Abstract
Background
Stroke is a leading cause of disability worldwide. Focused training of the impaired limb has been shown to improve its functional outcome in animal models. However, most human stroke survivors exhibit persistent motor deficits, likely due to differences in rehabilitation intensity between experimental (animal) and clinical (human) settings.
Objective
The current study investigated the effect of training intensity on behavioral outcome in a mouse model of stroke.
Methods
Mice were trained preoperatively on a skilled reaching task. After training, mice received a unilateral photothrombotic stroke. Postoperatively, animals received either daily rehabilitative training (traditional intensity), intermittent rehabilitative training (every other day), or no rehabilitative training (control). Assessment of the impaired limb occurred after 14 training sessions (14 days for the Traditional group; 28 days for the Intermittent group).
Results
Assessment of the impaired limb illustrated that traditional, daily training resulted in significantly better performance than no training, while intermittent training offered moderate performance gains. Mice receiving intermittent training performed significantly better than control mice but did not exhibit reaching performance as strong as that of animals trained daily.
Conclusions
The intensity of rehabilitation is important for optimal recovery. Although intermediate intensity offers some benefit, it is not intensive enough to mimic the performance gains traditionally observed in animal models. These results suggest that intensive training, which is often unavailable for human stroke survivors, is necessary to achieve an optimal functional outcome. The lower bounds of training intensity for functional benefit still need to be determined.
Introduction
Approximately 795 000 Americans each year suffer a stroke. 1 With advances in medical technologies, only 10% to 20% of strokes are fatal; however, survivors often face long-term impairment, 2 making stroke the leading cause of long-term disability in the United States. Approximately 50% of stroke survivors will suffer from chronic, moderate to severe loss of upper limb function. 3 While larger strokes can have a variety of long-term effects, the most common chronic deficits following stroke affect fine motor control. For instance, patients often have difficulty using their wrist, hand, and fingers on their impaired side. 3 These motor deficits impede on a person’s independent daily living and contribute to the rising cost of health care for stroke survivors. The United States spends an estimated $45.5 billion on care and treatment for stroke patients annually. 1 It is therefore imperative that we identify improved rehabilitative strategies to promote greater functional independence and overall functional outcome.
After stroke, the brain undergoes restorative plastic changes that result in minimal behavioral recovery, known as spontaneous recovery. These improvements occur within the first 6 months after stroke and generally involve gross motor function, as fine motor function is significantly more difficult to recover. 3 Therefore, scientific research tends to focus on fine motor control recovery, both to minimize the confounding effects of spontaneous recovery and to mitigate the difficulties in daily living. While functional recovery is often reported in rodent models of stroke,4,5 human stroke survivors fail to show complete restoration of motor skill after injury, with survivors achieving, on average, about 70% of their recovery potential. 6 There is a disconnect between the rehabilitation regimens used in experimental models and those used in clinical settings, and it is hypothesized that training intensity may be one of the critical factors accounting for this discrepancy.
In rodent models, targeted training of the impaired limb greatly improves the functionality of that limb (eg, Krakauer et al 6 and Tennant and Jones 7 ). While training of the impaired limb is known to promote recovery of function in animals, it is slow and often incomplete in humans. After stroke, patients typically rehabilitate in a clinical setting for 2 weeks before being released to their homes. Rehabilitation facilities often focus more on establishing independence focusing on compensatory strategies. 6 Compensation refers to substituting a new behavior in place of a lost behavior. 8 A common compensatory strategy after stroke is a reliance on the intact limb because it is easier to use and yields faster results in terms of completing basic tasks. Unfortunately, results from animal studies suggest that training the intact limb has detrimental effects on the long-term recovery of the impaired limb.4,9,10 In fact, Kerr et al 4 demonstrated that this effect remains even after the impaired limb is trained for 28 days after early post-stroke compensation in a mouse model. Thus, stroke survivors face a potential conflict between ease of living using their intact limb and recovery of function of the impaired limb.
