Abstract
Background and objective. Unilateral spatial neglect (USN) can interfere with rehabilitation processes and lead to poor functional outcome. The purpose of this study was to determine whether prism adaptation (PA) therapy improves USN and functional outcomes in stroke patients in the subacute stage. Methods. A multicenter, double-masked, randomized, controlled trial was conducted to evaluate the effects of a 2-week PA therapy on USN assessed with the Behavioral Inattention Test (BIT), the Catherine Bergego Scale (CBS), and activities of daily living (ADL) as evaluated with the Functional Independence Measure (FIM). A total of 38 USN patients with right-brain damage were divided into prism (n = 20) and control (n = 18) groups. Patients were divided into mild and severe USN groups according to BIT behavioral test (mild ≥ 55 and severe<55). The prism group performed repetitive pointing with prism glasses that induce rightward optical shift twice daily, 5 days per week, for 2 weeks, whereas the control group performed similar pointing training with neutral glasses. Results. The FIM improved significantly more in the prism group. In mild USN patients, there was significantly greater improvement of BIT and FIM in the prism group. Conclusions. PA therapy can significantly improve ADL in patients with subacute stroke.
Keywords
Introduction
Unilateral spatial neglect (USN) is defined as a failure to report, respond, or orient to novel or meaningful stimuli presented to the side opposite the brain lesion. 1 USN occurs much more frequently with right-sided brain lesions than with left-sided lesions; it is reported to occur in 13% to 100% of patients with right-sided stroke. 2 Because the most important clinical problem in USN patients is interference with rehabilitation processes, resulting in poor functional outcome,3-10 various rehabilitation techniques for USN have been explored.11,12
Rossetti and colleagues 13 reported that short-term adaptation for rightward optical shift induced with prismatic lenses improved performance on various standard tests of left USN. In later studies, it was confirmed that the effects of adaptation could generalize across various clinical aspects of USN, 12 including wheelchair navigation,14,15 postural control,16,17 tactile extinction, 18 auditory extinction, 19 and mental imagery.20,21 Long-term effects (up to 5 weeks) were also reported following an intensive, twice-daily adaptation program for 2 weeks. 22 In recent studies, Shiraishi and colleagues 17 reported that the effects of a 50-minute daily program for 8 weeks were sustained for up to 6 weeks and over 2 years. 23 However, few randomized, controlled trials (RCTs) with sufficient sample size have examined the effects of prism adaptation (PA),24,25 and few studies have demonstrated improvement not only in neuropsychological examinations but also in activities of daily living (ADL) with PA.11,12,17,23-26
Furthermore, most previous studies have examined patients in the chronic phase, more than 3 months after stroke onset. Studies of patients in the subacute phase, which we define in this study as the period from 1 month to 3 months after onset, are few, despite the fact that USN is likely to be associated with poor functional outcome in the subacute phase.5,8-10 During this period, intensive rehabilitation is needed to obtain higher ADL scores. In rehabilitating USN patients, deficits of spatial recognition and body image prevent them from learning efficient body movements and increase the risks of falls and collisions. Furthermore, they tend to be unduly limited in their activities because of fall risks, and such limitations decrease the time and quantity of voluntary training. Thus, the functional outcome is lower for USN patients than for non-USN patients. If greater efficacy of rehabilitation can be brought about by early intervention for USN, patients with USN might achieve higher ADL goals.
The purpose of this RCT was to determine whether PA improves USN and functional outcomes for stroke patients with USN in the subacute stage.
Methods
Study Design
A multicenter, double-masked RCT was conducted to evaluate the effects of a 2-week PA therapy on USN and ADL in patients with right-brain lesions. Assessment took place on study entry; posttreatment and follow-up assessments were performed at discharge from the rehabilitation hospital. The study protocol was approved by the Medical Ethics Committee of the Keio University School of Medicine.
Participants
Participants were recruited from rehabilitation departments in the Tokyo Metropolitan Rehabilitation Hospital, Ichikawa Rehabilitation Hospital, Keio University Tsukigase Rehabilitation Center, Kizankai Memorial Hospital, National Hospital Organization Murayama Medical Center, Tsujimura Hospital, Inagi Municipal Hospital, and Asahikawa Rehabilitation Hospital. Patients were screened at admission to the rehabilitation hospitals. Inclusion criteria were as follows: (1) first-ever hemiparetic stroke, (2) admission within 3 months after stroke onset, (3) age 41 to 89 years, (4) no severe cognitive impairment (Mini-Mental State Examination, MMSE >15), (5) right hemisphere damage, and (6) at least 1 item scoring less than the cutoff value in the standard test of the Behavioral Inattention Test (BIT). 27 Exclusion criteria were as follows: (1) unable to sit on a wheelchair, (2) unable to understand the task because of aphasia or other cognitive impairment, (3) unable to understand Japanese, (4) extremely impaired eyesight, (5) severe hearing loss, (6) unable to reach with the right upper extremity because of restricted range of motion, (7) right upper-extremity amputation more proximal to half of the forearm, (8) severe position sense deficits of the right fingers because of peripheral neuropathy, (9) past medical history of head trauma or ventriculoperitoneal shunt, and (10) patients’ refusal to participate.
Outcome Measures
Behavioral Inattention Test
The BIT is a battery that is commonly used to assess spatial neglect. 27 It has conventional and behavioral tests. The conventional test (BIT-C) has 6 items, and the behavioral test (BIT-B) has 9 items. The cutoff scores are determined for each item and the total scores of the conventional and behavioral tests. The assessment could be separated into several sessions within 1 week if the participant felt fatigue to the extent of affecting the scores.
Catherine Bergego Scale (CBS)
The CBS is a validated and reliable ADL measure for USN patients. 28 It is based on direct observation of the patient’s functioning during 10 tasks that correspond to common everyday life situations, including grooming, dressing, and wheelchair driving. For each task, a 4-point scale is used, ranging from 0 (no neglect) to 3 (severe neglect), and a total score is calculated (range, 0-30). The same questions are used for patients and caregivers, thus allowing an estimate of anosognosia.
