Abstract
Introduction
Visual search refers to the capacity of a participant to find a target among simultaneously presented distracters, 1 and it is based on visual abilities such as a fast visual processing and an accurate control of ballistic eye movements (saccades) that guide the fovea to the target location.2-4 Many brain areas are involved in this type of task, in particular, the 2 major visual intracortical streams—the so-called ventral and dorsal pathways—that transmit information from posterior sites (the primary visual areas) to anterior cortical regions. During visual search, the 2 systems work in parallel, the ventral stream being involved in pattern recognition of the searched stimulus and the dorsal stream being responsible for its spatial localization.
Studies in adults have shown that unilateral damage to the postchiasmal visual pathway is often associated to visual search disorders 5 and to homonymous hemianopia—that is the loss, in part or completely, of vision in the visual field contralateral (CL) to the side of lesion. These individuals cannot process images in the same way as normal controls and usually have difficulties with reading, detecting stimuli, or finding objects in the visual space corresponding to the affected field. Their fixations typically dwell in the intact hemifield, and their search pattern is characterized by frequent exploratory saccades into the blind part of the visual field6-8 with repeated saccades and fixations to the same object, resulting in overall longer visual search times.6,9,10 This phenomenon has been defined by Zihl 6 as “slowness of vision” in the CL hemifield to the side of the lesion.
Few studies have explored the possible effects of brain damage on visual search abilities when the lesion is acquired during childhood. Netelenbos and Van Rooij 11 studied 7 school-aged children with acquired unilateral brain lesions but no visual field defect and sensory or motor deficits. They reported abnormal values on visual search tasks only in children with right hemispheric lesions. Different results were reported by Schatz et al, 12 who studied 33 children with acquired stroke secondary to sickle cell disease and found abnormally slow responses in the contralesional visual field, especially when the lesion involved the left hemisphere.
Animal studies suggest different effects of early brain damage, compared with postnatal injury, on visual orienting. Monkeys with unilateral surgical ablation of the striate cortex sustained at 5 to 6 weeks of age demonstrate residual abilities to detect and localize visual stimuli within the CL hemifield, whereas monkeys with lesions sustained in adulthood show a large impairment in visual orienting. 13 Analogous studies on cats have reported a considerable sparing of visual functions following unilateral lesions in infancy extending to the occipital, parietal, and temporal cortices, whereas in adult cats, the same lesion results in a dense blindness and incapacity to orient to CL visual stimuli. 14 It has been suggested that modifications in circuits within the superior colliculus (SC) or involving efferent pathways from the CL intact hemisphere to the ipsilesional SC underlie the observed visual sparing.15-17
Also, in humans, there is some evidence suggesting a more effective mechanism of functional compensation of visual functions following early brain damage and corroborating the idea that maturational status has a pivotal role in determining individual responses to cortical damage.18,19 For instance, patients who underwent hemispherectomy at 7 years of age or earlier showed greater spared visual capacities compared with patients who underwent surgery at 17 years of age. 20 In addition, single case studies document residual visual abilities in patients with bilateral damage to the visual cortex at birth 21 in patients who underwent hemidecortication in the first year of life 22 and in patients affected by congenital right hemihydranencephaly. 23
To date, however, no study has explored visual search abilities in children with congenital brain damage, with and without associated visual field defects, even though common anecdotal reports among pediatric neurologists and ophthalmologists describe better compensating strategies of environmental exploration and object localization in those with congenital, as opposed to acquired, hemianopia.
To systematically investigate the different patterns of visual search in a group of children with congenital and acquired unilateral brain lesions in relation to the presence/absence of visual field defects, 29 patients were tested with a visual search test battery. Based on previous literature suggesting the presence of functional compensation in the case of congenital lesions caused by a mechanism of cortical/subcortical plasticity, a more organized pattern of visual search is expected for patients with visual field defects caused by congenital as compared with acquired lesions. These findings may help in the design of visual search interventions.
Methods
Patients
Children with congenital or acquired unilateral brain lesions, admitted for developmental check to the Pisa and Roma clinical units, were screened for inclusion into the study. Patients with intellectual disability or with severe ophthalmologic abnormalities, including cataract, retinopathy, or optic nerve atrophy, were not included. Children with a visual acuity below 8/10, even if reached with correction, were also excluded to avoid abnormal performances at the visual field and visual search tests that could arise because of the presence of spectacles.
