Abstract
Background. Enriched rehabilitation (ER; environmental enrichment plus skilled reaching) improves recovery after middle cerebral artery occlusion (MCAo) in rats. Fundamental issues such as whether ER is effective in other models, optimal rehabilitation intensity, and underlying recovery mechanisms have not been fully assessed. Objective. The authors tested whether the efficacy of ER varies with ischemia model and assessed the importance of rehabilitation intensity and brain-derived neurotrophic factor (BDNF) in recovery. Methods. Rats in experiment 1 received 8 weeks of ER or remained in standard housing. Functional outcome was assessed with the staircase and cylinder tasks. Surprisingly, ER provided no functional benefit in any model. In this experiment, ER was delivered during the light phase, whereas other studies delivered ER in the dark phase of the light cycle. It was hypothesized that in the light, rats engaged in less rehabilitation or alternatively that BDNF was lower. Experiment 2 tested these hypotheses. Following MCAo, rats received ER in either the light or dark phase of the light cycle. Functional outcome was assessed and BDNF levels were measured in the motor cortex and hippocampus. Results. Recovery was accompanied by increased BDNF. This occurred only in rats that received ER in the dark and these animals reached more than those in the light condition. Conclusions. Data suggest that there is a critical threshold of rehabilitation, below which recovery will not occur, and that BDNF mediates functional recovery. The use of intensive rehabilitation therapies for stroke patients is strongly supported.
Keywords
Introduction
Stroke is a leading cause of mortality and disability, and many survivors never regain functional independence. 1 Although rehabilitation promotes motor recovery in many patients, there remains limited or conflicting evidence for best practice in many elements of stroke rehabilitation. 2 Animal studies have furthered our understanding of stroke pathology and mechanisms of recovery. 3 For example, enriched rehabilitation (ER), a coupling of environmental enrichment and daily reach training, improves forelimb use following middle cerebral artery occlusion (MCAo) in rats. 4 Greatest benefit occurs when ER begins within the first week after ischemia, supporting the idea of a critical therapeutic window. 5 Indeed, clinical data suggest that earlier rehabilitation is associated with greater functional improvement.6-8
Other fundamental parameters, including optimal rehabilitation duration and intensity are yet unknown. Retrospective studies suggest dose-dependent rehabilitation effects, with accelerated9,10 or improved outcome8,11,12 after more intense therapy. It is also possible that a critical threshold of rehabilitation intensity is needed to obtain functional benefit. A computational model suggests that poststroke therapeutic arm use below a “certain threshold” is ineffective.13,14 However, clinical and animal studies are yet to test this threshold hypothesis.
In addition to optimizing treatments, stroke studies aim to identify and enhance neuroplastic changes mediating functional benefit. Recovery of forelimb function is associated with increased dendritic length and complexity, 4 cortical motor map reorganization, 15 and upregulation of plasticity-associated growth factors. 16 Notably, administering brain-derived neurotrophic factor (BDNF) improves sensorimotor recovery following ischemia in rats,17-19 whereas blocking endogenous BDNF with antisense oligonucleotide completely prevents rehabilitation-induced skilled reaching recovery. 20 Similarly, humans with the val66met polymorphism of the BDNF gene display reduced cortical map reorganization after motor training, supporting the role of BDNF in activity-dependent reorganization. 21 BDNF levels may vary with rats’ circadian rhythm, with highest levels occurring at the beginning of the dark phase. 22 Thus, depending on when therapy is administered, diurnal BDNF production could influence the efficacy of poststroke rehabilitation.
