Abstract
Background
Robotic assisted movement has become an accepted method of treating the moderately-to-mildly impaired upper limb after stroke.
Objective
To determine whether, during the subacute phase of recovery, a novel type of robotic assisted training reduces moderate-to-severe impairment in the upper limb beyond that resulting from spontaneous recovery and prescribed outpatient therapy.
Methods
A prospective, randomized, double-blinded, placebo-controlled, semi-crossover study of 83 participants. Over 6- to 9-weeks, participants received 18, 30-min training sessions of the hand and wrist. The test intervention consisted of assisted motion, biofeedback, and antagonist muscle vibration delivered by a robotic device. Test Group participants received the test intervention, and Control Group participants received a placebo intervention designed to have no effect. Subsequently, Control Group participants crossed over to receive the test intervention.
Results
At enrollment, the average age (±SD) of participants was 57.0 ± 12.8 year and weeks since stroke was 11.6 ± 5.4. The average Fugl-Meyer baseline score of Test Group participants was 20.9, increasing by 10.8 with training, and in Control Group participants was 23.7 increasing by 6.4 with training, representing a significant difference (4.4) in change scores (P = .01). During the crossover phase, Control Group participants showed a significant increase in FMA-UL score (i.e., 4.7 ± 6.7 points, P = .003) as well as in other, more specific measures of impairment.
Conclusions
Robotic impairment-oriented training, as used in this study, can significantly enhance recovery during the subacute phase of recovery. Spontaneous recovery and prescribed outpatient therapy during this phase do not fully exploit the potential for remediating moderate-to-severe upper limb impairment.
ClinicalTrials.gov Registry: NCT00609115–Subacute stroke rehabilitation with AMES
Introduction
Each year in the United States alone, about 800,000 people have their first stroke, contributing to a population of about 3 million survivors with moderate-to-severe, chronic, sensorimotor impairment.1,2 The most commonly observed motor impairment associated with stroke is weakness in finger extension,3,4 a condition that severely restricts hand-opening on the affected side and limits the performance of many activities of daily living. 5 By 6 months post-stroke, about 50% of stroke survivors are challenged with upper limb (UL) dyscoordination, many never regaining any functional use of the affected UL.6–8
There are few therapeutic options for restoring normal motor function in persons with moderate-to-severe UL impairments post-stroke.9–11 Current therapeutic approaches for this group of stroke survivors focus on compensation and accommodation,12–14 rather than physiological restoration of normal or quasi-normal movement. This finding reflects the prevailing clinical view that patients whose affected UL remains severely impaired 1-month post-stroke are unlikely to recover any normal function in the long term.14–17
Robotic assisted motion devices for the UL developed over the last 2 decades may provide an alternative rehabilitative approach for patients recovering from stroke, as these devices can provide intensive, repetitive, task-oriented training.18,19 Previous studies have shown that the efficacy of robot-assisted training is generally comparable to that obtained with conventional approaches, 20 in patients with moderate-to-mild UL impairment. However, patients with more severe impairments remain underserved by available therapeutic methods,7,9 possibly due to the absence of effective restorative approaches and the current focus on accommodative training in these patients.
The robotic assisted motion device and intervention used in this study (AMES Technology, Inc., Portland, OR) were designed specifically to mitigate physiological impairments that limit hand and wrist function, including weakness, spasticity, and somatosensory deficit.9,18,21,22 Two impairment-addressing features of the investigational device used in this study distinguish it from other robotic assisted motion devices. First, high-frequency mechanical vibration (60 pulses/s) is applied to finger, thumb, and wrist tendons while the respective muscles are lengthening in order to augment proprioceptive input related to the robotically applied movement.23,24 Second, assisted motion involves the patient assisting the motion of the investigational device rather than the device assisting the motion of the patient, which allows for accurate measurement and feedback of the participant’s volitional contribution to UL motion. The movement imposed by the investigational device consisted of simple flexion and extension of the fingers and thumb or the wrist—in other words, the movement was simple and largely nonfunctional.
