Abstract
Background. Brainstem regions with descending axons could influence recovery after spinal cord injury, thus presenting potential targets for treatment. Neurons in the hindbrain raphe magnus respond to sensory and chemical concomitants of trauma (eg, pain, circulating cytokines) and release trophic substances (serotonin, neuropeptides) in widespread spinal regions. Objectives. The authors tested in rats whether intermittent stimulation of the raphe magnus or its major midbrain input, the periaqueductal gray, influences recovery from incomplete thoracic (T8) injury. Methods. After a moderate weight-drop injury, an implanted wireless stimulator intermittently delivered cathodal pulses (8 Hz, 30 µA) through a microelectrode for 12 hours daily over multiple days. Stimulators in controls were inactive or not implanted. Results. Raphe magnus stimulation, started 1 to 2 hours after injury, enduringly improved open-field motor performance (measured weekly for 8 weeks) and footprint and gridwalk performance (measured in the 9th week). These improvements increased with days of stimulation (1-7). Myelinated axons in perilesional white matter and serotonin-containing terminals in gray matter, quantified 14 weeks postinjury, also increased. In separate parametric studies that examined open-field behavior following 14 days of stimulation, starting 2 days after injury was better than 7 days or 2 hours; starting at 8 weeks appeared ineffective. The periaqueductal gray, an established, safe stimulation target in man, similarly facilitated recovery of motor performance and myelination (but not serotonergic terminals) when stimulated for 4 to 7 days. Conclusions. Raphe magnus neurons mediate restorative feedback in acute spinal cord injury. Their interim activation, direct or indirect (via periaqueductal gray), offers a basis for possible treatments in patients.
Introduction
After spinal cord trauma caused by a blunt impact, disruption of signal transmission in the long axis is often incomplete, and function gradually recovers over the ensuing weeks. This recovery necessarily involves many interrelated adaptive molecular processes occurring in neurons and glia in and around the injury zone, as well as systemic processes such as immune responses and revascularization. Some processes in nervous tissue, for example, apoptosis or inflammation, function to dispose of injured or extraneous tissue and others to promote or guide neuronal regrowth and reconnection. 1 The disparate neural repair processes could be controlled, to some extent, by descending activity from certain brainstem regions. This possibility, if borne out, would have promising implications for treatment. One candidate region is the nucleus raphe magnus (NRM) of the medial medulla. The NRM has highly branched axons that terminate in all spinal laminae and segments.2,3 It is a major source of the spinal cord’s serotonin, 4 which is a critical molecule for both development and repair.5,6 Moreover, it co-releases various peptides, such as substance P, thyrotr pin-releasing hormone, and galanin, which also have trophic influences.7-9 We therefore examined how electrical stimulation of the NRM in adult rats affects behavioral and anatomical recovery following a moderate thoracic contusion injury. From a translational perspective, the relatively inaccessible location of the NRM in humans makes it a risky, impractical target for direct stimulation. However, the midbrain’s periaqueductal gray (PAG), a major input to the NRM, 10 has been safely stimulated with few adverse effects in many hundreds of patients for the treatment of drug-refractory pain. 11 Hence, a comparison study of PAG stimulation is also reported here.
Methods
Injury and Stimulator Implantation
Adult female rats (Sprague-Dawley strain, 250-275 g) were used in all experiments, under a protocol approved by the Institutional Animal Care and Use Committee of the University of Miami, Miller School of Medicine. To produce injuries, rats were maintained anesthetized with isoflurane (1.2% in oxygen) delivered through a face mask, following induction in a closed chamber. A laminectomy revealed spinal thoracic segment T8. A moderate, incomplete bilateral contusion was made by dropping a 10-g rod with a 2.5-mm diameter flat, circular tip a distance of 12.5 mm onto the midline, under the control of a MASCIS impactor. 12 Animals were excluded from the study if transducer readings showed compression times outside the range of 45 to 55 ms and depths from the spinal cord surface outside the range of 1.25 to 1.75 mm. For implantation of stimulators, rats under isoflurane anesthesia were placed in a stereotaxic holder in the flat-skull orientation. A monopolar tungsten microelectrode (approximately 500 kilohm impedance at 1 kHz; AM Systems Inc, Sequim, WA) was lowered to 1 of the 2 stereotaxic targets through a small aperture. The microelectrode and a bare stainless steel wire anode emerged directly from the epoxy encapsulation of a wireless, self-powered electrical stimulator. The stimulator, approximately 8-mm wide, 5-mm high, and 17-mm long, was fixed to the dorsal surface of the skull by 2 small screws and dental cement (Figure 1A and B). The anode was wrapped around one of the screws.
