Abstract
Neuroprostheses (NPs) are electronic devices that stimulate nerves to improve bodily function after damage to the central nervous system. They generally comprise hermetically sealed electrical stimulators with leads terminating in epimysial or nerve cuff electrodes.1,2 Command signals, and in some cases energy, are transmitted at radiofrequencies from an external control unit to the implanted stimulator.
Over the past 45 years, there has been an enormous proliferation of NPs to ameliorate disorders as diverse as pain, deafness, paralysis, epilepsy, Parkinson disease, tremor, spasticity, depression, and bladder dysfunction. 3 We have developed a new type of externally controlled NP, the Stimulus Router System (SRS). 4 The only component implanted in the body is a passive lead (Figure 1A), one end of which, the “pick-up” terminal, is implanted under the skin, and the other end, the ”delivery” terminal, is tunneled to a target nerve (Figure 1B). A surface electrode is placed over the implanted pick-up terminal, and a second electrode is placed nearby. Current pulses are now passed through the skin between the 2 surface electrodes. Some of the current is “captured” by the implanted pick-up terminal and delivered via the lead to the nerve. The basic properties of the SRS were explored in 2 series of animal experiments.4,5 Results from the studies showed that the SRS is reliable as a long-term NP and can selectively activate deep-lying nerves in a graded manner over the full physiological range. The first human proof of principle of SRS was shown during an acute human intraoperative procedure, 6 where functional muscle contraction was elicited through the SRS at surface current amplitudes below sensory perceptual threshold. In a separate pilot human study that evaluated the use of the SRS system for chronic pain treatment, 7 10 percutaneous SRS leads were implanted temporarily (5 days) in 8 patients with carpal tunnel syndrome and chronic pain. We describe a case report of the first permanent human implant of an SRS for restoration of hand function.
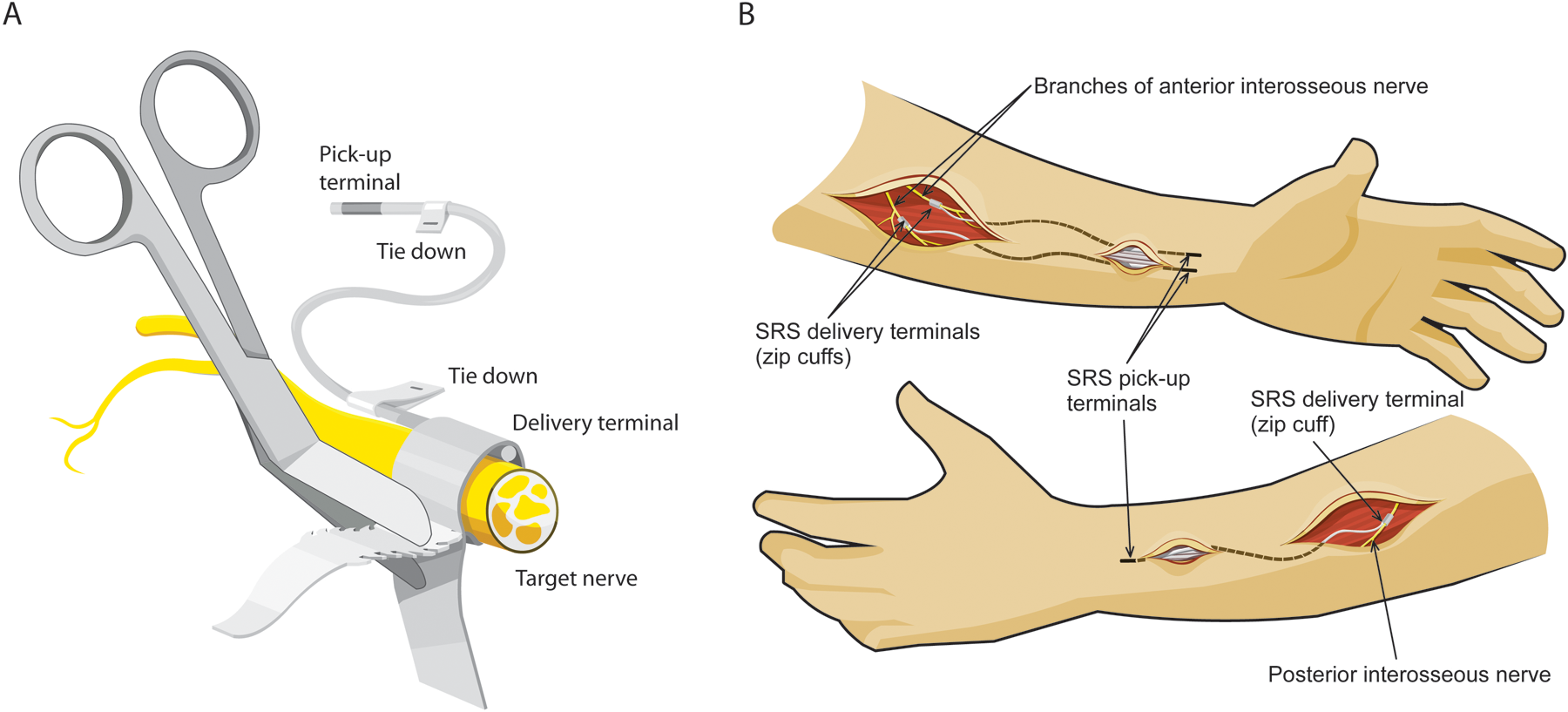
(A) Each Stimulus Router System (SRS) lead consisted of multistranded platinum–iridium wire loosely coiled inside 1.2-mm diameter silicone tubing. At both ends wire protruding from the tube was tightly wound back onto the surface of the tube to form an electrically conductive terminal. The pick-up terminal was 15-mm long, corresponding to a surface area of 0.9 cm2. The delivery terminal was 5-mm long, corresponding to a surface area of 0.3 cm2. A silicone strip with a structure similar to a tie-wrap was attached with silicone glue to the delivery terminal to form a nerve cuff. Once the cuff was secured on the target nerve, the excess strips of silicone were trimmed. Tie-downs attached to the leads at each end were sutured to the surrounding connective tissues to act as anchors. (B) The delivery terminals of 2 leads were implanted on branches of the anterior interosseous nerve innervating flexor digitorum profundus (FDP) and flexor pollicis longus (FPL) through skin incisions each about 3 cm long. The delivery terminal of a third lead was implanted on the branch of the posterior interosseous nerve innervating extensor digitorum communis (EDC) and abductor pollicis longus (APL). The pick-up ends of the leads were tunneled under the skin from the incisions and anchored subcutaneously about 3 to 4 cm proximal to the posterior and anterior wrist creases. Abbreviations: SRS, Stimulus Router System; FDP, flexor digitorum profundus; FPL, flexor pollicis longus; EDC, extensor digitorum communis; APL, abductor pollicis longus.
