Abstract
Introduction
Cognitive impairment, cardiovascular deconditioning, and muscle weakness are well-established sequelae of stroke and contribute to the impairment that has made stroke one of the leading causes of serious, long-term disability in North America.1,2 Cognitive impairment occurs in up to 64% of people who have had a stroke 3 and has been associated with a 3-fold increase in risk for mortality, 4 increased rates of institutionalization, 5 and decreased instrumental activities of daily living function. 6 Moreover, it places individuals who have had a stroke at a greater risk of developing dementia. 7 Data from older adult and animal models provide compelling evidence that exercise can improve cognition and promote neurogenesis. 8 In the healthy population, it is becoming increasingly evident that both aerobic training (AT) and resistance training (RT) have the potential to improve cognition. A meta-analysis of 18 studies showed that programs of both AT+RT resulted in significantly greater improvements in cognition than programs of AT alone. 9 Recent evidence has linked brain atrophy and poor cognitive performance with reduced fat-free mass in people with early Alzheimers disease, 10 suggesting that exercise such as RT that provides anabolic stimuli11,12 may have the potential to ameliorate cognitive deficits. However, few studies have examined the impact of an exercise program on cognition in individuals following a stroke, and the focus has largely been on examining the effect of AT alone.13,14 Moreover, to our knowledge, no study has explored the association between change in cognition and change in fat-free mass as a result of an exercise training program.
Accordingly, the aim of this investigation was to evaluate the effects of a 6-month exercise program of AT+RT on cognition and to explore associations between change in cognition and change in physiological outcomes attributed to AT (cardiovascular fitness) and RT (fat-free mass). Examining the benefits of a combined training approach (AT+RT) and the interaction between cognition and physiological outcomes would aid in selecting optimal treatment strategies for improving physiological and cognitive well-being poststroke.
Methods
Patients were referred to Toronto Rehabilitation Institute’s Risk Factor Modification and Exercise Program following Stroke (TRI-REPS) from outpatient stroke rehabilitation programs, primary care physicians, and from the community. The TRI-REPS program is a substream of the institution’s cardiac rehabilitation program. Consecutively enrolled patients ≥10 weeks poststroke, with a stroke-related motor impairment score of <7 on the Chedoke-McMaster Stroke Assessment scale of the arm, hand, leg, or foot (described below) were approached for participation in this study. All patients were able to ambulate ≥10 m independently with/without an assistive device, with no significant limitations caused by pain and were excluded if they had other neurological conditions, contraindications to maximal exercise testing such as severe aortic stenosis, significant cardiac arrhythmia, or uncontrolled resting severe hypertension. 15 Only 1 patient declined to participate.
Outcome measures were conducted at baseline and following 6 months of exercise training in the same order. The Centre for Epidemiological Studies Depression (CES-D) Scale questionnaire was completed on the same day but prior to the cardiopulmonary exercise test. The Montreal Cognitive Assessment (MoCA) was conducted on a separate day prior to other assessments to avoid the effect of fatigue. On a subsequent visit, a dual-energy X-ray absorptiometry (DXA) scan was conducted. The study was approved by the institution’s research ethics board, and written informed consent was obtained from all participants.
Adapted Cardiac Rehabilitation Program
The TRI-REPS service for stroke patients is similar in design to the traditional cardiac rehabilitation program. 16 Patients attended a 90-minute exercise class once per week for 6 months. Nutrition and psychosocial counseling, education sessions, cardiac exercise assessments, and plasma glucose and lipid monitoring were offered. The case manager prescribed an individualized AT program in the first weekly session, followed by an RT program in the following session. When patients were ready to perform RT at home (typically after 2-3 supervised sessions), they were provided with a booklet that included instructions for lifting weights safely, how to progress with the exercises, as well as pictures and a written description of each exercise. Patients were advised to complete 4 additional AT and 1 to 2 RT sessions at home. Patients were required to keep a detailed record of each exercise session, noting the precise distance walked, duration (walk/cycle), resting and peak heart rate, and any symptoms experienced during exercise. RT records included the amount of weight lifted and the number of repetitions and sets performed for each workout. This log was submitted and cross-validated by a cardiac rehabilitation supervisor at the patient’s weekly visit to the center. Patients were trained to measure resting and exercise heart rates at orientation to the program, and accuracy was checked at each weekly visit to the center.
