Abstract
Background and purpose. The identification of a genetic role for cognitive outcome could influence the design of individualized treatment in poststroke rehabilitation. The aim of this study is to determine whether brain-derived neurotrophic factor (BDNF) Val66Met polymorphism is independently associated with poststroke functional outcome. Methods. A total of 775 stroke patients with genomic data were identified from the Partners HealthCare Biobank, which contains a large number of genotypes from Biobank’s consented patients. Of 775 stroke patients who met the inclusion/exclusion criteria, 86 were enrolled. Functional outcomes were assessed using the Functional Independence Measure scores at the time of admission and discharge. Logistic and linear regression models adjusted for covariate variables, including age, sex, and medical conditions, were used to evaluate the association between BDNF Val66Met and functional outcome. Results. We detected a significant correlation between Met alleles and lower cognitive function at discharge in both ischemic and hemorrhagic stroke patients. Genotyping findings confirmed that BDNF Met allele frequency was higher in contrast to Val/Val allele frequency in lower cognitive functional recovery. Furthermore, after adjusting for covariate variables, BDNF Met alleles were found to be associated with lower cognitive outcome [P = .003; odds ratio (OR) = 5.95 (1.81-19.52)] and recovery [P = .006; OR = 3.16 (1.4-7.15)], especially with lower problem solving, expression, and social recovery in all stroke patients. Conclusions. Met allele carriers exhibited impaired poststroke cognitive function. The BDNF genotype may be a useful predictor of cognitive function in inpatient poststroke rehabilitation.
Introduction
Stroke is the fifth leading cause of death in the United States, and its global burden is increasing dramatically, with an incidence rate of 250 per 100 000 per year. 1 It is frequently associated with significant disability and impaired quality of life. Despite a decline in mortality in the past 2 decades,2,3 the number of people who have remained disabled from stroke, or have been affected by stroke, has increased significantly as the population ages. 4 Recovery after stroke varies considerably between individuals. The etiology and pathogenesis of stroke are complex, and it is suspected that genetic factors may be involved in stroke recovery. To develop effective interventions, it is important to have a clear understanding of the genomic factors influencing functional recovery.
Many studies have attempted to investigate connections between specific genes and poststroke recovery.5-7 The primary gene is BDNF, which leads to a substitution of valine to methionine at codon 66 (Val66Met). Brain-derived neurotrophic factor (BDNF) is a key molecule involved in neuronal survival, differentiation, and synapse plasticity, which are processes implicated in learning and memory.8,9 Previous studies have found cognitive impairment to be associated with changes in peripheral BDNF levels. 10 BDNF Val66Met (rs6265) single nucleotide polymorphism (SNP) is considered a viable gene polymorphism that leads to decreased BDNF secretion and impairment in specific forms of learning in humans.11,12 Some genetic studies have found that BDNF Val66Met was significantly associated with the occurrence and long-term outcomes of stroke 13 ; However, for poststroke recovery, the effect of heterozygous or homozygous Val66Met alleles on functional outcome is controversial.14-18
Although there is convincing evidence for the role of BDNF in cognitive performance, there is little known about the association between BDNF Val66Met and cognitive performance in poststroke patients. Understanding the effect of BDNF Val66Met for poststroke cognitive recovery can refine existing treatment and may lead to genotype-based personalized medicine. In this study, we aimed to investigate the effects of BDNF Val66Met polymorphism on cognitive recovery after stroke during subacute inpatient rehabilitation.
Materials and Methods
This was an observational study that was approved by the institutional review board of Spaulding Rehabilitation Hospital, a teaching affiliate of Harvard Medical School. Functional status was assessed using Functional Independence Measures (FIMs) on admission to the rehabilitation hospital and at discharge. All patients received a standardized inpatient stroke rehabilitation program at this acute rehabilitation facility. The standardized inpatient stroke rehabilitation program consisted of 3 hours per day of physical therapy, occupational therapy, and cognitive and speech therapy, and 24 hours per day, 7 days a week nursing and medical care. The therapy was administrated at the discretion of the therapist based on the patient’s functional evaluation. The medical care was guided by current stroke guidelines.
Participants
The 775 patients included in the study were identified from the Partners HealthCare Biobank, a biorepository of consented patients sampled at Partners HealthCare (the parent organization of Massachusetts General Hospital and Brigham and Women’s Hospital) between March 1, 2014, and September 30, 2017. These participants were screened using the following inclusion and exclusion criteria, and patients were eligible if they were (1) aged >18 years, (2) had a diagnosis of a first or recurrent stroke that was confirmed by brain computed tomography or magnetic resonance imaging, (3) linked to Spaulding Rehabilitation Hospital, and (4) scored the FIM within 48 hours of admission and discharge. Exclusion criteria included individuals with (1) any accompanying neurological or psychiatric disorder or (2) length of hospital stay of fewer than 6 days.
Genetic Analysis
Data regarding the BDNF Val66Met polymorphisms were obtained from the Partners HealthCare Biobank, a biorepository of consented patients sampled at Partners HealthCare. Patients were classified according to BDNF genotype into 1 of 3 groups: the Val/Val, Val/Met, or Met/Met group. Genotype data from the Partners HealthCare Biobank was entered into Plink software V1.07 to select SNP rs6265. Allele frequency = [(Number of homozygotes × 2 + Number of heterozygotes × 0.5)/n]. We grouped human Met allele carriers together (Val/Met and Met/Met) for analysis because the rarity of the human Met allele homozygotes does not provide for enough observation to produce meaningful analyses.