Methods
Participant
The study was performed with approval from the Human Ethics Committee of the University of Alberta and the informed consent of the participant. Inclusion criteria included the following: complete or incomplete cervical spinal cord injury (SCI) with tetraplegia for more than 1 year, loss of voluntary grip and release, functional electrical stimulation (FES) elicits enough hand opening and grip to lift a 4-cm-diameter ball, neurologically and medically stable, able to tolerate FES, and sufficient movement of shoulder and elbow.
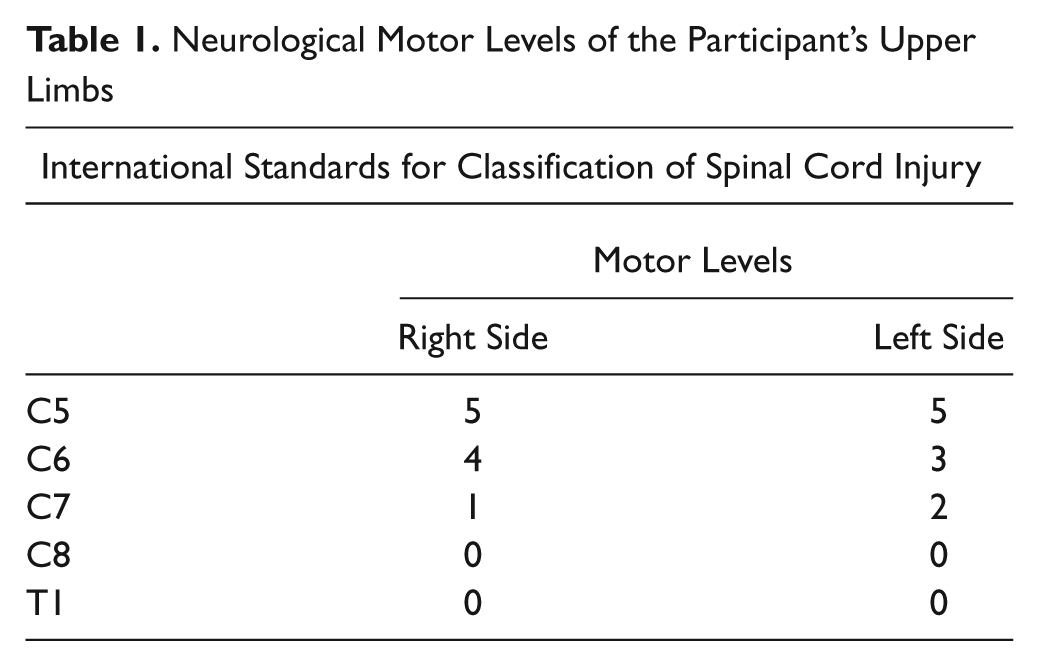
The participant was a 52-year-old man who suffered a C6/7 SCI in 1998 due to a sports injury. He had voluntary control of shoulder, elbow, and wrist and a weak tenodesis grip. His neurological motor levels are shown in Table 1. Two-point discrimination was measured at the pulp of the thumb. His pinprick and light touch sensations were normal except for diminished touch at C8 level on the right hand side.
Neurological Motor Levels of the Participant’s Upper Limbs
Surgery
The participant chose to have leads implanted in his right, less functional arm. Just prior to surgery, motor and sensory thresholds were determined by applying trains of current pulses between moistened pad electrodes 5 cm in diameter, placed at the optimal stimulation points of the long finger flexors and extensors and a similar sized reference electrode located on the posterior aspect of the forearm 15 cm proximal to the wrist crease. Grip force with and without stimulation was measured with a hand dynamometer (Jamar; Sammons Preston, Nottinghamshire, UK).
