Abstract
Keywords
Introduction
Motor neuroprostheses are devices that use electrical stimulation to elicit functional contractions in paralyzed muscles. Neuroprosthetic devices have been developed to restore functions such as hand grasp, 1 standing, 2 walking, 3 respiratory function, 4 and bowel and bladder control 5 in individuals with paralysis. To date, hundreds of individuals have received systems to restore a single function, and a few have systems to restore bilateral hand grasp or hand grasp and either bladder control, trunk control, or standing.
Although not yet clinically deployed, the recent development of a modular, fully implantable neuroprosthetic system was designed to enable restoration of multiple functions in a single individual.6,7 The challenge is how to control each restored function. At minimum, each requires one logical command signal. Logical command signals are used to turn systems on and off, trigger a specific function or set of functions (such as hand grasp configuration), and lock stimulation at certain levels. More complex systems require more advanced command inputs; Peckham et al
1
developed a hand grasp neuroprosthesis that uses both a logical and a graded “proportional” command signal. Proportional command signals provide graded actions, such as in hand opening and closing. Ideally, both logical and proportional command signals are easy to perform, inconspicuous, and do not interfere with other tasks.
8
Current successful implantable command sources include electromyographic (EMG) activity from muscles above the injury level that remain under voluntary control.
9
However, in individuals with cervical spinal cord injury (SCI), these muscle signals are limited. This study considers alternative sources for myoelectric command signals from
Anatomical and electrophysiological studies show that some cells remain intact across the injury even in cases of clinically complete SCI.10-12 At autopsy, Kakulas 13 reported that approximately 90% of SCI cases have some preservation of neural tissue across the lesion, even when half of injuries are considered clinically complete. 14 A recent study found 24% of more than 200 subjects studied had “discomplete” injuries, indicating clinically complete status according to the American Spinal Injury Association (ASIA) examination but neural tissue continuity at the injury level on autopsy. 11 A smaller case study showed that 45% of the motor complete injuries included had evidence of intact motor tracts anatomically. 15 Bunge et al 12 found that 50% of study participants classified as clinically complete retained some neural continuity at the lesion site.
Electrophysiological studies support the anatomical findings. Dimitrijevic et al
16
demonstrated the presence of intact descending pathways in someone with clinically complete SCI using electromyography more than 25 years ago. He coined the term
The goal of this study was to determine if muscle signals from the lower extremities of patients with motor complete cervical SCI could be detected in response to voluntary movement attempts and to consider the feasibility of using these signals as a command source for motor neuroprosthetic control. Specifically, we examined the muscles below the knee in people with chronic SCI and a lesion to the cervical region who were classified with either a complete injury (ASIA A) or a motor complete/sensory incomplete injury (ASIA B) for evidence of EMG activity in response to movement attempts.
Methods
Research Participants
A total of 12 individuals were enrolled with injury to the spinal cord at the cervical level. All participants had chronic injuries of at least 1 year. Table 1 describes the level, extent, time postinjury, and cause of injury. Each participant was classified as either ASIA A (motor/sensory complete) or ASIA B (motor complete/sensory incomplete) based on an examination performed by an experienced clinician trained in applying the ASIA testing criteria. All participants gave their informed consent and all study procedures were approved by the local institutional review board.