In experiment 1, we used several rodent ischemia models to determine whether ER is effective regardless of injury location and severity, an essential step in translational research. 23 In experiment 2, we tested the competing hypotheses that (a) a critical threshold of rehabilitation intensity must be reached to obtain functional benefit or (b) recovery does not depend on the intensity of rehabilitation per se, but occurs only when ER is paired with diurnal peaks of BDNF.
Materials and Methods
The efficacy of ER was tested in 4 models of focal ischemia in experiment 1. All procedures were done during the light phase (lights on 8:00
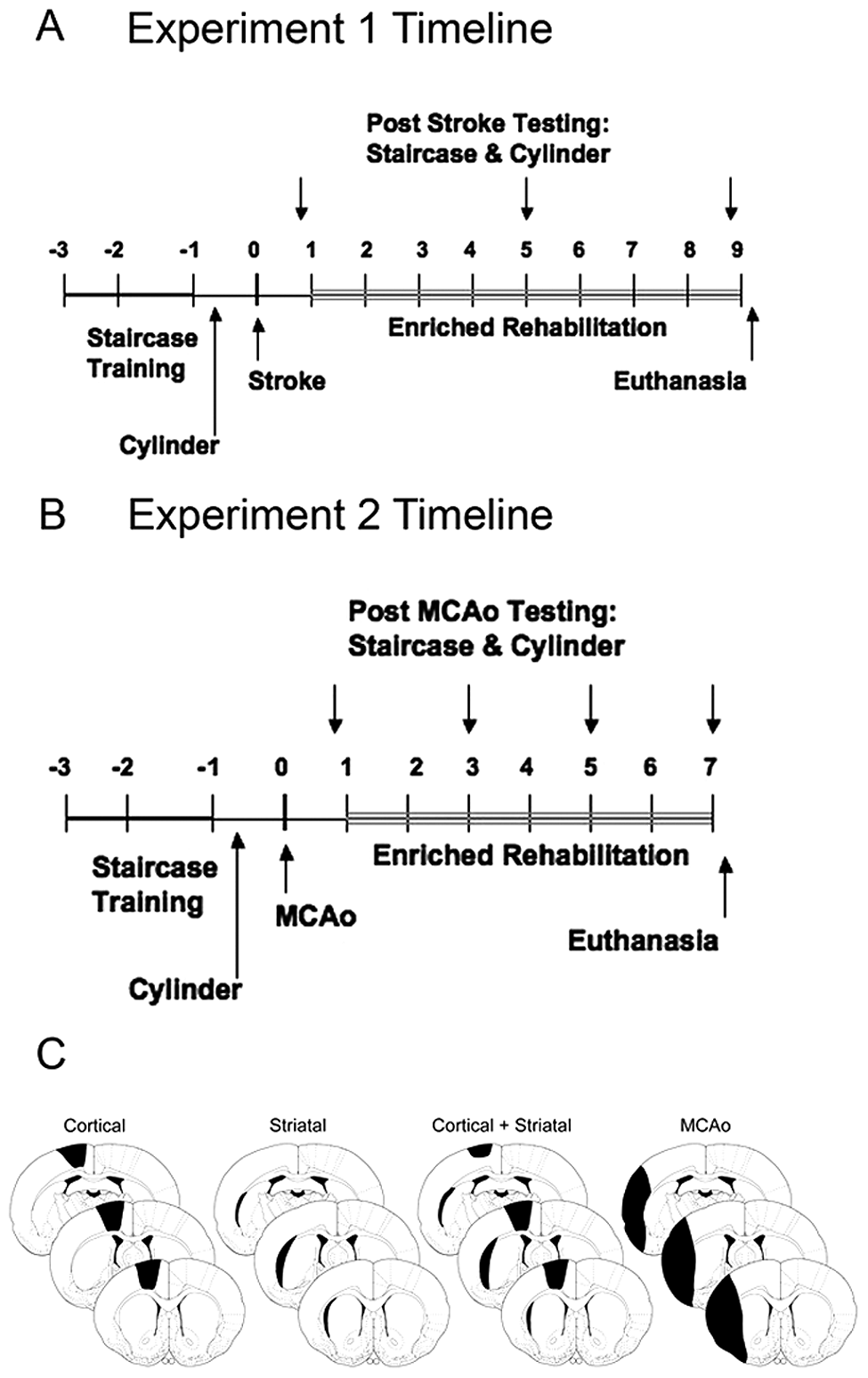
Timeline of experimental procedures
Experiment 2 determined why ER failed to provide benefit in experiment 1. We hypothesized that nocturnal rats would be more active during the dark phase and engage in more rehabilitation. This hypothesis suggests that there is a critical threshold of ER intensity, below which no benefit occurs (threshold hypothesis). Alternatively, BDNF levels, which promote recovery, may vary with the light cycle 22 and may be higher in the dark (BDNF hypothesis). In experiment 2, all rats were subjected to MCAo. This was chosen as our previous work on rehabilitation and BDNF was conducted using this model. Starting 7 days later, some received 6 weeks of ER during the light phase (Light ER) or remained in standard housing (Light STD). Others received ER during the dark. For some, reaching was limited such that it was equivalent to that of rats reaching in the light (Dark ER Lim) whereas others were allowed unlimited access to pellets during ER (Dark ER Unlim). Control rats remained in standard housing (Dark STD). Functional outcome was measured using several tests, and tissue loss and BDNF levels were measured at 7 weeks (Figure 1B).
Subjects
A total of 104 male Sprague–Dawley rats (Charles River Laboratories, Montréal, Québec, Canada) weighing ~200 to 225 g were used (experiment 1, n = 65; experiment 2, n = 39). Unless otherwise specified, rats were housed in groups of 2 or 3 with food and water provided ad libitum. Animals were handled for several days prior to experimentation, and all procedures were approved by the Memorial University Animal Care Committee and conformed to the Canadian Council on Animal Care guidelines. All procedures were done by experimenters blind to group identity.
Behavioral Training
Staircase task
Rats were trained to retrieve food reward pellets in the staircase reaching task as previously described.20,24 For 10 days, rats received 15-minute training sessions twice daily, separated by ~5 hours. To ensure rats had reached a plateau in performance such that no further increase in performance was likely, rats had to retrieve ≥12 pellets out of a possible 21 with either forelimb (most retrieved 17-20 pellets) over the last 8 trials. 25
Cylinder test of forelimb use asymmetry
Three to 5 days before ischemia, animals were placed in a Plexiglas cylinder and the number of independent (ipsilateral or contralateral to ischemia) or bilateral touches was recorded. Contralateral forelimb use was calculated as follows: ([contralateral contacts + 1/2 bilateral contacts]/total contacts) × 100%.26,27
Focal Ischemia
Surgery was performed aseptically. Focal ischemia was created by injecting endothelin-1 (ET-1, 400 pmol/µL; CalBiochem, La Jolla, CA) into the hemisphere opposite the preferred paw as determined in the staircase task. For each model, stereotaxic coordinates (relative to Bregma) 28 and volume of ET-1 injected are listed in Table 1. ET-1 infusions were at a rate of 1 µL over 2 minutes, and the needle remained in place for 3 minutes following infusion to minimize backflow. 25 Body temperature was maintained at ~37°C.