Surgery was performed in 2008 under general anesthesia without muscle relaxation. Incisions were made on the anterior and posterior aspects of the forearm (Figure 1B). Three SRS leads (Synapse Biomedical, Cleveland, Ohio) were implanted, 2 on branches of the anterior interosseous nerve innervating flexor digitorum profundus (FDP) and flexor pollicis longus (FPL) and 1 on a branch of the posterior interosseus nerve innervating extensor digitorum communis (EDC) and abductor pollicis longus (APL). The nerve branches were identified using a bipolar hook electrode connected to a nerve stimulator. In each case, about 2 cm of the nerve was dissected free of connective tissues and a custom silicone nerve cuff 8 containing the delivery terminal of an SRS lead was attached to the nerve by inserting the tongue of the cuff into a slit (Figure 1A) and pulling on the tongue until the cuff had an internal diameter approximately 30% greater than that of the nerve. To verify the placement, a pulse train was applied between the pick-up terminal and a 3.8 × 7.6–cm hydrogel reference electrode attached to the arm proximal to the elbow prior to surgery. The tongue portion of the cuff was trimmed. Wing-like tie-downs were sutured to subcutaneous fascia about 4 cm distal to the cuffs (Figure 1A). The pick-up ends of the leads were then tunneled under the skin to sites proximal to the wrist on the anterior and posterior aspects of the forearm.
After the surgical incisions had been sutured shut, 3.8 × 3.8–cm hydrogel electrodes were placed on the skin, one over the FPL and FPB pick-up terminals and another over the EDC/APL pick-up terminal. Stimulation was applied in turn between these electrodes and the reference electrode to elicit grasp and hand opening. The surgery took 2 hours, and the participant returned home the next day. He wore a splint on his operated forearm and hand for 10 days.
Study Design
Six weeks after the operation, the participant was provided with a wireless earpiece and wristlet stimulator (Figure 2A). The stimulator generated trains of pulses that were delivered through 4 wettable pad electrodes within the wristlet. Three of these were located over the 3 pick-up terminals. The fourth, the reference electrode, was centered 12 cm proximal to the wrist crease on the posterior aspect of the forearm. Positioning of the electrodes within the wristlet was achieved as follows. First, the reference electrode was held in position on the bare forearm with an elastic strap. Next, pulse trains were delivered through the skin in the regions of the pick-up terminals with a 2.5-cm pad electrode to identify optimal stimulation points. Snap connectors with Velcro backing were now positioned at corresponding positions inside the wristlet, so that when the pad electrodes were attached to the snaps and the wristlet was donned, the electrodes pressed against their respective optimal stimulation points. The EDC/APL and reference electrodes were 5 cm in diameter and those over the 2 flexor pick-up terminals (FDP and FPL) were each 2.5 cm in diameter. The stimulator generated pulses of maximally 10 mA and 100 V.
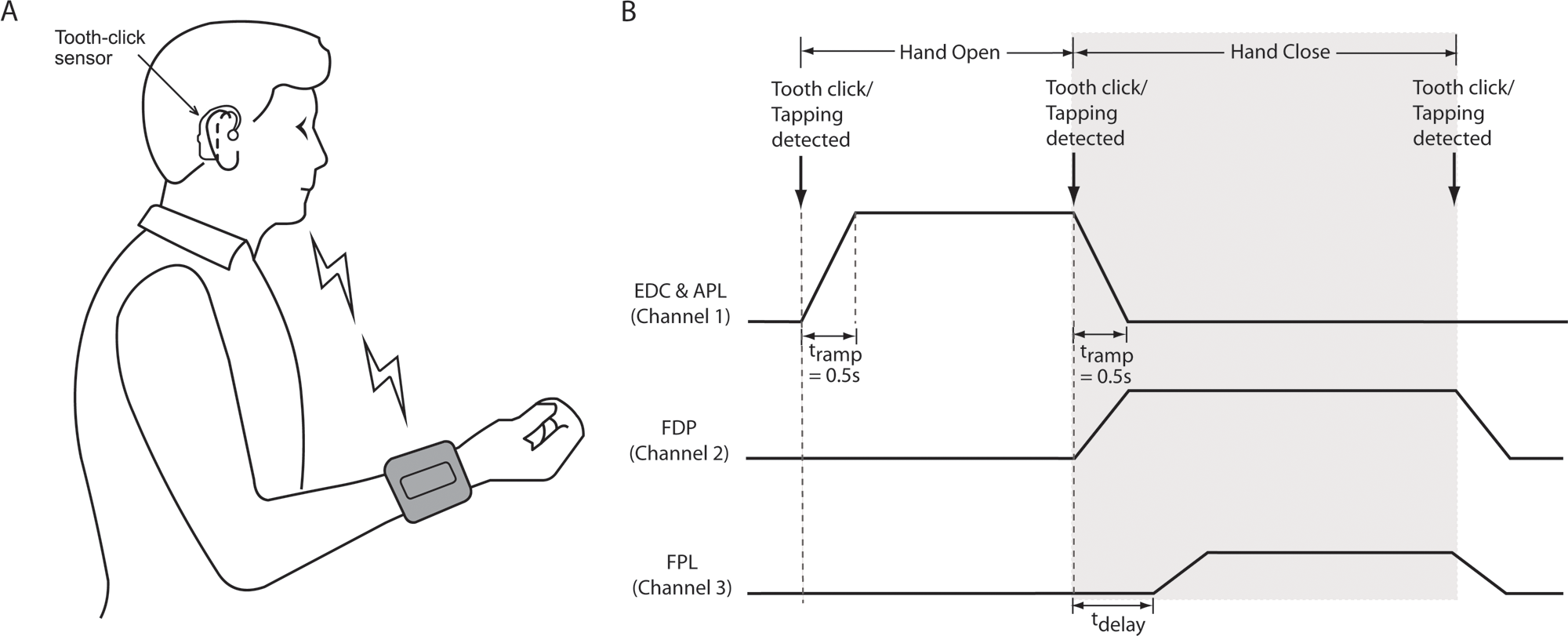