Computerized Visual Field
Visual fields of patients were assessed by means of an automated perimetry system (KOWA AP 340). Each eye was tested with full threshold and full field (237 points), incorporating fixation monitoring.
Visual Search Battery
This battery consisted of 4 tests that are modified versions of those of Zihl 24 : the apple test, frog test, smile test, and E–F test. These 4 different tests targeted a wide range of perceptual abilities because the tests differed in terms of the quality and saliency of the features discriminating the target stimulus from the distracters (eg, shape and color). Participants were shown the stimulus arrays (52° × 45°, horizontally and vertically, respectively) projected on slides at a distance of 120 cm, and they were required to actively explore the visual field by using eye, but not head, movements to search for visual targets. They were instructed to fixate a red cross located in the center of each slide (ie, fixation point) and to search, after its disappearance, for a single target embedded among distracters. They were asked to press a “yes” key as response if the target was present and a “no” key as response if the target was absent. Responses and reaction times (RTs) for each hemifield were recorded.
Apple test
Each stimulus array contained 10 stimuli, distributed at random over the array. The stimuli consisted of red apples projected on a black background. A total of 10 trials were presented: 8 trials in which the target (yellow apple) was present (4 in the right and 4 in the left hemifield) and 2 in which the target was absent.
Frog test
Each stimulus array contained 13 stimuli, distributed at random over the array. The stimuli consisted of green trees, projected on a black background. A total of 11 trials were presented: 8 trials in which the target (green frog) was present (4 in the right and 4 in the left hemifield) and 3 in which the target was absent.
Smile test
Each stimulus array contained 9 stimuli, distributed at random over the array. The stimuli consisted of little smiling faces projected on a black background. Here, 10 trials were presented: 8 trials in which the target (sad face) was present (4 in the right and 4 in the left hemifield) and 2 in which the target was absent.
E–F test
Each stimulus array contained 22 stimuli distributed at random over the array. The stimuli consisted of green “E” letters projected on a black background. In this test, 20 trials were presented: 16 trials in which the target (green “F” letter) was present (8 in the right and 8 in the left hemifield) and 4 in which the target was absent.
Statistical Analysis
For the statistical analysis, the patients’ RTs and percentage of correct responses in trials where the target was present were considered. All the analyses were carried out using a 3-way ANOVA with Group as between factor and Test and Field of Visual Presentation (ipsilateral [IL] or CL to the lesion) as within factors. Whenever necessary, pairwise comparisons were conducted using the Newman-Keuls test.
Results
Participants
In all, 29 children with unilateral or predominantly unilateral brain damage and no mental retardation, aged 6 to 16 years, were enrolled in this study (see Table 1 for the description of the general characteristics of the participants). Of these, 14 children had a congenital lesion (8 on the left and 6 on the right), and the remaining 15 had an acquired lesion (8 on the left and 7 on the right). In those with acquired brain damage, the time elapsed since lesion occurrence was at least 12 months. All participants had at least 1 brain MRI scan (see Figures 1A and 1B for a graphic reconstruction of the brain lesions according to Damasio and Damasio’s atlas 25 ); a detailed description of the type of lesion and the side, site, and timing of the insult is given in Table 1. Overall, patients with acquired brain damage demonstrate smaller lesions compared with patients with congenital lesions, as shown in Figure 1. The motor development was normal only in 2 participants, whereas all the others had a diagnosis of hemiplegia according to Hagberg et al 26 criteria.
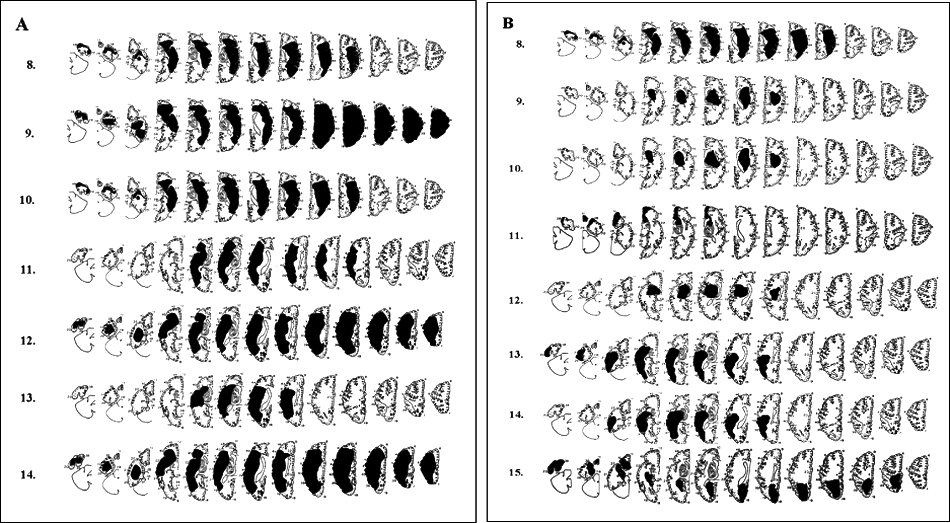
The figure depicts the graphical reconstruction of the lesion according to the Damasio and Damasio atlas25 for children with visual defects caused by congenital unilateral (A) and acquired (B) brain lesions.