The Infusion Coordinates (Relative to Bregma) and Volume of ET-1 Injected at Each Location for Each Focal Ischemia Model a
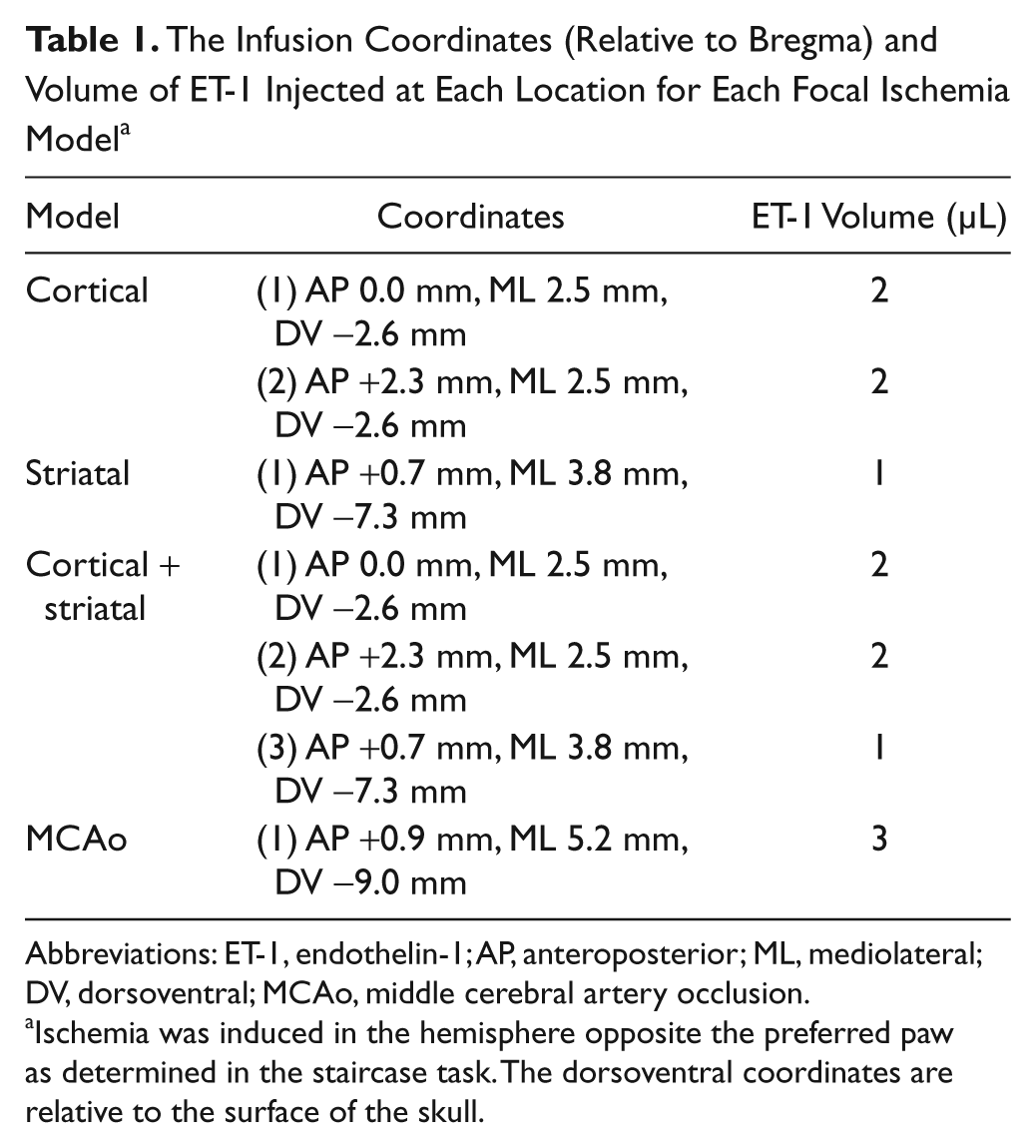
Abbreviations: ET-1, endothelin-1; AP, anteroposterior; ML, mediolateral; DV, dorsoventral; MCAo, middle cerebral artery occlusion.
Ischemia was induced in the hemisphere opposite the preferred paw as determined in the staircase task. The dorsoventral coordinates are relative to the surface of the skull.
Enriched Rehabilitation and Experimental Conditions
Rats were tested in the staircase (4 trials over 2 days) and cylinder (1 session) tasks 5 and 6 days after ischemia. To ensure that postischemia reaching deficits were similar among groups and that differences in performance could be attributed to treatment intervention, pairs of rats were pseudo-randomized into treatment condition as described below.
Rats in experiment 1 were housed on a standard light cycle in groups of 2 or 3 in standard housing (STD; n = 8 to 9 per model) or received ER (n = 8 to 9 per model). This consisted of living in groups of 4 to 6 in large multilevel cages containing tubes, ramps, and toys. In addition to environmental enrichment, rats received skilled reaching training (6 h/d, 5 d/wk for 8 weeks) using a modified staircase apparatus as previously described.4,5
In experiment 2, some rats were housed on a standard light cycle in groups of 2 or 3 in standard housing (Light STD; n = 7) or received ER with skilled reaching during the light phase as described above with the exception that reaching lasted 4 h/d for 6 weeks (Light ER; n = 8). Others were housed on a reverse light cycle. Control rats remained in groups of 2 or 3 in standard cages (Dark STD; n = 7), whereas others received reaching rehabilitation during the dark phase. To control for the possibility that rats engage in less rehabilitation in the light compared to the dark, one group of ER rats (Dark ER Lim, n = 8) was given limited access to pellets so they retrieved the same amount of pellets as the Light ER rats. These rats had access to the reaching apparatus for the entire 4-hour rehabilitation period, but the reward pellets were removed from the apparatus after ~2 to 3 hours to limit the number obtained. Others (Dark ER Unlim, n = 9) were allowed unlimited access to the reaching apparatus for the 4-hour reaching rehabilitation session.
Behavioral Testing
Functional recovery was assessed in the staircase and cylinder tasks as previously described 5 and 9 weeks after ischemia in experiment 1, and at weeks 3, 5, and 7 in experiment 2.