Outcome Measurement
Functional outcomes were evaluated with the FIM. The primary outcome is the gain of cognitive FIM score during inpatient rehabilitation. Secondary outcomes include gain of subscore of cognitive FIM score, motor FIM score, total FIM score, and discharge FIM motor and cognitive score. The FIM is a widely used, standard functional assessment to categorize a patient’s condition. This instrument is administered using the Uniform Data System for Medical Rehabilitation protocol, 19 and it provides indices for the level of assistance required for a patient to accomplish activities of daily living (ADLs). 20 The FIM comprises 18 items: 13 motor items (motor FIM subscale) and 5 cognitive items (cognitive FIM subscale). Each item is given an ordinal score from 1 to 7. The minimum score of 1 represents complete dependence on others for that task, and the maximum score of 7 represents full independence. Ranges for total, motor, and cognitive FIM scores are 18 to 126, 13 to 91, and 5 to 35, respectively; higher scores reflect greater independence. Cognitive FIM scores are calculated by items that measure comprehension, expression, social, problem solving, and memory tasks. The FIM instrument’s motor and cognitive scales can enable clinicians to monitor progress during rehabilitation.21,22 The FIM assessment was administered by trained therapists and nurses on admission and discharge to the rehabilitation hospital. Gain of FIM (FIM score at discharge − FIM score on admission) is commonly used to assess the degree of improvement in function. The reliability and validity of the FIM for stroke patients are well established.23,24 The FIM demonstrates acceptable reliability across a wide variety of settings, raters, and patients. In a quantitative review, a median interrater reliability for the total FIM was 0.95, and median reliability coefficients for FIM motor items and cognitive items were 0.97 and 0.93 respectively. 25 The FIM can be used as a predictor for outcomes. 26
Covariates
Covariates used in the regression models were assessed on admission and included sociodemographic information, comorbidities, and FIM severity or scores on admission. Other important factors influencing the outcome of stroke rehabilitation such as stroke lesion (right or left, supratentorial or infratentorial), first-ever or recurrent stroke, hospital stay, duration from stroke onset to comprehensive rehabilitation and aphasia were recorded. The following sociodemographic information was evaluated: age, gender, race, history of smoking and alcohol use, and employment. Medical history of vascular conditions, including hypertension, atrial fibrillation, coronary artery disease, diabetes mellitus, and stroke type, was recorded.
Statistical Analysis
Categorical variables were expressed as counts and percentages, and continuous variables as mean value ± SD. Genotype frequency was assessed for Hardy-Weinberg equilibrium using a χ2 test. 27 The χ2 test was used for dichotomous and categorical variables.
In logistic regression, we used the median value of FIM scores at discharge and FIM gain (FIM at discharge − FIM at admission) for stratification. FIM scores at discharge and FIM gain were stratified into 2 groups: low versus high. In each group, the median value and above of FIM scores at discharge and FIM gain were defined as the high group; below median was defined as the low group. Univariate logistic regression was performed to examine the association between BDNF Val66Met and functional outcome to determine the odds ratios (OR) with corresponding 95% CIs. Subsequently, linear regression was performed to test the association between BDNF Val66Met and FIM scores at discharge. The prediction of functional outcome at discharge and FIM gain was analyzed using 3 models. The crude model was not adjusted for covariate variables. Model I was adjusted for age, sex, and race. Given the association of admission functional status with functional outcome as well as the association of vascular condition with stroke recovery, 28 model II was adjusted for variables in model I plus medical vascular conditions, including hypertension, atrial fibrillation, coronary artery disease, diabetes mellitus, FIM severity or scores on admission, stroke lesions, first-ever or recurrent stroke, aphasia, hospital stay, and duration from stroke onset to comprehensive rehabilitation. These regression models were used to investigate the effects of BDNF Val66Met and the other variables on the functional outcome.
All analyses were conducted in SPSS for Windows 7, version 21 (IBM Corporation, Armonk, NY). Values of P <.05 were considered statistically significant in all tests.
Results
Figure 1 shows the study profile. A total of 775 stroke patients with genomic data were identified from the Partners HealthCare Biobank, which contains a large number of genotypes from Biobank’s consented patients. Of 775 stroke patients who met the inclusion criteria, 86 were enrolled. No deviation from the Hardy-Weinberg equilibrium was observed for the BDNF genotype (P > .05).
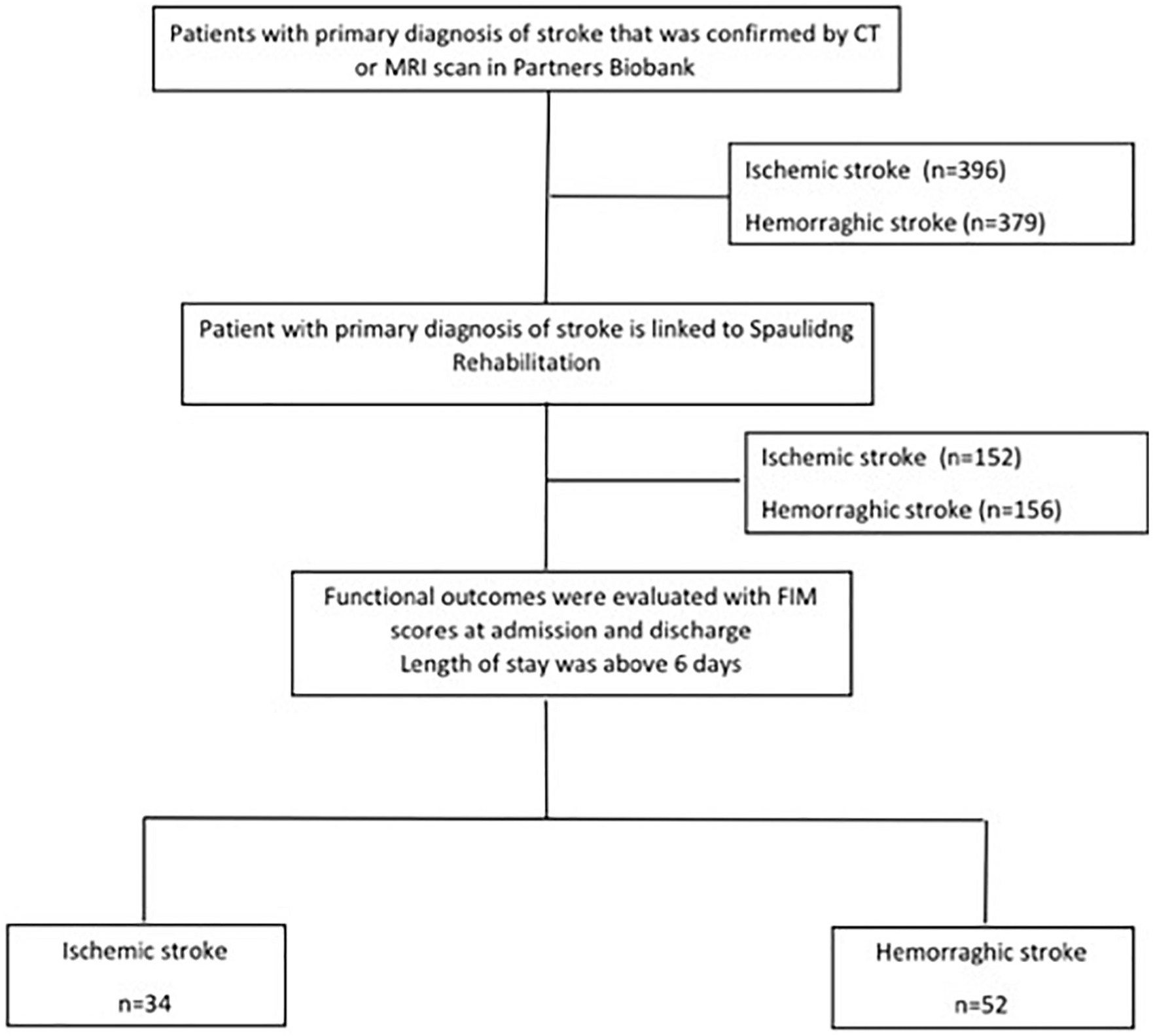
Flow diagram of subject selection procedures.