Clinical Data
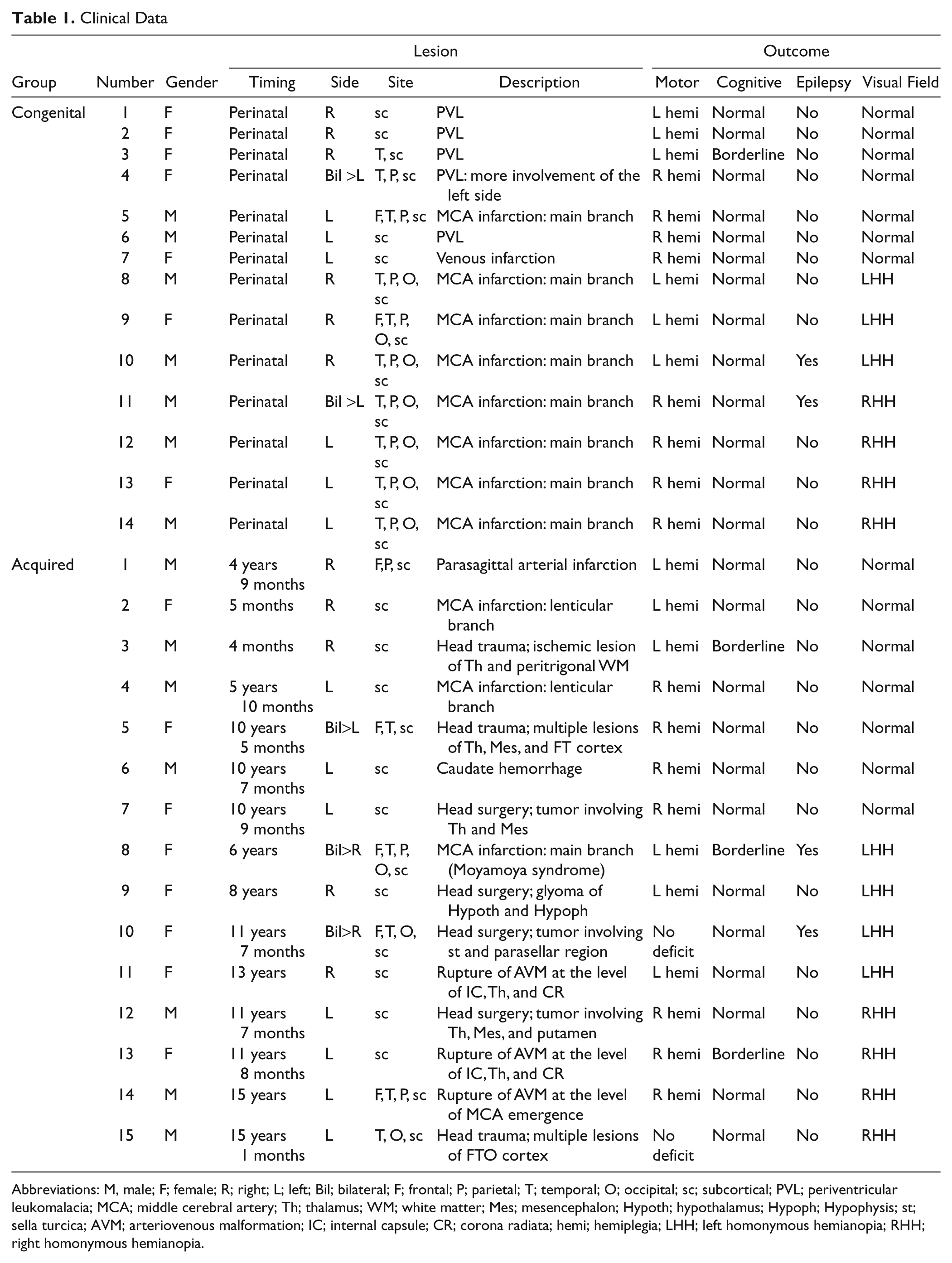
Abbreviations: M, male; F; female; R; right; L; left; Bil; bilateral; F; frontal; P; parietal; T; temporal; O; occipital; sc; subcortical; PVL; periventricular leukomalacia; MCA; middle cerebral artery; Th; thalamus; WM; white matter; Mes; mesencephalon; Hypoth; hypothalamus; Hypoph; Hypophysis; st; sella turcica; AVM; arteriovenous malformation; IC; internal capsule; CR; corona radiata; hemi; hemiplegia; LHH; left homonymous hemianopia; RHH; right homonymous hemianopia.