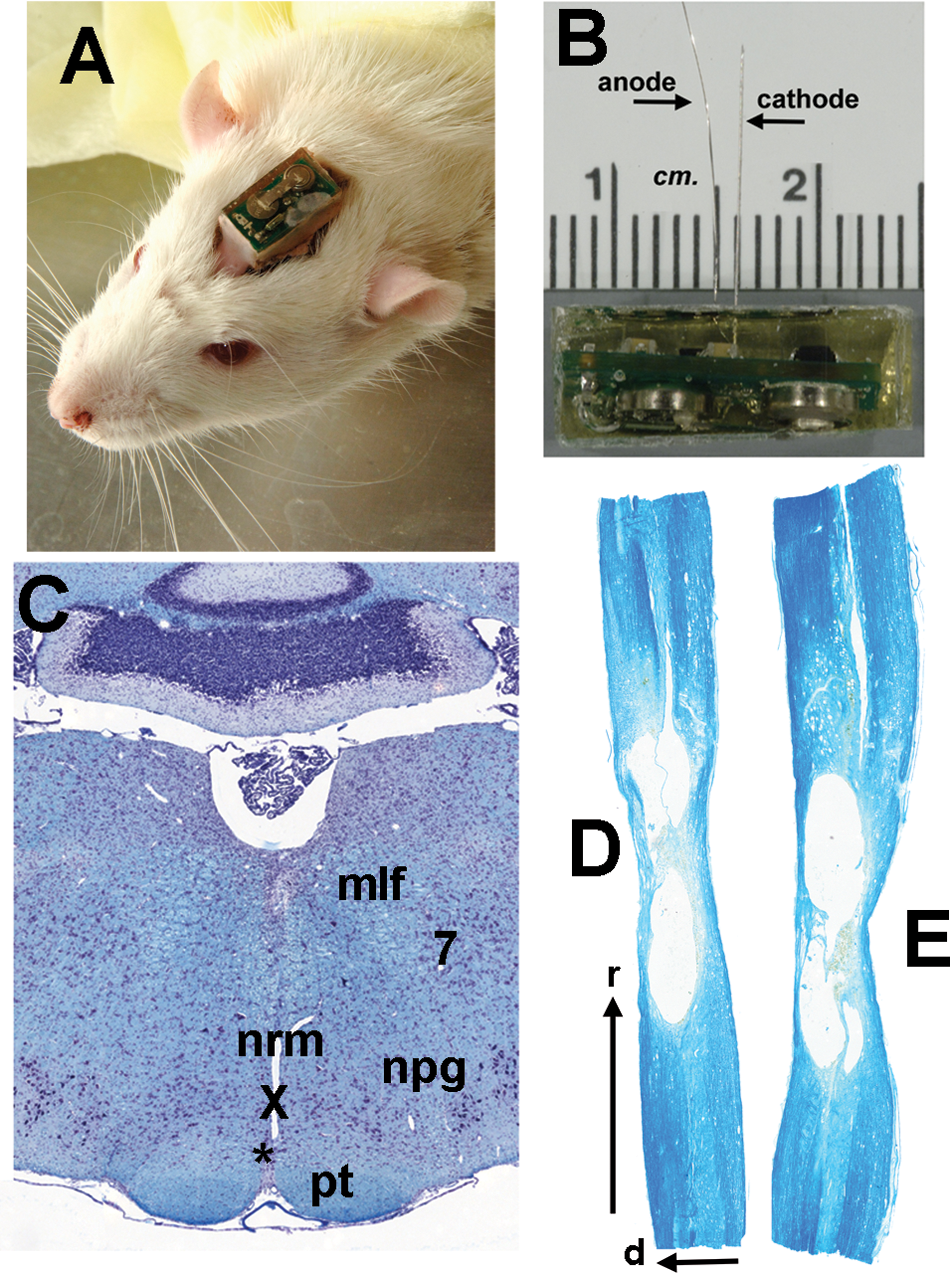
The stimulator, stimulation site, and injury site
Stimulation
Stimulation was given daily between 0600 and 1800 hours in 5-minute trains alternated with 5 minutes of rest. Trains consisted of 8 Hz, 1 ms, 30 µA cathodal constant-current pulses. The charge-density per pulse at the microelectrode surface was approximately 1.5 mC/cm2. The stimulator, via a miniature reed switch, could detect a pulsed magnetic field that encoded commands to start or halt the stimulation or to alter its timing parameters (pulse width, frequency, and clock time). The status of these parameters was signaled remotely by infrared pulses to a customized detector with an input wand held nearby.
Untoward aversive reactions, such as overt twitches or other motor responses, were never seen during stimulation in the NRM or PAG. Stimulation parameters were chosen to match the following aims and constraints: (a) to limit the spread of above-threshold current for cell body excitation to a radius of roughly 250 µm, as given by published threshold–distance relations, 13 pulse amplitude was fixed at −30 µA; (b) to match the relatively slow firing rate of NRM neurons, 14 a stimulation frequency of 8 Hz was used; (c) to allow recovery of serotonin release mechanisms, 15 5-minute pauses were alternated with 5-minute bouts of stimulation; (d) to provide a respite from possible effects of NRM or PAG stimulation on general arousal, 16 stimulation was turned off for 12 hours at night. Targeting of the NRM was assisted by symmetrical facial movements seen during temporarily applied stimulation above the target zone (between 3.2 ± 0.11 and 1.4 ± 0.17 mm above). If these movements were absent or asymmetrical, a fresh electrode track was made. In pilot animals, the microelectrodes were moved 1 mm ventral to the target, into the pyramids, which then evoked twitching movements, presumably due to excitation of the corticospinal tract. Absence of movements at the target in experimental animals, as well as the threshold–distance considerations mentioned above, suggested minimal activation of the corticospinal tract.
Experimental Sets and Treatment Groups
Three sets of experiments were performed. In set 1, the stimulation target was the midline NRM, 2.3 mm caudal and 0 mm ventral to the interaural line. Stimulators were either activated 2 hours after injury (n = 13) or left inactive as controls (n = 12). Some injured rats (n = 7) lacked stimulators to control for the physical implant in studies of open-field motor performance. Stimulation was stopped after variable numbers of days (range = 1-7 days, mean = 3.4). After 56 days, half of the inactive implant group had their stimulators turned on for 7 days, in order to examine influences on older injuries. In set 2, different starting times after injury for the NRM stimulation were compared: 2 hours (n = 6), 2 days (n = 7), or 7 days (n = 6); stimulation always lasted 14 days. In set 3, half of the rats received PAG stimulation (n = 10), with electrical parameters and injuries identical to those of the first 2 sets. The microelectrode was positioned unilaterally (right) in the ventrolateral PAG: 0.7 mm lateral to the midline, 1.2 mm rostral, and 4 mm above the interaural line. This site within the PAG was chosen for its ability to evoke acute analgesia without aversive reactions. 17 A concurrent control group (n = 9) received the same injury but were not given inactive stimulators, since long-term effects of implants had been already excluded in the NRM study. Stimulation was applied for 4 to 7 days (mean = 5.6 days, SEM ±0.60); this range was narrower than used in set 1, to improve statistical power.