Baseline characteristics are shown in Table 1. Of the 86 participants included in the analysis, 64 (74.4%) were carriers of the BDNF Val/Val genotype, whereas 22 (25.6%) carried the BDNF Met alleles. Val/Val and Met allele carriers had no significant difference in demographic characteristics; comorbidities; or FIM motor scores, cognitive severity, or scores on admission (P > .05).
Demographic and Clinical Characteristics. a
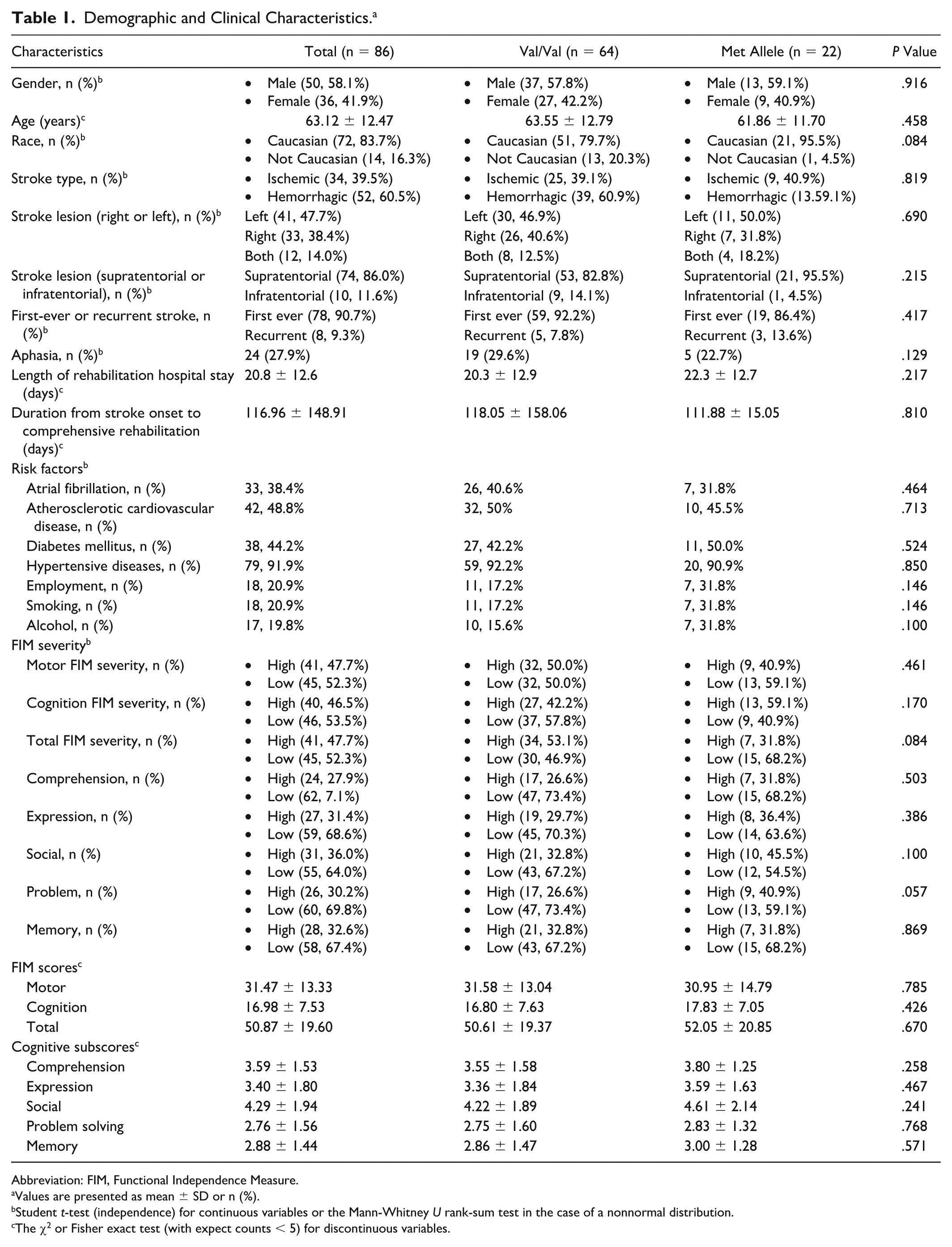
Abbreviation: FIM, Functional Independence Measure.
Values are presented as mean ± SD or n (%).
Student t-test (independence) for continuous variables or the Mann-Whitney U rank-sum test in the case of a nonnormal distribution.
The χ2 or Fisher exact test (with expect counts < 5) for discontinuous variables.