Visual Field
According to the results of the visual field test, patients were divided into 4 groups: C(NO-VFD), congenital lesions without visual field defect (7 patients); C(VFD), congenital lesions with visual field defect (7 patients: 3 left VFD and 4 right VFD); A(NO-VFD), acquired lesions without visual field defect (7 patients); and A(VFD), acquired lesions with visual field defect (8 patients: 4 left VFD and 4 right VFD). In Figure 2 (A and B), we show a graphic representation of the visual field assessment in children with visual field defects. The 2 groups of hemianopic patients were similar in terms of severity of the visual field defect: in fact, the results of a
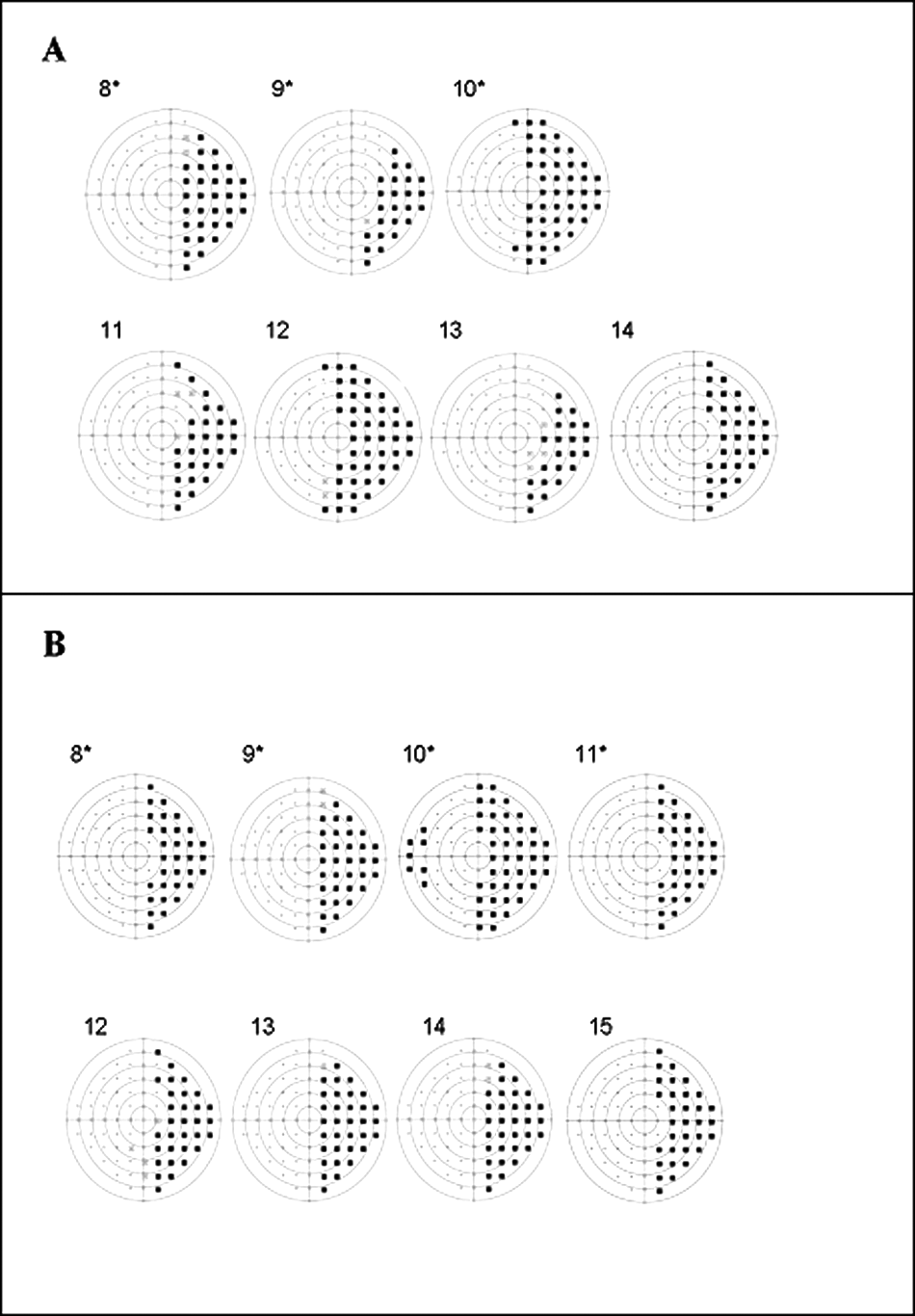
Visual field maps of the children with field defects: A. Children with congenital brain lesion. B. Children with acquired lesion. Maps of the children with left visual field defects were flipped horizontally to favor comparability and are indicated with an asterisk.