Functional Independence Measure (FIM)
The FIM, which was developed to ensure uniformity in assessing ADL, has motor and cognitive scores and is subdivided into 18 subcategories. 29 The total score ranges from 18 to 126; higher scores indicate greater independence in ADL.
Stroke Impairment Assessment Set (SIAS)
The SIAS is a comprehensive instrument for assessing stroke impairment, with well-established psychometric properties.30,31 It assesses various aspects of impairment in stroke patients, including motor function, tone, sensory function, range of motion, pain, trunk function, visuospatial function, speech, and sound side function. Motor scores of the SIAS are composed of 5 items that assess arm, finger, hip, knee, and ankle functions and are rated from 0 (severely impaired) to 5 (normal).
Randomization and Masking
Participants were randomly allocated to the intervention or the control group, with a computerized block randomization scheme, including prestratification according to 2 variables: total score of the BIT behavioral test (dichotomized to above or equal to 55 or below 55) and participating hospital. In the previous study that used BIT to evaluate the improvement of USN by PA therapy, 22 the average score of the BIT behavioral test was 54.2. After obtaining written informed consent, baseline assessments were performed within 3 months after stroke onset. All data were sent to the central office at the Department of Preventive Medicine and Public Health, School of Medicine, Keio University, before allocation. The participants, physicians, and hospitals were all unaware of group assignment.
Patient screening and the SIAS assessment were administered by physiatrists. Other outcome measures were rated by physiatrists or therapists trained to use them, masked to the treatment allocation and otherwise uninvolved in the participants’ treatment. The intervention was administered by occupational therapists or speech therapists who were uninvolved in the assessment of the participant.
Interventions
Patients were seated at a desk on which a wooden table (height 30 cm) was placed. The top of the table had a semicircular shape with a radius of 60 cm, and 3 targets were marked at the center, right, and left corners of its edge. Patients sat vertically during each session, with assistance if necessary. The distance between each patient and the targets was adjusted for the patient’s arm length. Patients were asked to point to the 3 targets through the bottom of the table with the index finger of the right nonparetic hand, according to the instructions of the therapists, who randomly indicated 3 points to the patients. The reaching trajectories were hidden by the table. Patients could see their own finger only at the edge of the table. Patients were asked to reach and bring back their hand as quickly as possible without correction of pointing error. First, they repeated the pointing task 30 times without prism glasses. Then, they wore prism glasses that shifted their visual field 12° to the right (Fresnel lens: Koyo Corporation, Tokyo, Japan) and repeated pointing 90 times. After PA, they took the prism glasses off and repeated pointing 60 times. During this postexposure period, patients could recognize and correct rightward pointing error.
Patients underwent 2 daily training sessions that took about 20 minutes each, 5 days a week for 2 weeks, for a total of 20 sessions. 22 Control group patients underwent the same training sessions with neutral plastic glasses instead of prism glasses.
For both groups, the interventions occurred during regularly scheduled rehabilitation sessions; all other routine interdisciplinary stroke rehabilitation was provided as usual.
Data Collection
Measurements were done by masked raters (Figure 1) on study entry (T0 = baseline), just after the 2-week intervention (T1 = posttreatment), and at discharge (T2 = follow-up). The assessments are performed within 1 week before the study entry, after the end of intervention, and before discharge.
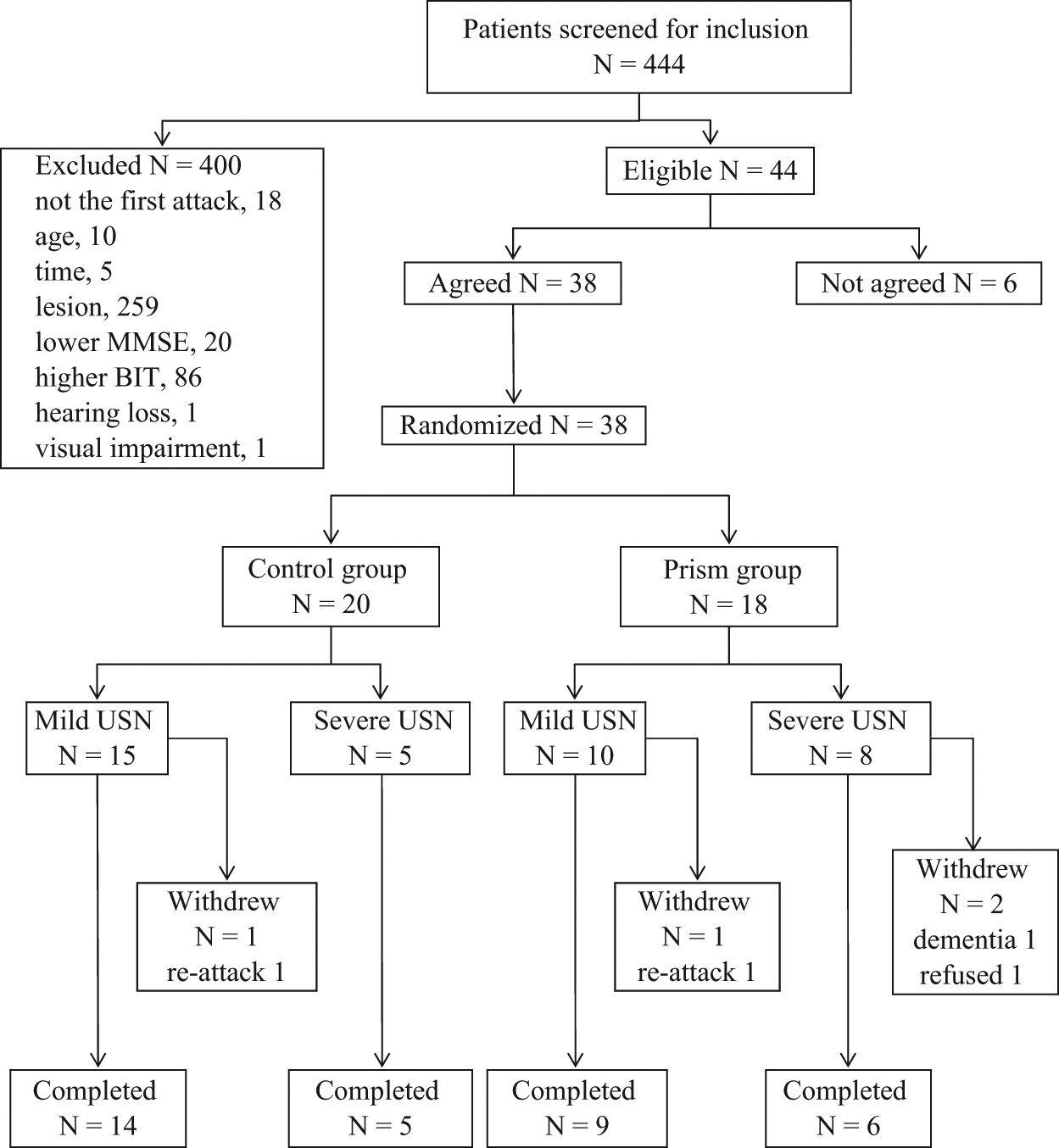
Data collection and rehabilitation protocol. Abbreviations: MMSE, Mini-Mental State Examination; BIT, Behavioral Inattention Test; USN, unilateral spatial neglect.