Walking, stationary recumbent, or upright cycling were the modes of AT prescribed depending on individual ability and access to equipment when away from the center. Treadmill or overground walking was considered for those who could sustain high enough speeds and durations to achieve aerobic benefit. Cycle ergometer exercise was prescribed to patients when stroke-related deficits precluded walking. The AT intensity was based on data from the cardiopulmonary exercise test. The goal was to progress patients to 20 to 60 minutes of exercise, 17 5 times per week, 16 at an intensity considering a combination of the following: 40% to 70% of heart rate reserve or VO2peak, 17 the heart rate achieved at the ATge, 18 and/or a rating of perceived exertion of 11 to 16 (“light” to “hard”) on the Borg 6-20 Scale. 19 Prescriptions were initially progressed by increasing duration to ≥20 minutes and then increasing intensity to 70% of heart rate reserve and/or the heart rate achieved at/above the ATge (maximum duration of 60 minutes). Thereafter, training intensity was adjusted to maintain a heart rate equivalent to 70% of heart rate reserve and/or that achieved at the ATge on the graded exercise test. Exercise diary information, heart rates measured at the center, and communication with the patient assisted the case manager in deciding when to increase the prescription.
For RT, the exercises were task specific, incorporating muscle actions that are performed during daily activities. Resistance was provided by hand-held dumbbells, exercise bands (wrist/ankle attachments), or patients’ body weight. A weight load equivalent to 50% to 60% of 1 repetition maximum (1 RM) was prescribed on the nonaffected limb. On the affected limb ≥50% of 1 RM and/or a resistance rated as 13 to 14 on the Rating of Perceived Exertion Scale on the last repetition of the set was prescribed. 12 Patients gradually progressed from 10 to 15 repetitions and then increased resistance by 1.6 to 5 kg or increased the exercise band level and then reduced repetitions to 10 and repeated this process.
Cognitive Testing
The MoCA was designed as a screening instrument for mild cognitive dysfunction. 20 The MoCA scores range from 0 to 30 points, and the score has a high sensitivity (77%) and specificity (83%) for detecting mild cognitive impairment (MCI) in patients in the chronic stage of stroke as determined by a score of <25.21,22 However, to ensure that the results were qualitatively similar, a threshold of <24 was also used because this threshold has been reported as being the most accurate for predicting MCI in individuals with cardiovascular disease who are at greater risk of MCI than those in the normal population. 23 The MoCA assesses 7 different cognitive domains, including the following: visuospatial and executive function with the adapted Trail Making B task, 3-dimensional cube copy, and clock drawing (5 points); naming with confrontation naming (lion, rhinoceros, and camel; 3 points); attention and concentration, including digits forward and backward, vigilance (finger tapping at the letter “A” in a list of letters), and serial subtraction (6 points); language, including sentence repetition and verbal fluency (3 points); abstraction, including similarities between 2 items (2 points); delayed recall, including short-term memory recall of a list of 5 words (5 points); and orientation, including date, month, year, day, place, and city (6 points). A point was added to the total score for those with 12 or fewer years of education. Koski et al 24 have shown that the MoCA is a reliable and valid quantitative estimate of cognitive ability and can be used to track changes in cognitive ability over time in a clinical setting. The assessment was conducted in a quiet room without distractions by 2 individuals trained in administration of the MoCA questionnaire.
Cardiopulmonary Exercise Testing
Medical history and anthropometric measures were collected prior to each cardiopulmonary exercise test. A symptom-limited cardiopulmonary exercise test on a recumbent cycle ergometer with specialized pedals to secure feet (Ergoline Select 1000, Germany), upright cycle (Ergoselect 200P, Germany), or a treadmill was performed at baseline and after 6 months of training. The type of ergometer was chosen based on balance and control of leg/foot position in pedals. Work load was increased by either 8.3 or 16.7 W every minute. Breath-by-breath gas samples were collected via a calibrated metabolic cart (SensorMedics Vmax Encore, California) with continuous monitoring of 12-lead ECG (Marquette Case 8000). Blood pressure was measured at 2-minute intervals. The expected VO2peak (mL·kg−1·min−1) was calculated from established age and sex norms 25 for comparison of cardiopulmonary fitness.
Gas Exchange Anaerobic Threshold (ATge)
We chose change in oxygen uptake (mL·kg−1·min−1) at the ATge as the independent variable reflecting AT performance rather than peak oxygen uptake (VO2peak). Although improvements in VO2peak are important for prognostic outcome, delaying the point at which lactate accumulates during exercise has important clinical implications for improving submaximal endurance performance. 26 More important, measurement of maximal cardiovascular fitness may be limited by motor and neurological impairments that may prevent individuals from reaching a “true” physiological maximum 27 (VO2peak) on the cardiopulmonary exercise test, thereby mitigating the utility of VO2peak measures in correlation analyses.