Description of Participants in Order of Duration of SCI
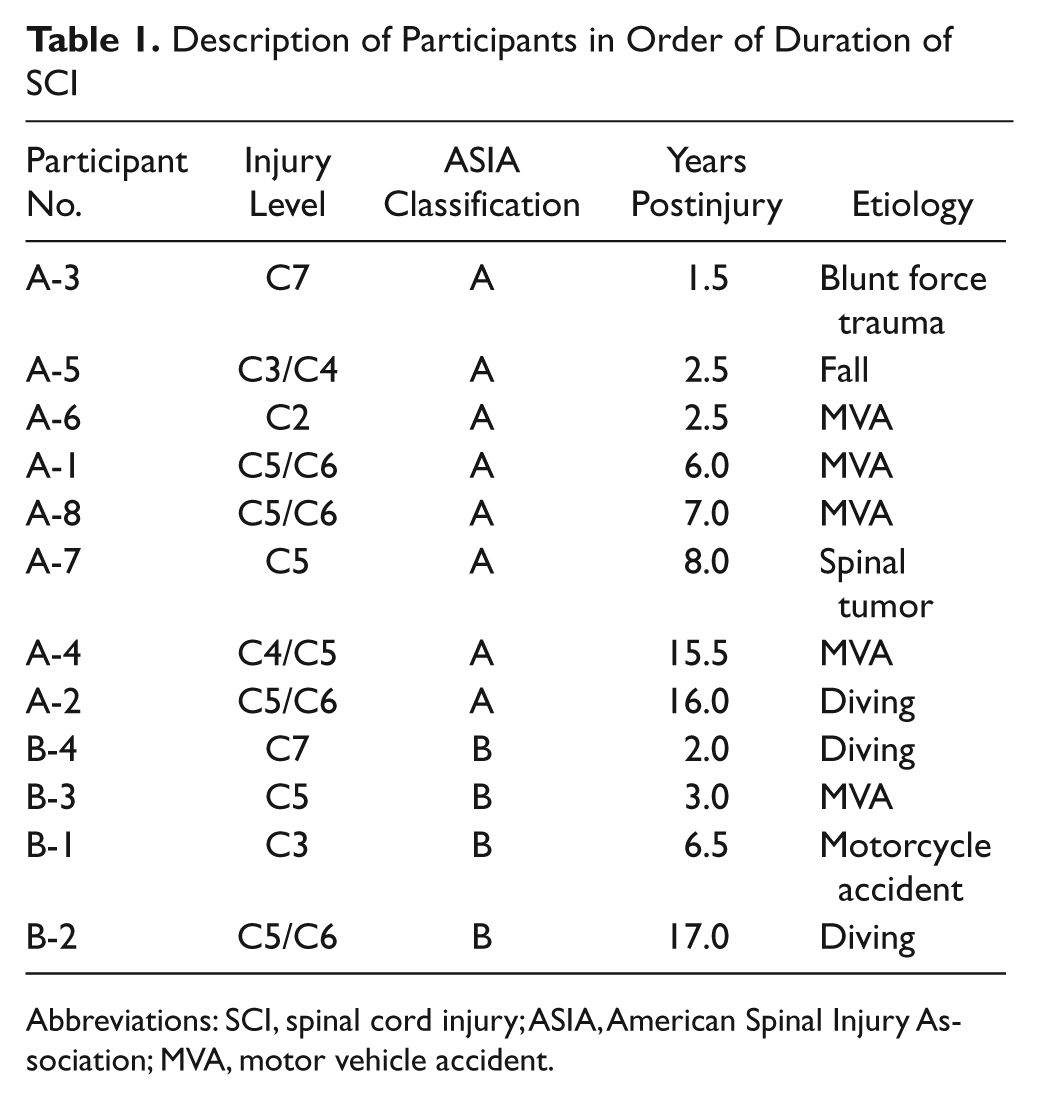
Abbreviations: SCI, spinal cord injury; ASIA, American Spinal Injury Association; MVA, motor vehicle accident.
Instrumentation
EMG signals were recorded from 8 muscles in each limb using pairs of surface electrodes positioned over a set of muscles below the knee. Preamplifiers located inches from the electrodes provided an initial gain of 100 and a high pass filter at 10 Hz to minimize any movement artifact. Programmable amplifiers (Cambridge Electronic Designs Limited, Cambridge, England) were used for further amplification with a low-pass anti-aliasing filter at 1 kHz and a 60 Hz notch filter. Signals were sampled at 2.5 kHz and displayed on a computer screen to the participant in real time using XPC Target and Matlab (The MathWorks, Inc, Natick, MA).
Protocol
EMG activity was recorded from 8 muscles located either in the foot or in the lower leg below the knee on each side of the body. The muscles recorded in the foot were the abductor hallucis (AbH), extensor digitorum brevis (EDB), and flexor digitorum brevis (FDB), and in the lower leg the soleus, tibialis anterior, medial gastrocnemius, lateral gastrocnemius, and peroneus longus. In the absence of significant palpable muscle tissue, bony landmarks were used to estimate the positions of the surface electrodes. 19 Two surface electrodes were spaced approximately half an inch apart over each muscle for differential recording.
Each participant performed at least 19 trials on each leg, each lasting 30 seconds. Muscle activity was recorded from 8 muscles in a single limb during each trial. Two types of trials were performed: (
Minimal feedback of these muscle signals was displayed to the participants. The participant could see 2 displays on a computer screen. The first showed scrolling full-bandwidth EMG from all 8 muscles at once. The second displayed all 8 signals rectified and low-pass filtered. One muscle was not singled out or highlighted in a given trial. Because this study was designed to consider any type of activity in response to voluntary movement attempts and literature suggests that single joint movements may elicit activity in multiple muscles, 20 activity from any of the 8 muscles was considered for each type of movement.
Analysis
Analysis was performed offline to determine if significant EMG activity was present during volitional movement attempts. In some instances, a substantial EMG signal was identifiable during volitional movement attempts without additional processing. For each channel, a threshold scheme was used to detect the presence of voluntary EMG activity. If significant activity was present, a second threshold was used to discretely classify the data as either “movement” or “rest.”
The initial threshold, known as the amplitude threshold, was determined as an integer multiplier times the standard deviation of the mean EMG signal at rest (multiplier stepped from 3 to 23 in unit increments). Offline analysis evaluated classification accuracy as a function of threshold level. Each data point with a rectified value greater than the threshold value was counted as a “threshold crossing.” The Wilcoxon rank sum test was then used to determine if the number of threshold crossings counted during movement attempts was significantly different from the number counted during rest segments. A significant difference in threshold crossings was used to determine whether significant activity was present in that muscle during attempted movements.