(A) The subject controlled hand opening and grasp with a wireless earpiece and a wristlet device. The wristlet held a detachable, rechargeable stimulator. Insulated wires led from the stimulator to Velcro-backed snaps that were positioned within the wristlet so as to overlie the pick-up terminals of the implanted leads when the wristlet was donned. Prior to use, pad electrodes were moistened and attached to the snaps. The wristlet had straps, buckles, and loops that allowed the participant to don and doff it unaided within 2 minutes. The earpiece contained an accelerometer that detected tooth-clicks. The stimulator also contained an accelerometer that detected taps applied to the forearm. (B) Radiofrequency trigger signals transmitted from the earpiece to the stimulator advanced it through a cycle of hand opening, closing, and relaxation. On detection of the first command (tooth-click or tap applied to the stimulator), stimulus pulses were delivered through the EDC/APL lead, causing hand opening. The pulse amplitude ramped up in 0.5 seconds (
The participant triggered stimulation with small tooth-clicks that were detected by the wireless earpiece, which contained a 3-axis accelerometer (Figure 2A). A microprocessor in the earpiece ran a pattern recognition algorithm that differentiated tooth-clicks from other vibrations such as those associated with speech.9,10 On detecting a tooth-click, the earpiece transmitted a radiofrequency signal to a receiver/stimulator located in the wristlet. This allowed the participant to advance the stimulator through the sequence hand-open, grasp, relax, with successive tooth-clicks, as shown schematically in Figure 2B. A 3-axis accelerometer in the stimulator provided an alternative means of advancing through the sequence by tapping or bumping the forearm. A ReJoyce workstation was installed in the participant’s home, enabling tele-supervised exercise sessions 5 d/wk for 6 weeks. 11 The sessions initially lasted 15 min/d, gradually increasing to 1 h/d after 3 weeks. Tele-supervised exercise sessions were then conducted at monthly intervals for 6 months.
Outcome Evaluation
Starting from day 10 postoperatively, the following parameters of the SRS implants were measured every week for 2 months, then monthly for 10 months. (
Results
Preoperative Measurements
Without FES, the participant was unable to generate a measurable grip force on the dynamometer. With voluntary flexion at the wrist, he achieved an aperture of hand opening of 4 cm between the tips of the forefinger and thumb.
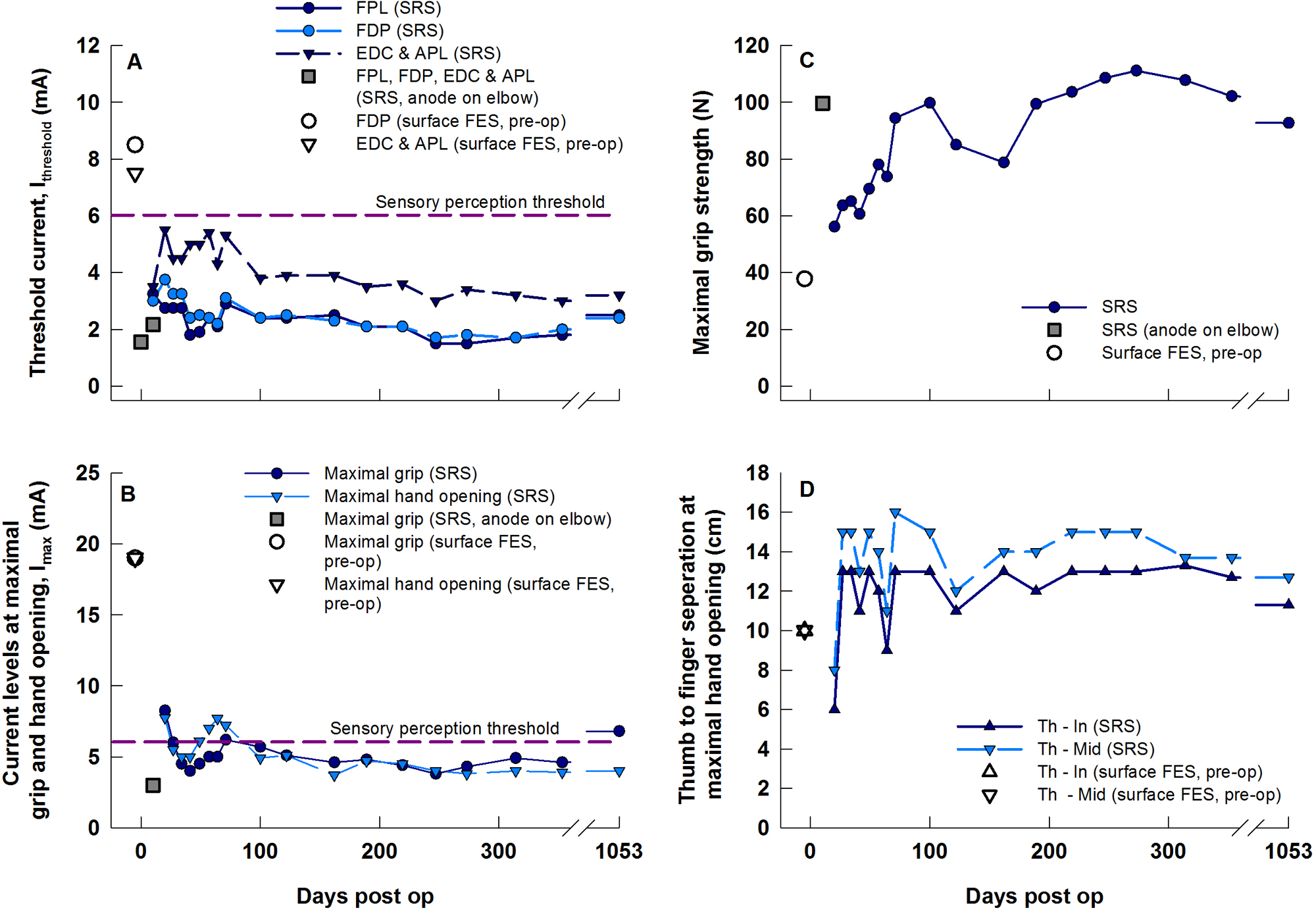
Stimulation parameters before surgery with surface stimulation (unconnected data points) and those monitored periodically over 356 days after the 3 SRS leads had been implanted. (A) minimal surface current (
Postoperative Data
After the wounds were sutured at the end of surgery, a self-adhesive surface electrode (3.8 cm × 3.8 cm; Sterile Post Operative Electrodes, Uni-Patch, Wabasha, Minnesota) was placed on the skin over the 2 pick-up terminals on the anterior forearm to co-activate FPL and FDP. A similar electrode was placed over the pick-up terminal on the posterior forearm to co-activate EDC and APL. Preoperatively, a 3.8 cm × 7.6–cm reference electrode had been placed proximal to the elbow and left in place for intraoperative and postoperative testing.