The purpose of this study was to determine whether the test intervention, combined with prescribed outpatient training, would augment recovery during the subacute stage of stroke recovery compared to prescribed outpatient training only. The primary measure of impairment in this study was the Fugl-Meyer Assessment for the upper limb (FMA-UL). Test Group FMA-UL scores were expected to increase during the training period significantly more than those of the Control Group, and Control Group scores were expected to show a significant improvement during the crossover training period. Such an outcome would suggest, firstly, that prescribed outpatient therapy does not fully exploit peoples’ potential to recover from moderate-to-severe impairment during the first 6 months post-stroke, and that robotic assisted motion devices such as the one used in this study can potentially provide the means to exploit this unused potential.
Methods
This study was conducted at the Rehabilitation Institute of Chicago (Shirley Ryan Ability Lab), University of California San Francisco, Emory University (Atlanta, GA), Legacy Health System (Portland, OR), Northwest Medical Rehabilitation (Spokane, WA), and Eisenhower Medical Center (Rancho Mirage, CA). Figure 1 presents a flowchart of the study design. The study protocol and associated documents were reviewed and approved by the Human Subjects Review Board at each study site. Study design and overall procedures.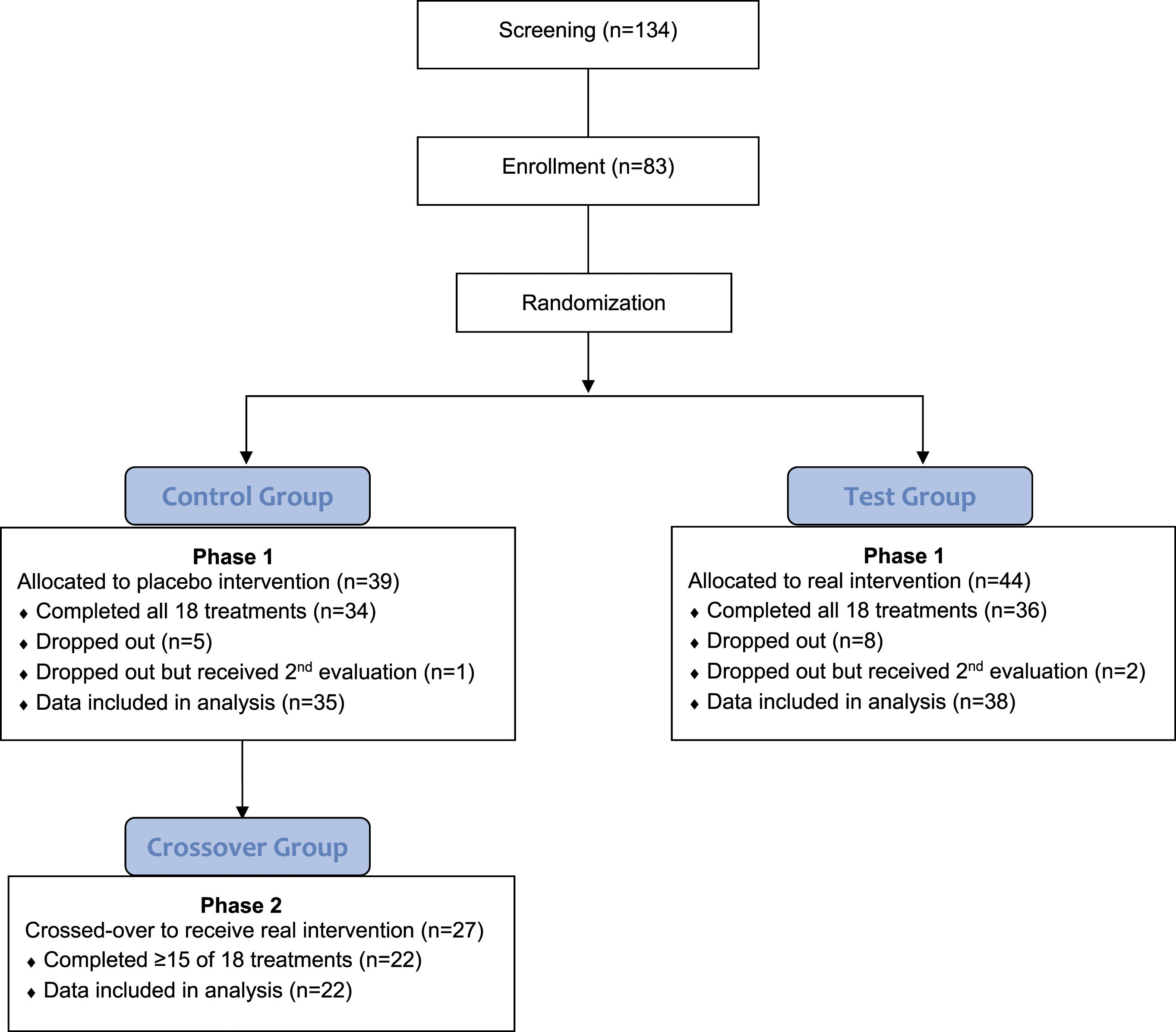
Participants
Based on data reported by Hesse et al. (2005) 25 and a MCID change score of 10 points for the FMA-UL,26,27 a power analysis provided a recruitment target of 90 participants with 45 in each group assuming a SD = 16 25 with an α = .025, a power = .8 and a correlation between baseline and post-training of .5. Prospective participants (n = 134) were recruited from inpatient stroke facilities, advertisements, and physician referrals. Once medical approval was granted by candidates’ personal physicians, the Study Physician at each study site screened each candidate to determine eligibility.