Statistical Analyses
Data were analyzed with SPSS (version 17.0, SPSS Inc). The delays between the end of treatment and discharge were compared between the 2 groups (control and prism) using the Mann-Whitney test. Changes from T0 to T2 of the BIT-C, BIT-B, and CBS were compared between the 2 groups using the Mann-Whitney test. An analysis of variance (ANOVA, P < .05) was conducted with the 2 factors of time (T0, T1, and T2) and group (control and prism) in the BIT-C, BIT-B, CBS, and FIM. Bonferroni post hoc analysis was performed when the Group × Time interaction was significant. Total FIM gains, calculated by subtracting T0 (FIM gain 1) or T1 (FIM gain 2) from T2, were compared between the 2 groups (control and prism) using the Mann-Whitney test. P values less than .05 were considered significant (Figure 1).
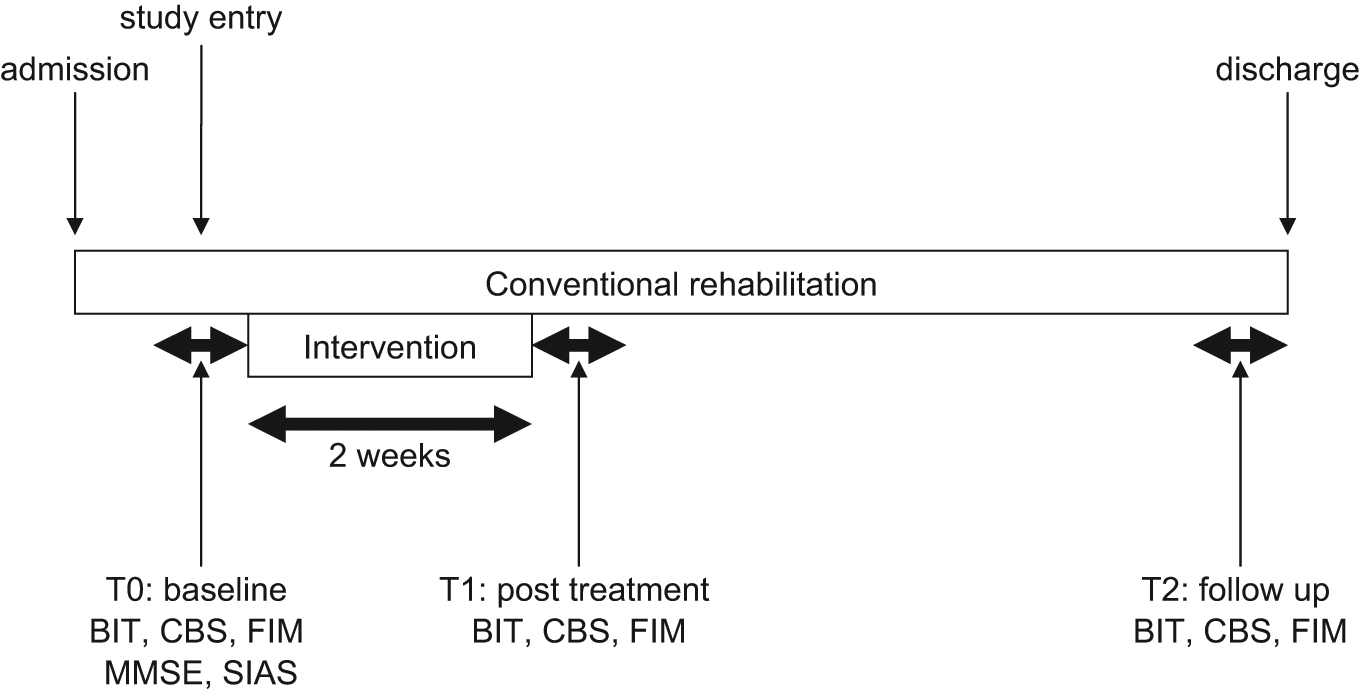
Study design: baseline assessments (T0) were performed in the last week before intervention, and posttreatment assessments (T1) were performed 2 weeks after intervention. Follow-up assessments (T2) were performed just before discharge. All tests were performed within 1 week. Conventional rehabilitation was provided throughout hospitalization. Abbreviations: BIT, Behavioral Inattention Test; CBS, Catherine Bergego Scale; FIM, Functional Independence Measure; MMSE, Mini-Mental State Examination; SIAS, Stroke Impairment Assessment Set.
Results
Participants
Between August 2005 and March 2007, 444 patients were screened; 38 patients (27 men and 11 women) met the criteria and agreed to participate in the study, of whom 34 (89.5%) completed the intervention and follow-up FIM evaluation. Participants were allocated to the prism (n = 20) and control (n = 18) groups. Before allocation, participants were classified into mild (n = 25) and severe (n = 13) groups according to the BIT-B score. There was no patient who could not complete the reaching task because of fatigue or cognitive dysfunction. Four participants, 1 in the control group and 3 in the prism group, dropped out: 2 had a stroke relapse; 1 rejected the treatment during the intervention period; and 1 developed delirium, which precluded the intervention (Figure 2).