Within 10 days of implantation, the participant was able to perform handshakes of increasing strength as the amplitudes of pulse trains were gradually increased. He could crush an empty soft drink can and hold a jug containing a liter of water by the handle. His only conscious sensation during stimulation was a “pulling” of the muscles as they were activated. He did not feel the parasthesiae he had felt during surface FES prior to the implant.
Figure 3 shows measurements of
Two factors may have contributed to the lower forces in the weeks after day 10. First, compression within the nerve cuffs and surgical trauma may have had adverse effects on nerves and muscles. This is addressed in the Discussion. Second, on day 10, to avoid the forearm incisions, the reference electrode was placed proximal to the elbow joint, whereas on all subsequent occasions and also for the surface stimulation tests prior to surgery, it was placed on the forearm. We had shown previously that the placement of the reference electrode with respect to the implanted nerve cuff can affect the force elicited. 5 This raised the question whether the reading on day 10 provided an appropriate baseline. To address this question, 35 months postoperatively, we compared grip forces elicited with the reference electrode placed either on the forearm or above the elbow. For the forearm location, the mean peak force elicited in 3 repetitions was 92.7 N. For the above-elbow location, it was 113.7 N, that is, 20% higher. So the force at day 10 in Figure 3C was probably about 20% higher than it would have been had it been elicited with the reference electrode on the forearm. However, this would still only account for about a third of the drop after day 10.
Figure 3D shows a 14-cm thumb–finger separation during maximal stimulation after 4 months, which was 10 cm greater than the separation he could achieve with passive tenodesis and 4 cm greater than the separation achieved with surface FES prior to surgery. At the time of submitting this report, the participant had been using the SRS in activities of daily life (ADLs) for 37 months. He had sufficient residual hand function in his left (nonoperated) hand to adjust the stimulation intensity of each channel and the delay between the onset of FDP and FPL stimulation without external assistance. He typically set the intensities to about 4.5 mA for EDL and APL, 3.5 mA for FDP, and 3.0 mA for FPL. He reported sometimes increasing the intensities for tasks requiring more force, or when muscle fatigue had set in.
The ability to grade activation levels with increasing amplitudes of stimulation was verified 34 months after implantation by delivering interleaved pulse trains through electrodes overlying the FPL and FPL pick-up terminals (Figure 4). The grip force thus elicited rose and fell smoothly as the amplitudes of the pulse trains were slowly ramped up and down. Force began increasing when the pulse amplitudes reached 2.5 mA (FDL) and 3.7 mA (FDP) and increased smoothly, albeit nonlinearly, to 90 N at 3.6 mA (FDL) and 5.6 mA (FDP).