Inclusion criteria for the study included the following: (1) first cortical or sub-cortical hemispheric stroke (ischemic or hemorrhagic); (2) 18–80 years old; (3) baseline FMA-UL score of ≥6 and ≤43 28 ; obtained no earlier than the fifth week post-injury, 14 (4) visible voluntary movement of the affected wrist and at least 1 digit in at least 1 direction; (5) at least partially functioning proprioception from the affected UL (Joint Position Test29,30 ≥70 correct); (6) Beck Depression Inventory score of <19 out of 63; and (7) physically and cognitively able to complete all 18 training sessions.
Exclusion criteria included the following: (1) flaccidity of the hand or wrist; (2) UL limitations not associated with the stroke; (3) severe loss of sensation in the UL; (4) cardiopulmonary compromise; (5) major active psychiatric disorder; (6) severe apraxia; (7) size of arm incompatible with the investigational device; (8) severe contracture or skin condition of the UL; (9) progressive neurodegenerative disorder of the UL; (10) uncontrolled seizure disorder; (11) abuse of alcohol or drugs; (12) terminal illness with anticipated survival of <12 months; (13) current or planned concurrent participation in another study; (14) NIH Stroke Scale, with the following scores: Item 1a >0; Item 1c > 0; Item 2 > 0; Item 3 > 0; Item 11 > 2; Item 12 > 1; Item 13 > 0 (i.e., consciousness, vision, and attention) based on an exam administered by the Study Physician; (15) Rankin Scale score 0–2; and (16) intent to receive Botox injections, to begin antispastic medication, or to use any other robotic or stimulation device during the study period.
Enrollment into the study was based on the Study Physician’s review of the candidate’s medical records and screening exam. A total of 83 candidates were enrolled in the study and randomized by the Study Coordinator to a test training group (“Test Group”) or a control training group (“Control Group”). Each site was provided a unique computer-generated randomization table to follow without replacement for dropouts. All but 2 Control Group participants and 2 Test Group participants received prescribed outpatient therapy in parallel with the study. Typically, this UL rehabilitation included passive stretching, strengthening, and accommodative training.
Overall Study Design and Procedures
The study was carried out in 2 phases (Figure 1). In Phase 1, Test Group participants received the real intervention, and Control Group participants received a placebo intervention, both groups using the same robotic device. In addition, both groups were allowed to pursue any ongoing outpatient therapy in parallel with the study. Records were kept at each site for each participant’s time spent in prescribed outpatient therapy.
At the time of enrollment, all participants were informed that they would be randomized to either a “real” intervention or a placebo, but that participants randomized to the Control Group in Phase 1 would, subsequently, be allowed (but not required) to continue in Phase 2 with a full 18 training sessions with the real intervention. The usual interval between the last (placebo) training session of Phase 1 and the first crossover session of Phase 2 was 1–2 weeks, but Phase 2 for some participants extended into the seventh or eighth month post-injury.