BDNF Val66Met Genotypes and Poststroke Outcome and Recovery
Genotyping findings confirmed that Met allele frequency was higher in contrast to Val/Val allele frequency in lower cognitive functional recovery, especially in lower expression (P = .032), social function (P = .033), and problem-solving function (P = .019; Table 2, Figure 2). A regression model was used to distinguish the effect of predictive value of the Met allele from comorbidities and clinical characteristics in cognitive function outcome. As shown in Figure 3, lower cognitive recovery is not associated with Met alleles in the crude model without adjusting for covariate variables [P = .275, OR = 1.47 (0.74-2.92), or in model I after adjusting for age, sex, and race only [P = .185; OR = 1.62 (0.79-3.30)]. However, in model II after adjusting for age, sex, race, comorbidities, stroke characteristics, hospital stay, and so on, lower cognitive recovery was significantly associated with Met alleles [P = .006; OR = 3.16 (1.40-7.15)]. A similar pattern was found for the Met allele for lower problem-solving recovery [P = .021, OR = 2.41 (1.14-5.08) in a crude model; P = .017, OR = 2.50 (1.18-5.32) in model I; P = .000, OR = 10.94 (4.03-29.73) in model II]; for lower expression recovery [P = .037, OR = 2.65 (1.06-6.64) in the crude model; P = .02, OR = 2.95 (1.16-7.55) in model I; P = .001, OR = 6.67 (2.17-20.52) in model II], and for lower social recovery [P = .036, OR = 2.15 (1.05-4.41) in a crude model; P = .02, OR = 2.44 (1.15-5.14) in model I; P = .003, OR = 3.49 (1.54-7.93) in model II].
BDNF val66Met Genotype Percentages for Functional Outcome at Discharge and Recovery. a
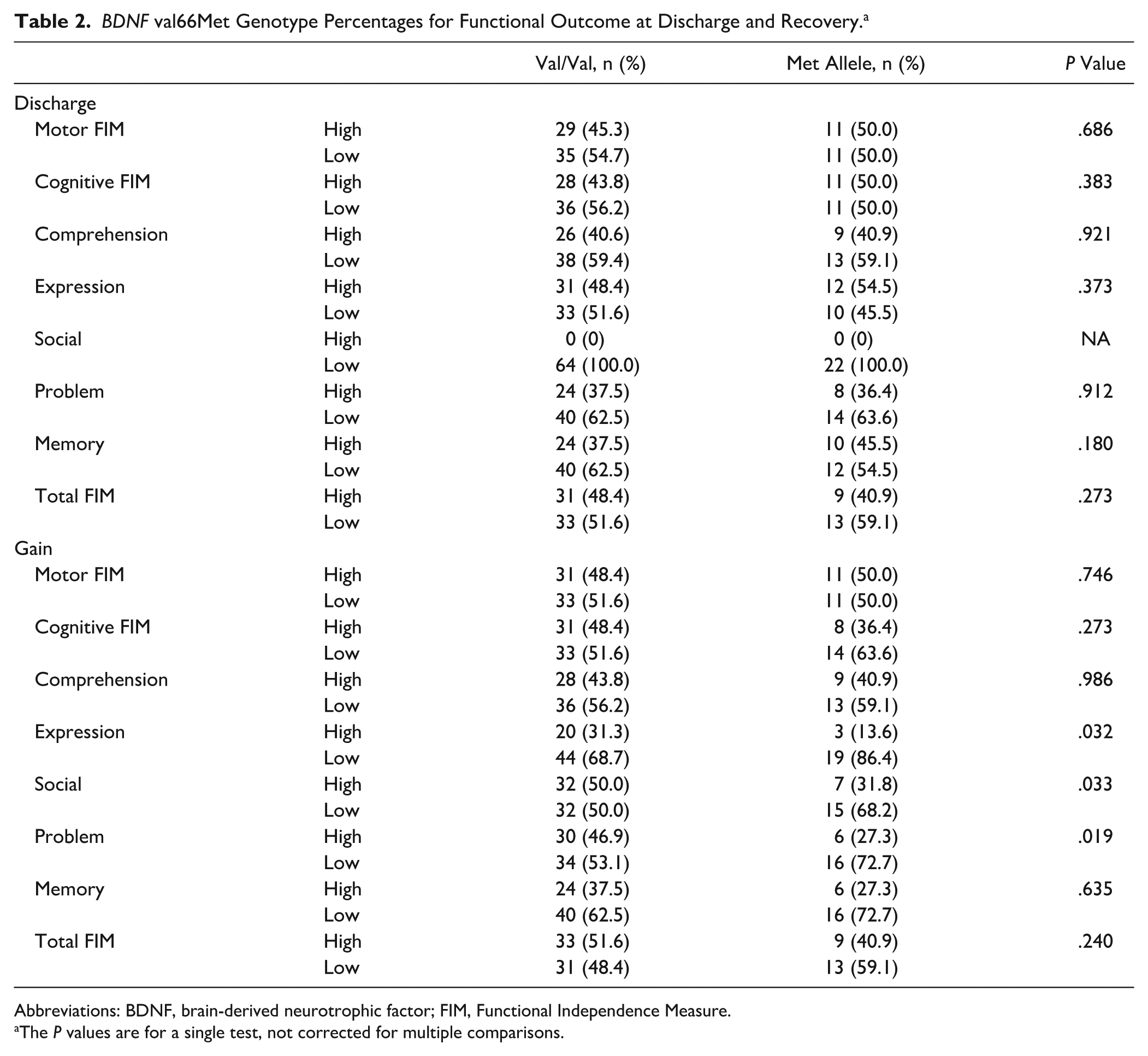
Abbreviations: BDNF, brain-derived neurotrophic factor; FIM, Functional Independence Measure.
The P values are for a single test, not corrected for multiple comparisons.