In all, 31 participants completed the follow-up BIT test at discharge; 3 participants, 1 in the control group and 2 in the prism group, could not complete the follow-up because of poor physical condition. For the CBS assessment, 33 participants completed the follow-up test. One control group patient was not evaluated with the CBS because of assessor’s error.
There were no significant differences in baseline characteristics and scores between participants who did and did not complete the intervention and follow-up evaluation (unpaired t test, P > .05). Therefore, analyses were conducted using data sets containing T0, T1, and T2 scores of the BIT (n = 31), CBS (n = 33), and FIM (n = 34). Masking was preserved in all cases.
Group Characteristics
No significant differences were found at baseline between the prism and the control groups with regard to the mean days from onset to intervention, mean hospital stay, MMSE score, and SIAS motor score (Table 1).
Baseline Characteristics and Outcome Scores a
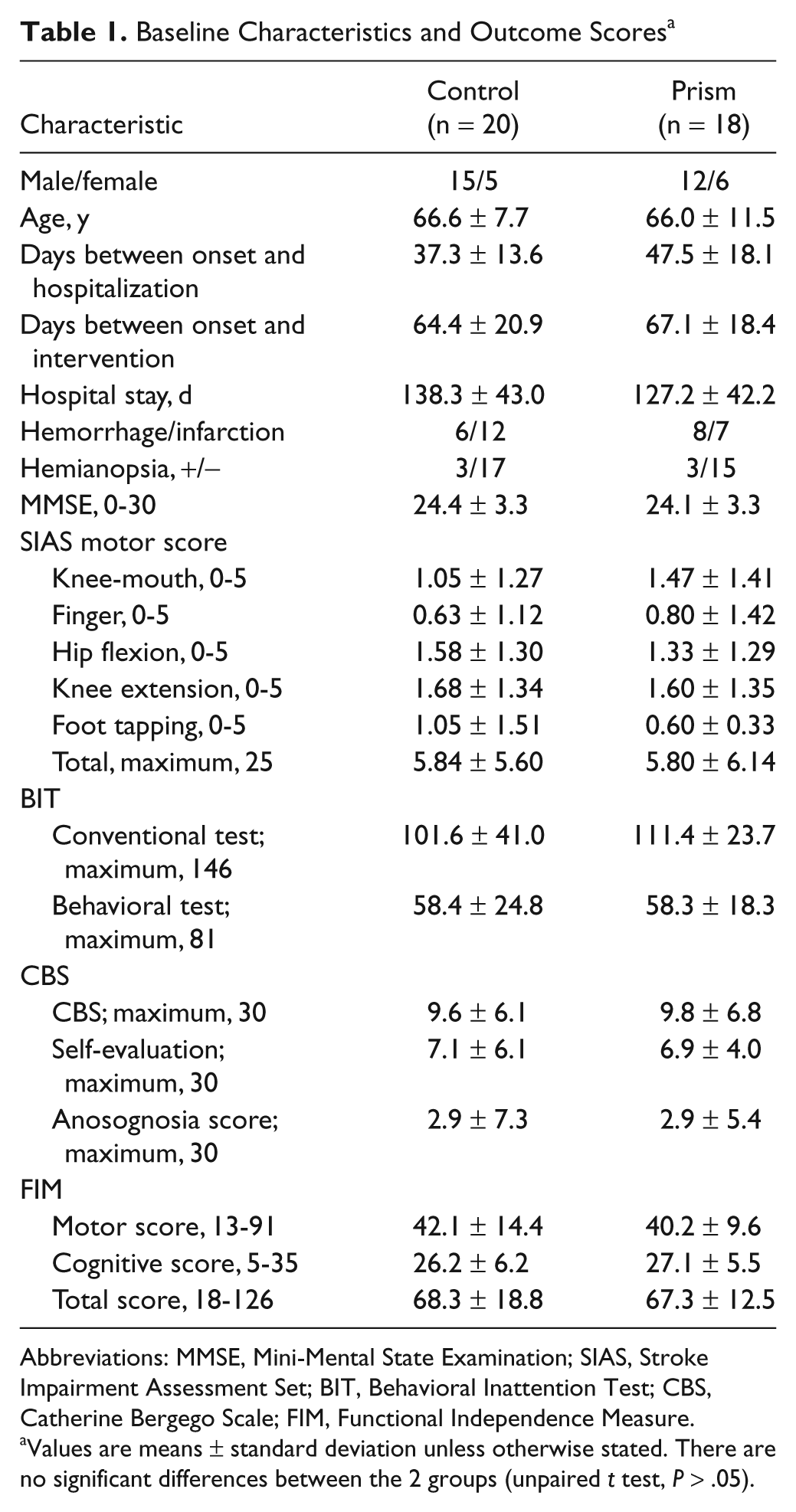
Abbreviations: MMSE, Mini-Mental State Examination; SIAS, Stroke Impairment Assessment Set; BIT, Behavioral Inattention Test; CBS, Catherine Bergego Scale; FIM, Functional Independence Measure.
Values are means ± standard deviation unless otherwise stated. There are no significant differences between the 2 groups (unpaired t test, P > .05).
Delay Between the Treatment and the Release
The mean delays between the end of the PA and discharge were 99.3 ± 49.0 days in the control group and 95.5 ± 41.2 days in the prism group. In the mild USN patients, they were 83.9 ± 32.9 days in the control group and 101.1 ± 43.4 days in the prism group. In the severe USN patients, they were 142.4 ± 64.3 days in the control group and 82.1 ± 40.0 days in the prism group. In the severe USN patients, there was a tendency for the delay to be shorter in the prism group than in the control group. However, no significant differences were found in all the severities (Mann-Whitney test).
Effect of PA on Specific Tests for Neglect
The results of the BIT and the CBS are shown in Table 2. The first upper part demonstrates the results for all participants, and the lower 2 parts demonstrate those belonging to the mild and severe groups (BIT-B ≥ 55 or <55).