Prior to enrollment of the first participant, one or more therapists or therapy assistants at each study site were assigned the role of trainer, and staff was standardized in the use of the investigational device for training and administering the Strength and Active Motion Tests. In addition, a different therapist at each study site was assigned the role of evaluator and was standardized in administering the FMA-UL, 28 Stroke Impact Scale (SIS), 31 Modified Ashworth Scale (MAS), 32 and Rancho Los Amigos (RLA) 33 assessment tools to persons with UL hemiparesis.
All participants and evaluators were blinded as to the participants’ treatment group assignments until the Phase 1 post-training evaluation was completed. At that time, the blinding was lifted for that participant. While the trainers were not blinded, they were instructed to avoid disclosing the participants’ treatment groups to either the participants or the evaluator. No trainer reported accidental disclosure of a treatment group assignment.
After being randomized to a treatment group, each participant received a baseline assessment, which was followed by the Phase 1 training of the UL. Training for the Test Group participants consisted of eighteen, 30-min sessions with the investigational device. The Control Group participated in eighteen, 30-minute placebo training sessions with the same investigational device. Participants in both groups were trained 2 or 3 times/week.
Phase 1 Training
All participants were randomized and initiated Phase 1 training no sooner than the beginning of the fifth week post-stroke 15 and no later than the 26th week post-stroke. The participants completed Phase 1 over a 6- to 10-week period, with most participants completing all 18 sessions. Training of crossover participants in Phase 2 often continued into the post-subacute period (i.e., >6 mo post-stroke).
Each participant sat next to the investigational device (Figure 2A), facing a video monitor for the duration of each session. The affected limb was placed in the forearm fixture of the investigational device, and 2 vibrators were applied to the forearm, ∼4 cm proximal to the wrist joint, over the extensor and flexor tendons of the hand (Figure 2B) and a third over the thumb tendons forming the thenar snuffbox. The vibration pattern consisted of a brief burst at 80 pulses/s followed by a ∼6-strain of 60 pulses/s (2–3 mm stroke) similar to the pattern naturally produced by muscle spindle Ia afferents to passive stretch (Figure 2C),34,35 but at a higher frequency than naturally produced during the speed and extent of joint rotation used in this study. Investigational device and intervention. In A, a participant sits along side the investigational device with the right UL placed in the device while looking at feedback on the video monitor. In B, 2 of the vibrator probes contact the tendons to the finger/thumb flexors and extensor. In C, records of vibration (from an accelerometer attached to the forearm), active finger–thumb torque, and MCP angle are shown. Note the higher frequency burst of vibration (80 pulses/s) at the beginning of each train of stimuli at 60 pulses/s.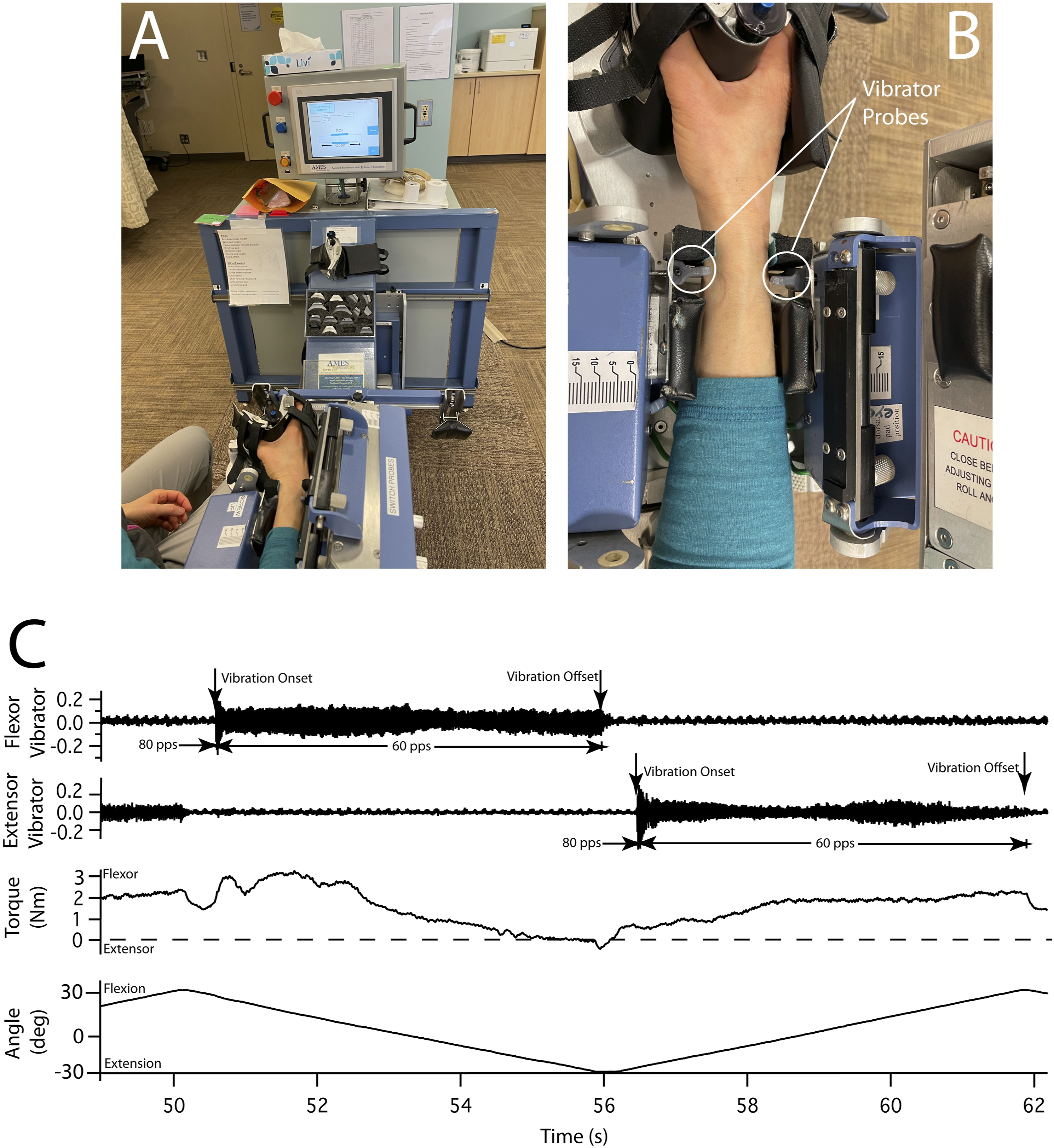
