Abstract
Introduction
Physical activity (PA) improves cardiovascular health and decreases the occurrence of obesity, diabetes mellitus, and other diseases. 1 Recently, attention has focused on the ability of exercise to enhance brain health.2,3 For example, in animal studies, physical exercise was found to increase levels of growth factors, 4 angiogenesis, 5 and neurogenesis and improve learning and memory. 6 Furthermore, in humans, physical exercise attenuates cognitive decline associated with aging.7-9 One difficulty in extrapolating exercise data derived from animal studies to humans is that animals are typically housed in isolation throughout experimentation and provided with 24-hour exposure to wheel running. This is problematic for several reasons. Rodents are highly social animals and individual housing results in isolation stress, decreased neurogenesis, and structural changes in cortical and hippocampal neurons.10,11 Additionally, isolated rats given 24-hour access to running wheels sometimes develop pathological patterns of running, whereby they may run as much as 48 km in one 24-hour period. 12 Clearly, there is a serious mismatch between commonly used animal exercise paradigms and exercise guidelines in humans that recommend 30 to 45 minutes of daily aerobic exercise.
Methods to enhance cognitive activity (CA) are attracting considerable attention in the treatment of mild cognitive impairment and dementia. Several retrospective reports indicate that individuals who actively participate in CA such as board games, reading, and writing are less likely to develop dementia.8,13,14 A few animal studies indicate that learning in the Morris water maze increases mRNA levels of brain-derived neurotrophic factor (BDNF), TrkB, and basic fibroblast growth factor (FGF2), all of which are thought to enhance neuroplasticity and improve learning and memory.15,16 It is interesting to note that BDNF is also increased by PA4,17 and may increase the proliferation and division of neuronal precursor cells, whereas CA appears to promote the survival of these cells, indicating an important link between both forms of activity.18-20
Unfortunately, it is very difficult to accurately determine the amount and intensity of PA and especially CA in human participants because most studies rely on self-reports. 2 However, in animals, these variables can be more precisely regulated, and potential beneficial mechanisms investigated. Here, we examined a novel combination of PA and CA and its effects on BDNF and phosphorylated cyclic AMP response element binding protein (pCREB) because these proteins are thought to be critically involved in neuroplasticity processes, including learning and memory.21,22 We found that combining PA and CA significantly enhanced learning and working memory abilities, independent of an action on BDNF or pCREB protein levels.
Materials and Methods
Participants
A total of 57 male Sprague-Dawley rats (Charles River, Quebec, Canada) weighing approximately 250 g (~2 months) on arrival were used in this study. Animals were socially housed on a 12:12 hour reverse light:dark cycle (lights off at 08:00) and fed food and water ad libitum, unless otherwise indicated. All procedures were approved by the Memorial University of Newfoundland Animal Care Committee and conformed to the Canadian Council on Animal Care guidelines.
Experimental Conditions
Experiment 1
There were 5 experimental conditions in experiment 1 (Table 1). The PA group (n = 8) was exposed to voluntary running wheels (115 cm diameter) for 2 h/d, 5 d/wk. Running distances were recorded with CatEye cyclocomputers (CC-MT400, Osaka, Japan). An additional 8 animals underwent similar PA training and were assessed in the radial arm maze (RAM) to measure the effects of PA alone on learning and memory abilities. The CA group (n = 9) was placed in novel Hebb-Williams mazes each day for 2 hours, 5 d/wk (similar exposure durations as the PA group). Following 2 weeks of daily novel Hebb-Williams exposure, animals in the CA group were trained in an 8-arm baited version of the RAM for an additional 2 weeks (2 trials/d for 10 consecutive days) as previously described. 23 Following the RAM procedure, CA animals were again exposed to novel Hebb-Williams mazes as above for 2 additional weeks, then exposed to a 4-arm baited configuration of the RAM (see below). It is important to note that RAM testing served both as a training paradigm as well as an outcome measure. Animals in the PA/CA group (n = 8) were exposed to both procedures as outlined above: that is, these animals were exposed to 4 hours of daily treatment for 5 d/wk (unless otherwise indicated), counterbalanced for treatment (Table 1). Sedentary control animals (n = 8) were housed in pairs in standard cages and handled similarly as animals in the other groups.