Behavioral Measurements
Open-field motor performance was evaluated by a standard 0 to 21 point score, the BBB score, 18 with respect to various postural and locomotor parameters. Two trained observers, blinded to the different treatments, obtained the BBB scores of rats placed in a 5-ft-diameter arena. Scores were taken 2 days prior to injury, 1 day after injury, and then weekly. Several parameters making up the BBB score have separate left and right limb components, which were averaged (with one exception, noted in Results). On 2 days in the 9th week after injury, footprint and gridwalk tests were performed. Bilateral footprints made by inked paws on paper were recorded for 5 consecutive step cycles. Four parameters were measured: hind paw toe spread, hind limb stride length, base of support, and hind foot rotation.19,20 In the gridwalk test, rats made 2 traverses, 1 in each direction, on a 3-ft horizontal ladder with irregularly spaced rungs.12,21 The number of footfall errors and the traverse time were determined by blinded observers viewing videotaped recordings.
Anatomical Quantification
Euthanasia was carried out 98 days (14 weeks) after injury with an overdose of a mixture of ketamine (120 mg/kg) and xylazine (20 mg/kg), followed immediately by intracardiac perfusion with 4°C phosphate-buffered saline and then 4% paraformaldehyde solution. The brain was dissected carefully away from the stimulating microelectrode and its skull attachment to minimize damage to the stimulation site. A cavity was left in the tissue where the body of the electrode had been (see Figure 1C). All tissue was embedded in paraffin and sectioned at 12 µm thickness.
The ventral medial medulla was visualized in coronal sections stained with hematoxylin–eosin and Luxol fast blue. Electrode tips were always found to be located inside the target region within 0.3 mm of the target (see Figure 1C). Signs of gliosis or trauma near the electrode tip were minimal compared with the estimated radial spread of effective stimulus current (0.25 mm). 13
The spinal cord was analyzed within ±1 cm of the injury center. Equivalent segments of non-injured adult female Sprague-Dawley rats were studied for comparison (n = 4). Sagittal serial sections were made from the inner 1-cm length of tissue centered on the injury. Sections at 60-µm intervals, stained with hematoxylin–eosin and Luxol fast blue, were used to estimate the volumes of the injury cavity, the penumbra of damaged tissue and the total tissue. Tissue 0.5 to 1 cm rostral or caudal to the injury was sectioned coronally. These portions contained T11-T12 and T5-T4 segments. Their quantification was centered on locations 7.5 mm (±0.5 mm) rostral and caudal to the injury center, just beyond the maximum longitudinal extent of the injury cavity observed in the sagittal sections (Figure 1D). Sections at 120-µm intervals were stained with rabbit anti-serotonin antibody 1:1000 (S5545, Sigma-Aldrich, St Louis, MO) and with goat anti-NeuN 1:250 (MAB377, Chemicon, Billerica, MA). The secondary antibodies used were donkey AlexaFluor 488 anti-rabbit and 594 anti-mouse (Molecular Probes, Carlsbad, CA). A second series of sections, also at 120-µm intervals, was stained with a modified Bielschowsky’s silver-impregnation method.
All quantitative analysis employed commercial software for microscope-based stereology (Stereo Investigator, MBF Bioscience, Williston, VT) and was performed by users blinded to experimental treatments. Volumes in sagittal sections were estimated by 3-dimensional reconstruction of contours. The total length of fluorescent serotonin-containing fibers in gray matter was estimated by an “isotropic virtual planes” probe (area and height sampling fractions, respectively, 0.25 and 0.8) under a Zeiss Axiovert microscope, with magnification set at 63× and numerical aperture at 1.4. The coefficient of error for the sampling was held to <0.1. Myelinated fibers traversing the plane of section in white matter were sampled by planimetry with a physical fractionator at 63× magnification. They were identified in silver-stained sections by their brown cores surrounded by unstained regions enclosed in turn by stained outer rings.
Statistical Analysis
Statistical testing on behavioral or anatomical variables employed SPSS version 17.0 (SPSS Inc, Chicago, IL). Repeated-measures analysis of variance (ANOVA) was used on time courses, and individual weeks were separately tested by 1-way ANOVA, using Bonferroni post hoc comparison. Other tests included multivariate ANOVA (Wilks’ Λ), and rank order testing (Kendall’s τ), as specified in the Results and figure legends. A 2-sided level of P < .05 was accepted as significant.
Results
Effect of Early NRM Stimulation on Motor Performance After Spinal Cord Injury (SCI)
When NRM stimulation was initiated 1 to 2 hours after injury and continued for up to 7 days (set 1), open-field motor performance underwent significant long-term improvement, as shown by higher bilateral BBB scores 18 compared with implanted or non-implanted controls (repeated-measures ANOVA, post-hoc Bonferroni P < .001). Before injury, all rats scored the maximum of 21. Definite improvements existed by the 14th day after the injury and gradually improved over the remaining 6 weeks of BBB testing (Figure 2A). Most weeks also showed significant differences between groups in 1-way ANOVA (Bonferroni comparison, P < .05). At the 7-day point, but not later, the implant itself may have hindered performance, since only the non-implanted group differed significantly from the stimulated group. The BBB scores at 6 to 8 weeks in the stimulated rats reflect consistent weight-supported stepping and good coordination between forelimbs and hindlimbs during locomotion, characteristics that were weak or lacking in injured, non-stimulated controls. The BBB subscore, 22 which represents a subset of postural and locomotor variables (foot rotation, toe clearance, trunk control, and tail position), also showed a strong divergence between stimulated rats and their control groups (Figure 2B; repeated-measures ANOVA, Bonferroni comparisons P < .001). After the first week, both the BBB score and the subscore were correlated positively with the number of days of NRM stimulation (Figure 2C and D, Kendall’s τ P < .05).