Another challenge with matching experimental research to clinical outcomes is that stroke patients do not receive the same level of rehabilitative training as laboratory animals. Laboratory animals train 5 to 7 days a week exclusively on their impaired limb and reach hundreds of times in a session.8,11 Human patients rarely receive therapy for more than 3 days a week. Also, rehabilitation facilities tend to focus more on achieving independent living through compensatory strategies, and therefore only half of the sessions target upper limb rehabilitation of the impaired limb with motions (ie, reaches) practiced an average of 32 times. 12 Birkenmeier et al 13 demonstrated that it is possible to translate training dosages typically used in animal studies to humans, but there are limitations relating to patient compliance (pain and discomfort may limit how much rehab a patient can, or is willing to, complete) and funding (expenses associated with rehabilitation limit how many sessions a patient can attend). To understand the impact of impaired limb training on humans, we need to understand the varying effects of differing training intensities on functional outcome. In addition, since training at high intensities is difficult for humans, it is important to define the lower bounds of effective training intensity to balance recovery with real-world limitations.
We know that training intensity can influence rehabilitative outcomes, yet this has only been systematically investigated in terms of increasing intensity in animals. When rats were allowed longer access to a reaching apparatus post-stroke, they showed improved reaching success over rats that had limited access. 14 Bell et al 15 demonstrated that increasing intensity of training to 2 training sessions a day leads to greater functional improvement in mice. This effect was true both when comparing performance across training days as well as training sessions. Notably, the low intensity in this study was defined as daily training trials of 15 minutes or 100 reaches. However, this level of training has been demonstrated to be more intensive than traditional rehabilitation in humans 6 and is therefore too high an intensity to accurately mimic human rehabilitative training.
In humans, time spent training the impaired limb and the number of repetitions of a training task are associated with improved outcomes.16 -18 Timing and duration of rehabilitative training also seems to be important, although these effects are less clear. 18 In a clinical study addressing the effects of daily training, patients that trained 7 days a week scored higher on measures of functional independence as compared to those who trained less frequently 19 ; however, there were no measures of repetitions in training sessions, so daily training intensity is unknown. It has been demonstrated that the amount of time scheduled for therapy is a significant predictor of improved recovery when controlling for the time after stroke. 18
The lower bounds of intensity have yet to be defined. Generally speaking, more rehabilitation is linked to better functional outcomes, but high intensity therapies are rarely practical. It is therefore essential to determine how much rehabilitation is needed to attain specific recovery goals. As previously mentioned, humans typically receive lower rehabilitative intensity than rodents. Developing a rodent rehabilitation model that more closely resembles current clinical practices will aid in the development of new and improved treatments. The current study aims to determine if decreasing training intensity of the impaired limb (ie, every other day) will still have the same functional benefits as more intense (ie, daily) training. A skilled reaching task was used to train and assess the function of the impaired limb under differing training intensities in laboratory mice. Following unilateral ischemic stroke, animals either trained every other day (intermittent training), daily (traditional rehabilitation), or not at all for a total of 14 training sessions. Performance was then assessed using the same reaching task. Results indicate that intermittent training has moderate effects on functional outcome: Mice receiving intermittent training displayed better-skilled reaching performance than untrained control mice but did not improve as much as mice receiving daily rehabilitative training sessions.
Methods
Subjects
The study began with 31 well-handled, 9-month-old male C57BL/6 mice (Jackson Laboratories) that were housed in groups of 3 to 4. All housing units were supplemented with a cardboard roll and PVC (polyvinyl chloride) pipe as part of standard enrichment practices. 7 Animals were on a 12:12 light:dark cycle and were mildly food deprived (2.5-3.5 g/mouse/d) throughout the study. Daily food allotments were adjusted to ensure that no animal lost more than 15% of its original, free-feeding weight. Animal use was in accordance with a protocol approved by Illinois Wesleyan University’s Institutional Animal Care and Use Committee.
Preoperative Behavioral Methods
All mice were trained on the Pasta Matrix Reaching Task (PMRT). 7 For this task, mice were trained to reach through a 0.5 cm slit in the center of a 20 cm tall, 15 cm long, and 8.5 cm wide Plexiglas chamber for pieces of uncooked Capellini pasta that were cut into 3.2 cm pieces and placed vertically in a 10 × 10 matrix. The pasta pieces were placed 2 mm apart in a block located outside the chamber.