ATge was determined by a combination of the V-slope method and the ventilatory equivalents methods (Figure 1).28,29 The V-slope method was defined as the point of departure from linearity of carbon dioxide output plotted against oxygen uptake. The ventilatory equivalent method was defined as the level corresponding to the rise in ventilatory equivalent of oxygen that occurs without a concurrent rise in the ventilatory equivalent of carbon dioxide. The ATge is considered a valid measure demonstrating high reproducibility and low interobserver variability when established standards are followed.30,31 ATge was determined by 2 independent assessors experienced in threshold analysis blinded to the objectives of the study. The final ATge was the average of that obtained by the readers if the values differed by <10%; otherwise, they were resolved by consensus. The ATge of both readers were either within 10% of each other or the readers were in concordance that the ATge was indeterminate for 75.6% of the tests at baseline and 80.5% of the tests at 6 months. ATge was discernible at both baseline and 6 months in 68.3% (n = 28) of patients.
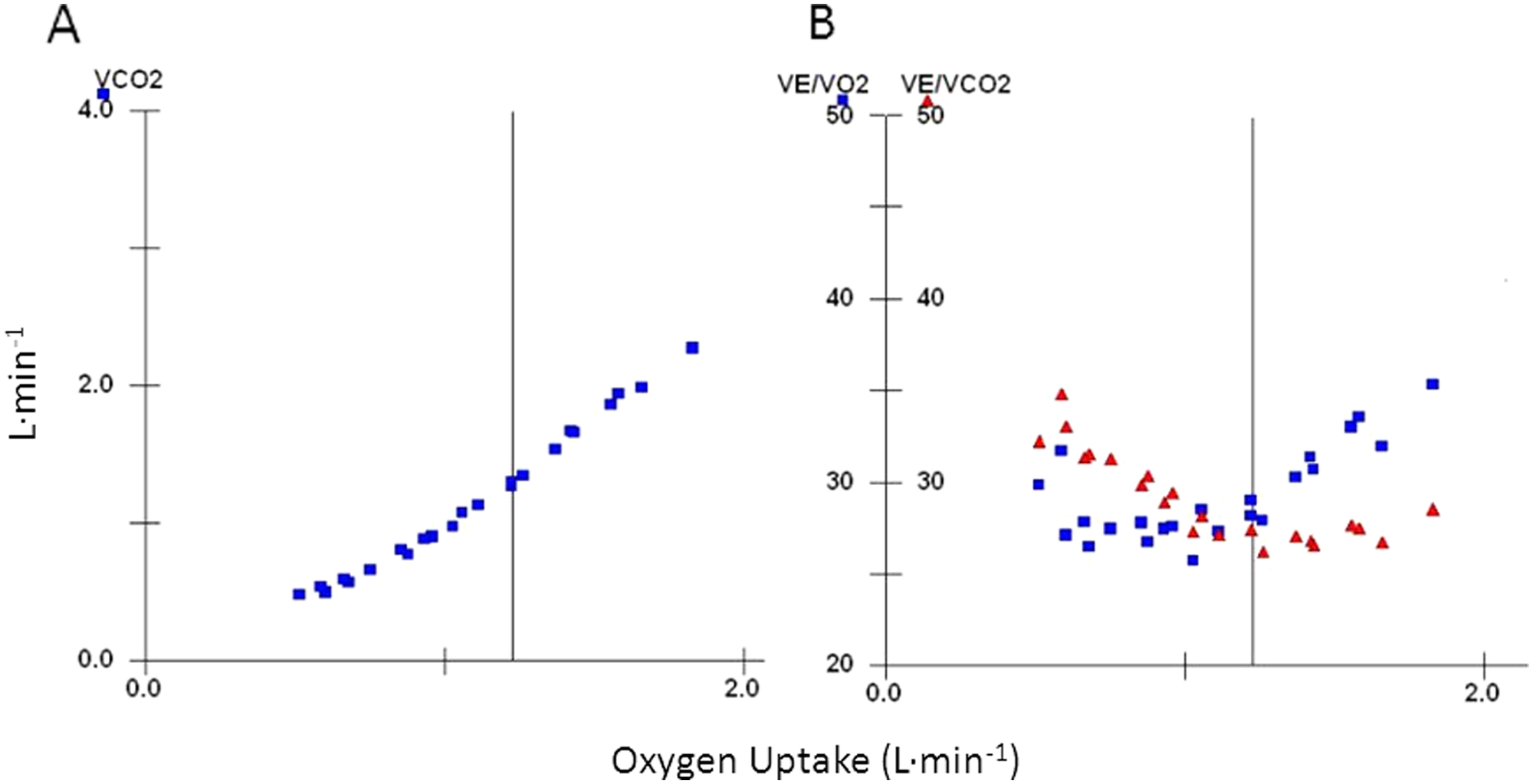
The gas exchange anaerobic threshold determined for 1 patient using the V-slope method (A) and ventilatory equivalent methods (B). VE/VO2, ventilatory equivalent of oxygen; VE/VCO2, ventilatory equivalent of carbon dioxide.