Visual Search
Responses were grouped, according to the side of the target, IL to the lesion or CL to the lesion (Table 2).
Mean Total Reaction Times and Percentage of Hits in the Ipsilesional and Contralesional Hemifields
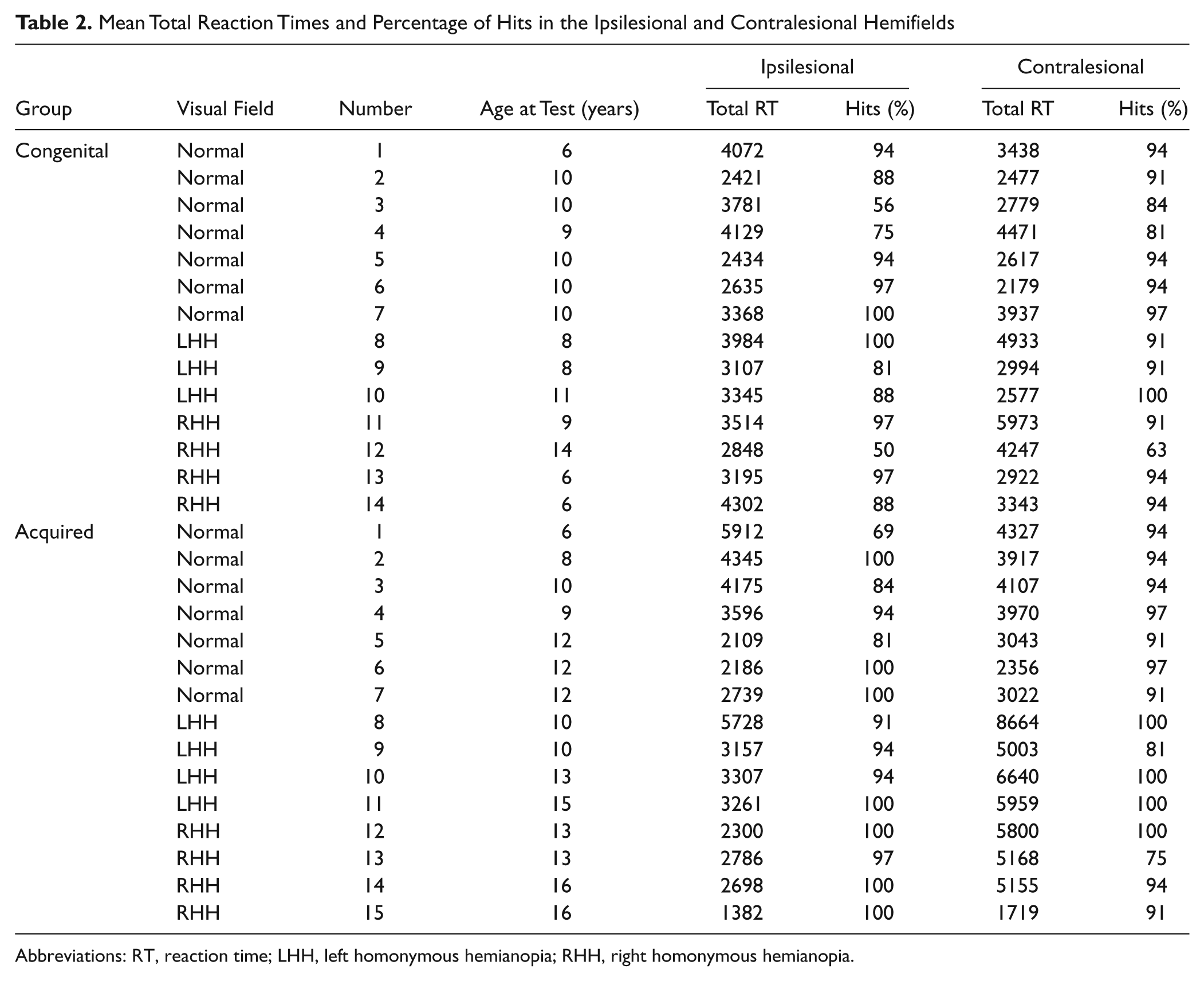
Abbreviations: RT, reaction time; LHH, left homonymous hemianopia; RHH, right homonymous hemianopia.