Mice were shaped for 9 days to determine limb preferences. Shaping consisted of placing the animals individually in the chamber with the pasta matrix stage full of pasta, allowing animals to reach with both limbs. Each trial lasted either 10 minutes or until the animal completed 10 reaches. Limb preference was determined after the animal used a single forelimb for a minimum of 70% of their reaches in a given trial.
After each animal’s limb preference was determined, all mice were trained on the PMRT to establish skilled motor performance. For training, the matrix stage was filled on the contralateral side to the animal’s preferred limb (5 rows of 10 pasta pieces), forcing the animal to reach only with the preferred limb. All mice were trained 5 days a week for 4 weeks (20 total days). Each training day consisted of a single trial that lasted for either 15 minutes or 100 reaches, whichever occurred first. To determine preoperative performance levels, the number of pasta pieces broken over each of the final 3 days of preoperative training were averaged. Twenty-six mice met the preestablished criterion (breaking an average of at least 9 pasta pieces over the final 3 training sessions) and received photothrombotic surgery to create an ischemic stroke contralateral to the preferred reaching limb.
Photothrombotic Stroke Induction
Following preoperative training on the PMRT, the mice that met the reaching criterion of nine pieces broken (n = 26) received a focal photothrombotic stroke affecting the preferred reaching limb. Mice were first given an intraperitoneal (i.p.) injection of Rose Bengal (100 mg/kg; Sigma Aldrich) that was administered at least 10 minutes prior to laser illumination. Immediately after Rose Bengal injection, mice were anesthetized with ketamine (100 mg/kg; i.p.) and xylazine (10 mg/kg; i.p.) and placed in a stereotaxic frame. A midline incision was made in the scalp to expose the skull and a 532 nm, 20 mW laser (approximately 1 mm in diameter; Beta Electronics) was focused 0.3 mm anterior to Bregma and 1.5 mm from midline (ie, forelimb reaching area) on the hemisphere contralateral to the preferred limb. The laser remained on for 20 minutes. The wound was then sutured and treated with antibacterial ointment. Animals recovered in a heated chamber and were given buprenorphine (3 mg/kg at 0.015 mg/mL in sterile saline, subcutaneously) before returning to their home cage. A total of 5 mice were excluded from the study due to surgical complications: 4 mice did not recover from surgical anesthesia and an additional mouse recovered from anesthesia but did not survive beyond 48 hours after insult.
Postoperative Behavioral Methods
The remaining 21 mice were divided into 3 postoperative training groups: the Traditional group (n = 7) trained using the impaired limb for 14 consecutive days, the Intermittent group (n = 7) trained using the impaired limb on alternate days for 14 total training days, and the Control group (n = 7) did not receive training. Groups were matched based on preoperative performance levels.
All mice received assessment of their impaired limb four days after stroke (postoperative day [POD] 4) and prior to beginning their postoperative training regimen. On POD 5, rehabilitative training began with animals separated into their respective groups. Traditional and Intermittent animals were trained to reach with their impaired limb on the same PMRT used for preoperative procedures. Control animals were placed in identical Plexiglas chambers but were not provided with a pasta matrix. Each Control animal was yoked to a trained animal such that equal numbers of pasta pieces broken by the trained mouse were dropped into the Control animal’s chamber and Control animals were returned to their home cage at the same time as their training partner. Impaired limb performance was assessed for all animals at the end of rehabilitative training. Because Traditional and Intermittent groups completed training at different times, Control animals received impaired limb assessment at both the end of Traditional training (POD 18) and the end of Intermittent training (POD 32).
Postmortem Analyses
Within 24 hours after the final testing session, mice were euthanized by carbon dioxide inhalation and perfused with 0.1 M phosphate buffer followed by 4% paraformaldehyde. The brain tissue was extracted and stored in 4% paraformaldehyde. Seventy-two hours before slicing, the tissue was stored in a 30% sucrose solution (made in phosphate buffer). The tissue was then frozen and sliced in 40 µm sections using a sledge microtome and stored in cryoprotectant.
To verify lesion size and location, a representative sample of tissue, every sixth slice, was mounted on a gelatin slide and stained with toluidine blue. Images of all slices were taken under a microscope at a total magnification of 20× and montaged in Photoshop (Adobe) such that the entire coronal section was visible.