Body Composition
Total body and regional measurements (arms, legs, and trunk) of fat-free mass, percentage body fat, and fat mass were determined by DXA (HOLOGIC QDR 4500 W, Software version 12.3). Prestudy and poststudy tests were conducted by the same operator. This method of body composition measurement has been shown to have a high level of precision. 32
Muscular Strength
The 1 RM test was performed at baseline and following 6 months of training on the bicep curl exercise using hand-held dumbbells and on the leg extension and leg curl exercise using a fixed-weight machine, as described elsewhere. 12 After 3 repetitions of a warm-up weight, a heavier weight was attempted once following a 2-minute recovery period. One repetition at a heavier weight was attempted every 2 minutes until the patient was unable to lift the weight with a proper technique.
Additional Baseline Measures
Baseline assessments included motor recovery stage of the arm, hand, leg, and foot of the stroke-affected side classified on the 7-point Chedoke-McMaster Stroke Assessment Scale. 33 A Chedoke-McMaster Stroke Assessment Scale motor impairment score of 1 indicates flaccid paralysis, 3 describes marked spasticity and weakness, 6 indicates near-normal coordination of patterns of movement and no spasticity, and 7 describes normal movement. 33 Depressive symptoms were assessed using the CES-D scale, with scores ≥16 demonstrating high sensitivity and specificity for detecting depression 34 ; this is a valid and reliable measure for the stroke population. 35 The 6-minute walk distance (6MWD), as described elsewhere, 36 was conducted preprogram and postprogram.
Statistics
Data are presented as means ± standard deviation (SD) unless otherwise indicated. Baseline and postexercise scores were compared using Student
Results