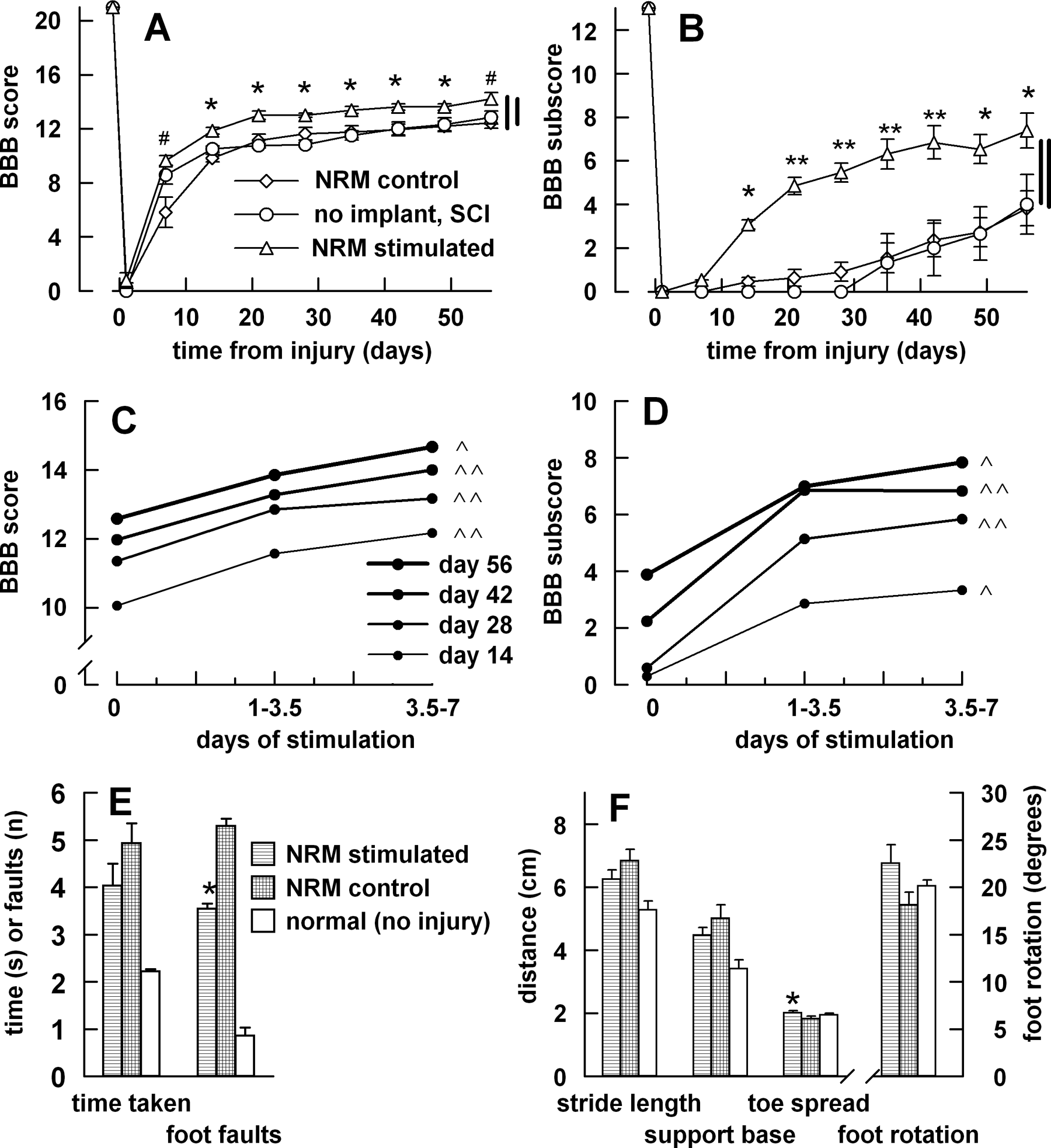
Effects of nucleus raphe magnus (NRM) stimulation on motor performance
Gridwalk testing on the 9th week after injury showed foot fall errors to be significantly lower (t test, P < .05) in the NRM-stimulated group (Figure 2E) than in non-stimulated, implanted, injured controls. The mean time taken to cross the ladder was unchanged by stimulation. For comparison, non-injured controls (n = 5) also underwent the gridwalk test; their scores were considerably lower than those of either injured group (Figure 2E). Rats that received NRM stimulation showed significant improvement in the overall characteristics of the bilateral footprint; thus, multivariate ANOVA on the combined 4 variables of toe spread, stride length, base of support, and foot rotation was significant in a comparison of stimulated rats with non-stimulated, implanted, injured controls (Wilks’ Λ, P < .05). All individual parameters were shifted by the stimulation toward normal values, more so in the case of toe spread and foot rotation than stride length or base of support (Figure 2F). However, in univariate analysis, only the toe spread showed statistically significant improvement (t test, P < .05), indicating improved dynamic stability. The individual gridwalk and footprint variables that improved statistically against controls all displayed a positive correlation with days of NRM stimulation (Kendall’s τ P < .05).
Effect of Different Treatment Start Times on Open-Field Motor Behavior (Sets 1 and 2)
Randomly selected rats (n = 6) from set 1 with initially inactive stimulators were used to assess the effect of NRM stimulation in chronic SCI. Stimulation was started 56 days after injury and lasted for 7 days. No subsequent difference was seen in the weekly BBB score or subscore between this later-stimulated group and the concurrent non-stimulated group; scores remained stable over the 4 weeks inspected (Figure 3A and B). In rats of set 2, NRM stimulation was started at 3 different times and lasted 14 days. Starting 2 days (n = 7) after injury, compared with starting at 2 hours (n = 6) or 7 days (n = 6), gave BBB scores (Figure 3C; repeated-measures ANOVA, Bonferroni comparison P < .01). Due to a lack of coordination between fore limbs and hind limbs, sufficient BBB subscores measured from set 2 gave insufficient numbers for analysis.