Experimental Paradigm and Timeline for Experiment 1
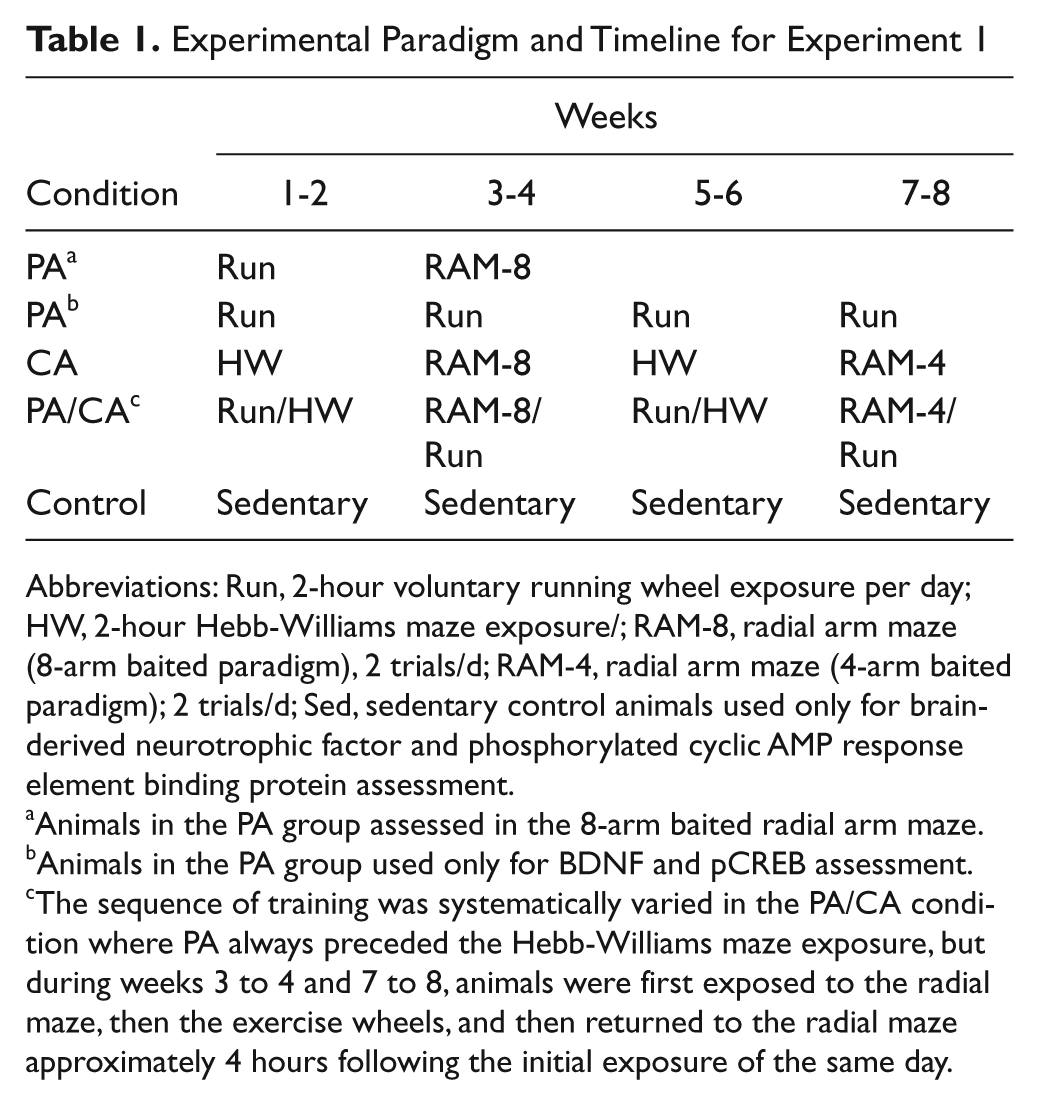
Abbreviations: Run, 2-hour voluntary running wheel exposure per day; HW, 2-hour Hebb-Williams maze exposure/; RAM-8, radial arm maze (8-arm baited paradigm), 2 trials/d; RAM-4, radial arm maze (4-arm baited paradigm); 2 trials/d; Sed, sedentary control animals used only for brain-derived neurotrophic factor and phosphorylated cyclic AMP response element binding protein assessment.
Animals in the PA group assessed in the 8-arm baited radial arm maze.
Animals in the PA group used only for BDNF and pCREB assessment.
The sequence of training was systematically varied in the PA/CA condition where PA always preceded the Hebb-Williams maze exposure, but during weeks 3 to 4 and 7 to 8, animals were first exposed to the radial maze, then the exercise wheels, and then returned to the radial maze approximately 4 hours following the initial exposure of the same day.
Experiment 2
There were 2 experimental conditions in experiment 2 (Table 2). The animals in the physical activity-2 (PA-2) group (n = 8) were exposed to the same paradigm as in experiment 1, consisting of voluntary wheel access for 2 h/d, 5 d/wk for 2 weeks. Animals in the physical activity-4 (PA-4) group (n = 8) were exposed to voluntary wheels for 4 h/d, 5 d/wk for 2 weeks. This condition controlled for the greater “enrichment” of the PA/CA condition in Experiment 1, where animals received 2 hours of running plus 2 hours of Hebb-Williams maze exposure. PA-2 and PA-4 animals were then tested in the 8-arm baited version of the RAM as above (2 trials/d for 10 consecutive days) to assess the effects of activity duration/intensity on learning and memory abilities.