Volume of Tissue Lost
Rats were sacrificed 1 to 2 days following the last functional assessment. In experiment 1, anesthetized rats were transcardially perfused with saline followed by 4% paraformaldehyde. Brains were frozen and coronal sections (40 µm) taken every 640 µm were stained with cresyl violet. Using Image J software (Scion Corporation, Frederick, MN), the volume of tissue lost was calculated from sections encompassing the injury as routinely done 29 :
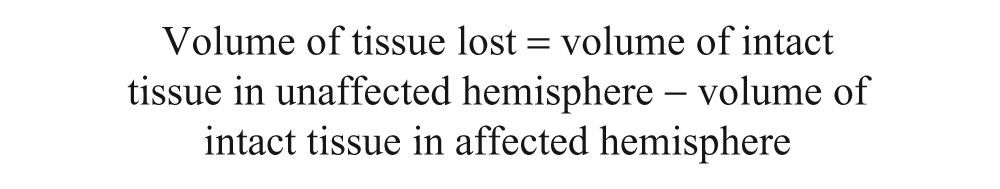

In experiment 2, rats were briefly anesthetized and decapitated. Brains were quickly removed and separated into hemispheres. Tissue from the contralesional hemisphere was used for BDNF assay (described below). The ipsilesional hemisphere was processed for lesion volume analysis as above. The volume of this hemisphere was estimated using sections from age-matched control tissue. This enabled calculation of the volume of tissue loss in this experiment, which could be compared with experiment 1.
Assay for Brain-Derived Neurotrophic Factor
Following decapitation, portions of the contralesional forelimb motor cortex and hippocampus were extracted, weighed, frozen in liquid nitrogen, and stored at −80°C. Using enzyme-linked immunosorbent assay (ELISA), duplicate samples were analyzed for BDNF (Chemikine BDNF Sandwich ELISA KIT, Millipore, Temecula, CA) following the manufacturer’s protocol. Concentrations of BDNF (pg/mL) were extrapolated from a standard curve.
Statistics
All analyses were done using SPSS version 13.0 (SPSS Inc, Chicago, IL). Data are presented as mean ± standard error of the mean (SEM). Behavioral data were analyzed using a 2-way repeated-measures analysis of variance (ANOVA). Subsequent 1-way ANOVAs were conduced following a significant interaction. The volume of tissue lost and BDNF protein levels were analyzed using 1-way ANOVA. Fisher’s post hoc tests and specific Huynh–Feldt (multivariate statistics) or Games–Howell (univariate statistics) corrections for unequal variances were used as appropriate. A P value ≤ .05 was considered statistically significant.
Results
Experiment 1
Enriched rehabilitation
The number of food pellets obtained during rehabilitation was similar among models (overall mean = 139 ± 9.78 pellets/d; P = .864; Figure 2A).
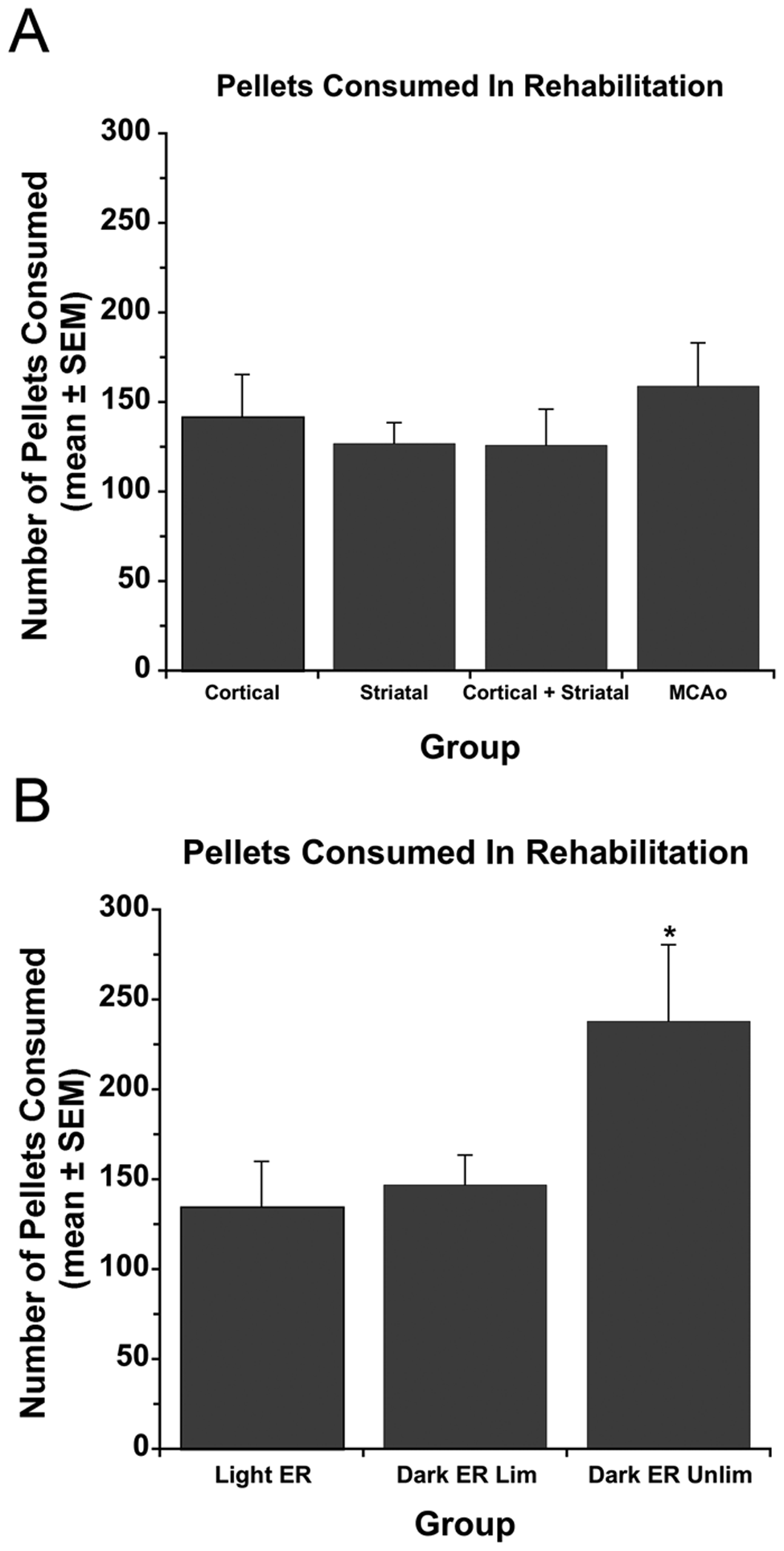
Food reward pellets retrieved during skilled reaching rehabilitation
Staircase task
For each model, groups performed similarly at baseline (overall mean = 17.32 ± 0.60 pellets; P ≥ .129). Similarly, ischemia caused impairments in all groups as they obtained fewer pellets on days 5/6 postischemia (P ≤ .014 vs baseline).