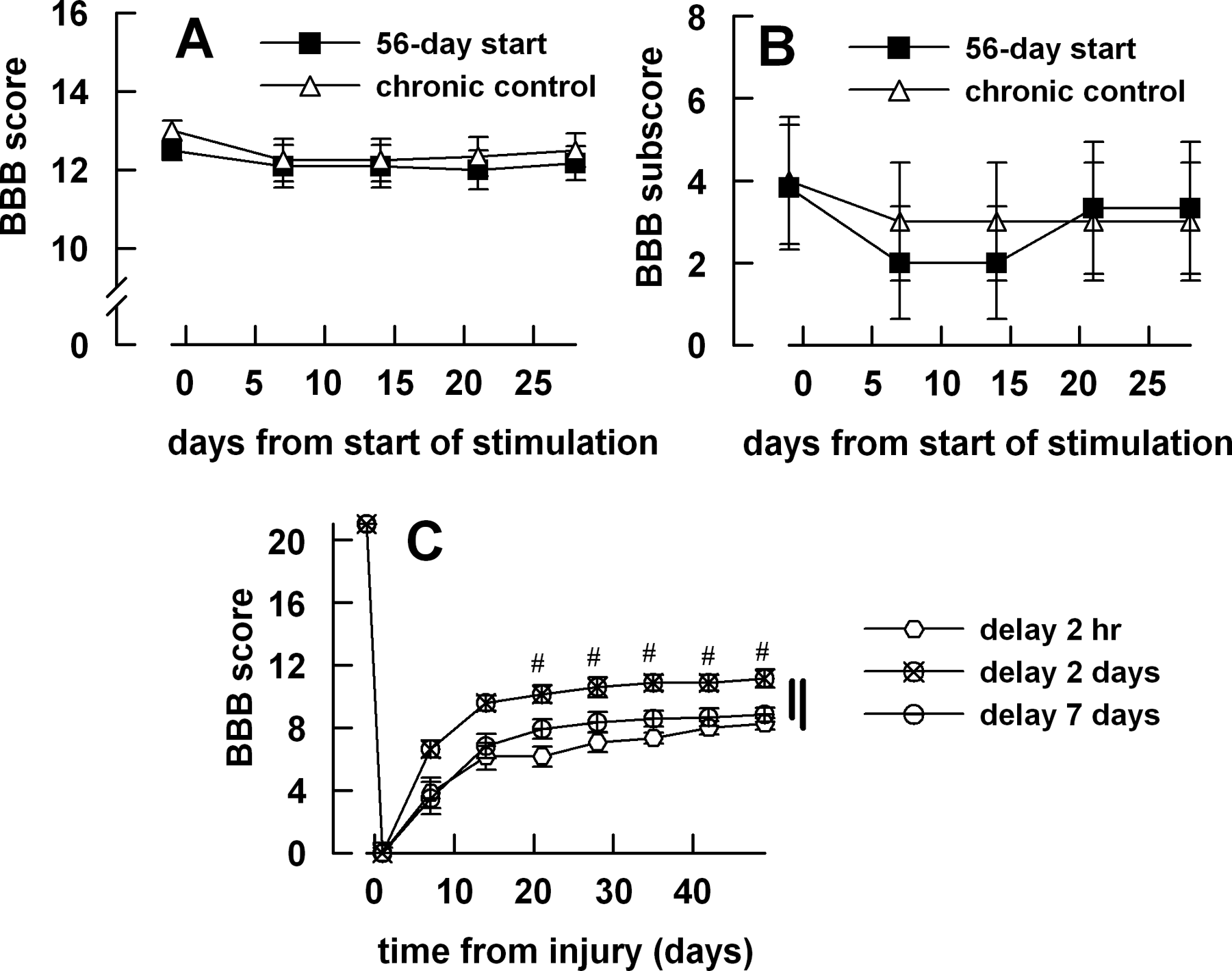
Parametric studies of start time for nucleus raphe magnus stimulation
Motor Performance After PAG Stimulation (Set 3)
PAG stimulation was applied for 4 to 7 days (mean = 5.6 days, SEM ±0.60), starting 2 hours after injury. The PAG-stimulated group (n = 10) exhibited improved bilateral BBB scores and BBB subscores compared with the injured control group (n = 9) that lacked implants (Figure 4A and B; repeated-measures ANOVA P < .05 for both BBB score and subscore). Certain individual weeks also showed significant differences in the BBB score and subscore (t test, P < .05). The off-midline stimulation target suggested the possibility of lateral asymmetry in the BBB score of the PAG-stimulated rats, but they proved to have identical left and right scores.
