Abstract
Background. The health benefits associated with physical activity–based rehabilitation in patients with no lower-extremity motor function after a spinal cord injury (SCI) is uncertain. Methods. The authors assessed signs of efficacy, safety, and utility associated with a novel pharmacological combination therapy to activate central pattern generator (CPG) activity and corresponding locomotor activity in complete thoracic Th9/10-transected mice. Results. Subcutaneous administration 4 times per week for 1 month of 1.5 mg/kg buspirone, 1.5 mg/kg apomorphine, 12.5 mg/kg benserazide, and 50 mg/kg L-DOPA induced episodes of weight-bearing stepping on a treadmill in nonassisted paraplegic mice for 45-minute sessions. Hindlimb muscle cross-sectional area and fiber area values as well as several blood cell constituent levels assessed at 30 days postinjury were positively affected by the combination therapy, as compared with controls. Episodes of locomotion remained effective on each treatment. Femoral bone mineral density loss was not prevented by triple therapy. Conclusion. Although translation of these findings needs further experimentation, similar pharmacological activation of the CPG offers a novel therapeutic target to provide some health benefits in motor-complete SCI patients.
Keywords
Introduction
A survey recently reported that 1.3 million Americans are currently living with a spinal cord injury (SCI). 1 Rehabilitation activity–based therapies such as body weight–supported treadmill training in which the legs are manually assisted on a treadmill by therapists, robotic assistive devices, and functional electrical stimulation are used to try to improve voluntary ambulation and exercise in motor-incomplete patients to at least the degree that more conventional physical therapies offer. 2,3 However, the metabolic outcomes and health benefits associated with these approaches remain unclear. 3,4 In turn, chronic SCI patients and especially motor-complete individuals (ASIA-A or ASIA-B) often experience secondary complications such as cardiovascular problems, muscle wasting, obesity, autonomic dysreflexia, deep vein thrombosis, urinary tract infection, immune system dysfunction, anemia, pressure sores, and so on. 5-8 Given that metabolically challenging activity-based therapies adapted for motor-complete SCI patients have not been clearly identified yet, we sought to study signs of efficacy and utility associated with a novel activity-based pharmacological approach recently developed in our laboratory. 9 It is a combination therapy that can trigger and endogenously generate, within 15 minutes, treadmill stepping movements for at least 30 minutes without manual assistance, harness-provided weight support, or other experimenter-provided sensory stimulation in motor-complete paraplegic animals. We took advantage of this murine model of complete paraplegia after transection (Tx) in which clinically relevant secondary complications were recently characterized, 10 to test the general hypothesis that this combination therapy can generate central pattern generator (CPG)-activating effects, metabolically challenging stepping movements, and corresponding health benefits. Specifically, we hypothesized that regular combination therapy–elicited step training (4 times per week for 1 month) will yield health benefits and prevent related biomarker changes in previously untrained and nonassisted Tx mice. Outcome measures included bone and muscle tissue loss and immune and blood cell counts based on profound deregulation of these systems reported after SCI in patients and animal models. 6-8,11,12
The locomotor CPG is a network located in the lumbar spinal cord that can generate, under certain circumstances, basic commands for walking in the absence of descending and peripheral inputs. 13 Based on a series of drug-screening experiments, we found a drug combination that could potently elicit significant CPG-activating effects, which included large-amplitude stepping movements with some equilibrium, plantar foot placement, and weight-bearing capabilities (ie, real stepping rather than sweeping, as initially reported with some drugs administered separately). 9,14-18 This strategy is supported by comparable findings in in vitro isolated spinal cord preparations, where compounds such as serotonin and dopamine, when combined, can elicit stable, coordinated, and fictive locomotor neurographic activity. 19,20 The combination therapy tested here included buspirone (a 5-HT1A receptor agonist), L-DOPA (a dopamine/noradrenaline precursor), benserazide (a decarboxylase inhibitor), and apomorphine (a dopamine receptor agonist). These drugs are available for clinical use as a 5-HT1A receptor agonist, dopamine/noradrenaline precursor, and decarboxylase inhibitor, respectively. 21-23 Although 5-HT1A agonists and dopamine/noradrenaline precursors and agonists have been shown separately to promote training-induced stepping, their synergistic effects on CPG activation and real stepping induction in untrained and nonassisted Tx animals has not been previously demonstrated.
