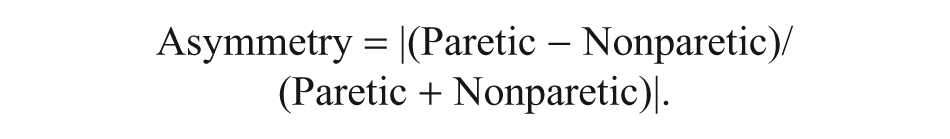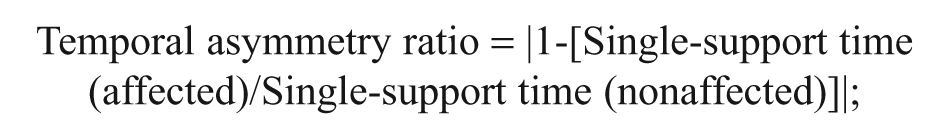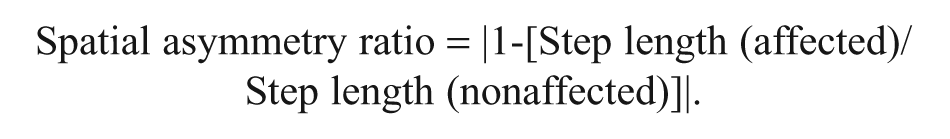Abstract
Background. The model of interhemispheric competition after stroke has been established for the upper but not for the lower extremity. Repetitive transcranial magnetic stimulation (rTMS) of the brain has been shown to modulate cortical excitability. Objective. The purpose of this study was to investigate the effects of rTMS followed by task-oriented training on cortical excitability and walking performance in individuals with chronic stroke. Methods. A total of 24 patients with average Fugl-Meyer lower limb scores of 17.88 ± 5.27 and average walking speeds of 63.81 ± 18.25 cm/s were randomized into an experimental group and a control group. Participants received rTMS (experimental group) or sham rTMS (control group) followed by task-oriented training (30 minutes) for 10 sessions over 2 weeks. Repetitive TMS was applied at a 1-Hz frequency over the leg area of the motor cortex of the unaffected hemisphere for 10 minutes. Outcomes, including motor-evoked potential (MEP), lower-extremity Fugl-Meyer score, and gait performance, were measured before and after training. Results. Decreased interhemispheric asymmetry of the amplitude of the MEP was noted after rTMS and task-oriented training. Improvement in spatial asymmetry of gait was comparable with increased symmetry in interhemispheric excitability. Motor control and walking ability were also significantly improved after rTMS and task-oriented training. Conclusions. rTMS enhances the effect of task-oriented training in those with chronic stroke, especially by increasing gait spatial symmetry and corticomotor excitability symmetry.
Keywords
Introduction
In the healthy brain, neural activity in motor areas of both hemispheres is functionally coupled and equally balanced. 1 After a monohemispheric stroke, the interhemispheric symmetry of brain excitability is altered in somatotopic reorganization of primary sensory and motor cortices, and the balance of cortical excitability between the motor areas of the 2 hemispheres is disturbed. 2 An enhanced excitability of the primary motor cortex (M1) of the unaffected hemisphere and an increased interhemispheric inhibition of the primary motor cortex of the affected hemisphere have also been observed.3,4 The model of interhemispheric competition has been established for the upper extremity. 5 Based on this concept of interhemispheric competition, it has been suggested that inhibition of cortical excitability in the unaffected hemisphere or facilitation of cortical excitability in the affected hemisphere normalizes the balance of cortical excitability between the 2 hemispheres, resulting in improvement of function of the affected hand.6,7
Transcranial magnetic stimulation (TMS) is a noninvasive means of stimulating nerve cells in superficial areas of the brain. 8 Repetitive TMS (rTMS) of the brain has been shown to modulate cortical excitability and cortical inhibition, with the effect depending mostly on the stimulation frequency. 9 Low-frequency rTMS appears to cause a transient reduction in cortical excitability, as shown by the motor-evoked potential (MEP), whereas high-frequency rTMS increases the size of the MEP and cortical excitability. 9 Based on the interhemispheric competition model, it has been suggested that rTMS be used to modulate abnormal brain excitability in 1 of 2 ways in patients with stroke: application of the high-frequency rTMS over the affected hemisphere or low-frequency rTMS over the unaffected hemisphere. 10 Butler and Wolf 11 further viewed rTMS as a tool that helps the brain to reach an optimal state of learning to facilitate subsequent training effects. A review study has shown that inhibitory (1 Hz) rTMS applied over M1 of the unaffected hemisphere is a safe and effective intervention to improve motor function of the affected hand after stroke. 5 Additionally, a combination with 1-Hz rTMS over M1 of the unaffected hemisphere during a motor training session improved motor performance. 12 However, whether inhibitory rTMS will be as effective in representative motor areas of the lower extremity remains unclear.