Comparison of Specific Tests for Neglect Between Control and Prism Groups a
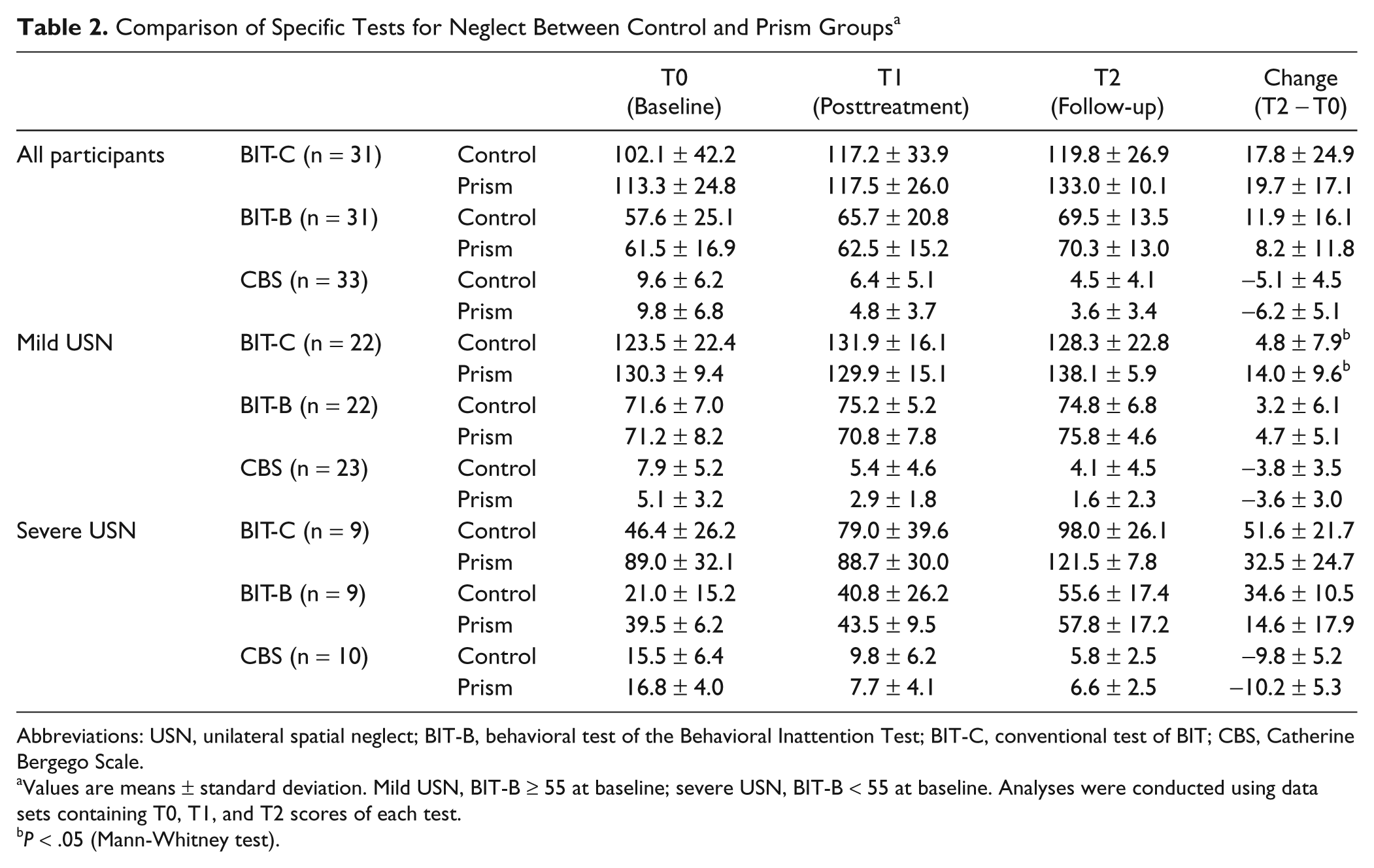
Abbreviations: USN, unilateral spatial neglect; BIT-B, behavioral test of the Behavioral Inattention Test; BIT-C, conventional test of BIT; CBS, Catherine Bergego Scale.
Values are means ± standard deviation. Mild USN, BIT-B ≥ 55 at baseline; severe USN, BIT-B < 55 at baseline. Analyses were conducted using data sets containing T0, T1, and T2 scores of each test.
P < .05 (Mann-Whitney test).
Both the prism and the control groups had improved total scores of the BIT-C, the BIT-B, and the CBS during hospitalization. Although the scores of BIT-C and CBS tended to improve more in the prism group than in the control group, no significant differences were found in the score at each time point and in the change from T0 to T2 (Mann-Whitney test). With a 2-factor ANOVA, there were significant effects of time in all tests (BIT-C: F[2, 58] = 13.505, P < .001; BIT-B: F[2, 58] = 12.337, P < .001; CBS: F[2, 62] = 33.879, P < .001). However, the 2-way Group × Time interaction was not significant for all tests.
In patients with mild USN (BIT-B ≥ 55), there was a significant difference in the change of the BIT-C from T0 to T2 (Mann-Whitney test, P < .05). Although the scores of BIT-B and CBS tended to improve greater in the prism group than in the control group, no significant differences were found in the score at each time point and in the change from T0 to T2 (Mann-Whitney test). There were significant effects of time in all tests (2-factor ANOVA; BIT-C: F[2, 40] = 7.366, P < .01; BIT-B: F[2, 40] = 5.515, P < .01; CBS: F[2, 42] = 20.931, P < .001). However, the 2-way Group × Time interaction was not significant in all tests.
In patients with severe USN (BIT-B < 55), the total scores of the BIT-C and the BIT-B tended to be higher in the prism group than in the control group at T0 (Mann-Whitney test: BIT-C, P = .065; BIT-B, P = .050). No significant differences were found in the score at each time point and change of all test scores from T0 to T2 (Mann-Whitney test). There were significant effects of time in all test scores (2-factor ANOVA; BIT-C: F[2, 14] = 15.193, P < .001; BIT-B: F[2, 14] = 18.091, P < .001; CBS: F[2, 16] = 21.120, P < .001). However, the 2-way Group × Time interaction was not significant in all tests.