Following injury to the forelimb motor cortex (cortical model), a Time effect (P = .003) indicated that rats retrieved more pellets at week 5 compared with days 5/6 (P = .004; Figure 3A). There was no Rehabilitation effect (P = .743) or Time × Rehabilitation interaction (P = .950). In the striatal model, there were no Time (P = .587), Rehabilitation (P = .643), or Time × Rehabilitation interaction effects (P = .644; Figure 3B). After combined injury to forelimb motor cortex and striatum (cortical + striatal model), there was a Time effect (P = .008) because rats obtained more pellets at week 5 (P = .004 vs days 5/6). However, there was no Rehabilitation effect (P = .443) or Time × Rehabilitation interaction (P = .775; Figure 3C). Finally, following MCAo, a Time effect (P < .001) proved that recovery occurred over 9 weeks (P ≤ .021). There was no Rehabilitation effect (P = .676) or Time × Rehabilitation interaction (P = .832; Figure 3D).
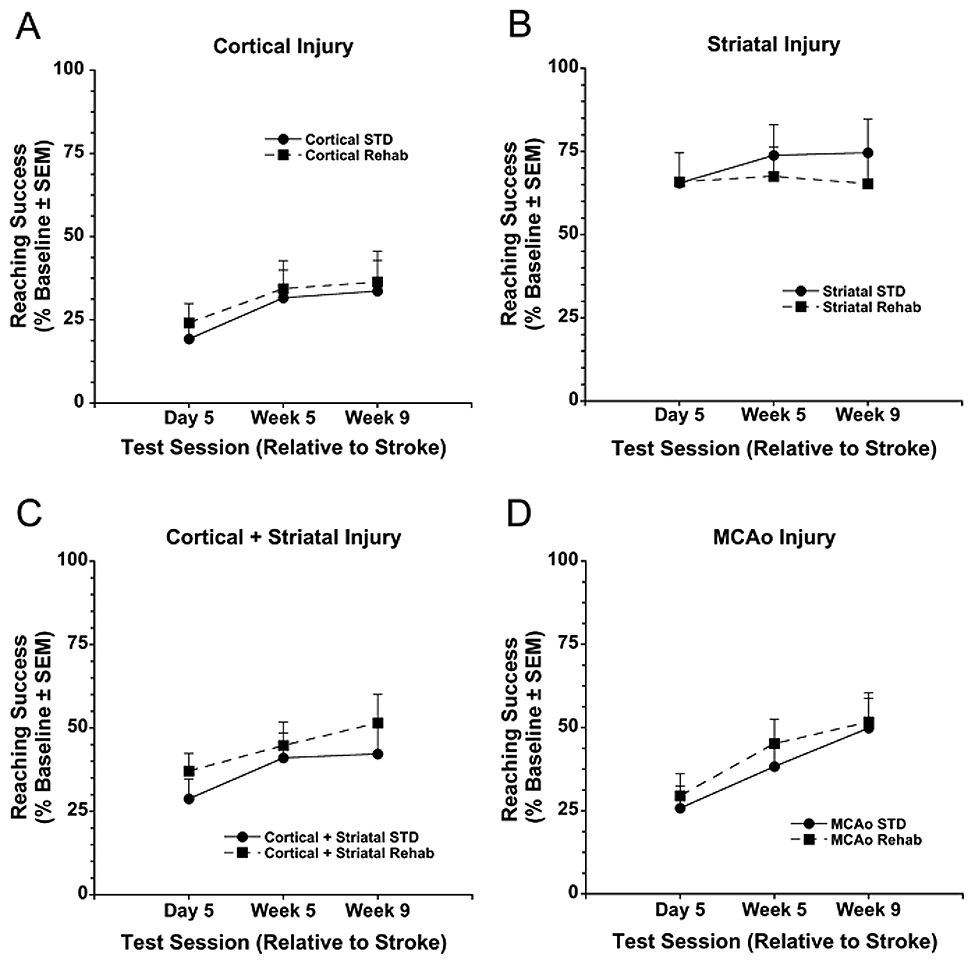
Contralateral forelimb reaching success (expressed as a percentage of baseline performance) in the staircase task in experiment 1
Cylinder task
All rats used the contralateral forelimb less during exploration after ischemia (P ≤ .001 vs baseline). The MCAo group recovered over time (days 5/6 vs week 5; P = .027; Figure 4D), whereas the cortical (Figure 4A) and cortical + striatal (Figure 4C) groups did not (P ≥ .221). There were no Rehabilitation effects (P ≥ .617) or Time × Rehabilitation interactions (P ≥ .160) in these models. Following striatal injury, there was no effect of Time (P = .054), Rehabilitation (P = .960), and Time × Rehabilitation interaction (P = .284; Figure 4B).