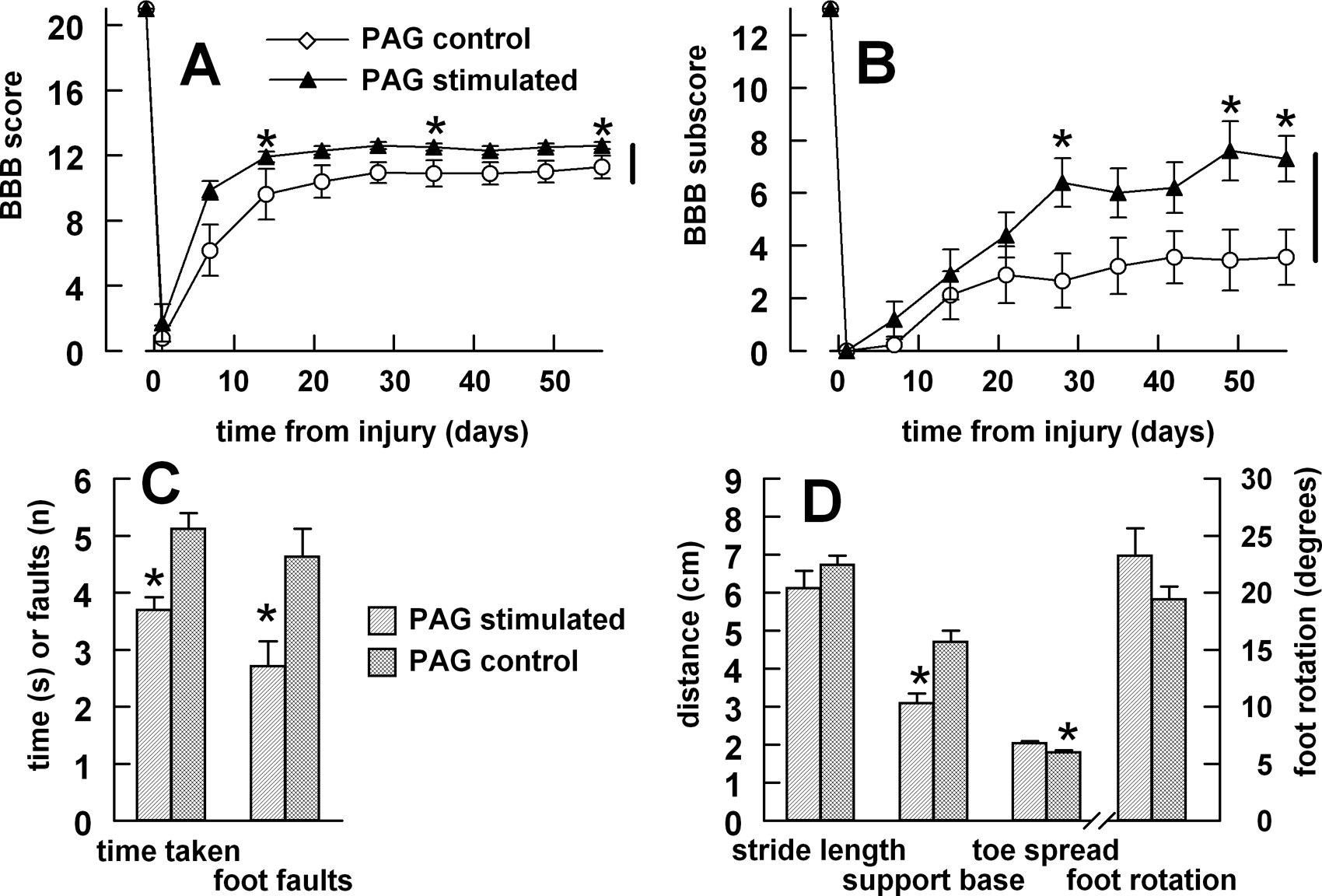
Effects of periaqueductal gray (PAG) stimulation on motor performance
In the gridwalk tests, both the number of foot faults and the traverse time were significantly reduced in the PAG-stimulated group (Figure 4C). Footprints showed significant improvement in overall characteristics (multivariate ANOVA on the 4 variables; Wilks’ Λ, P < .05), with all individual parameters shifted in the direction of normal values. Univariate ANOVA showed significant improvement in both the base of support and the toe spread (Figure 4D).
Anatomical Analysis (Sets 1 and 3)
Myelinated fibers in the spinal cord’s white matter (dorsal, lateral, and ventral columns) were counted approximately 7.5 mm caudal and rostral to the center of the injury. Myelinated fibers were significantly denser caudally but not rostrally in NRM-stimulated rats of set 1 (Figure 5A and B) compared with implanted injured controls with inactive stimulators (t test, P < .05). No significant effect of stimulation duration emerged in this analysis, in contrast to its effects on serotonergic terminals (see below). After PAG stimulation, statistically significant differences from the concurrent control group were found in both the rostral and the caudal directions from the injury (t test, P < .05). Both PAG and NRM stimulation restored the average level of myelination from the untreated values about half way toward the values of normal rats (n = 4), both rostrally and caudally. In volume analyses, neither the cavity nor the penumbra showed a significant difference in mean between NRM-stimulated rats and their implanted inactive-stimulator controls. The average total volume of the 1-cm lengths of tissue was significantly larger for the PAG treatment (156% of control, t test P < .05) but not significantly different for the NRM treatment (121%); this difference most probably arose from the greater myelination.
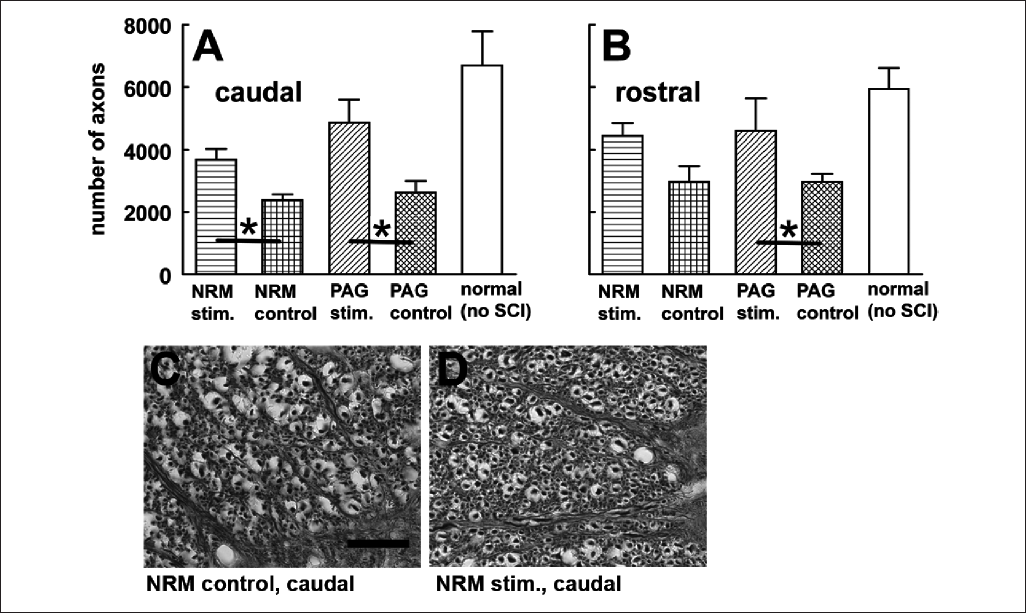
Recovery of myelinated fibers after nucleus raphe magnus (NRM) or periaqueductal gray (PAG) stimulation
Axons immunostained for serotonin were sampled in the gray matter. The total length of immunostained fibers in a fixed length (960 µm) of spinal cord, centered at the same segmental levels as the analysis of myelinated fibers, was also estimated. The NRM-stimulated group was divided into short and long periods of stimulation: 1 to 3.5 days (n = 6), 3.5 to 7 days (n = 7). Both caudally and rostrally, compared with injured controls with inactive implants, the total length of serotonergic terminals in the sampled region was significantly elevated in the rats that had been stimulated for the longer period (Bonferroni post hoc tests, P < .05) but not for the shorter period (Figure 6A and B). Values for normal rats (n = 4), not graphed in Figure 6, were 2.8 times greater than for NRM controls caudally and 5.7 times rostrally. Neither rostral nor caudal locations showed significant differences in serotonin-containing terminals after PAG stimulation.