The remaining cortical volume for each animal was estimated using the Cavalieri method.20,21 The cortical areas of nine coronal slices from approximately 2 mm anterior to 1.5 mm posterior to Bregma, each 240 μm apart, were measured by tracing the cortical boundaries of contralesional and ipsilesional cortex (Image J). The sums of section areas were then multiplied by the distance between sections and the difference between contralesional and ipsilesional cortices was determined.
Statistical Analyses
A series of planned comparisons were carried out using SPSS statistical software. Specifically, performance on the PMRT task (number of pasta pieces broken) was assessed for preoperative, postoperative and post training assessments. A separate analysis was also run to compare reaching performance on POD 18 (to compare group performance by training day as opposed to training session). Post hoc analyses with a least significant difference (LSD) correction were run when omnibus tests were significant. Statistical significance was defined at an α level of .05 for all comparisons. All error values reported represent the standard error of the mean (SEM).
Results
Traditional and Intermittent Training Result in Improved Functional Outcome Compared With Control Training
Prior to data collection, researchers predicted that all groups would exhibit similar reaching performance on both preoperative and initial postoperative assessments. Training groups were only expected to differ at the final post training assessment. Univariate planned comparisons were conducted for each assessment day to confirm these hypotheses.
Preoperative reaching performance did not differ between groups (n = 7 for all groups). A univariate analysis of variance (ANOVA) demonstrated that the Traditional (M ± SEM = 11.4 ± 0.63), Intermittent (M ± SEM = 11.8 ± 0.61), and Control (M ± SEM = 11.7 ± 0.67) groups all performed similarly when measured on the average number of pasta pieces broken over the final three days of preoperative training (F(2, 21) = 0.119, P = .889; Figure 1A and B). All groups also exhibited a similar deficit in reaching after stroke induction. A univariate ANOVA confirmed that all groups performed similarly on the initial postoperative assessment (POD 4: Traditional, M ± SEM = 5.57 ± 0.90; Intermittent, M ± SEM = 5.14 ± 0.91; Control, M ± SEM = 4.71 ± 0.61; F(2, 21) = 0.276, P = .762, Figure 1A and B).
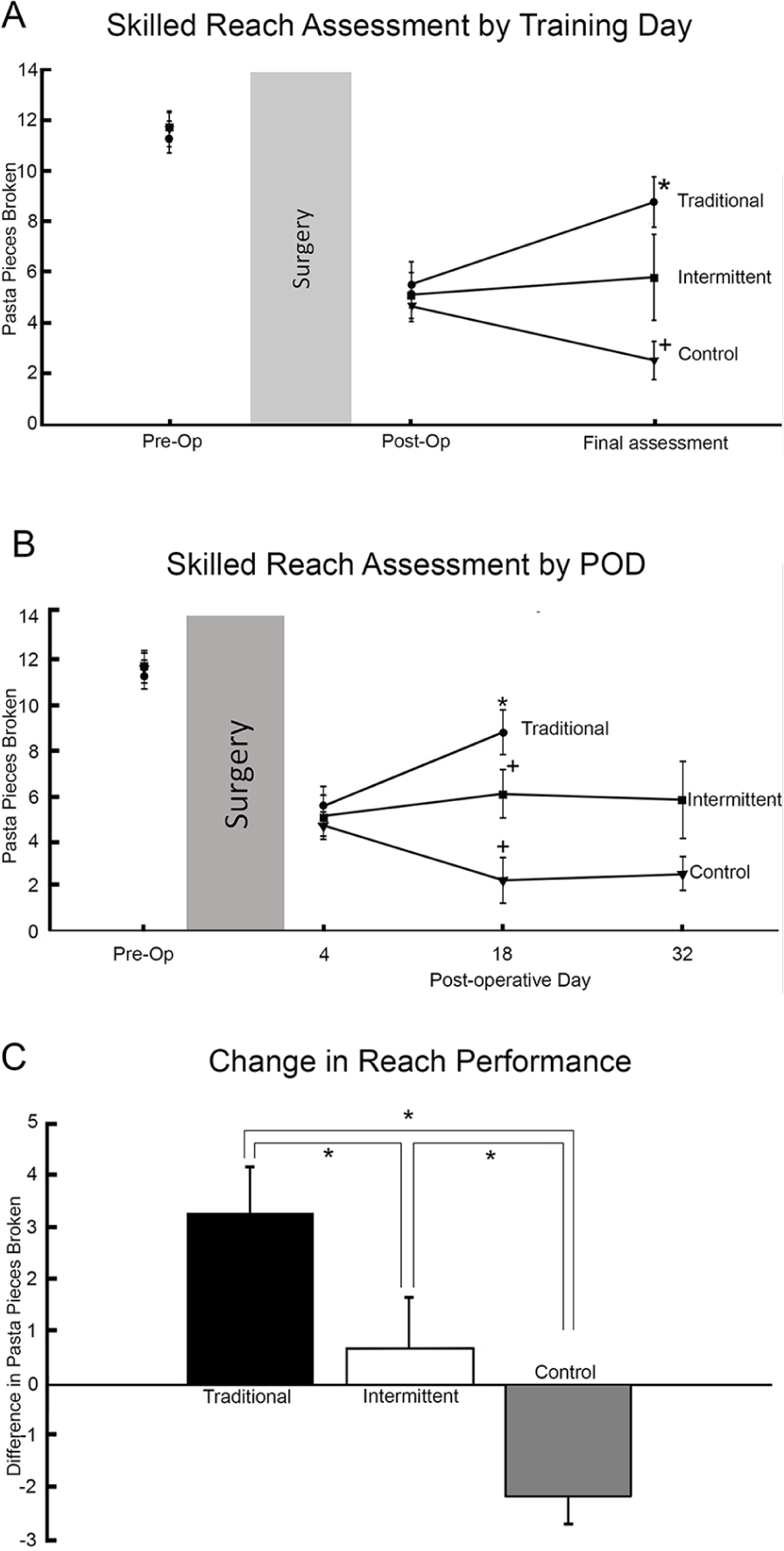