Figure 1 helps illustrate the analysis procedure. The top plot shows an example of full-bandwidth EMG data from the flexor digitorum brevis muscle from participant A-2. The bottom part of Figure 1 shows standardized EMG data illustrating the number of standard deviations away from the mean. The amplitude threshold is set at 4 standard deviations above the resting mean EMG in the bottom plot. The table in Figure 1 shows the number of threshold crossings within specific time segments. We included an additional second after the sound of the relax cue to allow for longer than normal reaction times because of lack of use of these muscles.
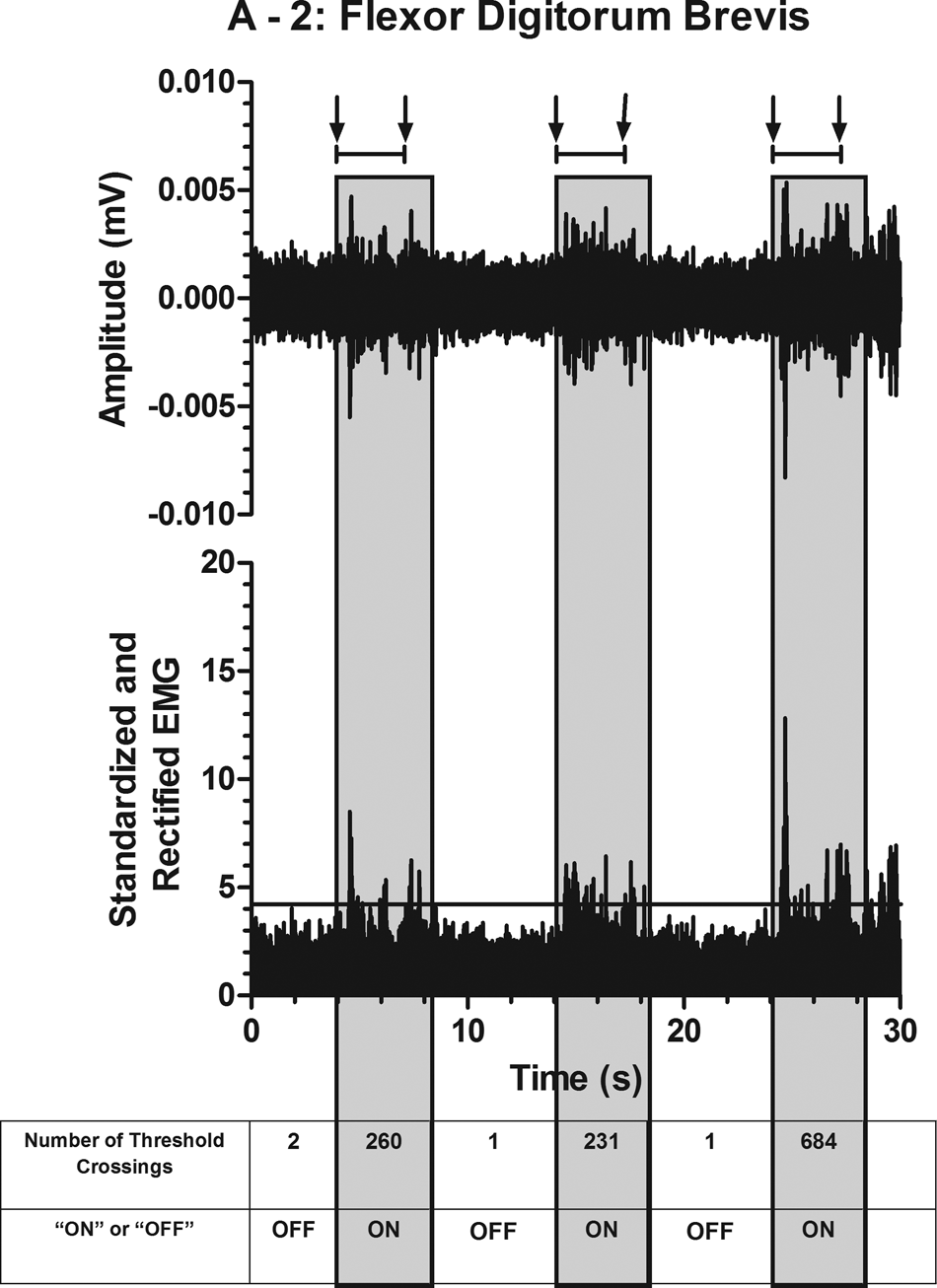
Top: Full-bandwidth electromyographic (EMG) data from the flexor digitorum brevis muscle during a single trial where ankle plantar flexion was attempted 3 times. Bottom: Same data rectified and standardized to illustrate the number of standard deviations from the mean. A threshold, shown on the lower graph at 4, helps illustrate the number of data points that cross the line during each 4-second movement time segment (shaded blocks). The first arrow above each shaded block represents the computerized audio cue to move and the second arrow indicates the timing of the second tone cuing the participant to relax. The number of threshold crossings before, during, and after movement attempts are listed in the table at the bottom.