Methods
Animal Model and Surgical Procedures
This work was performed in accordance with the Canadian Guide for the Care and Use of Laboratory animals. All procedures were approved by the Animal Research Committee of the Centre Hospitalier Universitaire de Québec. Experiments were carried out on 21 adult male CD1 mice (Charles River Laboratories, Montréal, QC) weighing 30 to 35 g prior to surgery. Mice were housed generally 4 per cage in a controlled-temperature environment maintained under a 12-hour light/dark cycle with free access to food and water. Preoperative care included administration of lactate-Ringer’s solution (1 mL, subcutaneously [SC]), an analgesic (buprenorphine; 0.1 mg/kg, SC, Schering-Plough, Pointe-Claire, QC), and an antibiotic (Baytril; 5 mg/kg, SC, Bayer, Toronto, ON). Spinal transection (Tx) at the low-thoracic level was performed under complete anesthesia with 2.5% isoflurane. The spinal cord was completely Tx intervertebrally using microscissors (no. 15000-08, Fine Science Tools, North Vancouver, BC) inserted between the 9th and 10th thoracic vertebrae. To ensure that complete Tx was achieved, the inner vertebral walls were explored and scraped several times with scissor tips to disrupt any small fibers that had not been previously severed. Opened skin areas were sutured, and animals were placed for a few hours on heating pads. Postoperative care included administration of lactate-Ringer’s solution (2 × 1 mL/d, SC), buprenorphine (0.2 mg/kg/d, SC), and Baytril (5 mg/kg/d) for 4 consecutive days. Bladders were emptied manually until a spontaneous return of some micturition function. Complete Tx was confirmed by flaccid hindlimbs during the first few days postsurgery and postmortem histological examination of the lesion using either coronal or longitudinal sections of the spinal cord stained with luxol blue and cresyl violet (see Figure 1A).
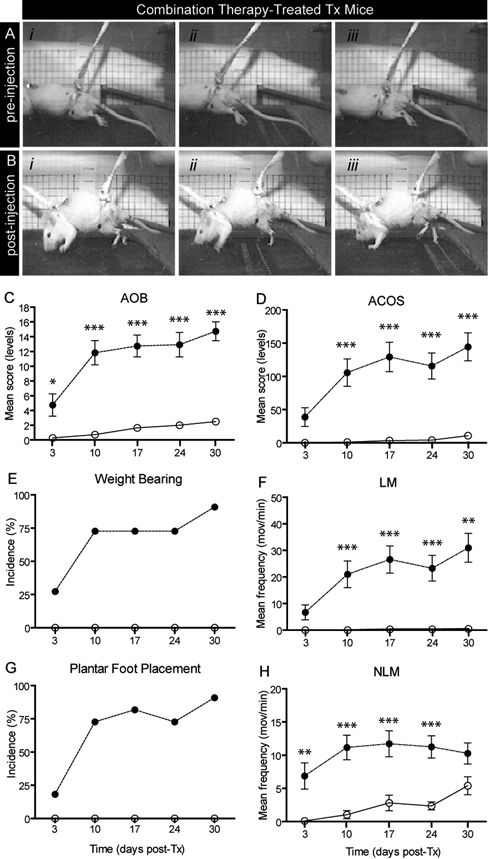
Hindlimb motor scores in Tx mice. (A) Absence of hindlimb movement prior to drug administration. (B) Powerful stepping induced within 20 minutes of post–combination therapy injection (i-iii). Pictures taken at different times during the step cycle or lack of step cycle. (C-H) Assessment of drug-induced hindlimb movements using the ACOS and AOB methods. LMs and NLMs, weight-bearing, and plantar foot placement capabilities were assessed in combination therapy–treated Tx mice (filled circles) and saline-treated Tx mice (open circles)a
Experiments
Animals were divided into 2 groups: a group of spinal Tx mice treated with the combination therapy (n = 11) and a group of spinal Tx mice treated with saline (placebo control, n = 10). The combination therapy consisted of 1.5 mg/kg buspirone, 1.5 mg/kg apomorphine and 50 mg/kg L-DOPA, combined with 12.5 mg/kg benserazide, a decarboxylase inhibitor to increase its central bioavailability. Doses were based on previous dose-dependent studies conducted in our laboratory (minimal effective dosage for inducing significant hindlimb locomotor movements and corresponding motor scores). Drug administration (combination therapy or saline) was performed SC 4 times per week from day 3 until day 30 post-Tx. Following SC injection, animals were placed on a motor-driven treadmill for approximately 45 minutes. All drugs were diluted in saline and purchased either from Sigma-Aldrich (St-Louis, MO) or Tocris (Ellisville, MO).