Of the 45 patients who agreed to participate in the study, 91% (n = 41) completed 6 months of exercise training. Reasons for discontinuing the program were reoccurrence of cancer (n = 1), transportation difficulties (n = 1), reoccurrence of stroke (third) unrelated to exercise (n = 1), and death of spouse (n = 1). Participant characteristics are presented in Table 1. Completers were a mean of 63.6 ± 13.5 years of age, and 74 ± 134.5 weeks (range = 10-650) poststroke, with the majority being >6 months poststroke (80.5%). Most patients were men (73.2%), postischemic stroke (65.9%), using an assistive device for ambulation (75.6%) with MCI (65.9%). At baseline, 22.5% of patients had depressive symptoms. Patients attended 83.5% ± 16% of weekly classes and completed 70% ± 37.5% of prescribed AT and 80.4% ± 46.4% of prescribed RT sessions. Although each RT program was individualized, the forward lunge, standing squat, and bicep curl exercises were common to most patients. Mean AT distance, time, intensity, and heart rate as well as the RT weight load lifted, ascertained by diary and cross-validated by in-class performance, increased significantly (Table 2). There were no associations between the change in cognitive outcomes and either accomplished work over time or change in 6MWD.
Baseline Characteristics. a

Abbreviations: AT, aerobic training; RT, resistance training; SD, standard deviation.
All data are presented as mean ± SD unless otherwise indicated.
Exercise Performance at Baseline and Following 6 Months of Training.
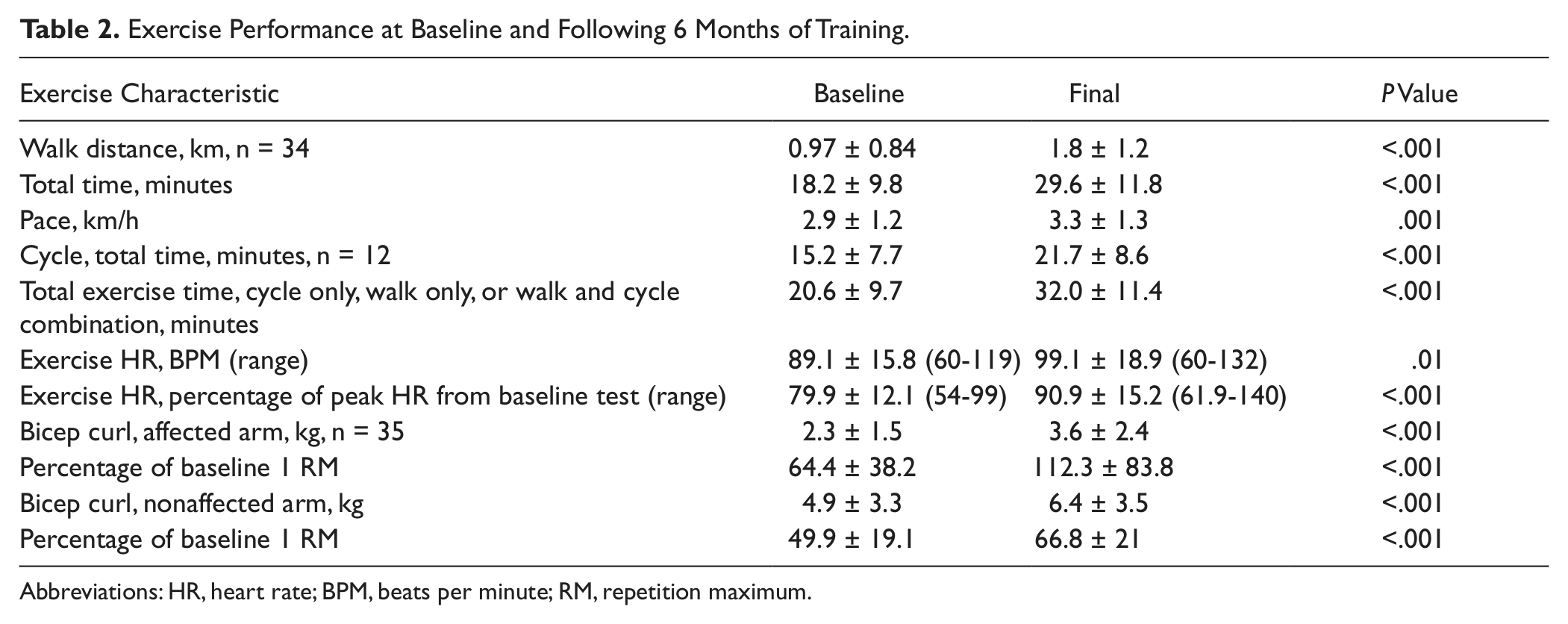
Abbreviations: HR, heart rate; BPM, beats per minute; RM, repetition maximum.
Following 6 months of AT+RT, there were significant improvements in VO2peak (
Change in Physiological and Depression Measures.
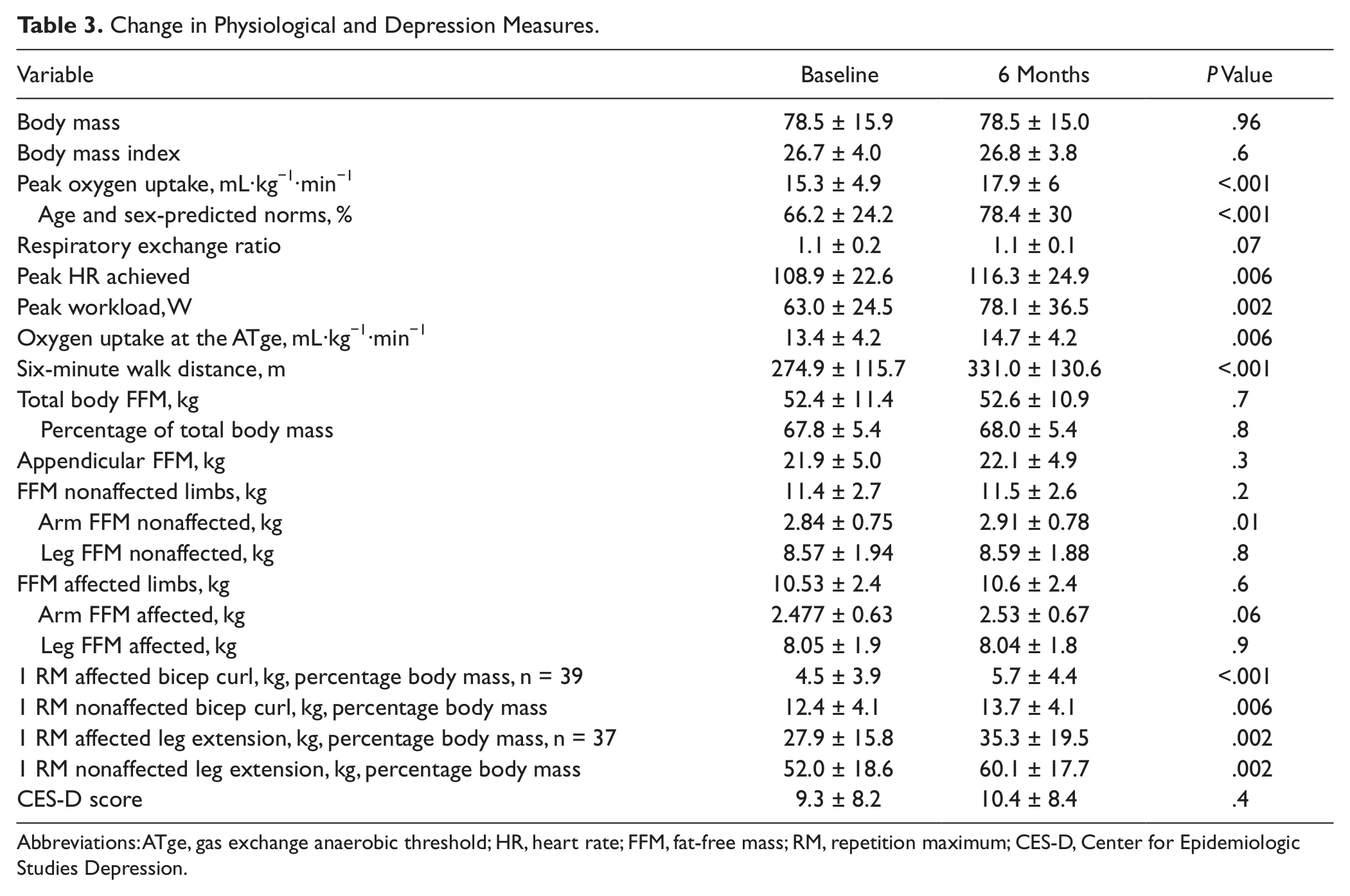
Abbreviations: ATge, gas exchange anaerobic threshold; HR, heart rate; FFM, fat-free mass; RM, repetition maximum; CES-D, Center for Epidemiologic Studies Depression.