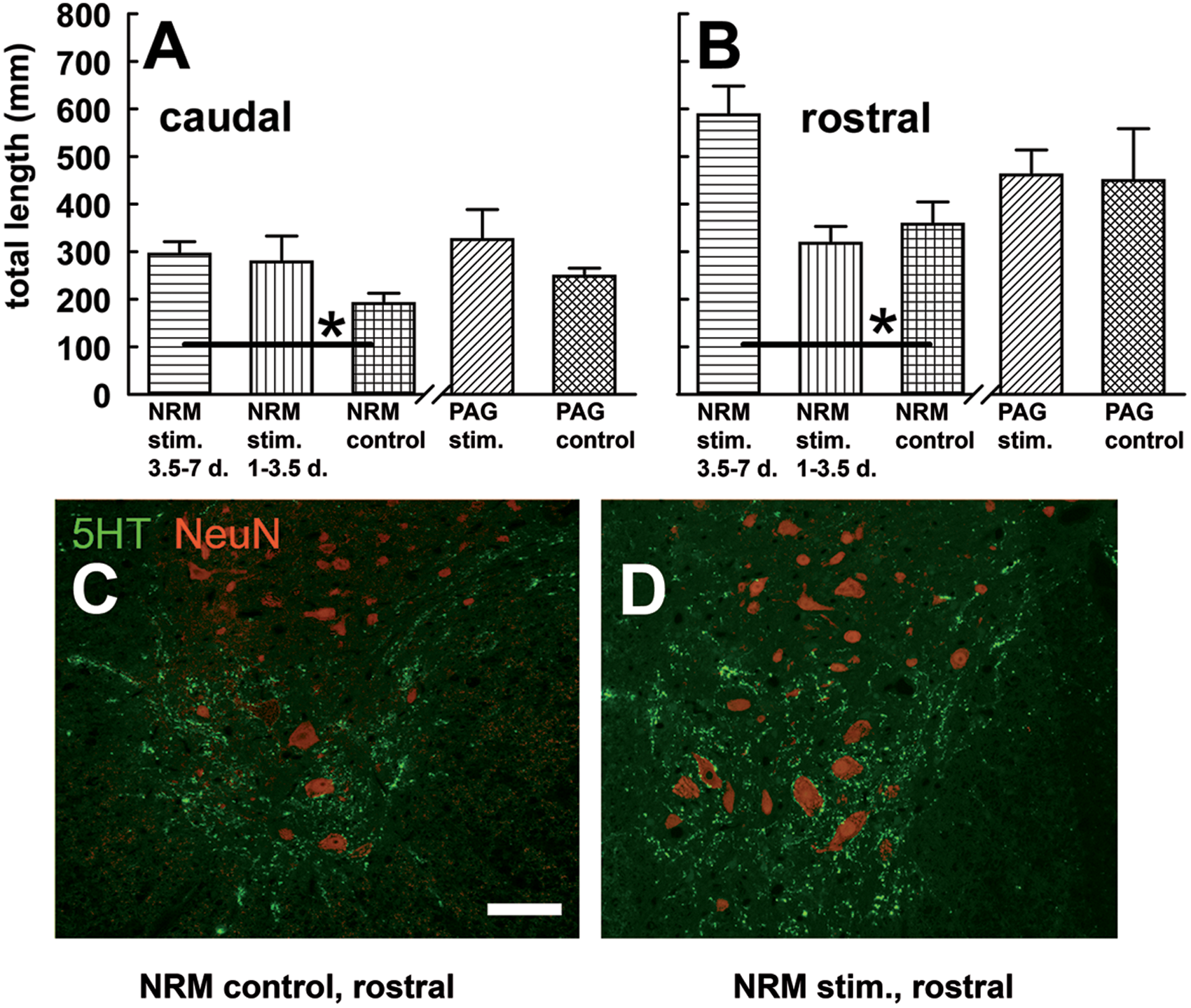
Recovery of serotonin-containing terminals in gray matter after NRM stimulation
Discussion
Overview
The results show that general motor and anatomical recovery from an acute, incomplete thoracic SCI can be lastingly enhanced by several days of NRM or PAG stimulation. Behavioral improvements were observed up to 9 weeks after injury, and anatomical improvements were measured 14 weeks after injury. NRM stimulation also normalizes pain sensation (reversal of forepaw allodynia), as described for the same SCI model in an earlier report. 23
Anatomic and Timing Parameters
The PAG lacks a strong spinal cord projection. Hence, its restorative effects are highly likely to have been relayed by the NRM, as previously shown for its antinociceptive effects. 24 Overlap in the restorative effects of NRM and PAG stimulation was imperfect; each site was superior in some respect. This may have arisen from the lower numbers used in the PAG study (set 3), which left its statistical power closer to threshold. Alternatively, firing in NRM neurons evoked indirectly by the PAG is probably unsynchronized, which could lower the peak spinal release of serotonin. The outcomes with PAG or NRM stimulation are unlikely to represent the maximum attainable benefits, because many stimulation parameters (current amplitude, pulse width and shape, frequency, rest pattern) were not optimized. Also, if bilateral (instead of unilateral) PAG stimulation had been used it might have enhanced the restorative effect. Both NRM and PAG stimulation probably interact with the dynamic cellular and molecular processes that occur in the first hours, days, and weeks after SCI,25,26 as is suggested by the variation caused by different total treatment durations and start times of NRM stimulation. Much more research will be needed to define the nature of this interaction. Part of the effect of NRM stimulation may be to prevent continuing secondary damage. However, its greater efficacy when started at 2 days, after considerable secondary damage has already taken place, 26 compared with starting at 2 hours, supports the idea that restoration of cellular damage is a major, possibly predominant effect.