Current approaches to stroke rehabilitation emphasize task-oriented training. Advocates of task-oriented training conduct a training program that focuses on specific functional tasks to engage systems. There is growing evidence that intensive task-oriented practice can significantly improve walking competency in individuals with stroke. 13 The protocols of task-oriented training in previous studies were 12 to 57 sessions, with an intervention period of 4 to 19 weeks. 13 However, the rate of recovery achieved with these studies may not be satisfying to patients and their families. rTMS is a potential approach to maximize motor recovery in a short period. Thus, combining the effects of task-oriented training with those of rTMS conditioning is attractive. It is possible that modulating cortical excitability by rTMS before task-oriented training would enhance the therapeutic effects and shorten the therapeutic period. The purpose of the present study was to investigate the effect of 1-Hz rTMS, applied to the unaffected hemisphere with task-oriented training for 2 weeks, on cortical excitability and gait performance in those with chronic stroke. We tested the hypothesis that rTMS improves the symmetry of interhemispheric corticomotor excitability and enhances the effect of task-oriented training in those with chronic stroke, thereby leading to improvement, especially in gait symmetry.
Methods
Participants
Participants with stroke were recruited from a medical center in Taipei. Information about the diagnosis, age, sex, stroke type, location of the lesion (the right or the left hemisphere), and onset duration of hemiparesis were obtained through patient interviews and from medical charts. The inclusion criteria for participants were as follows: (1) diagnosis of unilateral hemiparesis secondary to cerebrovascular accident with postonset duration of more than 6 months, (2) ability to walk independently for at least 6 m with or without the use of walking aids, and (3) ability to follow simple verbal commands or instructions. The exclusion criteria were as follows: (1) unstable medical conditions, (2) histories of other diseases known to interfere with participation in the study, (3) past history of a seizure, and (4) use of a cardiac pacemaker. In addition, participants were excluded if the MEPs of their ipsilesional hemisphere could not be induced by TMS.
Finally, 45 individuals were identified as potential participants for this study. Of these, 28 participants gave informed consent and participated in the study (Figure 1). This study was a double-blind, randomized controlled trial. Both the assessor and the participant were blinded. Block randomization (with a block size of 2) was used to assign participants to either the experimental or the control group; this was done by an independent person who selected one of a set of sealed envelopes 30 minutes before the intervention began.

Flow chart of participants recruited in this study. Abbreviations: MEP, motor-evoked potential; rTMS, repetitive transcranial magnetic stimulation.
Procedures
The study protocol was approved by the institutional review board of Taipei Veterans General Hospital and explained to all included participants before their participation. Outcomes, including corticomotor excitability and motor performance, were measured before intervention (pre) and after completing a 2-week training (post) by an assessor who was blinded to the participant’s group. The primary outcome measures were corticomotor excitability symmetry and gait symmetry. The secondary outcome measures were the Fugl-Meyer assessment (FMA) score and spatial and temporal parameters of gait performance.
Interventions
Participants in the experimental and control groups received rTMS and sham rTMS for 10 minutes, respectively, followed by task-oriented training during a 30-minute session every day for 10 consecutive weekdays. A well-trained and qualified physical therapist delivered rTMS and task-oriented training.