Change in Cognitive Measures and Correlation With Time From Stroke.
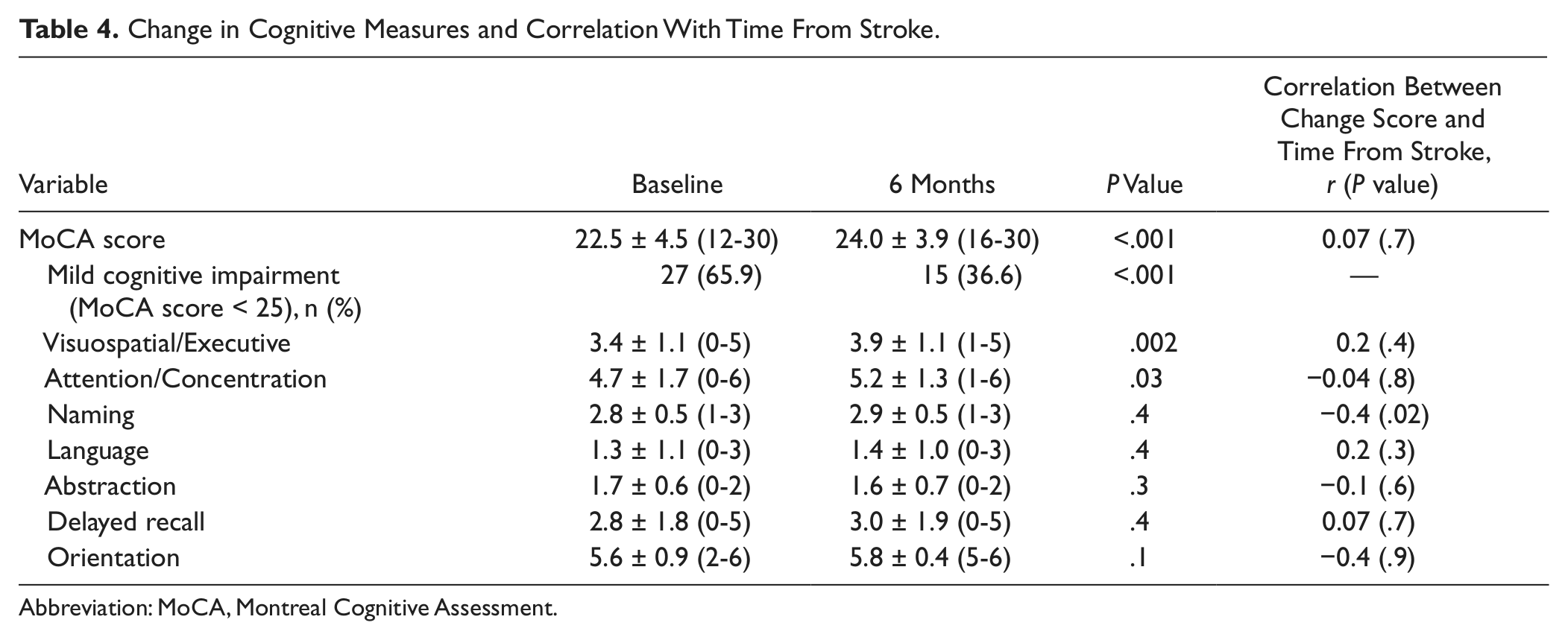
Abbreviation: MoCA, Montreal Cognitive Assessment.