If significant EMG activity is present on a muscle during volitional movement attempts, that muscle could potentially be used as a discrete command signal. To evaluate the muscles with significant volitional activity as a discrete command, a second threshold, referred to as the classification threshold, was used to separate “move” attempts from “rest” segments. A time segment was classified as movement if the number of amplitude threshold crossings was larger than the classification threshold. The classification threshold was also incrementally stepped to look at the effect of specific threshold values on classification accuracy. Classification of the data to determine the discrete state of muscle activity (“move” or “rest”) allowed for the calculation of the true positive rate (TPR) and the false positive rate (FPR) at specific threshold levels.
The TPR (sensitivity) and FPR (1 − selectivity) can be graphically displayed using receiver operating characteristic (ROC) curves. ROC curves were plotted to evaluate the effects different threshold values had on classification performance. These measures have been used to evaluate other types of discrete command signals21-23 and could be used to identify thresholds for implementation of EMG signals to control motor neuroprostheses.
A single measure that represents the overall ability to determine the muscle activity state and can be computed from the ROC plot is area under the curve (AUC). 24 If the muscle activation states were determined using a random guess, the AUC value would equal 0.5. An AUC value greater than 0.5 suggests a higher probability for successful classification.
Results
Figure 2A shows full-bandwidth EMG activity of the FDB muscle as an individual with an ASIA A SCI at the C3 level attempted to point his toes. The amplitude increase correlates with the three cues for voluntary muscle contraction. Small movements of the toes were visible during the movement attempts for this