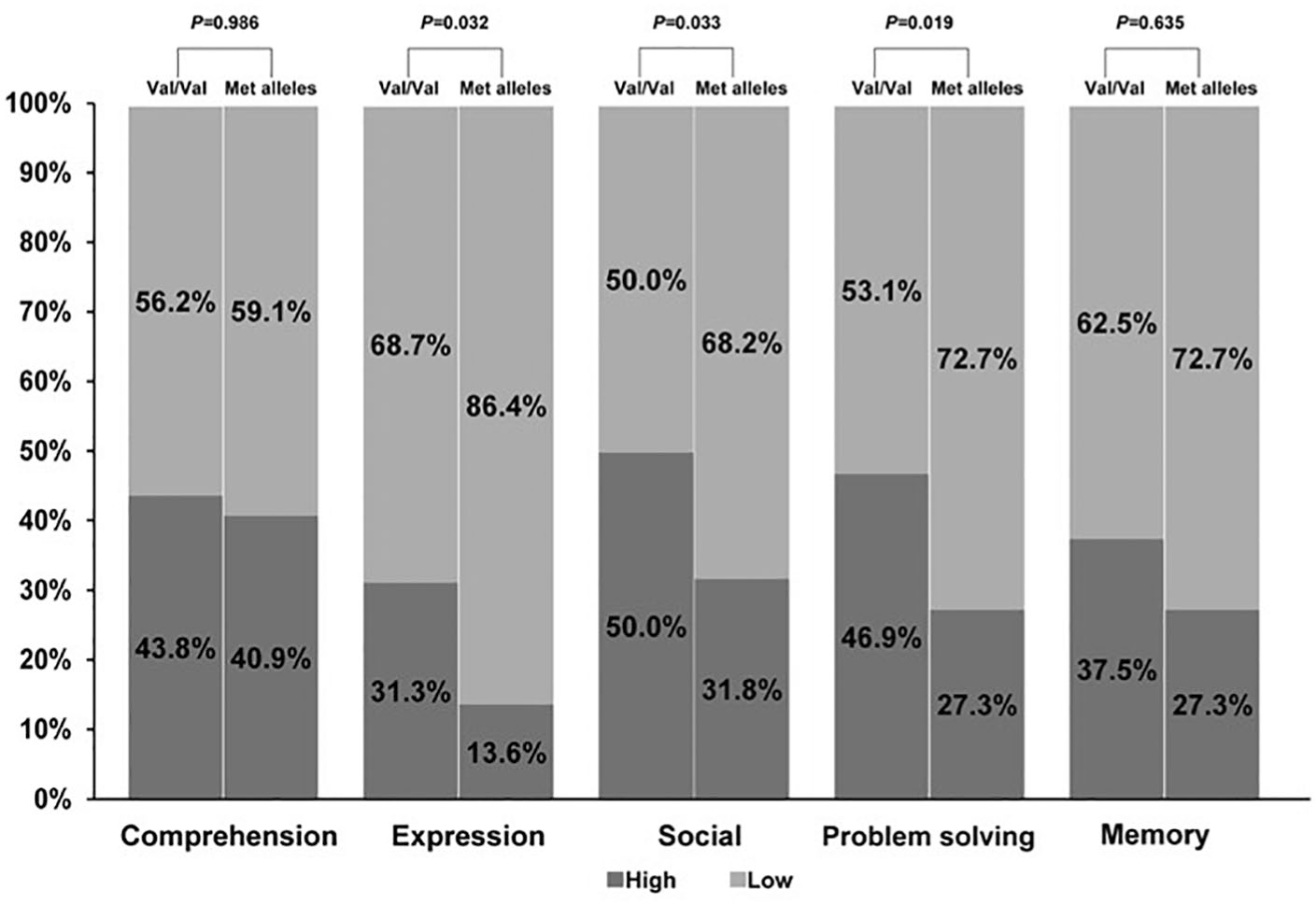
Genotype frequencies of the BDNF Val66Met for subcognitive recovery. The number of BDNF Met alleles showed a significant correlation with lower cognitive functional recovery, especially in lower expression (P = .032), social (P = .033), and problem-solving function (P = .019).
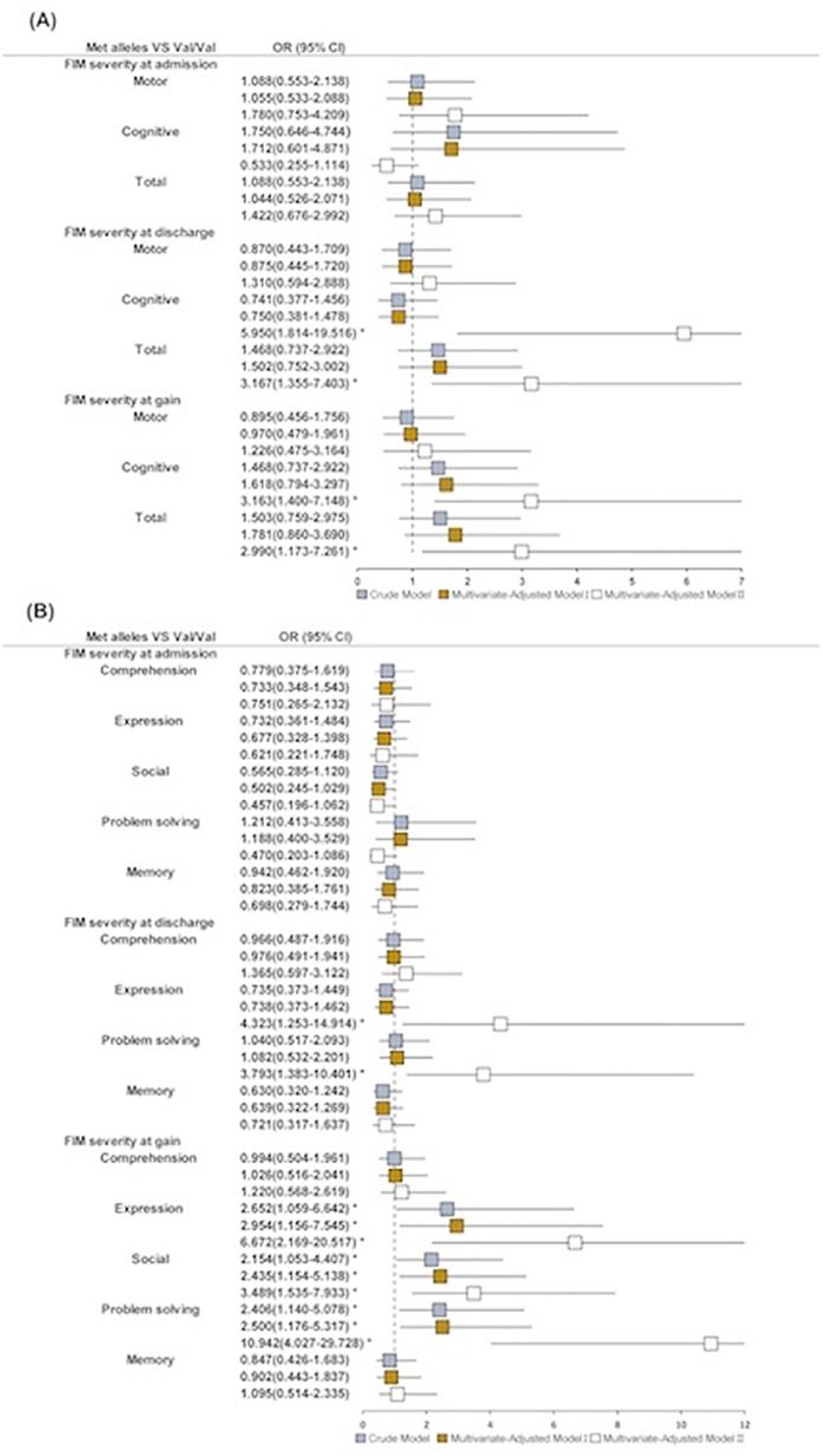
Odd ratios (ORs) and 95% CIs intervals for the association between BDNF Met alleles and lower functional outcome before and after adjustment for confounding factors: A. The association between BDNF Met alleles and motor, cognitive, and total functional outcome. B. The association between BDNF Met alleles and subcognitive functional outcome. Squares indicate the OR, with the size of the square inversely proportional to its variance, and horizontal lines represent 95% CIs. Blue boxes indicate a crude model. Gray boxes indicate model I, adjusted for age, sex, and race. White boxes indicate model II, adjusted for variables in model I plus medical vascular conditions, including hypertension, atrial fibrillation, coronary artery disease, diabetes mellitus, FIM severity or scores on admission, stroke lesions, first-ever or recurrent stroke, aphasia, hospital stay, and duration from stroke onset to comprehensive rehabilitation.