Experimental Paradigm and Timeline for Experiment 2

Abbreviations: PA-2, 2 hours of voluntary wheel running; PA-4, 4 hours of voluntary wheel running; Run, 2 or 4 hours of voluntary running wheel exposure per day; RAM-8, radial arm maze (8-arm baited paradigm), 2 trials per day.
Hebb-Williams Maze Training
Animals in the CA and PA/CA conditions were placed with a cage mate in modified Hebb-Williams mazes (100 × 100 × 20 cm 3 ) 24 for 2 h/d, 5 d/wk during weeks 1, 2, 5, and 6 (Table 1). Hebb-Williams maze configurations were changed daily, so that animals had only 1 repeated configuration throughout the entire study. Numerous objects were scattered throughout each maze, and these were replaced each week to increase novelty. Furthermore, Hebb-Williams maze training was conducted in a separate room from that used for RAM testing to control for room and cue familiarity during testing.
Radial Arm Maze Training
Two configurations (8- and 4-arm baited paradigms) of the RAM (70-cm arms, 12-cm width, 35-cm center platform, and 20-cm clear Plexiglas walls) were used as previously described.23,25 All animals, regardless of condition, were mildly food restricted (~90%-95% ad libitum body weight), and the animals in the PA, CA, and PA/CA groups underwent RAM training. A 45-mg food pellet (TestDiet AIN-76A Rodent Tablet, 45 mg; Richmond, Indiana) was placed at the end of each arm during testing (at the end of 4 arms for the 4-arm baited paradigm). Briefly, animals were acclimated to the maze for 2 consecutive days with food pellets scattered throughout the maze. Subsequently, each animal was placed in the center platform and allowed access to the maze until all 8 (or 4) arms were visited. Latency and working memory errors were recorded in the 8-arm paradigm (reentry into a previously visited arm), and latency, working, and reference memory errors (entering a nonbaited arm) were recorded in the 4-arm baited paradigm. Furthermore, the accuracy of the animals was determined by recoding the number of correct choices on the first 8-or-4 arm selections in the 8- or 4-arm RAM, respectively. The 8- and 4-arm paradigms occurred at weeks 3 and 7 of the experimental study, respectively (Table 1). CA consisted of 2 trials/d for 10 consecutive days.
Tissue Processing
Following completion of the behavioral training, animals were immediately removed from their respective training sessions, anesthetized (4.0% isoflurane in 30%:70% O2:N2O mixture), and decapitated between the hours of 10:00 to 14:00 to control for the diurnal effects of BDNF. 26 Brains were quickly removed, and the left hippocampus was dissected as previously described. 27 In addition, a wedge section of the cortex corresponding to the prefrontal and motor cortex extending ventrally to the corpus callosum 28 was also dissected, and both were weighed, rapidly frozen in liquid nitrogen, and stored at −80°C.