Mice that trained at a higher intensity show greater improvements on the Pasta Matrix Reaching Task (PMRT). Adult male mice were preoperatively trained on the PMRT and assessed 4 days after stroke induction. Postoperatively, mice either trained daily (Traditional intensity), every other day (Intermittent intensity), or did not train (Control). All error bars represent standard error of the mean (SEM). (A) Average group performance, measured in number of pieces broken during the PMRT, both preoperatively and postoperatively did not differ between the training groups. When assessed on the final training session (postoperative day [POD] 18 for Traditional and POD 32 for Intermittent), the traditional group significantly outperformed the control group. The Traditional and Intermittent groups did not differ significantly, and there was a trend (P = .073) for the Intermittent group to outperform the control group. *Indicates P < .05 from control group. (B) When the data are analyzed on POD 18 (halfway through training for Intermittent and the final training day for Traditional), there is a significant difference between the Traditional group and both the Control and Intermittent groups. There is a trend (P = .078) for the Intermittent group to outperform the Control group on POD 18. *Indicates P < .05 from Control group, +indicates P < .05 from Traditional group. (C) Difference scores were calculated by subtracting the number of pasta pieces broken at the initial postoperative assessment from the number of pieces broken on the final training day for each mouse. The Traditional group shows a significant improvement in performance over the Control group and the Intermittent group. The Intermittent group shows almost no improvement, but performance is still significantly better than the Control group, which declined in performance. *Indicates P < .05.
After 14 training sessions, mice were given a final assessment of reaching ability (POD 18 for the Traditional group and POD 32 for the Intermittent group). Mice in the Traditional group exhibited the best reaching performance (M ± SEM = 8.86 ± 0.99), followed by mice in the Intermittent group (M ± SEM = 5.86 ± 1.71) and finally those in the Control group (M ± SEM = 2.57 ± 0.75). A univariate ANOVA revealed a significant difference between groups on the final post training assessment day (F(2, 21) = 6.645, P = 0.016; Figure 1A). A post hoc analysis with an LSD correction indicated that the Control group performed significantly worse on the PMRT than the Traditional group (P = .002). The Intermittent group showed a trend of improved performance over the Control group (P = .073) and was not significantly different from the Traditional group (P = .099).