For cognitive functional outcome, Met alleles were significantly associated with lower functional outcome [P = .003, OR = 5.95 (1.81-19.52) in a multivariate-adjusted model II] at discharge, especially in expression [P = .020, OR = 4.32 (1.25-14.91) in a multivariate-adjusted model II] and problem solving [P = .010, OR = 3.79 (1.38-10.40) in a multivariate-adjusted model II]. We did not find any association between Met allele carriers and motor functional outcome.
BDNF Val66Met Genotypes and Poststroke FIM Scores at Discharge and FIM Gain
As shown in Table 3, we detected a significant correlation between Met alleles and cognitive scores [P = .002, OR = 2.11 (0.78-3.43) in a multivariate-adjusted model II], especially lower problem-solving subscores [P = .017, OR = 0.57(0.10-1.04) in a multivariate-adjusted model II] and expression subscores [P = .025, OR = 0.44(0.06-0.82) in a multivariate-adjusted model II] at discharge. The Met alleles genotype showed less recovery compared with the Val/Val genotype in cognitive and total functional recovery, and they were also significantly correlated with less total functional recovery [P = .018, OR = 6.07(1.05-11.09) in a multivariate-adjusted model II] and cognitive functional recovery scores [P = .009, OR = 2.30 (0.57-4.02) in a multivariate-adjusted model II], especially in expression recovery [P = .014, OR = 0.58 (0.12-1.05) in a multivariate-adjusted model II] and problem solving recovery [P = .000, OR = 0.71 (0.32-1.10) in a multivariate-adjusted model II].
Comparison of Functional Outcome Between Groups of BDNF Val66Met Polymorphism and Linear Regression Analysis Before and After Adjustment for Confounding Factors to Prognosticate Lower Functional Outcome and Recovery on the BDNF Val66Met Polymorphism.
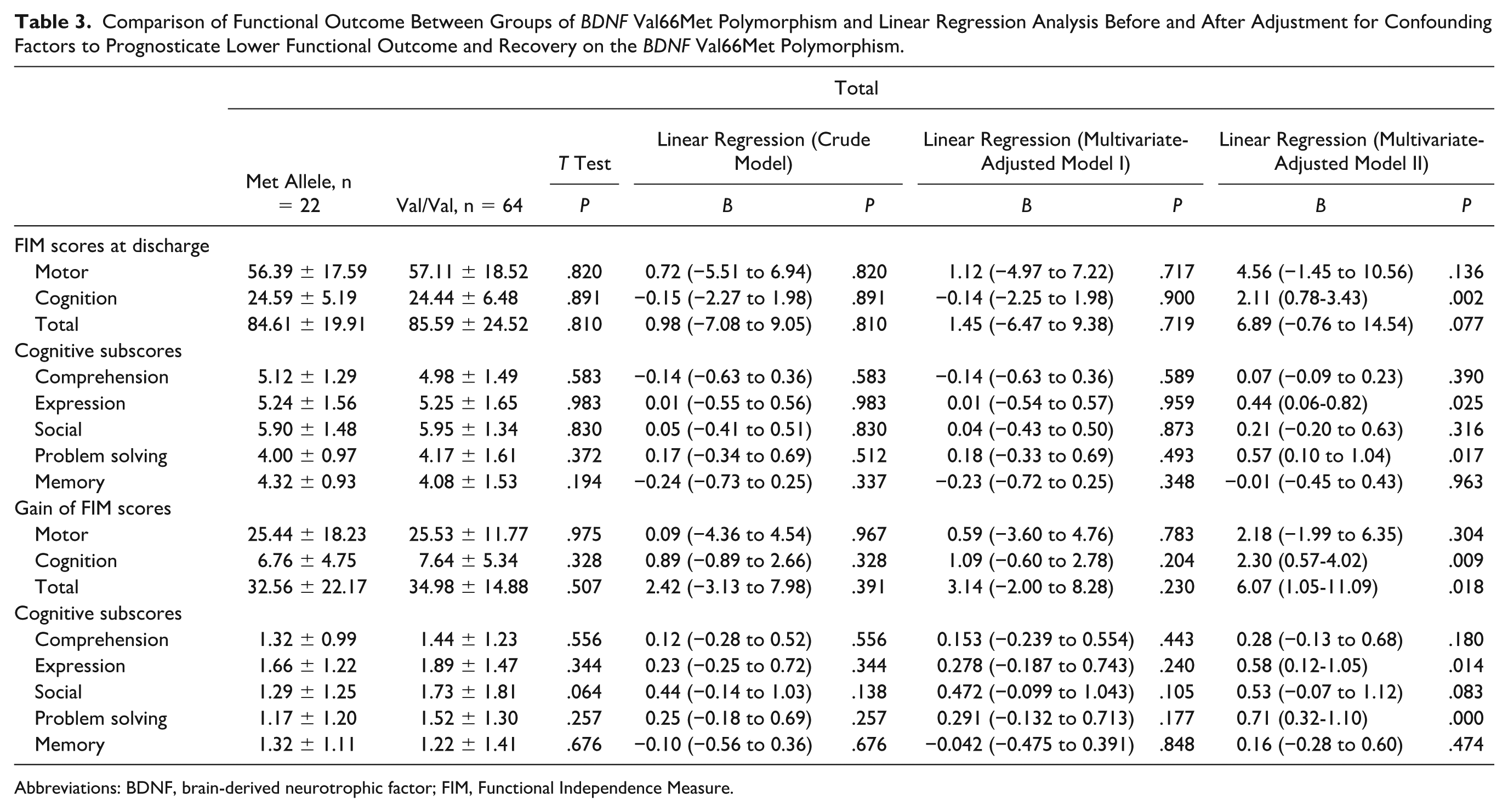
Abbreviations: BDNF, brain-derived neurotrophic factor; FIM, Functional Independence Measure.