participant. Figure 2B shows the power spectrum analysis of the signal at rest and during a single movement attempt. Notice the increase in frequency content during the volitional movement attempt over the expected EMG range. The full-bandwidth EMG activity from 7 other muscles recorded simultaneously to the signal shown in Figure 2A is displayed in Figure 2C.
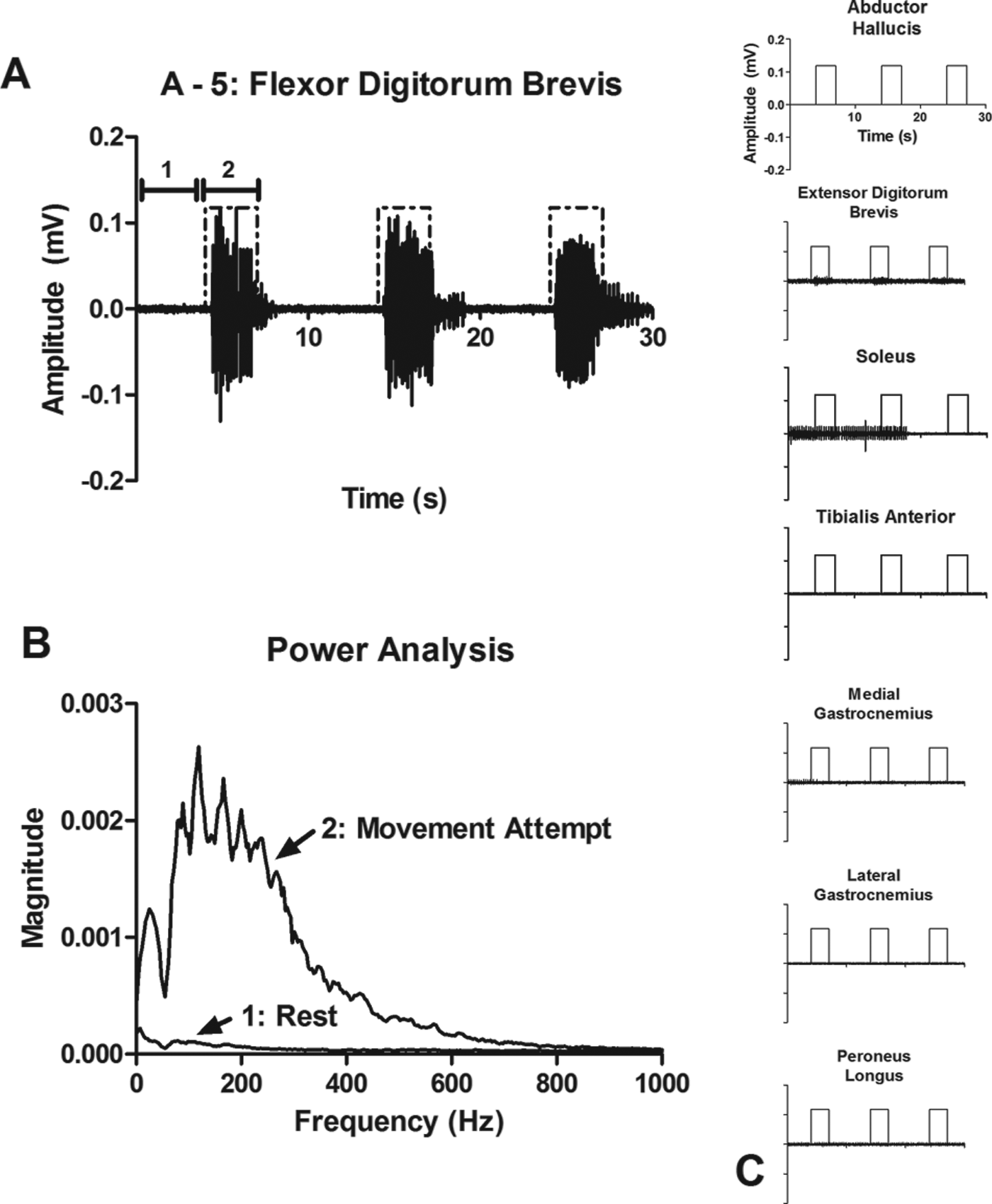
A) Volitional electromyographic (EMG) signal recorded from the flexor digitorum brevis muscle as an individual with ASIA A spinal cord injury at the C3 level attempted to point his toes. The square pulse represents the cues for volitional movement attempts. B) Power spectrum analysis of the EMG signal from A at rest (1) and during attempted movement (2). C) Full-bandwidth EMG data from 7 other muscles that were recorded simultaneously with the recording shown in A.
Varying the classification thresholds resulted in different TPR and FPR, illustrated by the ROC curves in Figure 3. Included is a graph of the curves with the AUC maximum (1.0), mean (0.807), and minimum (0.569) values from all the muscles with significant activity. Only one example, results from participant A-5’s medial gastrocnemius muscle, is shown for an AUC of 1.0. However, 9 muscles total had an AUC of 1.0. The mean curve (results from B-4’s EDB) provides a good illustration of the effect a changing threshold has on TPR and FPR. ROC curves will aid in the selection of proper threshold levels for the implementation of these signals to control a neuroprosthesis.
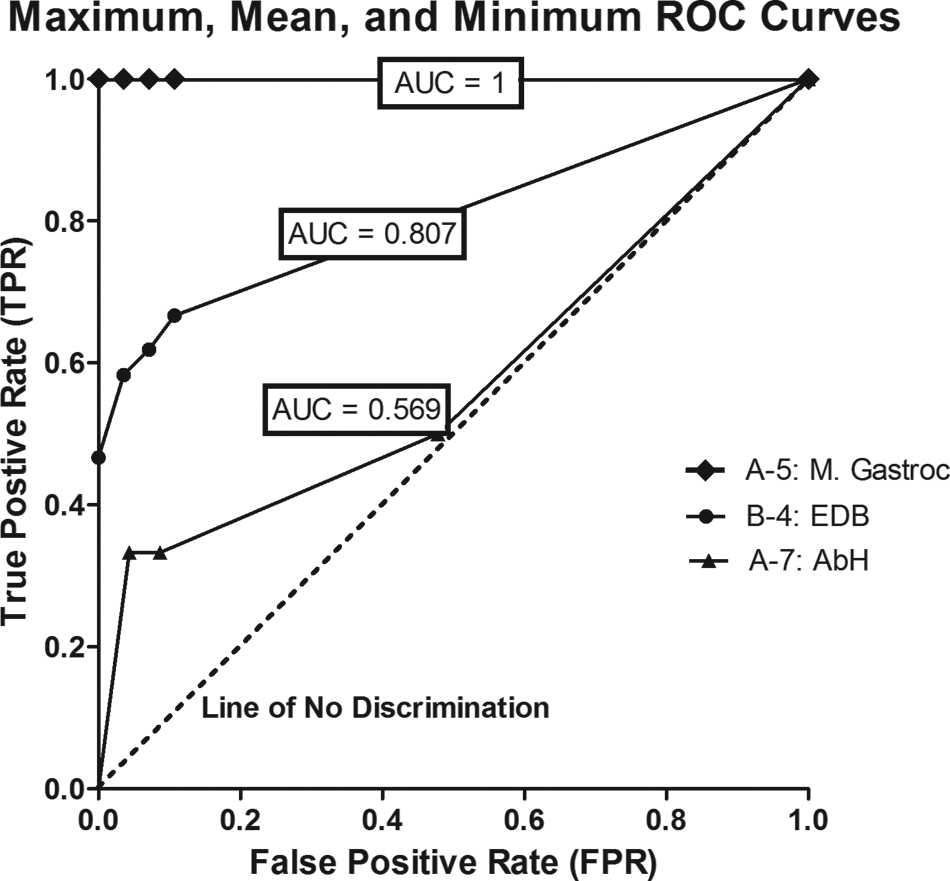
Receiver operating characteristic (ROC) curves from the muscles with the maximum, mean, and minimum area under the curve (AUC) values. These values were computed among all the muscles with significant activity during volitional movement attempts. The top line illustrates a perfect classification. The line of no discrimination represents random guess clarification, correlating to an AUC of 0.50.