Repetitive transcranial magnetic stimulation
rTMS was delivered to the scalp over the motor cortex of the unaffected hemisphere by using a MagStim SuperRapid stimulator (MagStim Co, Ltd, Carmarthenshire, Wales, UK) and a figure-of-eight coil.12,14-20 Real rTMS involved an intensity of 90% resting motor threshold and a train of 600 pulses (1 Hz) for 10 minutes during each session over the leg area of the motor cortex of the unaffected hemisphere. The motor cortex was stimulated by holding the figure-of-eight coil tangentially over the leg area at the optimal site for response from the rectus femoris muscle—typically, from 0 to 2 cm lateral to the vertex and from −1 to 2 cm posterior to the vertex. The location of the stimulation site was marked, referenced by vertex-centered coordinates, and recorded to maintain consistency among sessions. The coil was oriented at about 0° over the transverse plane with the handle pointing laterally to induce a posterior-to-anterior current flow in the cortex. Sham rTMS was performed with the coil held perpendicularly to the scalp of the unaffected hemisphere, using the same stimulation parameters (noise, time, and frequency) as used for real rTMS.
Task-oriented training
Task-oriented training emphasized active participation and individualized and task-oriented functional training. In this study, the program consisted of 6 workstations, each attended for 5 minutes, for a total of 30 minutes. The 6 workstations included (1) standing and reaching in different directions for objects located beyond arm’s length; (2) standing up from a sitting position in chairs of various heights (sitting-to-standing exercise); (3) stepping forward onto blocks of various heights (stepping-up exercise); (4) raising and lowering heel(s) while maintaining a standing posture (heel-lift exercise); (5) standing up from a chair, walking 3 m to a spot, turning around, and sitting back down (up-and-go exercise); and (6) focused walking as a part of ambulation training to improve symmetric gait pattern. Each individual participated in 1-to-1 therapy. Progressions included increasing the number of repetitions completed within 5 minutes at a workstation and increasing the difficulty of exercise performed at each workstation, including the distance reached in standing, height of the chair during the sitting-to-standing exercise, and height of the blocks during the stepping-up exercise.
Outcome Measures
Corticomotor excitability
The latency and amplitude of MEPs of the rectus femoris muscle elicited by TMS were used to indicate corticomotor excitability. 3 The MEPs of the bilateral rectus femoris muscles were recorded by an electromyographic machine in response to TMS (Magstim 200 electromagnetic stimulation) delivered through a figure-of-eight coil placed on the contralateral motor cortex. The MEP was found to be a sensitive measure for detecting residual corticospinal functions and to be predictive of motor recovery after stroke. 21
During measurements, participants sat comfortably in a chair with hips and knees in 90° flexion and wore a fitted cap marked with a coordinate system (distance, 1 cm). They were instructed to completely relax their leg during the evaluation period. Muscle activity was carefully monitored by real-time electromyography to confirm a relaxed state. The coil was positioned parallel with the coronal plane, and the handle of the coil pointed outward to induce a posterior-to-anterior current flow in the brain. The optimal scalp location (hot spot) was determined by moving the TMS stimulator over the scalp in 1-cm steps. Once the hot spot was identified, a single-pulse TMS was delivered to the location to determine the resting motor threshold, defined as the lowest stimulus intensity necessary to elicit MEPs greater than 0.05-mV peak-to-peak amplitude in at least 5 of 10 consecutive stimuli. 22 Single magnetic stimulations at 110% of the resting motor threshold were randomly administered over the motor hot spot of both hemispheres to determine the MEP latency and amplitude for rectus femoris muscles. In all, 10 sweeps of data were collected with a ≥5-s stimulus interval. The intensity used in the postassessment was the same as that used in the preassessment.23-25 The asymmetry ratio of MEP latency and amplitude were also calculated as follows 26 :
Motor performance
Motor control of the lower limb, including reflexes, synergistic patterns, and coordination, was assessed by FMA. 27 Each item was scored using a 3-point ordinal scale, from 0 (no performance) to 2 (complete performance), with a maximum of 34 points. This instrument has been reported to have good responsiveness and excellent validity and reliability.28,29
Walking performance
Spatial and temporal parameters of gait performance were obtained using the GAITRite system (CIR Systems, Inc, Havertown, Pennsylvania). The GAITRite walkway contains 6 sensor pads and 13 824 pressure-sensitive sensors and connects to a personal computer. The active area is 3.66 m long and 0.61 m wide. When the participant walks along the walkway, the contact time and location of each footfall are recorded, stored on a computer, and automatically translated into spatial and temporal parameters. The concurrent validity and reliability have previously been established.30,31 In the present study, participants were asked to walk along the walkway 3 times at a self-selected speed, and the average was used for analysis. In addition, temporal and spatial asymmetry ratios were calculated according to the following formulae32,33:
Statistical Analysis
All analyses were performed using the SPSS 17.0 statistical package. Descriptive statistics were generated for all variables, and distributions of variables were expressed as mean ± standard deviation. Intergroup differences among baseline characteristics were evaluated using an independent t test or χ 2 analysis. Change scores were calculated by subtracting baseline data from posttreatment data. To analyze intergroup improvement, change scores were analyzed using a 1-way multivariate analysis of variance with group as a factor. The statistical significance was set at P < .05.