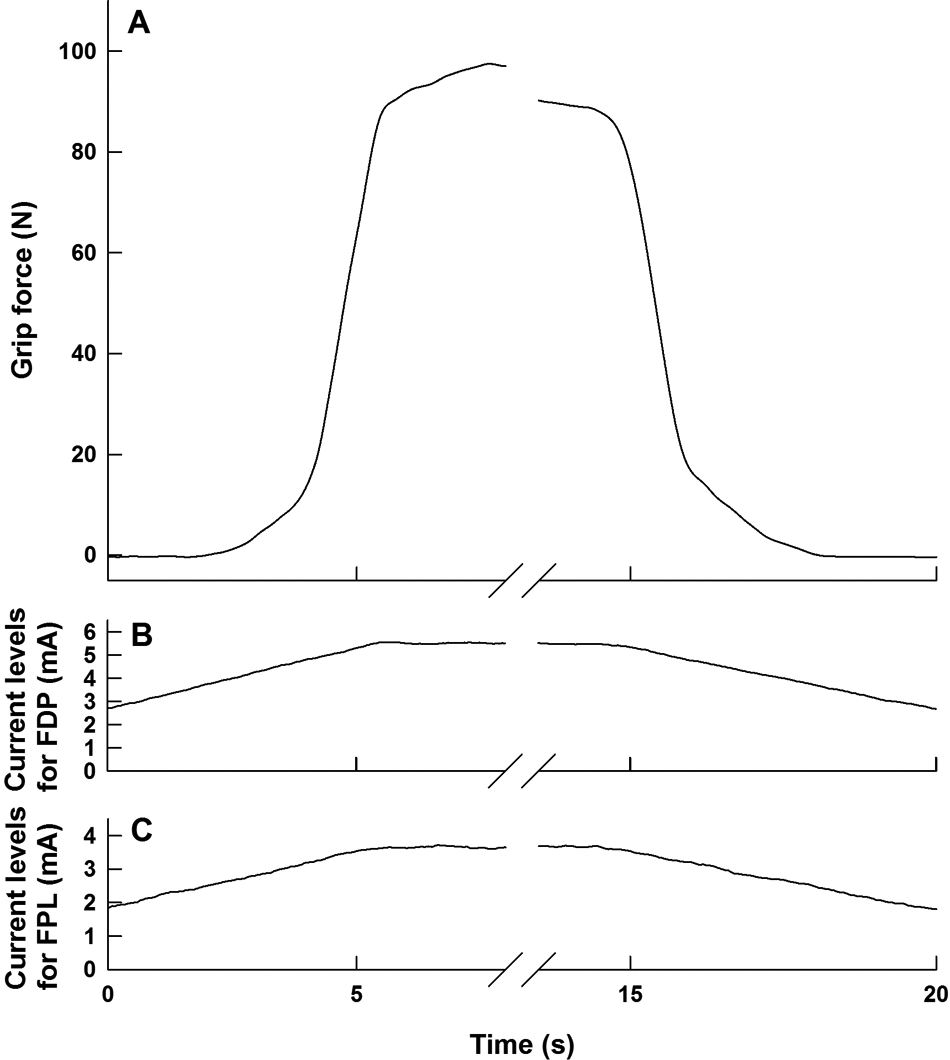
Smooth gradation of grasp force (A) elicited by slowly increasing and decreasing the amplitudes of pulse trains (B, C) delivered through surface electrodes located over the subcutaneous pickup terminals of the implanted FDP and FPL leads.
In a separate test, the selectivity of stimulation through surface electrodes located over the FDP and FPL leads was explored by delivering pulse trains (10 pulses/s) through a search electrode 2.5 cm in diameter applied to the skin at a series of points along a mediolateral line that passed over the FDP and FPL stimulation points (Figure 5). Stimulation at the FDP point elicited movements in the index finger and thumb, whereas stimulation at the FPL point elicited movements of the middle, ring, and index fingers.
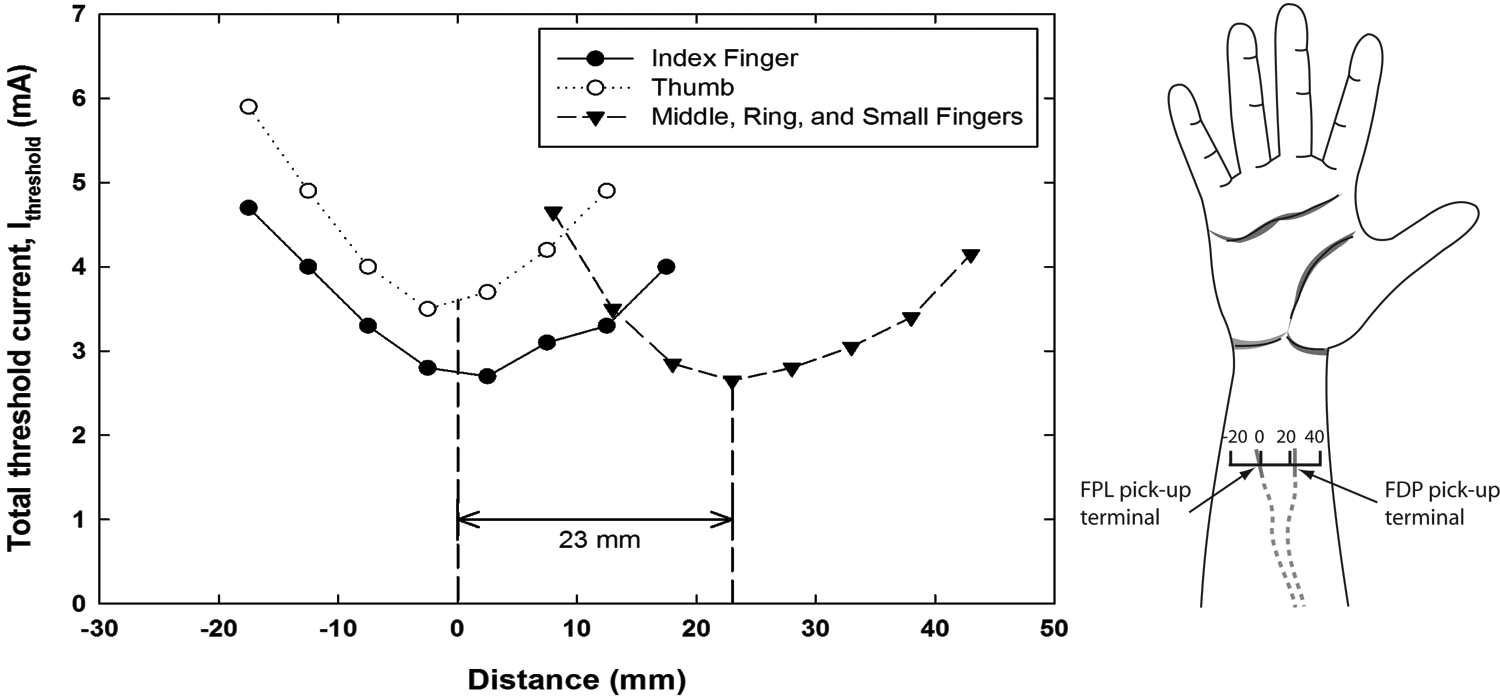
Selectivity of activation through the 2 flexor pick-up terminals implanted proximal to the wrist crease. Plots of threshold currents (
Usage was quantified by a counter in the stimulator that recorded the total duration of stimulation regardless of channel (Figure 6). To put the counter readings into context, in a session involving hand function tests lasting about 45 minutes, the counter read 16 minutes. Thus, a counter reading of 1 hour corresponds to manual activity of about 3 hours. The counter readings were recorded monthly up to 10 months after the participant received the stimulator and then at 12 and 15 months. The total duration of stimulation ranged from a few hours to 56 hours per month. Lower usage in the earlier months was attributed to self-reported muscle fatigue and to technical difficulties associated