There was a significant positive association between change in total cognition score and both change in fat-free mass of the nonaffected limbs (β = 0.002;
There were no differences in any of the cognitive outcomes when analyzed by sex, medications (aspirin, statins, β-blocker, and antidepressant), type of stroke (hemorrhagic vs ischemic), education, history of depression requiring medication, depression score, or comorbid diagnoses (
Discussion
In patients with residual motor impairment poststroke, a 6-month combined AT+RT exercise program resulted in significantly improved MoCA scores and a 44.5% reduction in the proportion of patients meeting threshold criteria for MCI. Improvement was characterized by gains in the subdomains of attention/concentration and visuospatial/executive functioning. These results are consistent with those from healthy, sedentary adult populations, in which AT and RT in combination resulted in significantly greater improvements than AT alone. 9 Previously, 1 study examining the effects of combined AT+RT exercise on cognition poststroke, reported significant improvement in working memory, a component of executive function, in 9 patients after 12 weeks of training. 37 In contrast, a pilot study randomizing 19 individuals poststroke to 8 weeks of AT alone resulted in greater improvements in motor learning but not executive function compared with those randomized to the control group, 14 suggesting that the addition of RT may be associated particularly with cognitive advantage.
Although the addition of RT to AT may offer incremental benefit, possible mediators of this relationship have not been studied extensively. In the present study, cognitive improvement was independently associated with fat-free mass accretion of nonaffected limbs. This finding is consistent with recent cross-sectional studies linking poorer cognitive performance with reduced fat-free mass. For instance, Nourhashemi et al 38 reported that women in the lowest quartile of fat-free mass, as measured by DXA, had an odds ratio of 1.43 for cognitive impairment compared with those in the highest quartile of fat-free mass after adjusting for confounders. Burns et al 10 evaluated 70 individuals with early-stage Alzheimer disease and 70 individuals without dementia, using brain magnetic resonance imaging, neuropsychological testing, and DXA scans. They found that there was a positive association between fat-free mass and whole brain volume in both groups. There was also a significant correlation between fat-free mass and global cognitive performance. Although these studies do not infer causation, an anabolic stimulus may enhance cognitive performance and, based on our findings, RT is feasible and potentially of clinical utility in patients poststroke. These findings strongly suggest the need for larger randomized trials.
Compared with fat-free mass accretion, cardiopulmonary fitness has been more extensively studied as a possible contributor to cognitive performance. 39 For instance, Kluding et al 37 reported a correlation between improved VO2peak and improved executive control on the Flanker test in individuals poststroke participating in AT+RT. However, an earlier meta-analysis and a systematic review of 10 and 11 studies, respectively, did not substantiate an association between changes in VO2peak and changes in cognitive function.40,41 The association we observed between change in attention/concentration and change in oxygen uptake at the ATge extends these findings. In clinical populations, factors that limit individuals in reaching VO2max on an exercise stress test, such as lack of motivation, depressive symptoms, 42 and musculoskeletal issues may reduce the utility of the VO2peak as a measure of cardiopulmonary fitness. More easily attained submaximal measures of oxygen uptake, specifically those occurring at the ATge, might provide a more reliable measure; in a previous study of 98 individuals poststroke, 67% reached ATge, whereas only 18% achieved VO2max. 27 ATge indicates the VO2 at which anaerobic energy production mechanisms contribute to the oxidative energy system, 29 with concomitant increases in ventilation and peripheral lactate concentrations. 43 Because cerebral lactate uptake is proportional to peripheral arterial concentrations, 44 the ATge might provide a more metabolically relevant measure than the VO2peak that also reflects cerebral metabolism. With a training-induced increase in oxygen uptake at the ATge, stroke patients are more likely to perform daily activities at a level where cerebral energy metabolism is predominantly aerobic. These possible implications require further investigation.
It is reported that patients following stroke with cognitive impairment will exhibit deficits in attention, executive function, and processing speed related to frontal network dysfunction. 45 Moreover, in healthy individuals, it is reported that the cognitive subdomains of attention and executive processes are the ones that will benefit most from an exercise intervention. 9 This may explain in part why there were significant improvements in the subdomains of attention/concentration and executive function and not in other subdomains in the current study.
Collectively, there is compelling evidence that a combined training approach (AT+RT) enhances cognition poststroke. Our findings indicate that accretion of muscle has the potential for cognitive gain. Although training of the affected limb is important and often the focus of treatment, RT of the nonaffected side should not be neglected. Given the link between improved oxygen uptake at the ATge and gains in cognition, exercise prescription methods should be based on results of a cardiopulmonary exercise test to ensure an exercise intensity that provides adequate cardiovascular stress to induce a training response.