Results
Participants
Of 28 participants, 14 were randomly assigned to the control group, and the other 14 were randomly assigned to the experimental group. Of 28 participants, 4 did not complete the intervention (2 in the control group and 2 in the experimental group; Figure 1). The 24 participants who completed the intervention attended all treatment sessions. None of the participants reported any adverse events. Demographic characteristics and cortical and clinical features of participants in both groups are presented in Table 1. The differences in the demographics of the 2 groups were insignificant. Moreover, differences in all the preintervention-selected outcome measures of the 2 groups were insignificant (Table 2).
Demographic Characteristics and Cortical and Clinical Features of Included Participants in Both Groups a
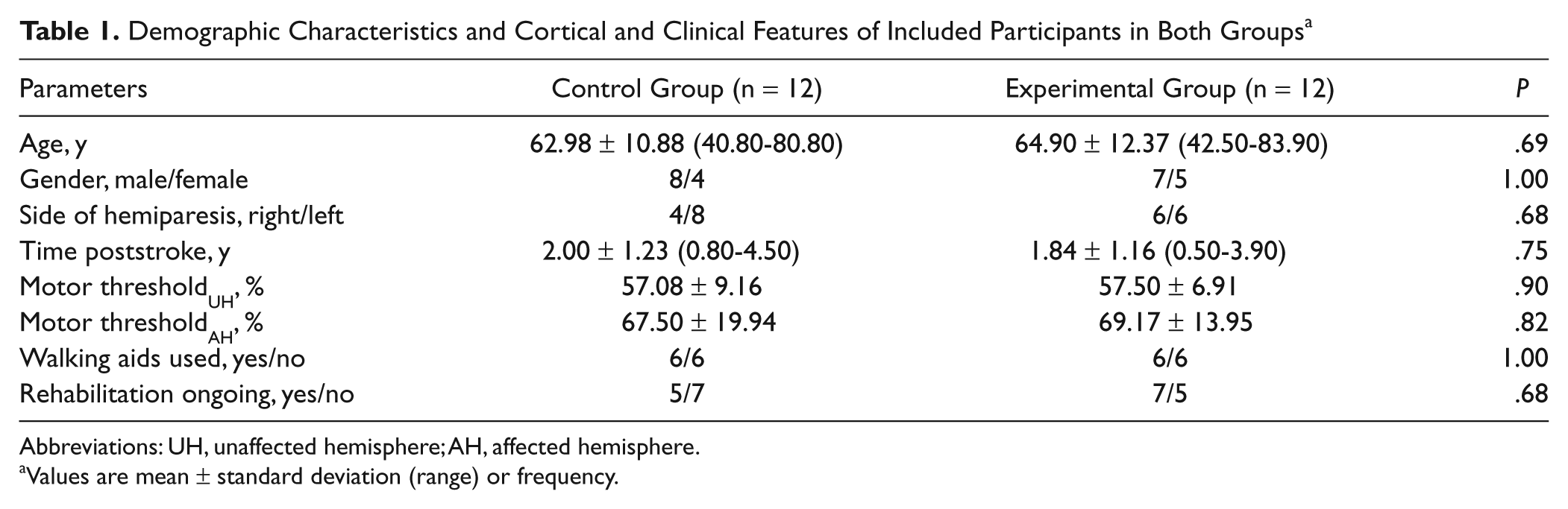
Abbreviations: UH, unaffected hemisphere; AH, affected hemisphere.