with the stimulator and wireless earpiece that were gradually overcome. Once the reliability of the external hardware had stabilized, the participant became more confident in using the device in ADLs. At month 7, he experienced a shoulder injury while doing unrelated exercises and his SRS usage declined. It resumed the next month. At month 9, stimulator repairs caused a hiatus of about 3 weeks. At month 10, the participant had a magnetic resonance imaging (MRI) scan for an unrelated condition and subsequently experienced mild muscle soreness in the implanted forearm. He stopped using the device until the soreness resolved 2 weeks later. At that point, stimulation thresholds were similar to those before the MRI scan (see parameters on day 356 postimplantation; Figure 3), indicating that the target nerve was not damaged. However, this does raise the issue of the safety of MRI scans in people with implanted SRS leads. Tests are underway in our laboratory to understand this better. 13
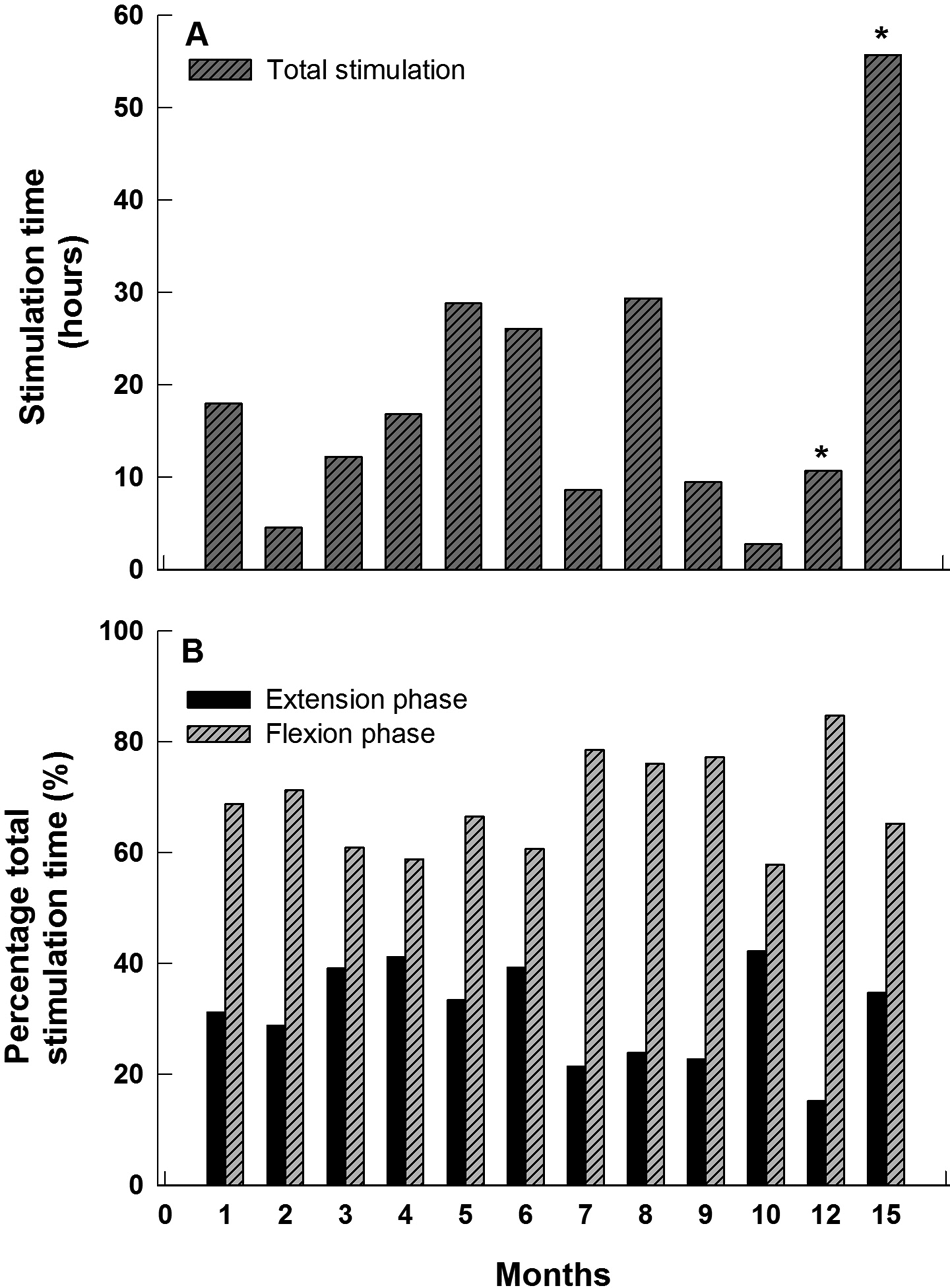
Usage of the Stimulus Router System. Monthly readings were obtained from a digital counter in the stimulator for 15 months after implantation. The readings represent the total stimulation time, not the total time the device was worn. (A) Monthly durations of flexion and extension stimulation. From the 10-month time point onward, only 2 readings were obtained (at 12 and 15 months, marked by asterisks). For consistency, we computed and displayed the mean monthly stimulation durations at these times. The drops in usage at 7 and 9 to 12 months are discussed in the text. (B) Percentage durations of flexion and extension stimulation.