Figure 4 indicates the number of muscles with significant activity present within each leg of each participant as determined by the Wilcoxon rank sum test. The darker bars in Figure 4 correspond to the number of muscles that achieved an AUC value of 0.90 or higher, suggesting potential success for use as a discrete command signal for a neuroprosthesis. Results show that all the 12 participants had at least one muscle with an AUC value of at least 0.90.
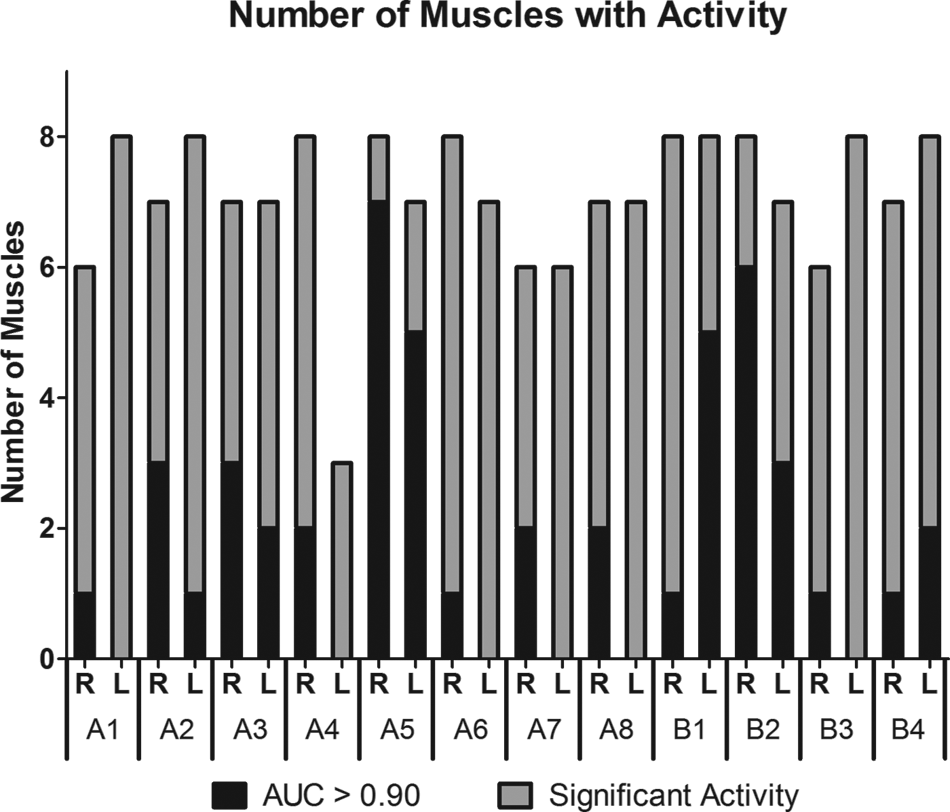
The top of the gray bar represents the number of muscles whose electromyographic (EMG) activity is significantly higher during attempted movements for each leg in each participant. The dark bar represents the number of muscles with an area under the curve (AUC) value of at least 0.90.
To demonstrate the utility of using attempted movements of foot/leg muscles as a command source for a motor neuroprosthesis, the 2 muscles with the highest AUC values were considered for each participant. The top 2 muscles were restricted to not being synergistic muscles in the same limb and to not being nearest neighbors in order to compensate for possible cross talk present on a channel because of the use of surface electrodes in this study. The mean AUC and standard deviation were then calculated across all participants for these top 2 muscle signals. The mean AUC value for the top muscle across all participants was 0.974 ± 0.033. The mean AUC value for the next highest muscle (that is not a nearest neighbor or synergistic partner) was 0.922 ± 0.061. All the participants had at least one muscle with an AUC greater than 0.90. The maximum AUC value possible, 1.0, was calculated for at least one muscle in 3 of the participants and for 9 muscles overall.
Table 2 indicates the number of legs that had muscles with significant activity. The total number of legs studied was 24. The last 2 columns highlight the number of legs with significant activity and an AUC value of at least 0.90 or 0.80, respectively, identifying these muscles as potential command signals for a neuroprosthesis. Of all 192 muscles evaluated, 46% had an AUC value greater than 0.80 and 25% had an AUC value greater than 0.90.