In each of the 30-min training sessions, the participants spent the first 20 min with training applied to the 5 digits of the affected hand (“hand training”) and the last 10 min with training applied to the affected wrist (“wrist training”). During hand training, the investigational device flexed and extended the participant’s thumb and fingers at the metacarpophalangeal joints between 15 deg flexion and 15 deg extension at a velocity of 5 deg/s (see Figure 2C). In the hand-closed position, the tips of the fingers and the thumb were separated by 1 cm—the combined thickness of the 2 rotating plates to which the hand was attached. The vibrator probes were positioned in contact with the affected forearm over the finger-and-thumb flexor and extensor tendons. During wrist training, the investigational device flexed and extended the entire hand at the wrist between 7.5 deg flexion and 37.5 deg flexion at 5 deg/s—that is, all wrist motion took place in the flexion hemispace. For wrist training, the vibrator probes were changed to ones designed to contact wrist flexor and extensor tendons.
During Phase 1 training, the Test Group participants’ task was to apply torque in the same direction as the currently imposed motion of the fingers and thumb or the wrist, and by doing so, the participant assisted the robot in performing the movement, in contrast to most other therapeutic robots where the robot assists the user. Regardless of how much assistance the participant provided to the investigational device in this study, the applied motion was always ± 15 deg at 5 deg/s. The participant’s task was to reach or exceed a target amount of active, assistive torque during each cycle of movement rather than to guide the position of the digits or hand to a target location or to perform any other type of functional task. The video monitor indicated, in real time, how much volitional torque the participant was producing in assisting with the motion as well as providing a torque target to reach or exceed during each flexion or extension half-cycle of motion.
During training, the participant’s volitional torque was extracted from the overall recorded torque by subtracting, in real time, the passive resistance profile to the same joint motion as during the training. This passive resistance profile was measured at the beginning of each training session with a series of calibration cycles on the investigational device while the participant kept the hand as relaxed as possible and could be repeated during the session if the trainer observed a significant and persistent change in the participants UL tone. The trainer adjusted the flexion and extension torque targets from 1 training session to the next, as needed, to keep the target at ∼25% of the participant’s maximum strength. During the Phase 1 training, vibration was applied to the antagonist (i.e., lengthening) muscles via their tendons, that is, to the extensors during flexion motion and to flexors during extension motion (Figure 2C).