Additional analyses were also conducted comparing performance 18 days after stroke. On this day, the Traditional group finished its last day of training, and the Intermittent group had completed 7 of 14 days of training. The same pattern of behavior was revealed: the Traditional group (M ± SEM = 8.86 ± 0.99) outperformed the Intermittent group (M ± SEM = 6.14 ± 1.08), which also outperformed the Control group (M ± SEM = 2.29 ± 1.02). A univariate ANOVA confirmed a significant difference between groups on POD 18 (F(2, 21) =10.32, P = .001). A post hoc analysis with an LSD correction revealed that the Control group differed significantly from both the Traditional (P < .001) and Intermittent groups (P = .016). The Traditional group showed a trend of improved performance over the Intermittent group (P = 0.078, Figure 1B).
Traditional Training Results in Improved Skilled Reaching Performance, While Intermittent Training Results in Conserved Reaching Performance
Visual inspection of the data suggested that mice in the Traditional group exhibited improved performance between the initial postoperative assessment and the final assessment day compared with Intermittent and Control mice. As such, difference scores were calculated for each animal by subtracting the number of pasta pieces broken during the postoperative assessment from the number of pieces broken during the final day of training (ie, final post training assessment). A repeated measures ANOVA revealed a significant day by group interaction (F(2, 21) = 11.099, P = .001; Figure 1C). The Traditional group broke an average of 3 (±SEM = 0.89) more pieces by the end of training, the Intermittent group remained about the same (±SEM = 0.71), and the Control group broke an average of 2 fewer pieces (±SEM = 0.51). An LSD-corrected post hoc analysis confirmed that the Control group was significantly different from both the Traditional (P < .001) and Intermittent groups (P = .023). The Intermittent and Traditional groups also differed significantly (P = .039).
Lesion Size Is Not Affected by Different Training Intensities
A univariate ANOVA revealed that the average lesion volume was not significantly different between Traditional, Intermittent, and Control groups (F(2, 21) = 0.360, P = .703; Figure 2). Lesions were localized to cortical regions in all animals, no white matter damage was observed.
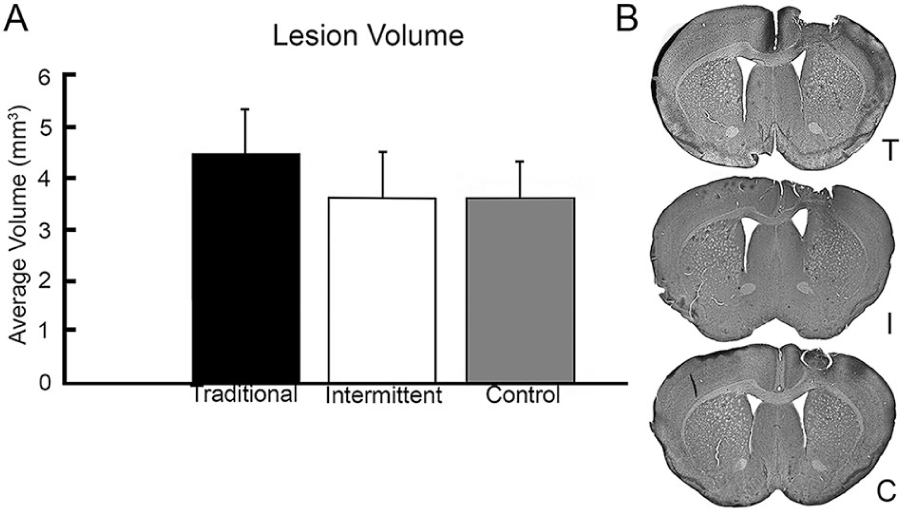
Lesion verification confirms that there is no difference in lesion volume between training groups. (A) Average lesion volume after stroke was 3.95 mm3 and did not significantly differ depending on training intensity. Error bars represent standard error of the mean (SEM). (B) Representative lesions from Traditional (T), Intermittent (I), and Control (C) mice taken from the center of the lesion (+0.26 from Bregma). Lesions were consistently located in the sensorimotor cortex and did not include white matter damage.