Values are mean ± standard deviation (range) or frequency.
Comparison of Cortical Measures Between Groups a
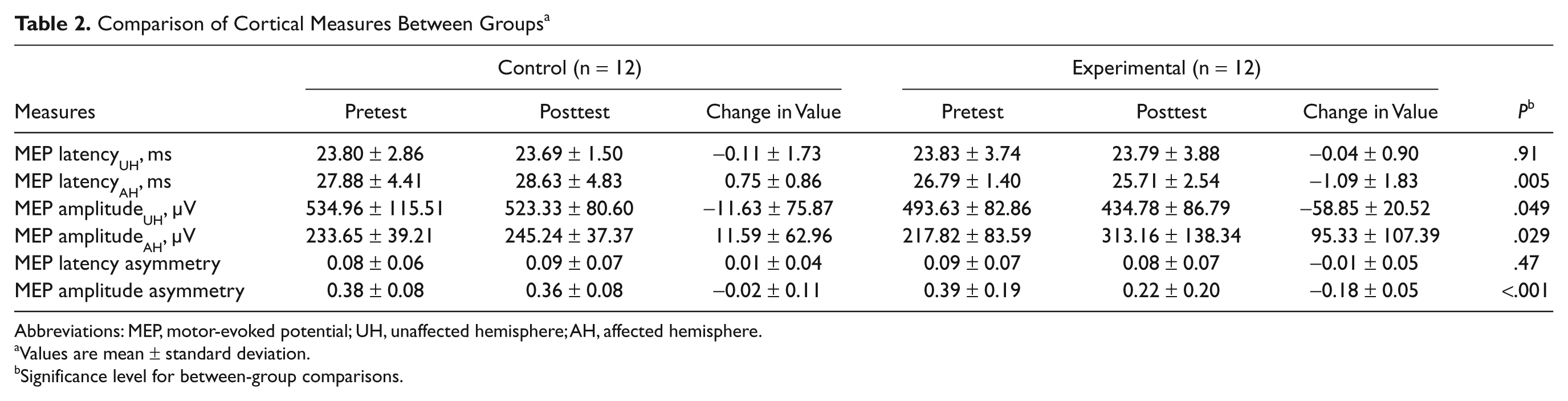
Abbreviations: MEP, motor-evoked potential; UH, unaffected hemisphere; AH, affected hemisphere.
Values are mean ± standard deviation.
Significance level for between-group comparisons.
Corticomotor Excitability
The results of corticomotor excitability are shown in Table 2. Between-group comparisons revealed significant differences between the 2 groups for MEP amplitude of the unaffected and affected hemispheres, MEP amplitude asymmetry, and MEP latency of the affected hemisphere.
Motor and Walking Performance
The results of motor and walking performance are presented in Table 3. Between-group comparisons revealed a significant improvement in motor control, as shown by FMA, and in walking performance, as shown by walking speed, cadence, bilateral step length, affected single-leg support time, double-leg support time, and spatial asymmetry ratio.