The NRM as a Repair Center
It is a common, useful practice to assign specialized functions (sensory, motor, motivational, etc) to brain regions, with-out implying exclusion of other functions. Given current knowledge, including the present results, it may be useful to regard the NRM as a repair-control center. Neurons in such centers are likely to have the following features: (a) good resistance to injury, (b) good axonal sprouting or regrowth, (c) responsiveness to a wide range of sensory and systemic inputs relevant to the initiation and continuation of repair, (d) highly divergent axons that cover many possible sites of damage, and (e) axons that release substances with neurotrophic or neuroprotective influences. Descending serotonergic fibers show superior survival and strong sprouting after damage.27-30 NRM neurons respond distinctively to various concomitants of neurotrauma, such as noxious cutaneous stimuli, 14 molecules produced early after neurotrauma (eg, cytokines, eicosanoids, and purines),31-33 and baroreceptor input that can signal hypotension caused by injury. 34 They furthermore release neurotrophic molecules, as in the Introduction. An interesting contrast can be made with the corticospinal tract, whose terminations are less divergent and maintain a highly topographic mapping. After partial corticospinal lesions at the medullary level, stimulation of the remaining, intact corticospinal axons causes increased sprouting into denervated spinal areas.35,36 This stimulation also increases the proliferation and differentiation of oligodendrocyte precursor cells, which explains how these newly sprouted branches become myelinated. 37 Perhaps unlike in NRM neurons, these compensatory growth responses are apparently restricted to recently active corticospinal axons and may play a part in normal functional plasticity. Further research is needed to resolve these questions.
A single mechanism is unlikely to account for the restorative effect of the NRM. Indeed, the complexity and variety of neurotrauma suggests that a repair-control center must influence many processes to be useful. One category of mechanism is the sprouting of axons or unmasking of synapses due to electrical activity in directly activated fibers, 38 as observed in corticospinal axons.35,36 Increases in serotonin-stained axons distal to the injury, seen following the more prolonged NRM stimulation, may reflect such an activity-dependent effect. This greater regrowth could have the positive secondary effect of accentuating other restorative effects of the raphe–spinal pathway. Another category resides in the behavioral effects of acute NRM stimulation. Descending activity from the NRM can acutely block pain24,39 and facilitate motor rhythms. 40 Rehabilitation is more successful in patients with recent SCI if their movements are free of pain. 41 A third category, probably the largest, includes trophic or neuroprotective effects of substances released by raphe–spinal terminals on adjacent neurons or glia, most notably serotonin. A key intracellular signaling molecule in this regard is cyclic adenosine monophosphate (cAMP), which is elevated when serotonin binds to 5-HT7 receptors. Several positive downstream effects are produced by cAMP following neurotrauma, such as augmented expression of trophic genes and a damping of the inhibitory effects of myelin-derived proteins on axonal extension and remyelination. 42 We recently found that NRM stimulation restores intracellular cAMP levels in the injured spinal cord, unless blocked by the 5-HT7 inhibitor pimozide (I. D. Hentall and M. M. Carballosa Gonzalez, unpublished data, 2011). Moreover, serotonin receptors (5-HT7 and 5-HT2) are present on astrocytes as well as on presynaptic fibers and postsynaptic dendrites in the spinal cord,43,44 which may reduce the activation of astrocytes, resulting in improvements in myelination 45 .
Clinical Implications
Electrical deep brain stimulation (DBS) is at present used mostly to obtain immediate symptomatic improvement in patients with chronic extrapyramidal motor disorders such as Parkinson’s disease. 46 For functional restoration after neurotrauma, the advantages typically cited for DBS still apply, including instantly adjustable dosage and restricted side effects. An added advantage specific to restorative DBS is that permanent cranial implantation is probably unnecessary. The PAG is well established as a site for DBS, especially with regard to safety. 11 Thus, its interim stimulation for SCI appears to offer a large translational advantage in the intermediate term over proposed interventions with cell transplants, manipulation or addition of genes, and administration of growth-promoting substances, many of which are of uncertain safety. Interim DBS, moreover, is well suited for combination with these other types of therapy, as they later become approved for clinical use.
Footnotes
Acknowledgements
We gratefully acknowledge the technical help and advice of Mr Scott S. Burns, Mr Paolo Diaz, Dr Beata Frydel, Dr Aldric Hama, Mr Clayton Jackson, Ms Denise Koivisto, Dr Alex Marcillo, Ms Ileana Oropesa, Dr Maria Luis Rodriguez, Ms Monica Stagg, and Mr Alberto Vitores. We are also grateful to Drs Coleen Atkins, Damien Pearse, and Jacqueline Sagen for comments on a draft version.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Craig H. Neilsen Foundation and the State of Florida.