Enzyme-Linked Immunosorbant Assay (ELISA)
Hippocampal and cortical tissue were homogenized in 3 times by volume of ice-cold lysis buffer for pCREB per manufacturer’s instructions, incubated on ice for approximately 30 minutes, and centrifuged at 14 000
Statistical Analyses
All analyses were conducted using the statistical package for the social sciences (SPSS; v 13.0.0 for Mac OS X, SPSS Inc, Chicago, Illinois). All behavioral data are presented as mean ± standard error of the mean and were analyzed using repeated measures analysis of variance (ANOVA), except trial 1 RAM-8 latency data, where a 2-tailed independent
Results
Experiment 1
Running distances
Repeated-measures ANOVA of animals’ running distances (Figure 1) revealed a significant effect of Time (
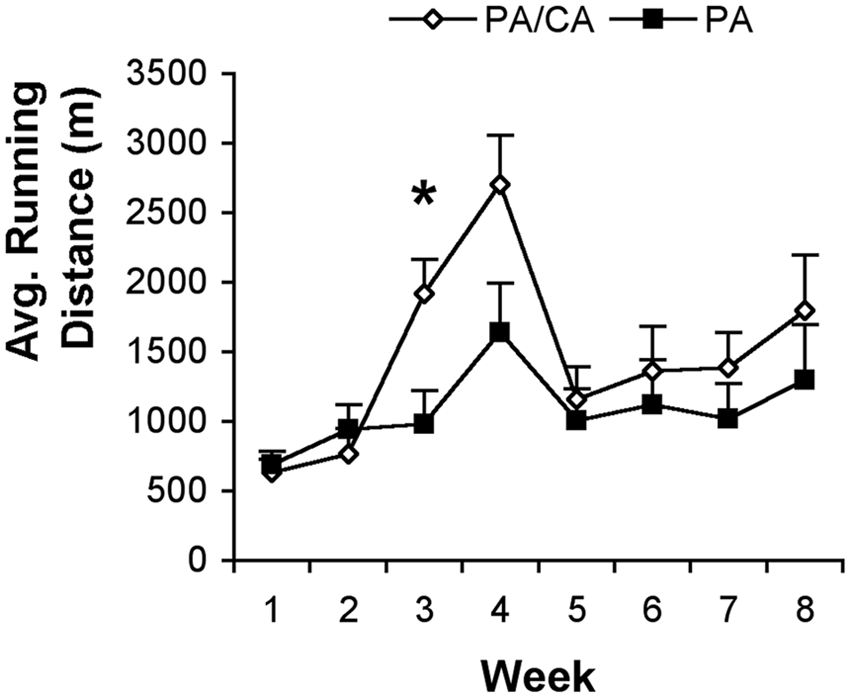
Average (±standard error of the mean) running distance in meters. Animals in the physical activity (PA) and the combined PA and cognitive activity (PA/CA) groups were exposed to voluntary running wheels for 2 h/d, 5 d/wk over a period of 8 weeks. Animals in the PA/CA group ran significantly greater distances during week 3 (*
Radial arm maze: 8-arm baited
The performance of animals was averaged over 2 consecutive days for the 10-day assessment period. Because there were 2 trials/d, each block of data represents 4 trials in the RAM and is presented as such. There were no differences among all conditions with respect to latency to complete the RAM-8 (
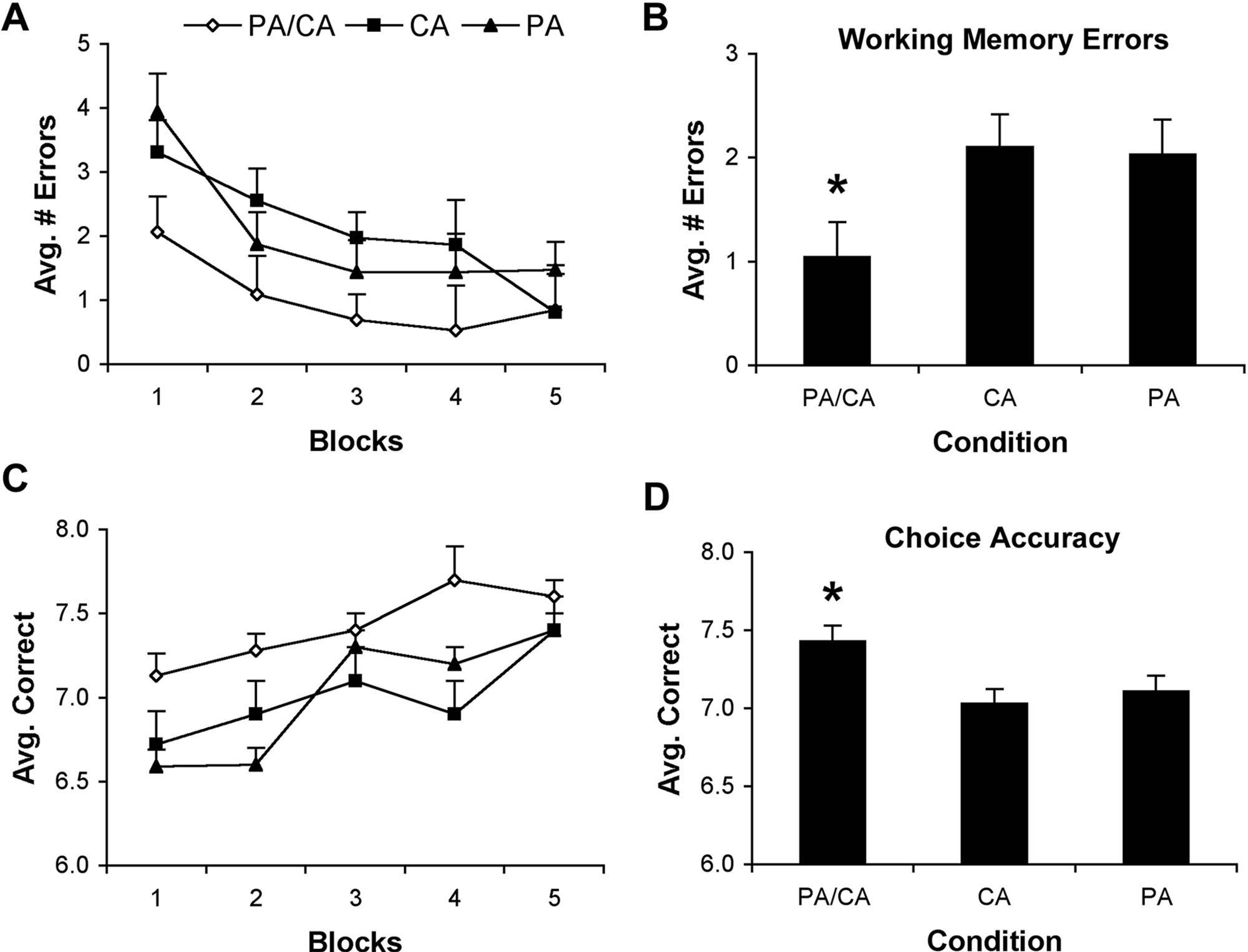
Performance (mean ± standard error of the mean) in the 8-arm baited configuration of the radial arm maze. (A and B). Animals in the physical activity and cognitive activity (PA/CA) group made significantly fewer working memory errors over the entire training period than those in the CA-alone or PA-alone groups (*
Analysis of animals’ choice accuracy revealed a similar effect, where there was a significant effect of Time (
To assess the effects of PA alone on cognitive abilities, an additional group of animals (n = 8) exposed to running wheels for 2 weeks prior to RAM (similar to rats in the PA group used only for protein measurements and those in the PA/CA group) were assessed in the 8-arm baited configuration of the RAM. Analyses revealed a significant effect of Time (
As above, analysis of the choice accuracy of animals in the PA group revealed a significant effect of Time (
Radial arm maze: 4-arm baited
There was no difference between animals in the CA and PA/CA groups with respect to latency to complete the RAM-4 (
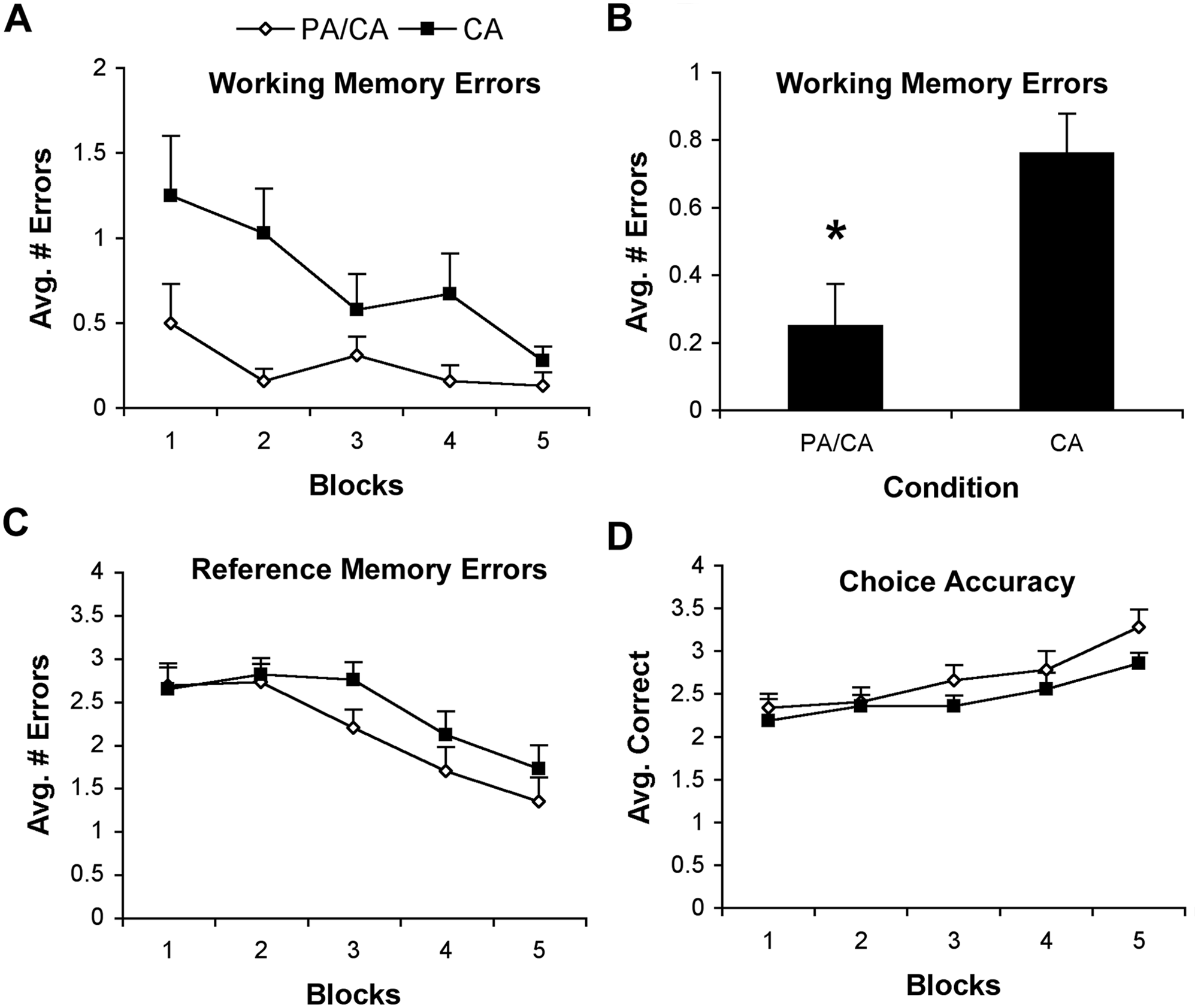
