor half centers of the locomotor CPG.
Clinical Implications
The results of this study and others14,15 indicate that vibration to the thigh muscles elicits step-like behavior in the legs. In this proof-of-concept study, we positioned participants in a side-lying posture, with the leg suspended to provide an environment wherein air-stepping could be most easily evoked. However, one might imagine a therapeutic setup in which vibration is applied while participants are partially suspended using a body-weight support system, as has previously been studied in ND individuals 15 ; as such, vibration may represent a novel therapeutic adjunct to locomotor training. Additional support for this idea comes from reports that the use of whole-body vibration is associated with improved walking function in individuals with MISCI. 17
Conclusions
Vibration to the muscles of the thigh elicits step-like responses in individuals with SCI, including those with and those without the ability to perform voluntary lower-extremity movement. Although responses in those with SCI were not as vigorous as those observed in ND individuals, the fact that these cyclic involuntary behaviors can be elicited at all indicates that the CPG-related circuitry is influenced by and accessible through afferent input, as has been shown in previous studies.2,4,12-14 The responses in those with SCI were not influenced by participation in a locomotor training program. Further investigation is warranted to assess the effect of vibration on walking function in individuals with SCI and to assess the use of vibration in combination with locomotor training.
Footnotes
Acknowledgements
The authors are grateful to The Miami Project to Cure Paralysis for the funding that supported this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