Discussion
Training intensity affects functional outcome. In the current study, Intermittent training resulted in differing outcomes from both Traditional daily training and untrained Control procedures when controlling for total number of rehabilitative training sessions. Mice in the Traditional group showed faster and better functional recovery than those in the Control group, as indicated by a greater number of pasta pieces broken. Mice that received Intermediate intensity rehabilitative training exhibited consistent reaching performance from the initial poststroke deficit through the entirety of their training regimen, while Control mice exhibited a decline in reach performance. These differences between groups replicate multiple studies finding that intense focused training of the impaired limb improves functional outcome after stroke (eg, Kerr et al, 4 Ballermann et al, 11 and Bell et al 15 ). These results further extend previous research to suggest that intermediate training is insufficient to recover functional outcome but may be protective against further functional decline. To our knowledge, this is the first study to investigate lower intensity training of this manner in the mouse.
It is important to determine the lower bound of effective training intensity necessary for optimal recovery for several reasons. First, limitations of human rehabilitation (including patient compliance and the expense of rehabilitation) mean that it is not always possible to deliver high-intensity rehabilitative training. Second, humans compensate on their own to regain independence in their daily lives, which can interfere with the recovery of the impaired limb.4,9,10 Therefore, it would be beneficial to know if they are receiving enough impaired limb training to allow them to promote functional recovery and perhaps use their intact limb for daily activities.
Some evidence suggests that functional recovery may be independent of training dosage. Lang et al 22 clinically investigated the effects of a high intensity intervention in human stroke survivors. Eighty-five patients practiced skilled reaching 32 000 to 96 000 times per 1-hour session 4 days a week and demonstrated no dose-dependent effect of training. However, the participant pool was composed only of chronic patients who experienced a stroke more than 6 months prior to the beginning of testing. It is well understood that training is most effective in the acute stages of stroke, less than 3 months after injury.16,18,23 -25 Therefore, it is possible that patient recovery was already limited due to the timing of rehabilitation in relationship to stroke onset. Also, even the lowest intensity group performed many more target behaviors than in a typical rehabilitation session, so effects between groups may have been obscured by a ceiling effect.
In the current study, animals’ performance was also compared by analyzing the data by the amount of time since animals sustained their stroke. On POD 18, Traditional mice had completed 14 training sessions, Intermittent mice had completed 7 training sessions, and Control mice were assessed for the second time following injury. Figure 1B demonstrates that these data show a similar behavioral pattern as when data are analyzed by training day. The animals that received higher intensity training show the most improved performance, but the animals receiving lower intensity (ie, Intermittent) training still outperformed Controls. Since the Control group did not show improved performance indicative of spontaneous recovery, neither rehabilitative training group would be expected to show improvements strictly related to time since injury.
Notably, Control animals showed a decline in their performance from the initial postoperative assessment to the posttraining assessment. This was unexpected, as previous work with untrained animals indicates some level of spontaneous recovery in Control mice (eg, Kerr et al, 4 Alaverdashvili and Whishaw, 10 and Kerr et al 26 ). This difference may be due to the use of the photothrombotic model of stroke induction in the current study rather than the Endothelin-1 (ET-1) model, which was used in previous behavioral studies. Previous work has shown that the photothrombotic model of stroke produces significantly larger lesions, and greater behavioral deficits, than the ET-1 model (Haan, Nemchek, Mavros, and Kerr, unpublished data). This suggests that smaller lesions may permit more spontaneous recovery and that the brain is more limited in its ability to recover after a larger area is damaged. However, the strokes given in both cases are quite small. Stroke produced by photothrombosis may also be more progressive in nature and negatively affect fine motor control over a longer period of time. This would explain why Control animals in the present study performed worse on the second assessment. Previous research suggests that Control animals undergo limited spontaneous recovery.4,5,15 For instance, Li et al 27 reported spontaneous recovery on 3 out of 4 fine motor tests in Control mice after photothrombotic injury. However, the tasks were untrained assessments of motor performance (eg, cylinder test, hanging wire test, pole test, and adhesive removal test), which typically show fewer deficits and more spontaneous recovery than skilled reaching tasks. 28 The animals in the study by Li et al 27 were also tested up to 8 weeks poststroke, and the majority of group differences were seen at this time point. According to these data, the Control mice in the present study should not demonstrate significant spontaneous recovery given that only 2 weeks of poststroke behavior were assessed.