Behavioral Assays
Mice were placed in specially adapted harnesses attached in front of the animals to maintain rostrocaudally their position on a treadmill for videoimaging purposes (ie, Sony DCR110, 30-50 frames/s, shutter speed 1/1000 placed sideways to capture hindlimb movements). No weight support, tactile assistance, or experimenter-provided stimulation was provided. A motor-driven treadmill (8-10 cm/s) was used for all behavioral tests. Two complementary methods developed to specifically assess hindlimb movements in Tx rodents were used to quantify drug-induced effects. The first assay called ACOS (average combined score) is routinely used in our laboratory and consists of a 4-minute assessment (and recording) in which numbers (per minute) of locomotor-like movements (LMs), non-locomotor-like movements (NLMs), amplitude, and incidence are quantified. 24,25 An ACOS is created by simple arithmetical combination of the collected values: ACOS = [NLM + (2 × LM)] × Amplitude. One LM was defined as an entire step-like cycle consisting of an extension phase followed by a flexion phase or vice versa occurring in both hindlimbs consecutively (ie, bilaterally alternating and out-of-phase movements). One NLM was defined as 1 non–bilaterally coordinated movement (ie, not followed by a flexion–extension on the other side) and included unilateral movements such as jerks, brief sequences of fast-paw shaking (typically lasting 1-2 s/episode and counted as one NLM), twitches, and kicks. Amplitude was characterized by assigning 1 of 3 values: 0, if no movement was observed; 1, if the amplitude of most movements was less than half the range of motion of normal steps; and 2, if the amplitude of most movements was at least more than half the range of motion of normal steps. Note that amplitude was scored for LM and NLM. Incidence corresponded to the number of mice (out of all mice tested in a group) in which NLMs or LMs were observed. LMs and corresponding ACOS scores do not include nor assess the presence of weight-bearing or plantar foot placement capabilities. 24,25
We also used a qualitative assay developed by Antri, Orsal, and Barthe, 26 referred to here as the AOB, that offers the advantage of also assessing weight-bearing and plantar foot placement capabilities. Similar to the ACOS, the AOB does not assess forelimb versus hindlimb coordination, which is considered irrelevant in assessing drug-induced hindlimb movements in completely spinal cord–transected animals. The AOB scale consists of 22 discriminative scores defined as follows: 0, no movement; 1, weak hindlimb jerks; 2, weak rhythmic hindlimb movements with no bilateral alternation; 3, large rhythmic hindlimb movements with no bilateral alternation; 4, weak rhythmic hindlimb movements with occasional bilateral alternation; 5, large rhythmic hindlimb movements with occasional bilateral alternation; 6, weak rhythmic hindlimb movements with frequent bilateral alternation; 7, large rhythmic hindlimb movements with frequent bilateral alternation; 8, weak rhythmic hindlimb movements with consistent bilateral alternation; 9, large rhythmic hindlimb movements with consistent bilateral alternation; 10, large rhythmic bilaterally alternating hindlimb movements with occasional body weight support; 11, large rhythmic bilaterally alternating (occasionally) hindlimb movements with occasional plantar foot placement; 12, large rhythmic bilaterally alternating (frequently) hindlimb movements with occasional plantar foot placement; and so on. Consistent means observed in >95% of the step cycles; frequent, 51% to 94%; and occasional, <50%. Additional levels (13 up to 22) are associated with increasingly consistent bilateral alternation, body weight support, and plantar foot placement (for further details, see Antri, Orsal, and Barthe 26 ). We also separately assessed weight-bearing and plantar foot placement regularity either as absent (0), occasionally (+), frequently (++), or consistently (+++) present. Each animal was weighed prior to Tx and before each behavioral assessment session post-Tx (once a week).