Performance (mean ± standard error of the mean) in the 4-arm baited configuration of the radial arm maze (RAM). (A and B). Similar to the 8-arm baited configuration, physical activity and cognitive activity (PA/CA) significantly increased the working memory abilities of animals compared with CA alone in the 4-arm baited RAM configuration (*
Experiment 2
Running distances
One animal was excluded (PA-2) from the running analyses because running distances were >12 standard deviations above the group mean. There was no effect of Time (
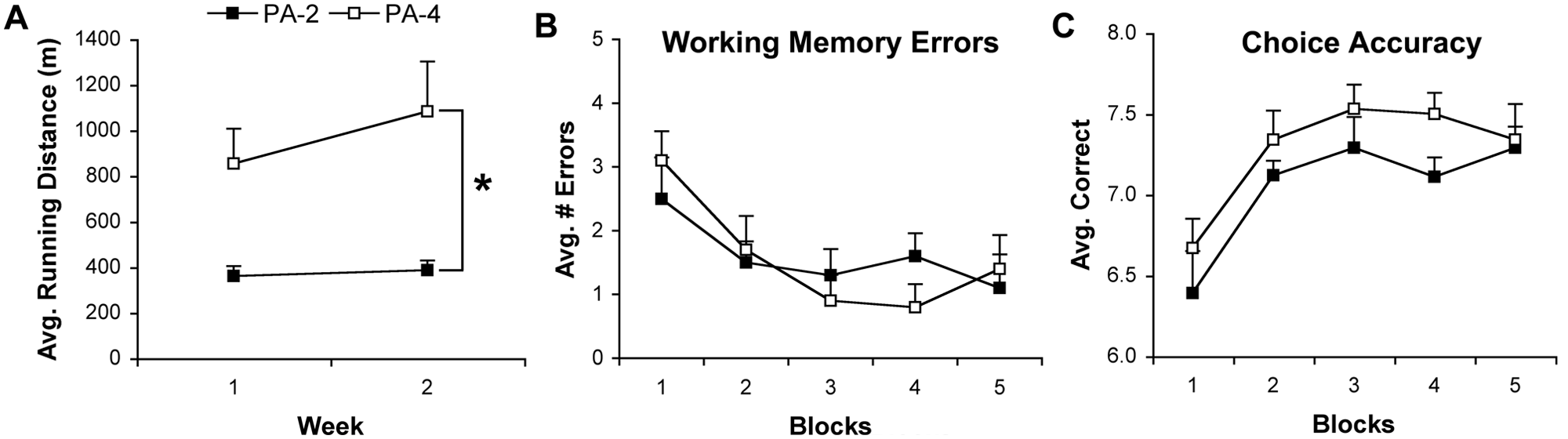
(A). Average running distance in meters (mean ± standard error of the mean [SEM]) of animals exposed to either 2 (physical activity [PA-2]) or 4 hours (PA-4) of running. PA-4 animals ran significantly greater distances than PA-2 animals (*
Radial arm maze: 8-arm baited
As mentioned above, the performance of animals was averaged over 2 consecutive days (4 trials) for the 10-day assessment. One animal was excluded (PA-2) because it did not move while in the maze. Repeated-measures ANOVA of working memory errors revealed a significant effect of Time (
Similar to the data above, repeated-measures ANOVA of the choice accuracy of animals revealed a significant effect of Time (
ELISA Measurements
Univariate ANOVA showed a significant difference among conditions in experiment 1 with regard to hippocampal BDNF concentrations (
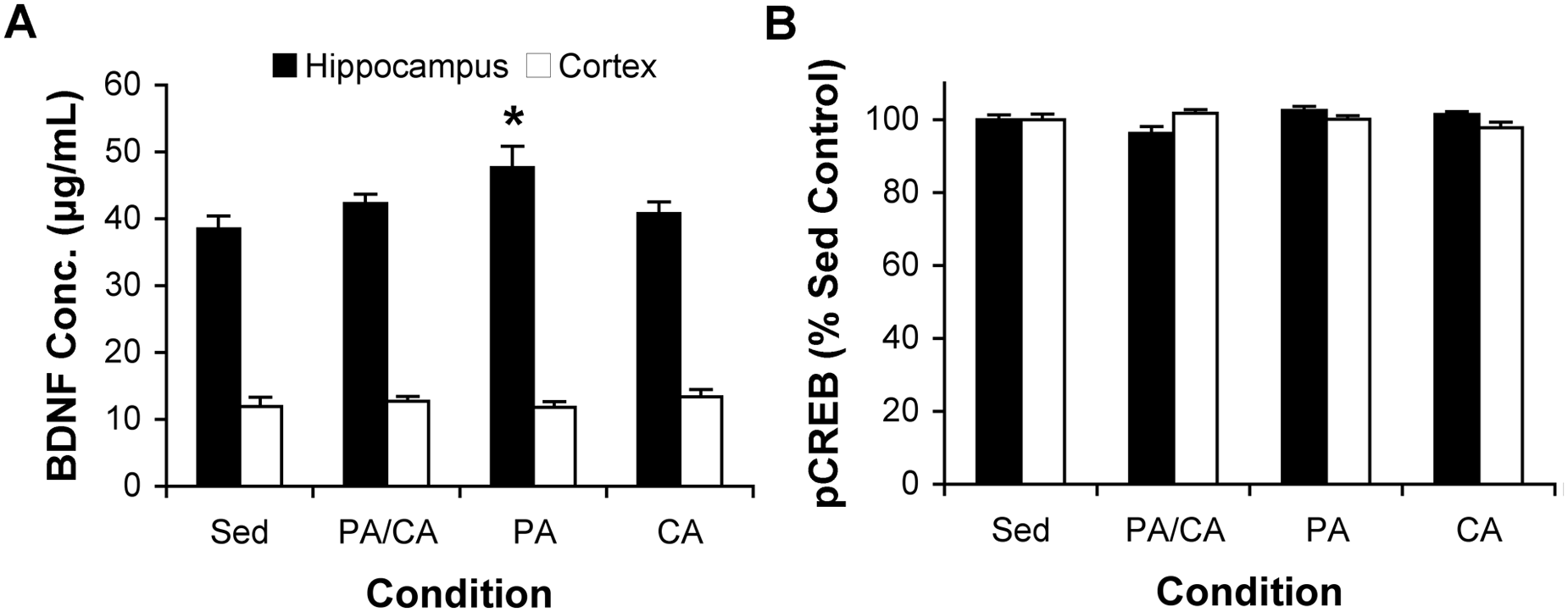
(A.) Average BDNF concentrations (in µg/mL) ± standard error of the mean (SEM) in the hippocampus and cortex of animals in experiment 1 as assessed using ELISA; 2 hours of physical activity (PA) significantly increased hippocampal BDNF levels compared with levels of sedentary controls animals (*
Discussion
Although different levels of exercise or cognitive stimulation can be controlled within the laboratory, surprisingly few animal studies have examined the effectiveness of a combination of PA and CA on brain health. 15 In the current study, we developed a novel PA and CA paradigm that resulted in superior performance in a task of learning and memory, independent of BDNF and pCREB elevations.
Animals exposed to a combination of 2 hours of PA and 2 hours of CA displayed superior performance in both working memory and choice accuracy in an 8-arm baited configuration of the RAM, compared with those exposed to either CA or PA alone, and working memory in a 4-arm baited paradigm, compared with those exposed to CA alone. Cortical and hippocampal BDNF levels were slightly elevated in all experimental groups compared with sedentary controls after 8 weeks of training; however, only 2 hours of PA significantly elevated hippocampal BDNF levels (no effect in the cortex). Furthermore, neither cortical nor hippocampal pCREB levels were altered by any of the treatments.