When successful pasta retrieval on the last day of training was assessed, animals that trained intermittently showed a trend of better functional outcome than Controls. When groups were compared using difference scores, the Intermittent group showed increased performance over Controls but less improvement than the Traditional group (Figure 1C). Notably, the Controls had a negative difference score indicative of a decline in performance, while the Intermittent group stayed about the same and the Traditional group improved. Intermittent training did not lead to the same improvements on the skilled reaching task as more intense training. Instead Intermittent training seemed to protect from the decline in performance seen in animals that did not train (Control). This suggests that rehabilitation is a beneficial practice even if high-intensity training cannot be achieved, though functional gains do require more intensive training.
Given that there was no difference in lesion size between groups (Figure 2), there must be other plastic changes driving the observed behavioral differences between groups. Based on previous work that suggests a correlation between rehabilitation and plasticity,5,29 -31 the authors predict that increased rehabilitation intensity would correspond to an increase in neural plasticity in the perilesion cortex. Both neurogenesis and angiogenesis are linked to reduced lesion volume as number of surviving cells increases, 32 therefore neither is an anticipated mechanism in the current study. One theory is that the amount of training that occurs during the window for optimal plasticity, 7 to 14 days poststroke,33,34 is important in determining the recovery potential. This is supported by the finding that performance on POD 18 follows the same patterns seen on POD 32, indicating that groups that continued training after POD 18 did not significantly improve after that day. Further studies need to be conducted in order to determine the relationship between training intensity and neural plasticity.
In this study, the authors elected to perfuse mice immediately after training. However, taking neural measurements after training lead to comparing final lesion size at 2 different postoperative days. It is possible that differences in lesion volume in this study were masked by euthanasia at different time points. While comparing lesion volume at POD 32 would eliminate this confound, it would introduce variability in the time elapsed posttraining. Future studies may be able to monitor stroke size and composition in living subjects and therefore repeat measurements across the rehabilitation period to answer questions about effects of rehabilitation on neural changes over time.
The current study begins to address issues associated with rehabilitation dosage; however, the data presented is not without limitation. First, the postoperative training period was relatively short. All animals completed 14 training sessions; however, 30 or more days of rehabilitation would capture long-term effects not assessed in the present study. While we attribute the failure of the Traditional group to regain preoperative performance to the abbreviated postoperative training, we cannot exclude the possibility that other systematic factors influenced behavior. Extending the training period would introduce a confound with recovery time, as the final assessment of Intermittent animals would be a full month after that of the Traditional animals. Future studies should consider a design that allows all groups to train until preoperative levels are achieved, eliminating issues associated with comparing relatively small changes in behavior that occur in brief periods of time.
Despite the small number of training sessions, data from POD 32 suggests little improvement in the Intermittent group after the first 2 weeks of rehabilitation. These results imply that differences in training intensity may have a greater impact during a critical poststroke window. Future studies should consider interactions between training intensity and time since injury in order to capture how early windows for neuroplasticity poststroke may influence rehabilitation. Last, rehabilitation dosage has yet to be investigated along with other factors known to influence poststroke recovery, such as exercise and age.
Conclusions and Implications
These behavioral results clearly illustrate the importance of intensity in rehabilitation. In the mouse, training at half of the traditional intensity leads to improvements compared with no training but does not lead to the same level of posttraining performance seen with higher intensity training. This indicates that rehabilitation need not be as intense as the dosage traditionally seen in rodent studies to mitigate certain poststroke effects. However, rehabilitation should be more intense than the model used in the present study (ie, every other day) in order to maximize potential recovery. Ultimately this study demonstrates the insufficiency of the low-intensity rehabilitation that stroke patients currently receive. Yet, the data allow for personalized treatment based on different goals: Return to prestroke performance or prevention of further declines. Both medical professionals and funding agencies should consider the disparity in recovery when deciding on a rehabilitation regimen for victims of stroke.
Footnotes
Acknowledgements
The authors would like to thank Amy Gourley for technical assistance with behavioral data collection and Violet Pavlik for technical assistance with image acquisition for lesion verification.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Illinois Wesleyan University Artistic and Scholarly Development Program.