In ischemic stroke, patients with a Met alleles genotype had a significant correlation with lower cognitive scores [P = .005: OR = 2.30 (0.73-3.87)], especially in problem-solving subscores at discharge [P = .003; OR = 1.08 (0.38-1.78)] and less cognitive recovery [P = .046; OR = 2.40 (0.04-4.75)], especially expression recovery [P = .025; OR = 0.72 (0.10-1.35)] and social recovery [P = .015; OR = 1.01 (0.20-1.82)] in a multivariate-adjusted model II (supplemental Table I).
In hemorrhagic stroke, Met alleles also had a significant correlation with lower cognitive scores [P = .004; OR = 2.77 (0.93-4.61)], especially in lower expression subscores at discharge[P = .002, OR = 1.15(0.45-1.86) in a multivariate-adjusted model II] and less cognitive recovery [P = .049; OR = 2.39(0.10-4.77)], especially problem-solving recovery [P = .001, OR = 0.81 (0.33-1.29) in a multivariate-adjusted model II; supplemental Tables II and III).
Although in hemorrhagic stroke inpatients with the Met alleles genotype showed a significant correlation with memory subscores [P = .043; OR = −0.65 (−1.28 to −0.20)] in crude models, there was no significant correlation with memory subscores adjusted for covariate variables [P = .059; OR = −0.61 (−1.24 to 0.02)] in a multivariate-adjusted model [P = .702, OR = −0.11 (−0.69 to 0.47) in a multivariate-adjusted model II; supplemental Tables II and III).
In supplemental Table IV, the ratio of the adjusted sum of squares for BDNF Val66Met polymorphism (BDNF) and total sum of squares was used to account for the proportion of variance in cognitive outcome explained by BDNF Val66Met polymorphism. The larger the ratio, the more important the BDNF Val66Met polymorphism is for explaining function outcome. As we show in this table, the ratio for memory function outcome is very small. It indicates that the effect of BDNF Val66Met polymorphism on memory function is less important.
Discussion
The Aim of Our Study Was to Explore the Role of BDNF Val66Met in Inpatient Poststroke Recovery
Our study suggests that the BDNF Val66Met genotype was a significant predictor of inpatient poststroke cognitive outcome and recovery. Currently, the predictive value of BDNF Val66Met polymorphism for functional outcome after stroke has been controversial, even though most studies suggest that BDNF Val66Met polymorphism affects neuroplasticity.
BDNF Val66Met polymorphism reduces intracellular packaging of proBDNF and secretion of mature BDNF.29,30 Mature BDNF interacts with TrkB (tropomyosin receptor kinase B) to play a major role in neuronal survival, synaptic plasticity, learning, and memory, whereas its precursor (proBDNF) promotes apoptosis in the peripheral nervous system by activating p75NTR.31,32 The balance of concentrations of proBDNF and mature neurotrophins is an important element of the cognitive process. 33 Also, BDNF Met allele carriers show disrupted intracellular trafficking of the BDNF-mRNA 34 as evidenced by impaired yet regulated secretion from neurons that results in a significant decrease in available BDNF via acute and chronic effects of neurotransmitter signaling.12,34,35
Some studies found no clear evidence of an association between BDNF Val66Met polymorphism and cognitive performance. 36 On the other hand, a few studies reported that BDNF Val66Met was related to cognitive function and brain morphology.28,36,37 The BDNF Val66Met is believed to correlate to the prefrontal cortex, anterior cingulate cortex, and hippocampal volume.38,39 It can influence neural circuits for cognitive control. 40 BDNF Met alleles played a role in activity-dependent learning rather than affecting brain structure. 41 Met allele carriers were found to increase interregional connectivity between the anterior cingulate cortex and prefrontal cortex and change cognitive network function 42 but decrease hippocampal gray matter volume 40 related to cognitive processes when compared with Val/Val homozygotes.12,40
In line with the latter concept, it was reported that Met alleles negatively affected stroke motor recovery, both in ischemic 43 and subarachnoid hemorrhage.44,45 In addition, rodent studies found that mice with the BDNF Val66Met variant in both Met alleles (BDNFM/M) displayed negative motor performance in the chronic phase of motor recovery.45,46 Furthermore, BDNF Met alleles showed poorer cognitive performance than did BDNF Val homozygote carriers in the Chinese population and in Caucasian adults,47-49 especially in learning and memory.37,41,50,51
This study offers preliminary supporting evidence that Met alleles may negatively affect cognitive performance in inpatient poststroke rehabilitation, mostly with regard to learning and expression and less with regard to memory, although previous research has reported that the BDNF Val66Met polymorphism influences the memory process and plays a prominent role in the regulation of memory. 52 Several other studies found no clear evidence of an association between BDNF Val66Met polymorphism and cognitive performance. 36 Cognitive skills decline with age, supporting the idea that age and vascular risk factors play a more important role in the memory process.53,54 Consistent with this, our results suggest that the above risk factors affect the predictive value of BDNF Val66Met polymorphism in cognitive function. This needs further verification in future research.