Effects of PA on Changes in ADL
The total FIM scores of the prism and control groups at T0, T1, and T2 are shown in Figure 3. As a whole (Figure 3A), both groups improved their total FIM scores during hospitalization. With 2-factor ANOVA, there was a significant effect of time: F(2, 64) = 104.231; P < .001. However, the 2-way Group × Time interaction was not significant.
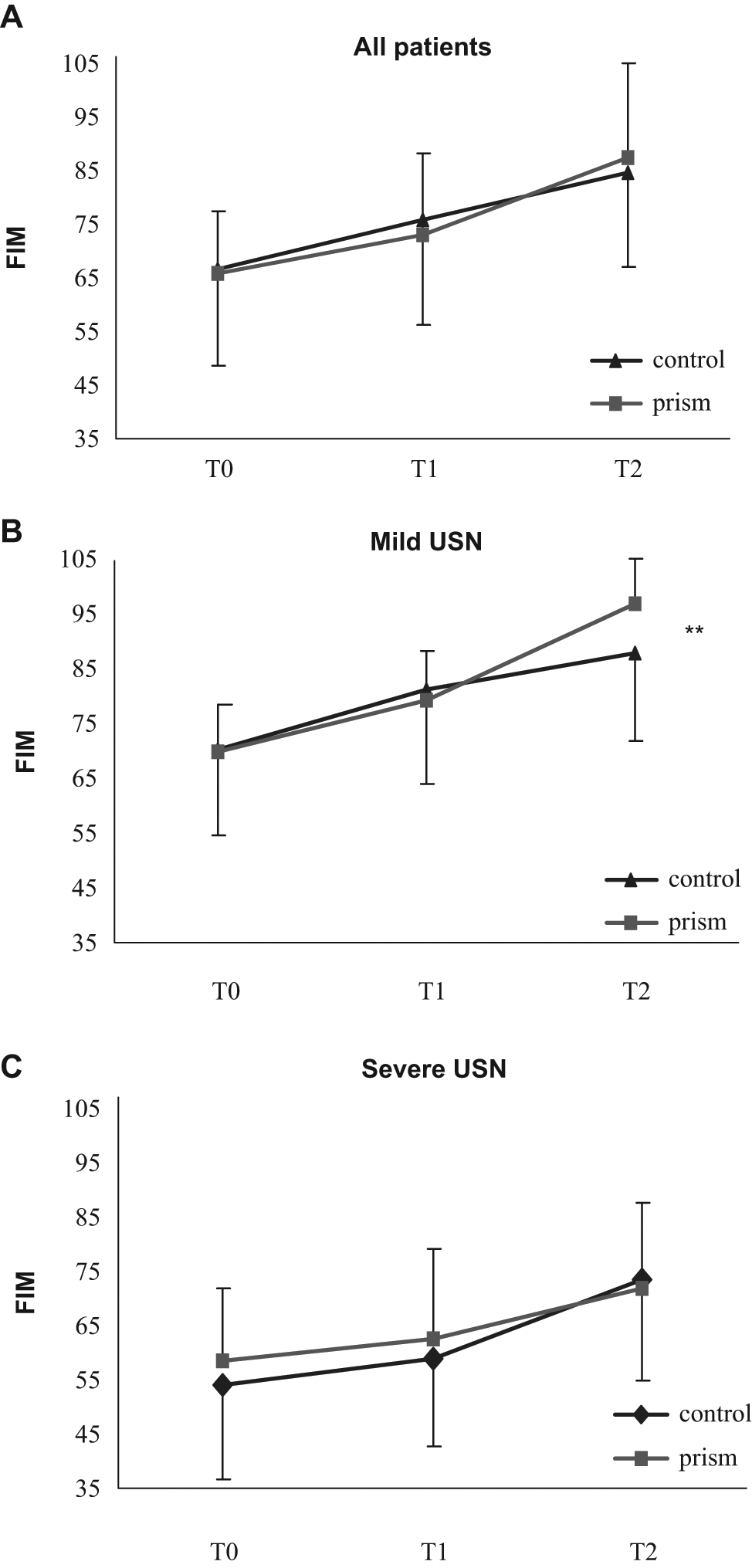
Total FIM scores of control group (filled triangle) and prism group (filled rectangle) participants. The FIM was rated at admission (baseline, T0), just after treatment (posttreatment, T1), and at discharge (last, T2). Error bars indicate standard deviation. A. Total FIM scores of all patients. B. Total FIM scores of mild USN patients (BIT-B ≥ 55 at baseline). C. Total FIM scores of severe USN patients (BIT-B < 55 at baseline). Abbreviations: USN, unilateral spatial neglect; FIM, Functional Independence Measure; BIT-B, behavioral test of the Behavioral Inattention Test.
In patients with mild USN (Figure 3B), both groups improved their total FIM scores during hospitalization, and there were significant effects of time (2-factor ANOVA, F[2, 42] = 113.749; P < .001) and Group × Time interaction (F[2, 42] = 7.761; P < .01). Post hoc tests showed a significant time effect between all pairs of time points in both groups.
In patients with severe USN, both groups improved their total FIM scores during hospitalization. With 2-factor ANOVA, there was a significant effect of time (F[2, 18] = 26.042; P < .001). However, the 2-way Group × Time interaction was not significant.
FIM changes in both groups are shown in Figure 4. There were no significant differences in the change from T0 to T2 (FIM gain 1; Mann-Whitney test; Figure 4A). The change from T1 to T2 (FIM gain 2) was significantly larger in the prism group than in the control group (Mann-Whitney test, P < .05). In patients with mild USN, FIM gains 1 and 2 were significantly larger in the prism group than in the control group (FIM gain 1, P < .05; FIM gain 2, P < .01; Figure 4B). In patients with severe USN, no significant differences were found between the 2 groups (Figure 4C).