Combining Hebb-Williams and RAM maze training with PA improved learning and memory. Although the RAM served primarily as a training paradigm, we were also able to use the data to assess the animals’ acquisition of information (learning) and retention as well as choice accuracy. Because there was a significant difference between animals in the PA/CA and CA-alone groups in the above dependent measures, we sought to determine whether this was solely a result of the effects of PA as has been shown with a short exercise paradigm (3 hours) in female mice. 29 A separate group of animals was exposed to voluntary running wheels for 2 weeks and then tested in the RAM (without simultaneous wheel exposure to control for activity combination effects). These animals performed similarly to animals in the CA-alone group with respect to the number of working memory errors and choice accuracy but significantly poorer than PA/CA animals. We also assessed whether the improved performance of the animals in the PA/CA group relative to the PA- and CA-alone groups was the result of a dose or intensity effect, whereby more activity is better. That is, the PA/CA group received a total of 4 hours of “enriching” activity, whereas animals in the PA and CA conditions received only 2 hours of activity. In experiment 2, we controlled for this possibility by exposing animals to either 2 or 4 hours of running. Although PA-4 animals ran more than twice the distance of PA-2 animals, this did not translate into corresponding improvements in working memory or choice accuracy in the 8-arm baited RAM. Clearly, these data indicate that the combination of PA and CA has a synergistic effect on learning and memory outcome that is independent of the dose or intensity of activity. This finding is interesting in light of a recent clinical study in which the intensity of CA was not associated with improved cognitive abilities; rather, it was the diversity or number of different cognitive activities that was a significant predictor of cognitive performance. 30 Furthermore, 2 clinical meta-analyes within the past 10 years have shown that low-moderate levels of physical exercise have similar effects as high levels in reducing cognitive decline in normal and “clinical” participants, potentially indicating a plateau effect of PA by itself.31,32 This plateau appears to be true for aerobic activity alone, where combining aerobic activity with strength training regimens results in greater cognitive improvements. 31
An additional important finding from this study is the transfer of improvements from training in the Hebb-Williams maze to performance of working memory in the RAM for the PA/CA group. The paradigm of modified Hebb-Williams maze training likely involved more reference memory training (eg, object recognition) 33 than working memory or spatial memory training as originally designed, 24 although we cannot exclude the possibility that these cognitive processes were indeed activated. 