Blood Cell Count
At the end of the study, all animals were killed by ketamine-xylazine overdose. Blood samples (~850 µL) were immediately collected by cardiac puncture, and 50-µL samples were diluted 1/10 in 5 mM phosphate buffered saline (PBS)-EDTA for counts. Individual blood samples were stored at −20°C. Blood leukocytes were counted for each mouse using a hematocytometer following acetic blue staining. Cytospin preparations (500 rpm for 10 minutes) of blood cells were analyzed after differential staining using the Wright-Giemsa stain kit. Counts were confirmed with a CELL-DYN 3700 automatic blood cell analyzer (CD3700; Abbott Laboratories, North Chicago, IL), which was also used for erythrocyte, hemoglobin, hematocrit, and platelet counts. Given that signs of anemia and altered immune cell counts have been found in Tx mice and SCI patients, 11 corresponding measurements were made here to provide a rationale for using them as convenient markers of combination therapy-induced health benefits for clinical trials.
Bone Densitometry
Femoral bones were dissected and cleaned of soft tissues. The femoral bones were wrapped in saline-soaked gauze and frozen at −20°C in sealed vials until testing. On the day of testing, the femoral bones were slowly (4 hours) thawed at 4°C. Dual-energy X-ray absorptiometry measurements (PIXImus 2, Lunar Corp, Madison, WI) were performed using the head and femora of animal from both groups. Calibration of the apparatus was conducted according to the manufacturer’s protocol. Bone mineral density (BMD) values (g/cm2) were measured within a predetermined metaphyseal common region of interest in the metaphyseal area for all specimens. These experiments were performed at the McGill Centre for Bone and Periodontal Research.
Muscle and Cell Histology
On sacrifice, left soleus (SOL) and extensor digitorum longus (EDL) muscles were dissected, frozen in melting isopentane, and stored at −80°C until further use. Serial cross sections, 12-µm thick, from the midbelly of the muscles were cut using a cryostat maintained at −20°C (2800E Frigocut, Leica Instruments, Germany) and mounted on Superfrost plus glass slides (VWR Canlab, Mississauga, ON). Sections were incubated for 2 hours in either myosin heavy chain slow or myosin heavy chain fast primary antibodies (specific for myosin heavy chain isoform type I and type II respectively; diluted 1/50 in 0.1 M PBS, Vector Laboratories, Burlingame, CA). Sections were then rinsed in PBS 0.1 M before incubation with a goat antimouse IgG (H + L) Alexa Fluor 488 secondary antibodies (diluted 1/500 in 0.1 M PBS; Molecular Probes, Eugene, OR). Slides were mounted with PBS–glycerol (50–50). Some sections were treated as above, except that the primary antibodies were omitted from the incubation solution. Theses sections served as negative controls. Immunofluorescent labeling was visualized with a 10× water-immersion objective on an Olympus BX61WI confocal microscope. Images were captured using FluoView 300 (Olympus Canada Inc, Markham, ON) and analyzed with ImageJ (ImageJ 1.40, Research Services Branch, National Institutes of Health, Bethesda, MD). Analyses consisted of determining SOL and EDL muscle fiber type composition, whole muscle cross-sectional area (CSA) and fiber CSA of each fiber phenotype. For the latter analysis, 50 fibers/isoform/muscle were used. In cases in which the number of fibers was less than 50 for an isoform, all fiber CSAs were averaged.