A limitation of this study was the relatively small sample size of nonrandomly selected individuals following stroke and the multiple comparisons made. A further limitation was the use of a single neurocognitive measure and the pre-post test design. To elucidate a cause and effect relationship between exercise and change in cognitive function, a randomized controlled study is required. However, the independent correlation between change in physiological measures and cognitive change support an effect of the intervention.
Although we did not have a comparison control group, the relative magnitude of the physiological change suggests that both the AT and RT interventions were effective. We observed a mean increase in VO2peak (mL·kg−1·min−1) of 18.8% ± 24.4%, which is greater than the 10.7% to 13.2% improvement reported in 3 studies46-48 and 17% gain reported in 1 study 49 in chronic stroke patients randomized to an exercise intervention—changes that were significantly greater than that in control groups. Moreover, the relative magnitude of change in the present study is similar to that reported in cardiac patients (9%-18%) participating in exercise programs tested at the same center12,50 and elsewhere.51,52 In addition, the 10.8% ± 18.6% change in the ATge in the present study was similar to relative gains experienced by cardiac patients following an exercise program; 4 studies yielded changes ranging from 2% to 11%,12,51-53 and 1 study reported a 15.2% 54 change. Also, exercise training yielded a gain of 56.1 ± 47.1 m in functional exercise capacity measured by the 6MWD test and was similar to the change reported by others (49.1-64.6 m)48,49 and was significantly greater than that in control patients with chronic stroke.
The significant improvement in muscular strength for upper- and lower-body affected and nonaffected limbs suggests effective RT. However, although there was no significant increase in overall fat-free mass as measured by DXA, regional measures showed a significant change in the fat-free mass of the nonaffected arm and trend for change in the affected arm. Similarly, Ryan et al 55 reported no change in overall fat-free mass measured by DXA as a result of an RT program in people following chronic stroke. However, the investigators reported a significant increase in paretic and nonparetic muscle area of the midthigh as measured by multislice CT scanning. This suggests that CT scanning may provide a more effective measure of change in fat-free mass in stroke patients. DXA-prediction models have been reported to overestimate total body skeletal muscle by the amount of intramuscular and intermuscular fat present, which may not be considered in the prediction model. 56 Ryan and colleagues 57 demonstrated that there were significantly greater intramuscular fat deposits in paretic compared with nonparetic thigh muscle and that RT resulted in reductions in intramuscular fat in both midthighs following stroke. 55 Thus, an accurate measure of intramuscular fat in the larger muscle groups would be important for detecting change in fat-free mass in this population and may explain in part the lack of change in overall fat-free mass measured by DXA in the larger muscle groups.
Furthermore, the lack of effect that stroke recovery time to initiation of the program had on change in overall cognition in this study suggests that spontaneous recovery played an inconsequential role in outcomes. Moreover, more than 80% of the patients started cardiac rehabilitation more than 6 months poststroke, and although there does not appear to be a consensus, previous research trends toward an indication that unappreciable changes in cognition occur after the first 3 to 6 months of recovery.58-60 In view of the home-program treatment model, the added attention and interaction from the 90-minute weekly appointment is unlikely to have had a significant effect on cognition. Rather, participants would likely have experienced less social contact than received from the 3 times weekly traditional stroke rehabilitation visits that many would have completed in the first 3 to 4 months poststroke. Also, owing to the heterogeneity of mobility function, minimal between-patient interaction occurred during stationary cycling or walking on the 200-m track.
The sample of 41 participants and exclusion of patients who were not able to walk ≥10 m with/without a gait aid limits the generalizability of this study. However, the physical profile is consistent with other studies, and the high prevalence of mild cognitive deficits is consistent with that previously reported in the stroke population. 3 Finally, although we did not formally assess intertester reliability of the MoCA, the training of the 2 administrators of the MoCA was conducted by a psychologist, and informal “calibration” meetings were conducted when a score required verification.
Conclusions
A cardiac rehabilitation model of AT+RT resulted in improvements in overall cognition and in the subdomains of attention/concentration and visuospatial/executive function. There was a 44.5% reduction in the proportion of patients meeting the threshold criteria for MCI. Change in cognition was associated with change in the ATge and fat-free mass of the nonaffected limbs, indices typically attributed to AT and RT, respectively.
Footnotes
Acknowledgements
We acknowledge the contribution of Susie Ward, Dr. Gail Kunkel, Rene Belliard, Brandon Zagorski, Daryl Dooks, Dr. Don Mertens, and patients and staff at Toronto Rehab.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Toronto Rehabilitation Institute who receives funding under the Provincial Rehabilitation Research Program from the Ministry of Health and Long-Term Care in Ontario. SM receives funding from the Heart and Stroke Foundation Centre for Stroke Recovery. DB holds a Canada Research Chair.