Muscles Listed With Total Number of Legs and Percentage of Legs With Significant Activity a
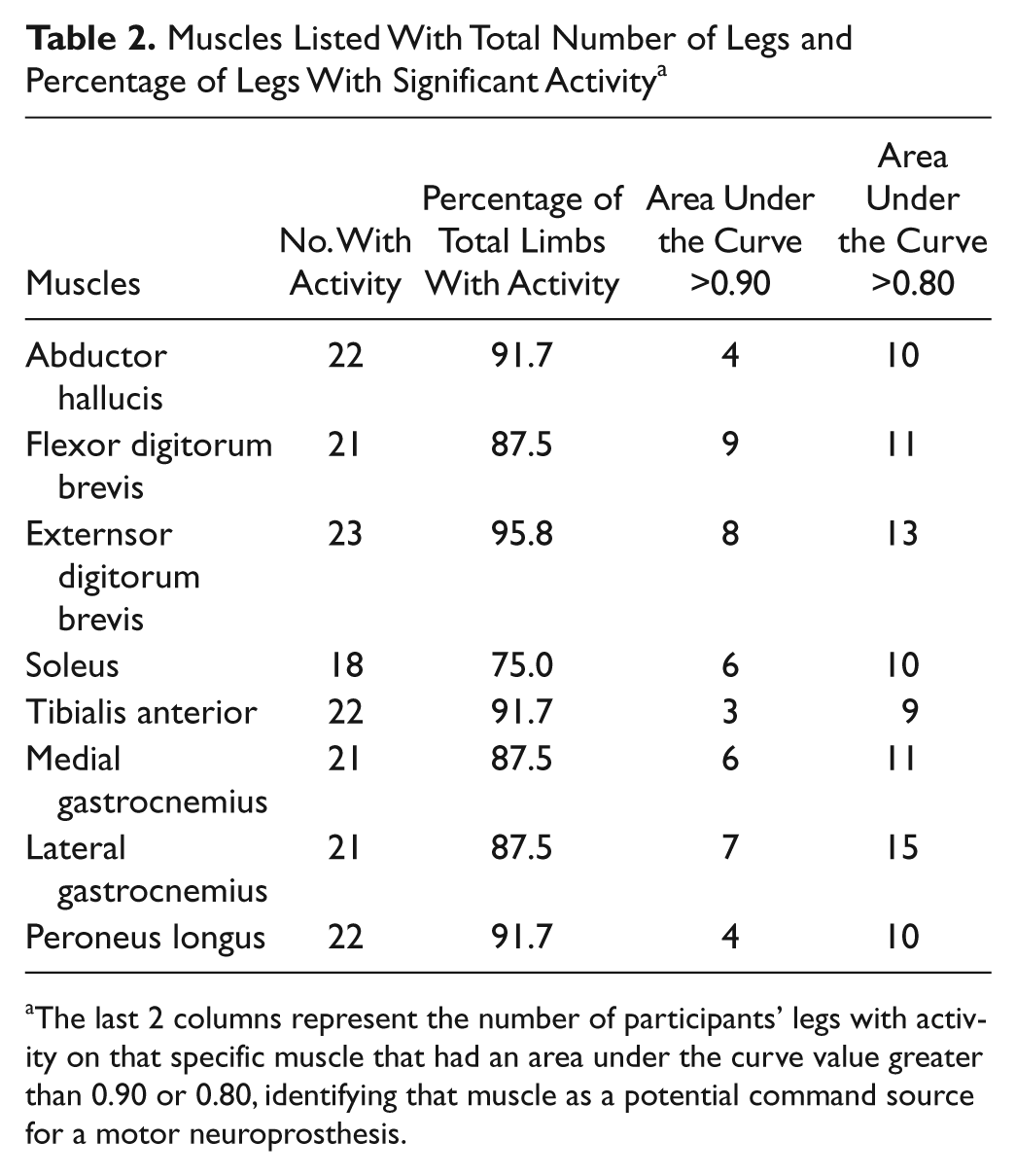
The last 2 columns represent the number of participants’ legs with activity on that specific muscle that had an area under the curve value greater than 0.90 or 0.80, identifying that muscle as a potential command source for a motor neuroprosthesis.