34 It is interesting to note that these improvements were not general in nature because they were not observed in the CA group compared with the PA condition but only in the PA/CA group, thus leading one to speculate that the combination of PA and CA had a positive effect on this transfer. Notably, our results contrast with clinical data where only cognitive training was used. In such studies, the transfer of abilities from one cognitive domain (eg, attention) to another (eg, working memory) are often difficult to demonstrate.35-37
Both retrospective and prospective clinical studies reported a lower incidence of cognitive deficits and dementia among individuals who regularly engaged in physical exercise.7-9 This was also true for a recent randomized controlled trial, where 6 months of exercise resulted in improvements in cognition at follow-up in patients with mild cognitive impairment. 38 Several animal studies have also shown that physical exercise promotes improvements in cognitive function.6,39 One difficulty in the translation between the animal and clinical data presented above is the definition of PA. PA in the clinical literature often refers to chronic activity over a 5-year period where moderate to high levels corresponded to once per week to ≥3 times per week of activity, respectively. 7 However, in preclinical studies, animals are often housed in isolation and have 24-hour access to running wheels throughout the entire study,4,39,40 sometimes running distances of up to 48 km per day. 12 These running paradigms induce changes in reward pathways that are similar to compulsive stimulant use 41 and therefore may have little in common with typical exercise patterns in humans.
Our PA paradigms included much shorter durations and less frequent access to running wheels to better approximate human exercise patterns. It is interesting to note that these exercise paradigms produced only slight elevations in neuronal BDNF levels in all experimental conditions. Based on previous work,12,42 longer running wheel exposure (eg, 24 hours) would likely produce larger increases in BDNF levels; however, these may not relate to human exercise patterns. Moreover, the observation that hippocampal BDNF was significantly increased in the PA but not in the PA/CA group suggests that elevation of BDNF is not a necessary condition for cognitive enhancement because only the PA/CA group showed improved learning and memory. CA is also thought to protect the brain by delaying or preventing the onset of dementia. Based on diary records, nuns who had higher linguistic abilities and idea and grammatical complexity in early life were less likely to suffer from dementia than their less intellectual counterparts. 13 Furthermore, more diverse and intense intellectual activity in midlife is associated with a lowered incidence of Alzheimer disease in later life. 8 Participating in leisurely activities later in life such as playing board games, reading, and playing musical instruments decreased the likelihood of developing dementia, 43 and CA diversity has been shown to predict cognitive performance. 30