Statistical Analyses
Repeated-measures 2-way ANOVAs followed by a Bonferroni post hoc correction were used for motor performances, whereas standard 1-way ANOVAs accompanied by a Fisher’s least significant difference (LSD) correction post hoc test were conducted for all other analyses using SPSS (version 11.0; SPSS Inc, Chicago, IL).
Results
Efficacy was evaluated weekly, comparing 4 minutes immediately preinjection to 4 minutes of stepping approximately 15 to 20 minutes postinjection. Locomotor performances (ACOS and AOB hindlimb motor scores) in combination therapy–treated and placebo (saline)-treated Tx animals were compared. The results showed that after each injection, significant differences in scores (P < .05) were found in combination therapy–treated mice. Pictograms in Figure 2 show the lack of hindlimb movement prior to drug injection in a typical paraplegic mouse placed on a motorized treadmill (Figure 2A). In clear contrast, large amplitude and powerful weight-bearing stepping movements with some equilibrium and plantar foot placement capabilities were displayed 15 to 20 minutes post–drug injection (Figure 2B). These effects lasted from 30 to 60 minutes, but were not specifically examined quantitatively in this study. Averaged data from all animals revealed that AOB scores above the 10th level (ie, the latter is the key level above which weight bearing stepping is found) and ACOS scores ranging between 100 and 150 were generally found (Figures 2C and 2D). Relatively poor AOB and ACOS scores were found at 3 days post-Tx. All scores assessed subsequently were significantly (P < .05 or P < .001) different from those assessed in placebo-treated animals (filled vs open circles, respectively, in Figures 2C and 2D). On the last assessment day (at 30 days post-Tx), AOB and ACOS scores reached 14.7 ± 1.2 and 144.5 ± 21.1, respectively. Examined in more detail, factors constituting the ACOS scores, such as movement frequencies, revealed that at 30 days post-Tx, LM and NLM frequencies reached 31.0 ± 5.4 and 10.3 ± 1.6 movements/min, respectively (Figures 2F and 2H). The number of animals in each group displaying weight-bearing and plantar foot placement values was significant and consistently found in most combination therapy–treated animals (eg, incidence of 90.9% at 30 days post-Tx; Figures 2E and 2G).
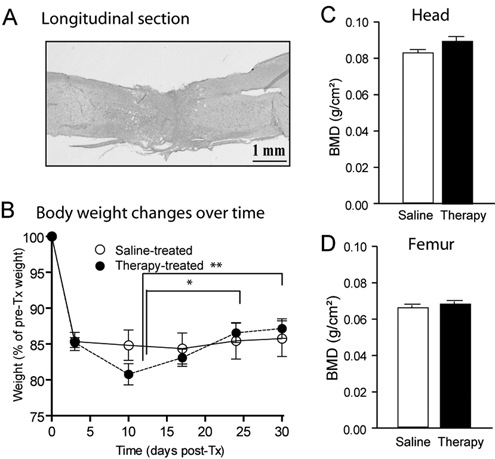
Histology, body weight, and dual X-ray absorptiometry (DXA) in transected (Tx) mice. (A) Histological confirmation of the completeness of the spinal cord transection was done using coronal or midsagittal spinal cord sections stained with luxol fast blue/cresyl violet for myelinated axons and Nissl substance, respectively. (B) Body weight expressed as percentages of initial values (g). DXA-measured bone mineral density (BMD) of the head (C) (control tissue) and femur (D). White and black bars represent data obtained from saline-treated Tx mice and combination therapy-treated Tx mice, respectivelya
Following an initial loss (−19%) of weight typically found in Tx mice caused by extensive muscular atrophy within a few days postsurgery, 12 body weight values remained relatively stable over time after 1 week post-Tx. Combination therapy–treated animals progressively displayed a moderate increase (P < .01) in body weight, suggesting that repeated administration of this combination therapy was well tolerated (Figure 1B).
No significant difference (P > .05) was found in BMD values in control tissues (Figure 1C, head) or femoral bones (Figure 1D) from combination-treated versus placebo-treated Tx animals (black and white bars, respectively).