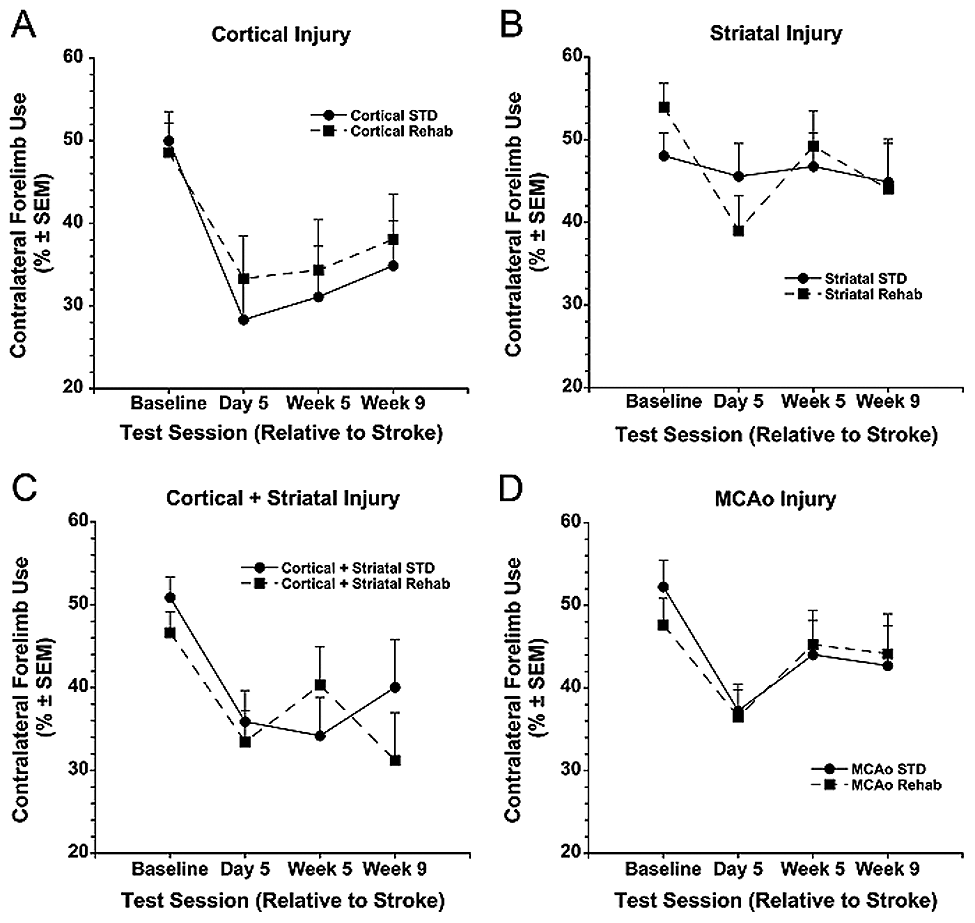
Spontaneous contralateral forelimb use in the cylinder task in experiment 1
Tissue injury
Enriched rehabilitation did not affect tissue loss in the cortical (overall mean = 58.41 ± 11.31 mm3; P = .393), striatal (overall mean = 26.03 ± 10.50 mm3; P = .721), cortical + striatal (overall mean = 37.79 ± 6.88 mm3; P = .393), or MCAo models (overall mean = 89.03 ± 11.846 mm3; P = .734). Representative illustrations of injury are depicted in Figure 1C.
Experiment 2
The Light STD and ER groups were included in experiment 2 to replicate our experiment 1 finding that ER administered in the light does not facilitate recovery and to serve as controls for the Dark groups. In both experiments, ER in the light failed to provide benefit on any outcome measure. Although the Light groups were included in all analyses and data are reported below, these groups were not included in most of the figures to simplify the graphs and highlight results from the groups of interest (Dark groups).
Enriched rehabilitation
Reaching rehabilitation in the Dark ER Lim group was restricted such that the number of pellets obtained by these rats was similar to the Light ER group (P = .768; Figure 2B). Reaching was not limited in the Dark ER Unlim group and these rats obtained more pellets than the Light ER group (P = .027). Although nonsignificant, there was a trend for the Dark ER Unlim group to obtain more pellets than the Dark ER Lim group (P = .052).
Staircase task
Groups performed similarly prior to MCAo (overall mean = 17.15 ± 0.64 pellets; P = .455), and were impaired afterward (to ~30% of baseline; P = .001). There was a Rehabilitation effect (P = .016) and a Time × Rehabilitation interaction (P = .025), but no Time effect (P = .565). Subsequent 1-way ANOVAs at each time revealed that all groups performed similarly 5/6 days post-MCAo (P = .931), and that group differences occurred at week 3 (P = .020), week 5 (P = .014), and week 7 (P = .004). Notably, at week 3, the Dark ER Unlim group obtained more pellets than Light ER, Dark STD, and Dark ER Lim groups (P ≤ .013; Figure 5A). Results were similar for the other times; however, by week 7, the Dark ER Unlim group was different from the Light ER (P < .001) and Dark ER Lim groups (P = .017) but not the Dark STD group (P = .103).