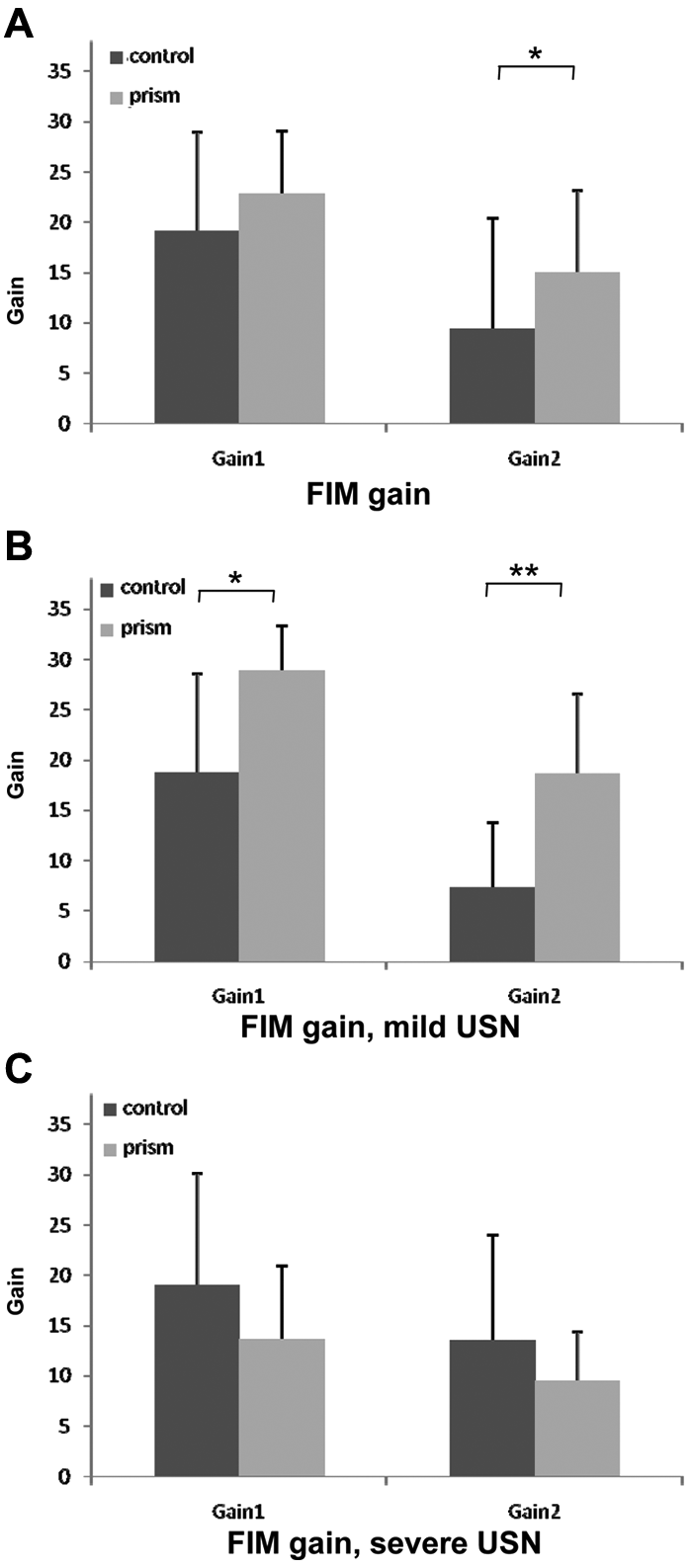
FIM gains of control group (black columns) and prism group (gray columns) participants: error bars indicate standard deviation. Gain 1: change between baseline (T0) and final assessment (T2). Gain 2: change between posttreatment (T1) and final assessment (T2). A. Total FIM scores of all patients. B. Total FIM scores of the mild USN patients (BIT-B ≥ 55 at baseline). C. Total FIM scores of the severe USN patients (BIT-B < 55 at baseline). Abbreviations: FIM, Functional Independence Measure; USN, unilateral spatial neglect; BIT-B, behavioral test of the Behavioral Inattention Test.
Discussion
This is the first RCT confirming the effectiveness of PA for USN in patients with subacute stroke; the 2-week PA therapy significantly increased FIM gain after finishing the PA therapy until discharge. The difference in FIM gain between the prism and the control groups was greater during this period than during the whole hospitalization period. This indicates that PA facilitated motor and cognitive relearning after the 2-week treatment, and the prism group achieved higher ADL goals than the control group. The sample size of this study is the largest among the studies on PA effectiveness reported so far, and it compares fairly well with studies reporting other USN treatments.11,12,17,22,24,25,32 In this double-masked RCT, the drop-out rate was small (10.5%), and baseline comparability of the PA and the control groups was relatively good.
The BIT and CBS scores showed trends toward greater improvement in the prism group than in the control group. However, the amount of improvement was not significantly different between the 2 groups when analyzed for the group as a whole. Most previous studies11-18,22,24,25,32 reported improvement of USN with PA in patients who were several months or more after stroke onset. Possible reasons for failing to demonstrate greater improvement of USN with PA as compared with the control group during the subacute phase are as follows: (1) the differences are masked because spontaneous recovery might have occurred in both groups; (2) repetition of the tests and conventional rehabilitation might have caused a treatment effect in the control group; and (3) there might have been ceiling effects of the outcome measures because of higher average baseline scores of BIT in the prism group than in the control group in patients with severe USN.
When analyzed according to USN severity, the BIT-C score improved more in the prism group than in the control group in patients with mild USN (BIT behavioral test score ≥55), whereas there was no significant difference between the 2 patient groups with severe USN (BIT behavioral test score < 55). PA may improve directional attention to leftward, and therefore, more improvement was observed with the BIT-C, in which cancellation tasks are weighted, as compared with the BIT-B and the CBS. Several previous studies17,33 reported that PA was more effective for cancellation and line bisection tasks than for visuoconstruction (ie, copying and drawing). These findings support our results. In addition, the difference in FIM gain between the prism and the control groups was greater, and a significant interaction effect was seen in the total FIM scores between the 2 groups with 2-factor ANOVA in patients with mild USN. These results suggest that PA is effective both for the neglect symptoms and improvement of ADL in mild USN patients. This may be because capacities for brain plasticity and learning were larger in mild than in severe USN patients.
No significant difference between the prism and control groups was found with regard to improvement of BIT and FIM scores in patients with severe USN. One possible reason is that severe USN patients are affected by other physical and cognitive impairments except for USN such as hemiparesis and other higher cortical dysfunctions. However, it is too early to conclude that PA is ineffective for them. The number of patients with severe USN in our study was small, and the possibility of selection bias cannot be ruled out because the BIT score of the prism group was higher than that of the control group despite comparable baseline FIM scores. In addition, the length of hospital stay after PA was longer in the control group with severe USN than in the prism group, though the difference was not statistically significant. This could have caused greater improvement of BIT and FIM in the control group.