Figure 6B shows that flexors were activated for longer durations than extensors, namely, 60% to 70% of total stimulation time. This indicates that in ADLs the time spent gripping objects was longer than the time taken to open the hand around objects. The participant reported that his hand opened more quickly and with a larger aperture with continued daily usage of the device. A video clip of the participant controlling hand opening and grip is provided as Supplementary Material.
Discussion
The main aim of this study was to evaluate the performance of the SRS as a new type of implantable NP. Conventional NPs consist of an implanted stimulator, controlled by radiofrequency transmission of commands and energy from a coil antenna worn outside the body. We chose to test the SRS in a motor application in the first permanent human implant, primarily because the activation of nerves innervating muscles elicits responses that are unambiguous and relatively easy to measure.
We found that the SRS was able to selectively activate the target muscles in a graded and controlled manner over their full physiological range at surface current levels below those eliciting sensory perception.
The
Grip force and hand aperture with SRS stimulation were larger than those achieved with surface FES prior to surgery (Figure 3). Grip force dropped from 100 N at day 10 to 60 N a week later and recovered to 100 N over the next 7 weeks. About a third of the drop can be accounted for by the differences in reference electrode positions (see Results). What could have caused the rest of the drop? The surgical separation of the nerves from their surrounding tissues or a build-up of pressure within the nerve cuffs might have caused some delayed denervation. 14 After day 10, the forearm became slightly swollen and sore, possibly because usage of the system in daily life had commenced too early. The swelling and tenderness gradually resolved over the next few weeks. The drop in force after day 10, and the gradual recovery, coincided with this period. This suggests that tissue edema may have reduced the ability of the muscles to generate force.
The amount of surface current needed to achieve useful muscle activation with the SRS was much lower than that required with surface FES prior to surgery and generally below sensory perceptual levels. The ratios of
The participant has continued to use the SRS on a daily basis. The counter readings ranged from 2 to 56 hours per month. According to our estimate, this corresponds to manual activity of 6 to 168 hours per month, or on average 0.2 to 5.6 hours per day. To our knowledge, this is the first time usage statistics have been obtained with electronic counters in NPs. Normative data on the mean duration per day of hand muscle activity in able-bodied people in different occupations do not exist but would certainly provide additional context.
The counter results suggest that the benefit of using the system outweighed the inconvenience. The participant described improved efficiency (eg, fewer phone handset drops, improved grip of documents, no need to scoop or “sweep objects into his lap, and ability to pick up heavier objects such as a large milk container, open his refrigerator without assistance, and shake hands at social gatherings). He found that the manipulation of small objects was better done without the SRS, as strong activation of finger flexors and extensors reduced dexterity. This FES action is not unique to the SRS. 15 He could don the wristlet in 2 minutes without assistance, but needed help with the earpiece, so on some occasions he used the alternative triggering method of tapping or bumping his forearm.
The SRS provides several potential advantages over existing systems. Greater selectivity is achieved compared with surface FES because current is routed directly to deep-lying nerves. Unlike surface FES systems, where the electrodes must be placed over motor points that may be widely distributed and in inconvenient locations, several SRS leads can be routed subcutaneously to a convenient location, The external garments containing the surface electrodes and stimulator are therefore smaller, easier to don and doff, more comfortable, and less conspicuous.
The surgical implantation of the SRS is potentially less invasive and faster than that of NPs that incorporate implantable stimulators, such as the Neurocontrol Freehand and the Finetech Stimugrip.1,2 The StimuGrip stimulator is implanted in the forearm and leaves a noticeable bulge under the skin. External components of the SRS are easy to service and upgrade. On the other hand, the SRS requires surface electrodes to make good electrical contact with the skin, which is less convenient than inductive coupling. The delivery terminals of SRS leads must be placed within 2 or 3 mm of a nerve. In addition, cuffs containing SRS terminals require open surgery. The SRS is limited in the number of individual stimulation channels. NP stimulators controlled by inductive coupling may therefore be preferable in more demanding applications, using, for example, percutaneously placed Bion microstimulators. 16 This study supports the potential utility of the SRS as a new family of neuroprostheses.
Footnotes
Acknowledgements
We thank Michel Gauthier for designing the stimulator and earpiece and Kelly Brunton and Kieva Richards for their assistance in clinical evaluations.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Arthur Prochazka is a stakeholder in Rehabtronics, Inc, a university spin-off company that holds the intellectual property in the SRS and ReJoyce systems. Jan Andrzej Kowalczewski holds intellectual property in the nerve cuff used in the SRS system.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank the Canadian Institutes of Health Research and the Alberta Heritage Foundation for Medical Research for funding.