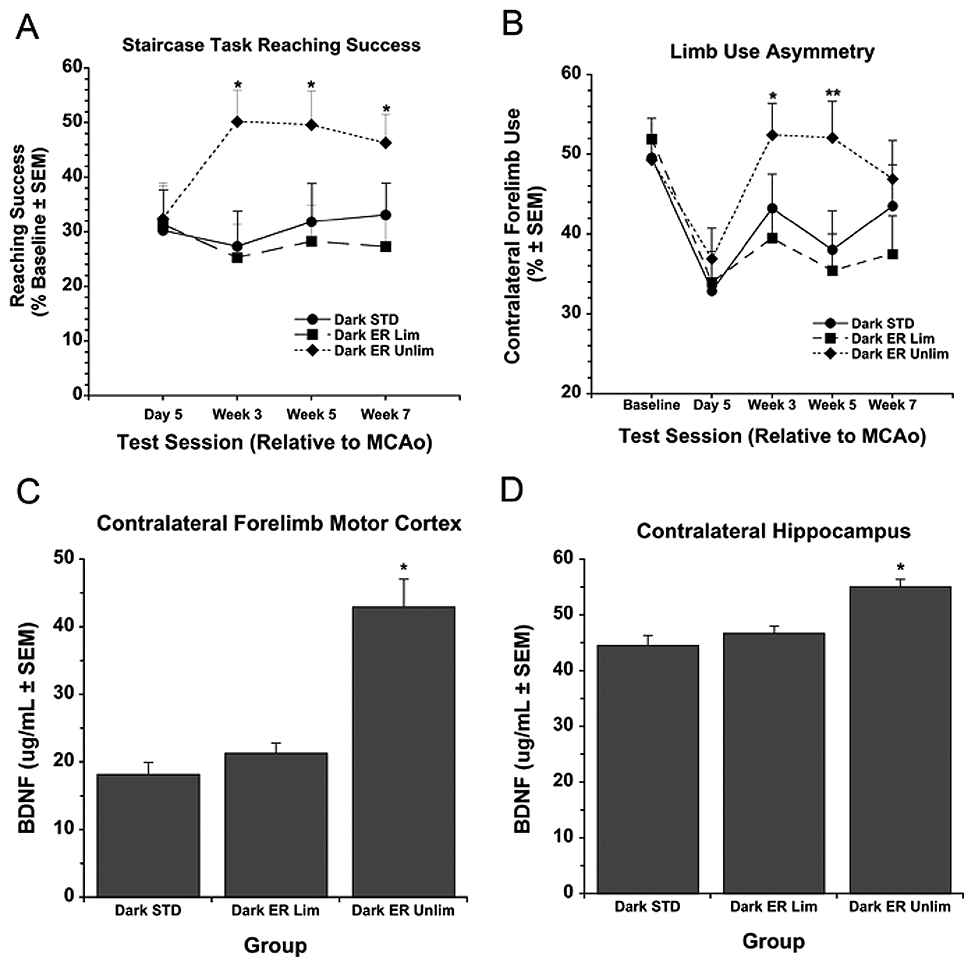
(A) Contralateral forelimb reaching success (percentage of baseline performance) in the staircase task in experiment 2. The Dark enriched rehabilitation (ER) Unlim group retrieved significantly more pellets than all other groups at weeks 3 and 5. By week 7, the Dark ER Unlim group retrieved more pellets than all groups except the Dark ER Lim. (B) Spontaneous contralateral forelimb use in the cylinder task in experiment 2. The Dark ER Unlim group used the contralateral forelimb significantly more than the Light STD, Light ER, and Dark ER Lim groups at week 3, and more than the Light STD, Dark STD and Dark ER Lim groups at week 5. (C) Brain-derived neurotrophic factor (BDNF) levels measured with enzyme-linked immunosorbent assay (ELISA) in experiment 2. The Dark ER Unlim group had significantly elevated BDNF levels (vs all other groups) in the forelimb motor cortex. (D) In the hippocampus, BDNF levels in the Dark ER Unlim group were significantly higher than the Dark STD, Dark ER Lim, and Light STD groups.
Cylinder test
One rat was excluded from this analysis because of being an outlier (>2 standard deviations from the mean) on days 5/6 post-MCAo. This rat did not display an asymmetry following MCAo but was severely impaired at subsequent times. An initial lack of asymmetry followed by severe “emerging deficits” occasionally follows severe injury, 30 which was confirmed histologically. A Time main effect (P < .001) revealed that compared with baseline (overall mean = 50.14% ± 1.20%), all groups used their contralateral forelimb less 5/6 days post-MCAo (overall mean = 34.41% ± 1.68%; P ≤ .032; Figure 5B). Although no main effect of Rehabilitation occurred (P = .076), there was a Time × Rehabilitation interaction (P = .018). Subsequent 1-way ANOVAs revealed group differences on week 3 (P = .030) and week 5 (P = .007) but not week 7 (P = .285). Notably, by week 5, the Dark ER Unlim group used the contralateral forelimb more than the Light STD, Dark STD, and Dark ER Lim groups (P ≤ .044).
Volume of tissue lost
The volume of tissue lost was similar among groups (overall mean = 74.96 ± 12.34 mm3; P = .801; Figure 1C).
Brain-derived neurotrophic factor assay
Contralesional forelimb motor cortex BDNF levels differed between groups (P < .001). Notably, BDNF levels in the Dark ER Unlim group were higher than all other groups (P ≤ .001), which were not different (P ≥ .546; Figure 5C). Similarly, analysis of contralesional hippocampal BDNF levels revealed a Group effect (P < .023). Subsequent post hoc tests revealed that the Dark ER Unlim group had higher BDNF levels than Dark ER Lim, Dark STD, and Light STD groups (P ≤ .025; Figure 5D).
Discussion
Skilled reaching during the light phase did not improve poststroke recovery in these experiments, despite testing different injury locations and severities. These results contrast with studies demonstrating substantial benefit of poststroke rehabilitation in rats using skilled reaching,4,5,31,32 running,33-35 and running/reaching combinations.20,36 In these studies, rehabilitation was administered during the rats’ dark phase, whereas rehabilitation was administered in the light phase in experiment 1. We predicted that because rats are nocturnal, they are more active in the dark and engage in more rehabilitation. Thus, we theorized that functional benefit occurs only if a threshold of rehabilitation intensity is achieved. In confirmation of this hypothesis, skilled reaching improved only in rats with unlimited access to the reaching apparatus while in the dark (Dark ER Unlim; experiment 2). These rats retrieved more pellets (~240 pellets/d) than those in the light or when reaching in the dark was restricted (~140 pellets/d). Despite retrieving many pellets, ER did not benefit the latter groups. Thus, we are the first to show that a critical threshold of rehabilitation intensity is required to obtain functional benefit.