Comparison of Clinical Measures Between Groups a
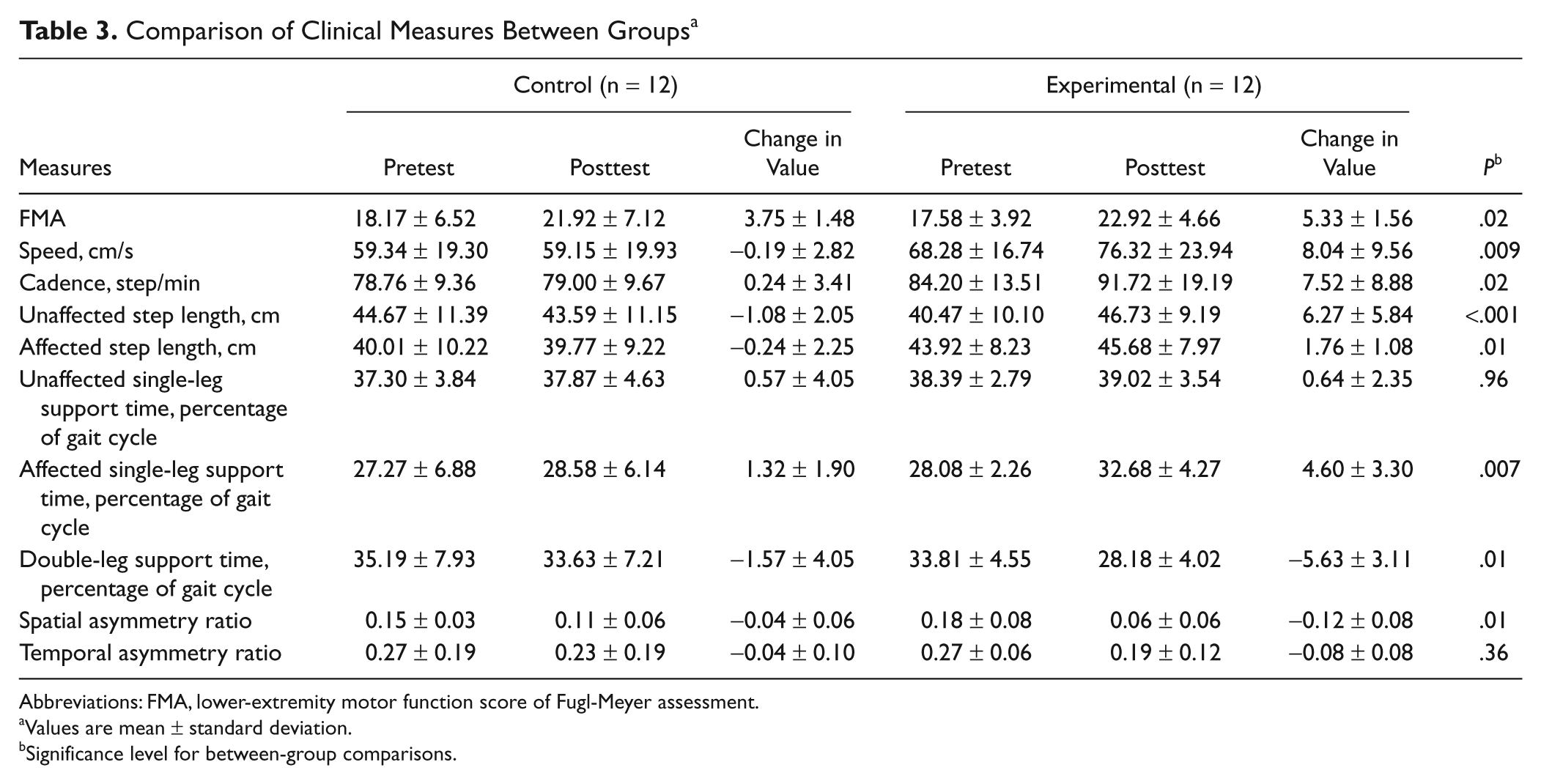
Abbreviations: FMA, lower-extremity motor function score of Fugl-Meyer assessment.
Values are mean ± standard deviation.
Significance level for between-group comparisons.
Discussion
In this study, for chronic stroke patients whose MEPs of the ipsilesional hemisphere could be induced by TMS, we found a significant decrease in MEP amplitude of the unaffected hemisphere and a significant increase in MEP amplitude of the affected hemisphere in the experimental group, as compared with the control group, after intervention. Such significant changes in MEP amplitude of bilateral hemispheres significantly improved MEP symmetry of the experimental group at posttraining. Application of low-frequency rTMS to the representative motor area of the lower extremity appeared to improve the balance of bilateral hemispheres in cortical excitability in a manner similar to that for the upper extremity.12,19
Our results support the concept of interhemispheric competition. Inhibitory rTMS decreased corticospinal excitability in the unaffected hemisphere and then increased corticospinal excitability in the affected hemisphere. Consequently, increased excitability in the affected hemisphere may contribute to improvements in motor function. Increased excitability of the affected motor cortex after rTMS appears to provide a more suitable environment for reorganization of the affected motor cortex by motor learning. 12 Takeuchi et al 19 suggested that improvement in motor function results not only from enhancing the excitability of the remaining neurons in M1 but also from unmasking local and distant neural networks.