Postmortem examination of muscle size (whole surface area and fiber CSA) measured from cryostat transverse sections prepared from 2 hindlimb muscles, SOL and EDL, was performed to assess the effect of combination therapy–induced training on muscular atrophy normally found after SCI. We found values corresponding with larger muscles and muscle fibers in the combination therapy–treated paraplegic animals compared with the placebo-treated paraplegic animals. Averaged data from all animals revealed that SOL surface area values significantly (P < .05) increased by 24% in combination therapy–treated paraplegic mice (0.61 ± 0.05 mm2) compared with placebo-treated ones (0.49 ± 0.03 mm2; Figure 3A). Comparable results were found in EDL (combination therapy treated, 0.91 ± 0.03 mm2 vs placebo treated, 0.77 ± 0.06 mm2; not shown). At the cellular level, comparisons between combination therapy–treated and placebo-treated animals revealed that type I fiber CSA values nonsignificantly (P > .05) changed (Figure 3B), whereas type II fiber and intermediate fiber (type I + II labeled) CSA values significantly (P < .05 and P < .01) increased subsequently, both by 8% (Figures 3C and 3D). An analysis of muscle fiber-type ratios (ie, proportion among all fibers of type I, type II, or type I + II fibers) indicated that nonsignificant (P > .05) changes were found between groups (Figures 3E to 3G). In turn, a large increase (50% increase) in SOL intermediate fiber-type ratio was found in paraplegic compared with nonparaplegic animals (not shown). Comparable changes were generally found in EDL muscles (a significant 18% and 22% increase of type II and type I + II fiber-type CSA values in combination therapy–treated compared with placebo-treated mice; not shown).
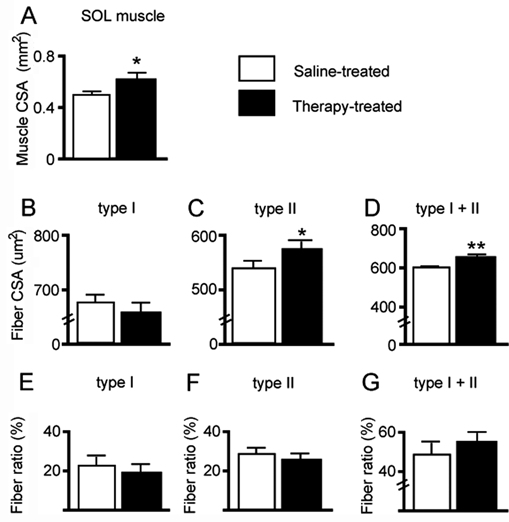
SOL muscle and single-fiber CSA and ratio in Tx mice. (A) CSA of SOL in combination therapy-treated (tritherapy, black bars) and saline-treated (placebo, white bars) Tx mice. (B-D) SOL fiber area (CSA) values. (E-G) SOL fiber-specific ratio. Type I, type II, and intermediate fibers (type I + II labeled) were labeled with MHCs or MHCf primary antibodies (specific for MHC isoform type I and type II, respectively)a
Levels of RBC, platelet, hemoglobin, and hematocrit increased (P < .05) by 11%, 19%, 10%, and 10%, respectively, in combination therapy–treated animals (Figures 4A to 4D, black vs white bars, respectively).