Although clinical studies suggest that intensity is key to stroke rehabilitation efficacy, 12 no study has systematically assessed whether a critical threshold of rehabilitation intensity must be reached to obtain benefit. A computational model of bilateral hand use following stroke proposes that patients will improve only if they exceed a particular threshold of arm use. If the threshold is not reached, recovery of the affected arm will not occur, and patients instead develop compensatory movements with the less affected arm.13,14 Clinical data strongly suggest that intensive therapy is most effective, but that implementing intensive rehabilitation programs is challenging given the cost of therapy and the low caregiver-to-patient ratio. Recent studies suggest that the amount of task-specific training in stroke rehabilitation is much less than in animal models and likely insufficient to promote reorganization required for optimal recovery.37,38 Importantly, a recent proof-of-concept study demonstrated the feasibility of delivering hundreds of repetitions (>300) of upper extremity task-specific therapy in standard 1-hour rehabilitation sessions and that some stroke patients may benefit from therapy. 39
Besides increasing rehabilitation intensity, one may prolong treatment duration. However, data from our laboratory suggest that extending ER during the light phase for an additional 4 weeks provides no added benefit (unpublished data). Similarly, rats that benefit from early ER do not further improve with subsequent periodic returns to therapy. 31 These “tune ups” also fail to affect dendritic complexity, which is a mediator of poststroke brain plasticity and recovery. 4 Benefit may have obtained in experiment 1 had we administered combination therapies, such as running and reaching, or rehabilitation programs that become more challenging each week.20,36 Although the optimal composition of rehabilitation is unknown, paradigms such as these may be more effective simply because they are more intense than the ER paradigm presently used.
In addition to assessing the impact of rehabilitation intensity, we tested whether reaching must be paired with peaks of BDNF that occur at the beginning of the dark phase.22,40 Unexpectedly, we could not confirm that BDNF varies diurnally, as levels in the early part of the light and dark phases were identical. Others have similarly failed to show diurnal BDNF changes in rats. 41 Variations in measurement techniques may explain contradictory findings. For example, we measured protein levels directly using ELISA whereas most studies measured mRNA. Furthermore, the timing or degree of mRNA translation into protein may vary in different brain regions. 42 Finally, whereas most studies measure BDNF in healthy, intact animals, ours were done following ischemia and BDNF increases following various brain insults, including ischemia. 43 Thus, given that all rats were subjected to MCAo in experiment 2, any circadian changes in expression were likely masked by injury-induced elevations in BDNF.
A limitation of this work is that multiple forms of BDNF (eg, pro vs mature BDNF) or the TrkB receptors were not assayed. Furthermore, BDNF was assayed only in the contralateral hemisphere. The ipsilateral hemisphere was used for lesion volume analysis to ensure that any differences in outcome were not due to injury severity. It is possible that BDNF levels may have been different in other regions such as the peri-infarct cortex or in plasma. Nonetheless, we found increased BDNF levels only in rats that received unlimited reach training in the dark (Dark ER Unlim group). Notably, only these rats displayed improved contralateral forelimb performance in the staircase and cylinder tasks, providing further support for the importance of BDNF in rehabilitation-induced recovery following stroke. Regardless of whether rats received rehabilitation in the dark or light, BDNF levels were identical in all other groups, and no functional benefit occurred. It is logical to predict that a greater effect occurs in the forelimb motor cortex compared with the hippocampus (effects were similar, but not as pronounced) as changes in the cortical representation of the forelimb likely mediate recovery in these tasks.15,44 Furthermore, our data support the view that rehabilitation should be task specific, such that recovery of fine digit use occurs with appropriate skilled reaching therapy.45,46
The current findings complement growing evidence that BDNF mediates poststroke brain plasticity and behavioral recovery. For instance, intravenous BDNF administration improves sensorimotor function following photothrombotic ischemia in rats.17-19 In addition, we showed that a graduated rehabilitation program consisting of wheel running and skilled reaching training significantly improved forelimb use in the staircase task. However, when endogenous BDNF was blocked with antisense oligonucleotide, beneficial effects of rehabilitation were negated, suggesting a critical role for BDNF in rehabilitation-induced reaching recovery. 20 Nevertheless, given that BDNF is released from glutamatergic neurons in a use-dependent fashion,47,48 one might argue that elevated BDNF in the Dark ER Unlim group is simply a by-product of increased physical activity during ER and does not mediate recovery per se. This is unlikely, however, because BDNF did not correlate with reaching activity in the ER groups. Furthermore, with the exception of the Dark ER Unlim group, BDNF levels were similar among all other groups despite some receiving ER and others remaining in STD conditions. Thus, it does not appear that physical activity alone increases BDNF. There is also no evidence that increased BDNF alone is enough to improve reaching ability. Although Schabitz et al17,18 and Muller et al 19 elegantly demonstrated that BDNF administration improves sensorimotor function, skilled reaching, which is notoriously difficult to treat, was not assessed. We suggest that both rehabilitative therapy and experience-dependent BDNF upregulation are required for poststroke skilled reaching recovery.
In summary, we are the first to show that a critical threshold of rehabilitation intensity must be reached to obtain benefit following MCAo in rats. These data support the development and use of intense task-specific rehabilitation paradigms, and illustrate the necessity of identifying this threshold in stroke patients. Furthermore, recovery occurred only in rats with elevated BDNF, supporting its important role in poststroke recovery. Additional research must identify the mechanisms by which BDNF promotes brain plasticity so that it can be manipulated (eg, pharmacological administration) to promote more complete recovery in patients. Understanding the nature of the interaction between rehabilitation intensity and BDNF, and how factors such as age, type of stroke, and comorbid conditions influence it, may offer insight into novel therapies for improving functional outcome in stroke patients.
Footnotes
Acknowledgements
The authors thank Drs Fred Colbourne and Kris Langdon for helpful comments on the article.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
This work was supported by a grant (to D. Corbett) from the Canadian Institutes for Health Research (CIHR). C. L. MacLellan was supported by a CIHR postdoctoral fellowship. M. Keough was supported by a Focus on Stroke summer scholarship from the Canadian Stroke Network. D. Corbett holds a Canada Research Chair in Stroke and Neuroplasticity.