In this study, we found greater improvement in FIM than in BIT and CBS. The FIM is a widely used assessment for ADL. Although the CBS is also a scale of ADL, it is used to assess only the difficulties resulting from USN. There are few rehabilitation techniques that have previously demonstrated significant improvement of ADL in patients in the chronic phase.11,12 It may be difficult to generalize the improvements of USN symptoms to ADL. However, in the earlier poststroke phase, patients have a larger capacity for brain plasticity and could obtain higher ADL by rehabilitation. Therefore, the improvement of USN might have been generalized to ADL in the prism group of this study. A recent neuroimaging study 33 supports our result. Luauté et al 33 reported that the therapeutic effect of PA is related to the activation of the corticocerebellar network, whereas Farné et al 34 demonstrated that patients with right-brain damage, including USN patients, also had impaired performance of ipsilateral hand movement. In the previous studies, it was reported that the effectiveness of PA generalized to the behavioral level, such as wheelchair driving14,15 and postural control.16,17 These generalized effects may be beneficial to daily rehabilitation training. It is likely that PA can improve not only spatial attention but also the motor performance of USN patients and enhance rehabilitation in the subacute phase.
The effectiveness of top–down approaches for USN has been demonstrated,11,12 but these approaches require patients’ awareness of their disorder and a longer training period.11,12,35 The advantage of PA is that the effects can be obtained with a shorter period of training (2 weeks), and it does not require patients’ awareness of the disorder.11-13,22,35 Therefore, PA can be integrated into the rehabilitation program in a practical manner, and patients can perform conventional ADL training concurrently with PA.
In this study, we selected patients at an earlier poststroke phase as compared with most of the previous studies. For patients in the earlier poststroke phase (mean days from onset = 57.3), in accordance with our study, Rousseaux and colleagues 36 reported the ineffectiveness of PA when measured using reading, cancellation, line bisection, and scene drawing tests. However, they compared only the effects of a single session with 10° prisms and with neural glasses in the same patients, and they did not estimate the cumulative effect of repetitive PA sessions. Nys et al 24 reported the effect of a 4-days-in-a-row PA session for acute patients (mean days from onset = 9.7). The patients in the experimental group demonstrated earlier recovery in cancellation and bisection than those in the control group. However, there was no significant difference in BIT at 1 month posttreatment. These 2 studies24,36 examined the effect of PA for patients in the early poststroke phase, similar to our study, but they only assessed neuropsychological measures. The long-lasting effect of PA on ADL was verified for the first time in our study.
Previous studies on the effectiveness of PA targeted patients in the chronic phase or a longer period,11-13,17,32 and no RCT demonstrated the improvement of ADL at discharge in patients in the early poststroke stage. USN is one of the most important predictors of poor rehabilitation outcome during this period.8-10 Nys et al 24 reported that the existence of USN within 3 weeks after stroke onset was a strong predictor of functional outcome at 6 to 10 months after the first evaluation, although most patients no longer demonstrated USN at that time. In this study, we evaluated the long-term effects of PA at discharge, whereas previous studies22-24,32,37 evaluated them at constant time points, such as 1 month after treatment. Therefore, the delays from treatment to follow-up were variable in this study. In the study with the acute patients, Nys et al 24 could demonstrate no significant difference in BIT scores between the prism and the control groups 1 month after the treatment. In the early poststroke period, the recovery rate of a patient is affected by various factors, such as age, brain lesion, stroke type, severity of paresis, and so on. It may be more valid to evaluate patients at a given landmark in their own functional evolution than at a constant delay after the intervention. In addition, the average delay postintervention of this study was more than 3 months. We therefore could demonstrate a remarkably long-term effect of PA compared with previous studies.22-24,32,37 This study suggests that rehabilitation outcome could be improved by integrating PA in a conventional rehabilitation program for early poststroke patients.
In our PA procedure, patients repeated pointing after the prism exposure with visual feedback of pointing error. In this phase, the visuomotor misalignment caused by PA was reduced forcedly. This misalignment may compensate the rightward motor bias of neglect patients. Therefore, therapeutic effects may be reduced by this forced reduction of the aftereffect. On the other hand, this effect could play a negative role in recovery for the patients in everyday life because it provides error signals that actions are biased to the left. Our method could avoid this negative effect. In addition, the reduction of visuomotor misalignment through the postexposure pointing may also favor the capacity of patients to adapt by a larger amount during the following session. The added pressure for adaptation resulting from the stimulated forgetting may produce longer-lasting retention of the positive effect of PA. Serino et al 37 reported that aftereffects of PA did not correlate either with neglect amelioration or the leftward deviation of eye movements, though error reductions correlated with both. Our method may reinforce the error reduction process of PA even though it reduces visuomotor misalignment. In future studies, it should be confirmed that other aftereffects of PA, such as straight ahead pointing and oculomotor deviation, would be retained even after such forced de-adaptation. In addition, changes in pointing errors and clinical tests throughout repetitive PA sessions should be compared between those with and without a forced de-adaptation process to validate whether this process is helpful in treatment or not.
A few limitations of this study warrant consideration. First, it was likely that spontaneous recovery influenced our results because the period of intervention was earlier than in previous studies. Second, because the number of patients with severe USN was small, the possibility of selection bias cannot be ruled out. Third, because of the sample size limitation, subgroup analysis of the responses to PA could not be sufficiently done, such as comparing ischemic versus hemorrhagic stroke, disease types, locations of lesion, and so on. In addition, because USN is classified into several subtypes based on patients’ symptoms, the difference in responses to PA among various subtypes needs clarification.
Despite these limitations, this is the first study to demonstrate the facilitatory effects of PA for ADL improvement in patients with USN in the subacute phase of stroke.
Footnotes
Acknowledgements
We thank all research staff who executed the treatment and assessment protocols.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by funds from the Tokyo Metropolitan Rehabilitation Hospital.