Percentage of Correct Responses
The 3-way ANOVA with Group as between factor and Test and Field of Visual Presentation as within factors revealed a significant effect of the main factor Test:
Reaction Times
The 3-way ANOVA on RTs with Group as between factor and Test and Field of Visual Presentation as within factors showed a significant effect of the main factor Test:
Moreover, a significant effect of the main factor Field of Visual Presentation—
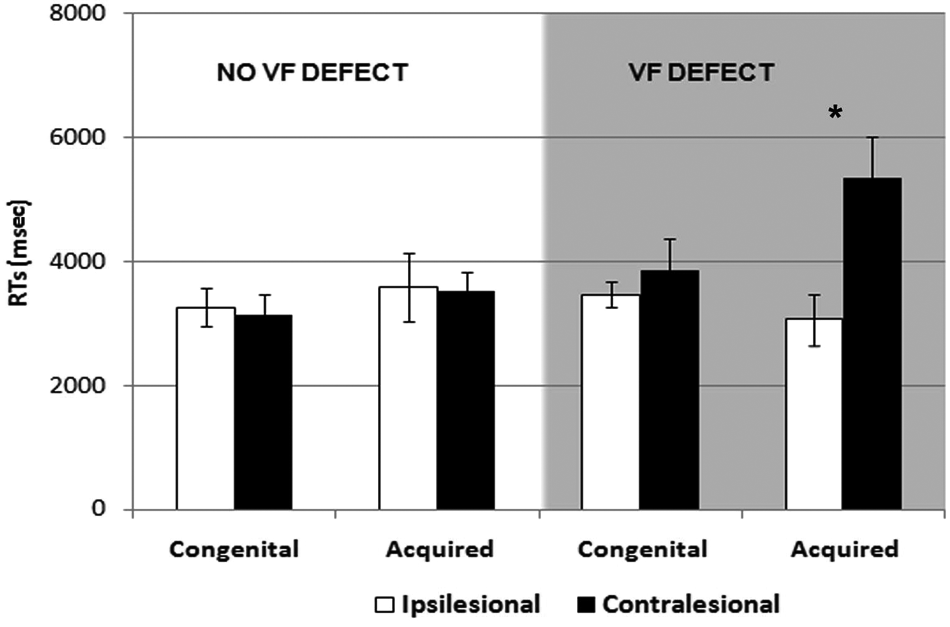
Mean and standard errors of visual exploration times (RTs) in children with congenital and acquired brain lesions, and with visual field defects (gray background) and without visual field defect (white background): white bars represent RTs in the ipsilesional field of presentation, whereas black bars represent RTs in the contralesional field
Discussion
Adult patients with hemianopia cannot properly process images in the blind hemifield, and they show frequent refixations and inaccurate saccades that result in unsystematic scanning, longer search times, and omission of relevant objects.6,9 This leads to significant difficulties in daily-life activities, such as driving, avoiding obstacles during walking, and reading.
The main result of our study is that children with congenital and acquired brain damage show different results on visual search tasks in the presence of a visual field deficit, supporting the idea of more effective compensation for the visual field defect after congenital brain damage. Children with acquired damage and visual field reduction show significantly longer RTs when the target is presented in the hemianopic field compared with the normal field. In contrast, children with congenital damage and visual field reduction do not show significantly different performances between the ispilesional and contralesional visual field. This striking result was corroborated by the finding that the performance of children with congenital lesions and visual field reduction is comparable with the performance of children without visual field restriction. The different patterns of results observed in patients with visual field defect with acquired and congenital lesions was not a result of differences in the severity of the visual field defect or in the extension of the lesions.