Reported as percentages of all leukocytes (Figure 5A), lymphocyte and neutrophil counts were 25% higher (Figure 5B) and 27% lower (Figure 5C), respectively, in combination therapy–treated versus placebo-treated Tx animals (black and white bars, respectively), whereas monocyte, eosinophil, and basophil counts did not change (Figures 5C, 5E, and 5F). Statistically significant levels were not reached in most cases possibly because of typical intersubject variability problems with immune cell counts in humans or mice. 11,27
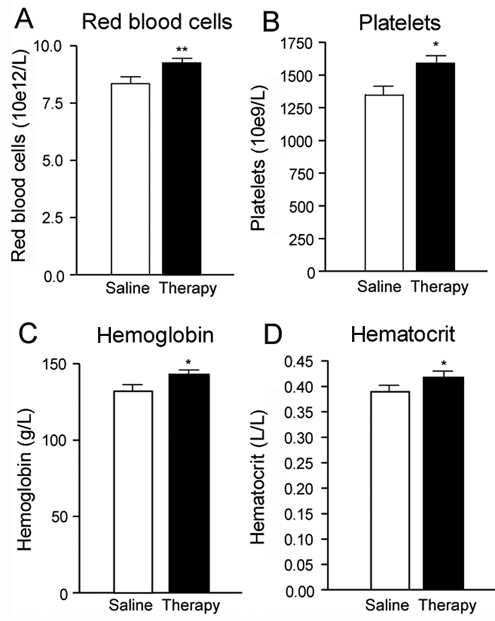
Red blood cell (RBC) count in transected (Tx) mice. RBC constituents expressed either as total erythrocyte (RBC) count (A), platelet count (B), hemoglobin (C), or hematocrit (D) concentrations (g/L and L/L). White and black bars represent data obtained from saline-treated Tx mice, and combination therapy–treated Tx mice, respectively a
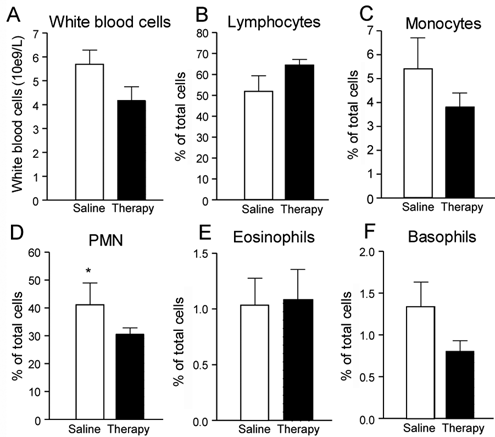
White blood cell count in transected (Tx) mice. White blood cell constituents (B-F) expressed as percentage of all white blood cells or leukocytes (A). White and black bars represent data obtained from saline-treated Tx mice and combination therapy–treated Tx mice, respectivelya
Discussion
Our results showed that repeated systemic administrations of a combination therapy composed of small molecules that have already been approved by the US Food and Drug Administration, including buspirone, L-DOPA/benserazide, and apomorphine, potently induced, after each injection, weight-bearing stepping on a treadmill in previously untrained, completely paraplegic mice. In addition, evidence suggests that regularly induced stepping (4 times per week for 1 month) with this novel pharmacological approach may be safe and may yield some health benefits because body weight did not decrease, and whole muscle CSA, single muscle fiber CSA, lymphocyte count, RBC count, and platelet, hemoglobin, and hematocrit levels all increased in combination therapy–treated animals as compared with placebo-treated mice. This is the first report of such potent pharmacologically induced, CPG-activating effects in vivo.
In comparison with control data from uninjured mice (R. V. Ung, P. Rouleau, P. A. Guertin, unpublished data, May 2010), the results presented here revealed that most parameters assessed in combination therapy–treated Tx mice were closer to values normally found in non-Tx CD1 mice. Except for BMD data, which did not change in therapy-treated versus placebo-treated mice, the greater SOL and EDL fiber area (CSA) values reported here in combination therapy-treated Tx mice (24% increase compared with saline-treated Tx mice; Figure 3) correspond, respectively, to 72% and 83% of normal CSA values in uninjured and untreated CD1 mice. Longer periods of training and anabolic treatments may be needed to completely prevent atrophy (R. V. Ung, P. Rouleau, P. A. Guertin, unpublished data, May 2010). Combination therapy–treated Tx mice recovered partially from the mild anemia found after transection. Erythrocyte, hemoglobin, and hematocrit levels found here corresponded to 93%, 90%, and 88% of normal levels. White blood cell levels were also partially restored toward normal levels given that lymphocytes and neutrophils reached 86% and 150% of normal levels, respectively. Because low concentrations of lymphocytes are typically associated with increased rates of infection after surgery or trauma, the re-increase of lymphocyte levels in therapy-trained Tx mice shown here may lower probabilities of infectious problems. In turn, neutrophils are typically known to be recruited to the site of injury following trauma and are the hallmark of acute inflammation. A return toward normal levels could be interpreted as a reduced probability of inflammatory problems in combination therapy–treated Tx mice.