It is difficult to determine the extent to which intellectual activity or PA independently contribute to improved cognitive outcome in older adults. 8 It is possible that both PA and CA interventions share underlying molecular mechanisms, given that PA and CA both improve cognitive functioning in humans and animals and also slow age-related cognitive decline.2,44 Although learning in the Morris water maze is associated with increases in BDNF, FGF2, synapsin-I, and TrkB mRNA levels in the hippocampus,15,16,45 it is unknown whether these changes correspond to changes in protein levels.
Clearly, additional studies are needed to identify the underlying mechanisms responsible for the cognition-enhancing effects of our combination therapy. For example, exercise increases dendritic length and complexity in the dentate gyrus 46 as well as neurogenesis.6,39,47 Such changes may also contribute to the benefits arising from our PA/CA combination.
A limitation of the current study is the age of the animals. As with most other preclinical studies,15,16,29,39 young adult animals were used in our experiments. Nonetheless, our results provide a template for future experimentation using aged animals with subsequent evaluation in a clinical population.
Conclusions
A growing body of evidence indicates that PA improves cognition in both animals and humans17,47,48 paralleled by anecdotal literature suggesting that similar benefits can be derived by engaging in lifelong intellectual activities (eg, reading, crossword puzzles). This is of paramount importance because, with an aging population, the incidence of cognitive decline will increase to epidemic proportions in the coming decades.2,49 Consequently, therapies that can reverse or delay cognitive decline are urgently needed. The present results strongly suggest that a more effective means for improving and maintaining cognition is a combination of PA and CA instead of a monotherapeutic approach.
Footnotes
Acknowledgements
The authors thank Garry Chernenko, Shirley Granter-Button, and Julia Curtis for technical assistance.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This research was supported by the Natural Sciences and Engineering Research Council of Canada. DC holds a Canada Research Chair in Stroke and Neuroplasticity. KDL was supported by a Heart and Stroke Foundation of Canada/CIHR/Canadian Stroke Network/Astra-Zeneca Focus on Stroke Doctoral Fellowship.