One possible interpretation of these findings is that the capacity for visual search might be differentially affected by the timing of insult, with more effective mechanisms of functional compensation observed when brain damage occurs at an earlier stage of development. This would be consistent with the general assumption of a more efficient reorganization following early brain damage, known as the Kennard principle, which has been extensively demonstrated for the visual system in the animal model, but less in humans. 14 In fact, studies on the reorganization of the human visual system following early brain damage are scarce. Recently, Batista and colleagues, 27 using PET, reported an increase in glucose metabolism in the visual cortex CL to the angioma in a group of children with Sturge-Weber syndrome. This finding was in particular present in the subgroup of children who showed hemianopia, suggesting that the increased metabolism had a compensating nature, although it was not sufficiently responsible for the sparing of conscious perception of the contralesional visual field. In analogy with animal studies, it might be hypothesized that the hypermetabolism observed in the intact visual cortex underlies vision-related functions such as visual orientation and visual search. Indeed, in hemidecorticated cats, CL cortical metabolism is largely preserved if the surgery is performed in young animals, whereas it is clearly decreased if the hemidecortication is performed at an older age.28,29 These findings are also in keeping with the increased glucose metabolism observed in human PET studies in remote regions after cortical injury. 30 Functional correlates of the observed metabolic increases, however, remain largely unknown.
Nevertheless, the neuroanatomical basis of functional reorganization in visual functions following early brain damage have been explored in animals, demonstrating a complex mechanism of plastic reorganization involving both cortical and subcortical structures. It is well known that cats with congenital unilateral ablation of the primary visual cortex show completely normal visually guided orienting behavior, whereas those with identical lesions acquired at later ages are severely impaired in the same task. 31 Anatomical, electrophysiological, and behavioral studies suggest that a major contribution to the sparing of visually guided behaviors is provided not only by the intact extrastriate cortical visual structures but also by the modifications in the circuitry of the SC, which provide good visual performance despite the massive retrograde transsynaptic degeneration of retinal ganglion cells and the absence of compensating cortical circuits. 14 In cats with unilateral lesions, the capacity to orient might also be augmented by additional direct excitatory crossed corticocollicular projections or by the release of inhibition from the intact hemisphere, which add to the excitatory boosts provided by IL corticocollicular expansions. 32 The expanded crossed corticocollicular pathway may be based on a relatively small pathway known to emerge from primary and higher visual regions in the intact brain. Alternatively, excitatory pathways from the intact hemisphere may expand their influence on the ipsilesional visuoparietal cortex and boost the signal transmitted onto the SC. These plastic mechanisms are not observed when brain damage is acquired in the adult stage.
The hypothesis that the retino-collicular-extrastriate pathway could at least partly account for functional compensation in visual functions in humans also is corroborated by the evidence that residual visual abilities in hemianopia are mediated by the retinotectal pathway. 33 In addition, it is very well known that the SC is a pivotal oculomotor structure and plays a crucial role in the execution and initiation of saccades, in target selection, and in visual search.34,35 Recent studies on adult hemianopic patients have exploited the ability of the SC to integrate stimuli from different sensory modalities and its role in oculomotor orienting behavior, demonstrating that a systematic audiovisual stimulation of the blind field can improve accuracy, search times, and oculomotor patterns in visual exploration.36,37
Our finding of a more effective visual search strategy in children with congenital brain damage as compared with that in children with acquired damage is in line with animal models and suggests the existence, in the case of early lesions, of a spontaneous functional reorganization by means of subcortical structures involving the SC. In contrast, in the case of acquired lesions, spontaneous visual compensation is not so effective. Unfortunately, we have no patient with early complete hemianopia and damage between birth and 72 months, so we cannot say at what time in development the efficacy of neuroplasticity decreases and whether this phenomenon is gradual or abrupt. The findings may contribute to strategies to improve vision.38,39
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:The research was supported by Mariani Foundation Grant 2006 and by PRIN 2007 from the Italian Ministry of University and Research to Giovanni Cioni, by PRIN 2007 Ser.In.Ar. to Elisabetta Ladavas, and by European Research Council advanced grant 229445 “STANIB” to Francesca Tinelli.