We cannot exclude the possibility that the effects induced in combination therapy–treated mice were in part caused by the drugs themselves and not by the activity-based training elicited by them. However, given that these drugs induce spinal stepping activity even in animals left in their cage (ie, effects do not depend on the motorized treadmill and related sensory inputs; unpublished observation), a control group in which such a hypothesis may be correctly tested may be difficult to design. For instance, a control group receiving the treatment but having its legs immobilized (eg, with a cast) may limit movement but not hindlimb muscle contractions or stress associated with this new condition (ie, additional manipulation, restraining movement). On the other hand, animals not receiving the treatment but getting passive exercise training (eg, motorized bikes used in Tx rats) lack most of the active muscular contraction aspect (ie, although some reflex-elicited EMG activity can be found in passively generated cycling) associated with the proposed drug treatment. 28
Researchers using isolated spinal cord preparations have provided evidence that drug combinations with specific neurotransmitters (eg, serotonin and dopamine), rather than separately administered compounds, can trigger superior CPG activities recorded from ventral roots or peripheral nerves in vitro. 19,20 However, in vivo adult animal models of SCI (eg, cats, rats, and mice) have generally been tested with separately administered small molecules (eg, 5-HT or NA receptor agonists or NA/DA precursors), which were occasionally shown either to modulate or trigger some LMs typically during experimenter-assisted interventions (eg, body weight–supported assistance, robotic devices, electrical stimulation, repeated-training-induced spinal plasticity, sensory-stimulated perineum, tail, sexual organs, abdominal skin, etc). 29-31 In fact, many of these experimenter-assisted interventions were shown without drugs to affect CPG activation per se: for example, in kittens, regular training with perineum stimulation and body weight support was reported to acutely elicit some nonpharmacological hindlimb stepping on a treadmill. 32
The reasons why combination treatments may augment pharmacological CPG activation and corresponding weight-bearing stepping in nonassisted completely paraplegic animals remain unclear. Although some of the neuronal elements constituting the CPG were recently identified genetically (eg, V0-V3 interneurons), many of their properties (eg, pharmacological and electrophysiological) are incompletely characterized. 33 Coactivation of specific sets of receptors may induce greater cellular activity within the network and optimal motoneuronal activation for robust CPG-generated locomotor movements.
Whether comparable effects will be found in SCI patients needs more specific study. Differences between humans and mammalian species with regard to locomotor control mechanisms are incompletely understood. For instance, compelling but still inconclusive evidence of a CPG in humans exists 34,35 ; the CPG in humans may be more complex or require different inputs for successful walking 36 ; and locomotion and specifically bipedal walking may largely differ biomechanically and kinematically from one species to another, 37 so CPG-activating drugs may generate species-dependent effects (eg, weight-bearing stepping in rodents but only stepping without weight-bearing capabilities in humans).
Additional preclinical experiments that need to be conducted include toxicology and full safety pharmacology. Long-term assessment of effects on a wide variety of markers and end points must also be examined, given the goal of chronic (eg, several months every year) activity-based treatment to induce and maintain health benefits.
Footnotes
Acknowledgements
We are grateful to Nicolas Lapointe for his help in some of the experiments.
The author(s) declared a potential conflict of interest (e.g. a financial relationship with the commercial organizations or products discussed in this article) as follows: Pierre Guertin was CEO of Neurospina Therapeutics between 2005 and 2008 and since 2009 is CEO of Nordic Life Science Pipeline, which owns the worldwide exclusive rights to develop and commercialize the proposed combination therapy described in this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This work was supported by funding from the International Foundation for Research in Paraplegia (Switzerland) and Neurospina Therapeutics.