Genetic effects on functional outcome differed between patients with ischemic and hemorrhagic stroke. 55 In supplemental Tables II and III, it can be seen that Met alleles have a significant correlation with lower cognitive outcome both in ischemic stroke and hemorrhagic stroke. However, there was a minor difference in the cognitive subscores outcome: the Met allele was associated with lower problem-solving subscores in ischemic stroke and lower expression subscores in hemorrhagic stroke. It is not clear if the BDNF polymorphism plays a different role in cognitive outcome between ischemic and hemorrhagic stroke.
Predictors of functional outcome after stroke rehabilitation are obviously multifactorial. Age has consistently emerged as an important predictor in poststroke rehabilitation.22,56 Gender is a well-established risk factor for rehabilitation, with studies reporting that male gender is associated with a better outcome than female gender after stroke. 57 Furthermore, several studies suggest that not only sociodemographic (such as age, gender), but also stroke-related characteristics (stroke lesion, time from stroke onset to rehabilitation admission, and aphasia) and vascular condition are important factors in predicting stroke recovery.22,28,58-60 In this study, we utilize 3 models (crude, model I, and model II) to investigate the effects of predictors for the outcome. The crude model did not adjust for any covariates; model I adjusted for age, gender, and race; and model II adjusted for age, gender, race, stroke-related characteristics, vascular comorbidities, duration from stroke onset to rehabilitation admission, and aphasia. Model II showed a significant predictive value of Met for lower cognitive function. Our results show that adding these covariates changes the predictive value of BDNF Val66Met polymorphism on functional outcome. This may provide insight into designing an individualized treatment and prevention plan with integration of gene and vascular/sociodemographic risk factors.
Taken together, this study shines light on the mechanism of cognitive outcome in inpatient poststroke rehabilitation and helps identify therapeutic targets to promote cognitive recovery. Individuals with BDNF Val66Met variants may have different responses to therapeutic drugs and/or physical and cognitive treatment. For example, BDNF gene polymorphism influences the effects of rehabilitation treatment such as repetitive transcranial magnetic stimulation on motor recovery and motor learning in stroke patients.61-63 However, more in-depth studies are needed to understand BDNF genotype effects on different aspects of cognitive recovery and long-term outcome. Further research along these lines would help design personalized rehabilitation programs according to BDNF genotype in poststroke patients.
Some limitations of the present study should be considered. This was a cohort study from 1 urban rehabilitation hospital, with the majority of individuals being Caucasian. The relatively small sample size may imply a lack of statistical power. Although the predictive value of BDNF Val66Met for functional outcome in stroke patients has been controversial, our data suggested a statistically significant relationship between Val/Val and the Met genotype in cognitive recovery. A large sample would be needed to illustrate the magnitude of clinical meaningfulness. Therefore, a larger, multicenter study is required to confirm our findings. Although the FIM is a simple, practical, and efficient measure of functional outcome in inpatient settings, this instrument has floor and ceiling effects in addition to shortcomings of being subjective, ordinal, and imprecise. In a future prospective study, it would be important to use a battery of instruments such as the NIH Stroke Scale, Modified Rankin Scale, and Fugl Meyer as well as more in-depth cognitive tests like the Mini-Mental State Examination and Montreal Cognitive Assessment for ADLs and cognitive function.
Finally, recent gene discoveries have found that approximately 35 genetic loci are associated with risk prediction and treatment of stroke. 64 The Apolipoprotein E genotype was associated with functional outcome in poststroke recovery. 65 Also, besides BDNF Val66Met, it is reported that BDNF rs11030119 was independently associated with poor functional outcome after stroke. 65 Therefore, further research using a genome-wide association study (GWAS) with a large sample size is needed to gain a fuller picture of how genetics affect stroke recovery.
In conclusion, this finding suggests that the BDNF Met allele may impede cognitive outcome in inpatients post–stroke rehabilitation. The results may provide insight into designing targeted therapies. Moreover, further studies using functional magnetic resonance imaging to investigate the mechanism of BDNF Val66Met on neural circuits are warranted.
Supplemental Material
Supplemental_Material_10.15_cleancopy – Supplemental material for BDNF Met allele Is Associated With Lower Cognitive Function in Poststroke Rehabilitation
Supplemental material, Supplemental_Material_10.15_cleancopy for BDNF Met allele Is Associated With Lower Cognitive Function in Poststroke Rehabilitation by Zhenxiang Han, Lili Qi, Qinfeng Xu, Mingzhu Xu, Lei Cai, John Wong, Xinjia Hu, Xun Luo, Jiening Wang, Yuling Zhang, Yapeng Li and Qing Mei Wang in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
Qing Mei Wang is the principal investigator of this study, overseeing study design, data collection, interpretation, and manuscript preparation. Zhenxiang Han had an important role in study design, data collection, interpretation, and manuscript preparation. Lili Qi contributed to data interpretation and manuscript preparation. Mingzhu Xu contributed to data collection and data analysis. Qinfeng Xu and Yapeng Li contributed to statistical analysis. We would like to thank Lei Cai, John Wong, Xinjia Hu, Xun Luo, Yuling Zhang, and Jiening Wang for their assistance with data interpretation and manuscript preparation.
This is an observational study that was approved by the Institutional Review Board of Spaulding Rehabilitation Hospital, the teaching affiliate of Harvard Medical School. Data analysis was fully anonymized and, as such, the Institutional Review Board of Spaulding Rehabilitation Hospital waived the need for approval. All the methods were performed in accordance with approved institutional guidelines.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was undertaken and completed at Spaulding Rehabilitation Hospital, the teaching affiliate of Harvard Medical School, MA. Funding to conduct the study was partially supported by the Harvard Catalyst/The Harvard Clinical and Translational Science Center, National Center for Research Resources and the National Center for Advancing Translational Sciences, National Institutes of Health (Award UL1 TR001102); Harvard University and its affiliated academic health care centers; and Fundamental Research Funds for the Central Universities (Grant12ykpy39). The content is solely the responsibility of the authors and does not necessarily represent the official views of Harvard Catalyst, Harvard University, and its affiliated academic health care centers or the National Institutes of Health. Zhenxiang Han was supported by the Outstanding Leaders Training Program of Pudong Health Bureau of Shanghai (Grant PWR12016-02).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