Previously, we showed that performing task-oriented training 12 times in 4 weeks improved muscle strength of the lower extremity, gait velocity, cadence, and stride length. 34 In the present study, task-oriented training performed 10 times in 2 weeks did not lead to significant effects, as observed in our control group. Walking ability of the participants in the present study was poorer (average walking speed = 63.81 cm/s, least-limited community walker) before training when compared with that of participants in our previous study (average walking speed = 80.94 cm/s, community walker). Lack of improvement in gait outcome indicates that those with poor walking ability are less affected during a short period (even more frequent with 10 times in 2 weeks) of task-oriented training. However, rTMS followed by task-oriented training can exert a significant effect on gait performance in a short training period. In other words, additional rTMS in the experimental group seemed to enhance training effects. In previous studies, the effect of rTMS in healthy participants lasted for a maximum of 30 minutes.35,36 Thus, those who practiced task-oriented training immediately after rTMS in our study may have benefitted from a priming effect on brain excitability for facilitating learning and motor performance. Motor function can be influenced by an imbalance of transcallosal inhibitory circuits between motor areas in both hemispheres. Our findings support the fundamental notion of a balance of rivalry controlling interhemispheric interactions critical to motor function.
Motor control ability, represented as the FMA score, improved after training in both groups. This result was supported by results from previous studies that showed that FMA scores indicated an improvement of motor function after task-related training.37,38 Because all participants received task-oriented training, improvement in the FMA score could be expected. Furthermore, rTMS would enhance improvements in FMA scores.
Our results show that the value of spatial asymmetry approached zero (greater symmetry) after rTMS was combined with motor training. Furthermore, we found that the bilateral step length increased only in the experimental group. Application of low-frequency rTMS over the unaffected hemisphere was beneficial for gait symmetry. Spatial gait asymmetry has been shown to be related to older age, poor motor recovery, and low walking velocity. 39 Through improvements in walking velocity and motor recovery, the spatial asymmetry ratio approached symmetry in this study. In addition, spatial gait asymmetry was associated with insufficient propulsion force of the paretic leg. 40 Heel-lift exercises, which represented one of the workstations in this study, were designed to strengthen the ankle plantarflexor muscle. Strengthening of the ankle plantarflexor may also help improve spatial gait asymmetry. Additional rTMS could enhance the effect of task-oriented training and result in a more symmetric gait pattern.
Because the sample size was small and the therapist was not blinded, intervention and reliability cannot be generalized. However, we used the same protocol for all procedures to minimize the possible bias produced. Additionally, generalizability of our results is limited by inclusion of patients with measurable MEPs in the rectus femoris. Subsequently, these patients could have a relatively intact corticospinal tract and are likely to be at a relatively high functional level. Therefore, the results of the present study may only pertain to patients with these characteristics. Another limitation of this study is the lack of follow-up. We could not demonstrate whether these changes were maintained in participants. These limitations imply that caution should be exercised when interpreting results.
In conclusion, our results indicate that rTMS combined with task-oriented training is beneficial for motor recovery and brain reorganization in chronic stroke patients. This study is still at the beginning stage, and further studies are needed to determine the optimal dose, best location, and type of rTMS stimulation as well as the appropriate type of motor training and the most responsive characteristics of stroke patients. In addition, the findings of this study need to be validated through a study in a larger sample population.
Implications
These results may contribute to understanding the effects of a long-term trial. The findings of this study have meaningful implications for clinicians and researchers who focus on improving gait performance after stroke.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Cheng Hsin Rehabilitation Medical Center (Grant No. 98F117CY04) and the National Science Council (Grant No. NSC 98-2314-B-010-004-MY3).