Discussion
The key contribution of this study is to expand the number of possible myoelectric control sources in an individual with complete cervical-level SCI. Although the study was not specifically designed to differentiate between direct voluntary control and indirect spastic control, the results strongly favor the hypothesis that the signal observed was under direct voluntary control and hence, have implications regarding the physiological extent of SCI. In all 12 individuals we were able to detect activity in multiple muscles below the level of injury in response to attempted volitional movements. These results suggest that many individuals with complete cervical-level SCI may retain voluntary activity in muscles considered clinically paralyzed. Current clinical evaluation of SCI only measures visible or palpable movement, while a more sensitive measure of muscle activity may be EMG testing. 10 Although preservation of a small number of axons alone may not be sufficient to produce functional movement, 25 these small signals are likely to be sufficient as an additional command source for a motor neuroprosthesis. Our results align with the concept of a “discomplete” injury, as suggested both anatomically 11 and physiologically. 18
For this study, we considered muscles that are not included in the standard ASIA neurological exam. Specifically, we looked at 3 muscles in the foot: the AbH, the FDB, and the EDB, based on the results of Calancie et al. 26 Large EMG activity in response to movement attempts was evident in 3 of the 12 participants. In all 3, the largest activity was present on the FDB muscle of the sole of the foot. Two theories may help explain these results. 26 One hypothesis is based on the pathology of SCI combined with the somatotopy in the lateral corticospinal tract. 19 If a ring of white matter is all that remains intact, 12 only axons in the most lateral regions of the lateral corticospinal tract would be expected to conduct, where distal muscles of the lower extremity are located. The second theory is based on the number, rather than the location of axons innervating distal muscles verses proximal muscles. Primate studies suggest that a larger number of axons innervate distal muscles. 27
Of the 3 participants with large amplitude EMG signals from the foot, 2 were classified as ASIA B. Sensation below the lesion is evidence of intact fibers at the injury level. It has been shown that individuals with ASIA B injuries are more likely to regain voluntary control over muscles below the injury level 28 and exhibit evidence of motor preservation by transcranial magnetic stimulation. 29 Two of the 3 individuals with large-amplitude EMG activity in this study were ASIA B. Since SCI results in 10% classified as ASIA B and 47% with ASIA A, 28 a much larger study would be required to confirm this preliminary observation.
Our study introduces the possibility of using below-injury muscles as command signals for neuroprostheses. The methods used here to evaluate EMG signals as a command source are applicable to the logical type of command signal, where only 2 states (on/off) are required. Although we did not specifically implement a system to evaluate the potential of below-injury muscle activity to be used as a proportional control source, based on our current neuroprosthesis designs, 9 at least 4 of the muscles evaluated in this study (2 in 1 participant, 1 each in 2 additional participants) appeared to have characteristics necessary for a gradable control signal. Future work will investigate these muscles further for proportional control of an upper extremity neuroprosthesis for hand grasp.
In every participant, we were able to identify at least 2 muscles with AUC values greater than 0.80. In the identification of these 2 muscles, nearest neighbor muscles, as well as synergistic muscle relationships, were taken into account: The 2 muscles were either in opposite limbs or in different regions of the same limb (foot/lower leg) with antagonist functions. This suggests at least 2 potential independent command signals in each participant. Ideally, an implanted neuroprosthesis will be able to restore multiple functions such as restoration of bilateral hand grasp. In this case, it would be advantageous to have 2 unique command signals on each side of the body (one for implementing opening/closing and one for selecting different grasp patterns 9 ), for a total of 4 command signals. Seven of the participants (58%) met this criterion for bilateral control (using AUC > 0.80).
Detected activity on different muscles in a single limb does not necessarily imply the commands are independent. Many of the muscles in the lower leg are activated in synergistic patterns and therefore the subjects may not be able to control each muscle independently. For example, during foot plantar flexion, EMG activity is frequently present on the gastrocnemius muscles and the soleus muscle in the lower leg, and the FDB in the foot. Additionally, surface electrodes were used for this initial feasibility study and crosstalk may be present between muscles located adjacent to one another. This initial study was designed to consider any muscle activity in the lower limb because we were unsure if recording this type of activity was feasible in individuals with complete SCI, hence no trials in this study were specifically performed to demonstrate independence.
Both independence and amplitude level are critical for use as a command signal, whether proportional or logical. In this study, minimal feedback was provided and there was no muscle training. Improved visual feedback of performance with repeated sessions may help improve the quality of the muscle signal. Previous studies suggest that training with operant feedback may be able to significantly improve muscle signal amplitude in an individual with SCI.30,31 Feedback training specifically looking at independence of muscle activity may be able to increase the number of independent command signals within a single limb.
Appropriate selection of classification thresholds is critical for success of a below-injury muscle as a command source. As illustrated by Figure 3, higher thresholds mean not only fewer false events but also a lower detection rate. The optimum threshold will vary based on the function the user intends to control. In some instances, false events may be tolerable and a preference would be toward a lower threshold that allows for a higher TPR. In other instances, such as control of bladder voiding, a low FPR is critical for user acceptance. Evaluation of the ROC curves in individual circumstances allows for proper selection of a threshold while taking into account user preferences related to the function under command.
Using signals from below the injury as a command signal begins to bridge the gap between the biological and substitutional approaches to find a cure for SCI. Combining a biological regenerative approach with a substitutional approach such as neuroprosthetics could affect the quality of life of individuals with SCI.
Footnotes
Acknowledgements
The authors would like to thank Elise Fijan for her help with data analysis and Elizabeth Kiracofe for her help with subject visits.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
This work was supported by the State of Ohio Biomedical Research & Commercialization Program (BRCP 06-22) and National Institutes of Health (T32-EB04314, U01 NS-069517).