The use of participant-assisted motion (i.e., to the investigational device) with biofeedback of volitional torque was designed to augment the level of cortical motoneuronal activation to the impaired muscles, and muscle vibration was designed to augment the proprioceptive response relevant to the ongoing movement. The intent of this intervention was to facilitate neural reconnection in the damaged sensorimotor pathway, to reverse somatosensory loss, and to reduce spasticity.
During Phase 1, Control Group participants were trained with a placebo intervention. The only differences between the placebo and the real intervention were (1) Control Group participants were instructed to keep the affected UL as relaxed as possible (i.e., as opposed to active assistance); (2) Control Group participants received vibration to the shortening muscle(s) at 10 pulses/s (i.e., as opposed to 60 pulses/s applied to the lengthening muscle); and (3) Control Group participants viewed photographs depicting attractive landscapes on the video monitor (i.e., as opposed to volitional torque biofeedback). Consequently, the placebo vibration was not expected to have any significant perceptual or motor consequences.23,24 In contrast, the Phase 1 interventions provided to participants in both treatment groups shared the following features: passive, continuous motion of the affected fingers and thumb or wrist, visual attention to a screen, mechanical vibration of muscles in the affected forearm, and equal lengths of time with the investigational device.
At the end of each training session, participants in both treatment groups also performed a Strength Test and an Active Motion Test with the investigational device. In the Strength Test, the participants performed 6 maximal, isometric efforts, 3 in the extension direction and 3 in the flexion direction. The Strength Test was performed twice, once for finger-and-thumb flexion and extension and again for wrist flexion and extension. The investigational device recorded the peak volitional joint torque applied by the participant during each effort. In the Active Motion Test, the participant was instructed to successively extend, flex, and again extend either the digits or the wrist in order to follow the motion (±15 deg, 5 deg/s) of a 3-deg wide target box on the monitor. The investigational device recorded the overall time that the participant was able to track the target motion by maintaining the digits or wrist within the limits of the target box.
Phase 1 Assessments
Each participant was assessed at baseline and, later, within 1 week of the last training session (i.e., “post-training assessment”). During the baseline assessment, the evaluator administered the SIS, MAS, and RLA. The FMA-UL scores obtained previously during the eligibility screening evaluations were used as the baseline scores for that test. In each post-training assessment, the evaluator administered the FMA-UL, SIS, MAS, and RLA. In the MAS, spasticity was assessed in all 4 fingers together and in the wrist, each in the flexion and extension directions. All 4 scores were summed to provide a single MAS measure. In the RLA, only the number of tasks completed was quantified.
The baseline assessment scores from the Strength Test (i.e., in Nm) and Active Motion Test (i.e., time in target in seconds) were defined as the average scores obtained during training sessions 1–3. The post-training Strength and Active Motion scores were defined as the average scores obtained during training sessions 16–18.
Questionnaire on Blinding
After completing the post-training assessment, each participant and his/her evaluator responded to a written questionnaire to determine whether the Phase 1 blinding was effective. Subsequently, the blind was broken for that participant. The questionnaire allowed each respondent to indicate whether the Phase 1 training was perceived as “real,” “placebo,” or “unsure.”
Phase 2 - Crossover
After breaking the blinding for the Phase 1 Control Group participants, they were offered the opportunity to receive eighteen, 30-min training sessions with the investigational device. The training protocol in Phase 2 was identical to that used on the Test Group participants in Phase 1.
To participate in the crossover phase, former Phase 1 Control Group participants were required to complete at least 15 of their Phase 2 training sessions. The Phase 2 baseline measures for crossover participants consisted of their post-training assessment scores from Phase 1. Participants originally randomized to the Test Group did not participate in the crossover phase of the study. A total of 27 (63%) Control Group participants elected to receive crossover training, with 22 completing the minimum of 15 training sessions. The other 5 participants dropped out after completing between 1–8 sessions.
Data Analysis
Participant Demographics.
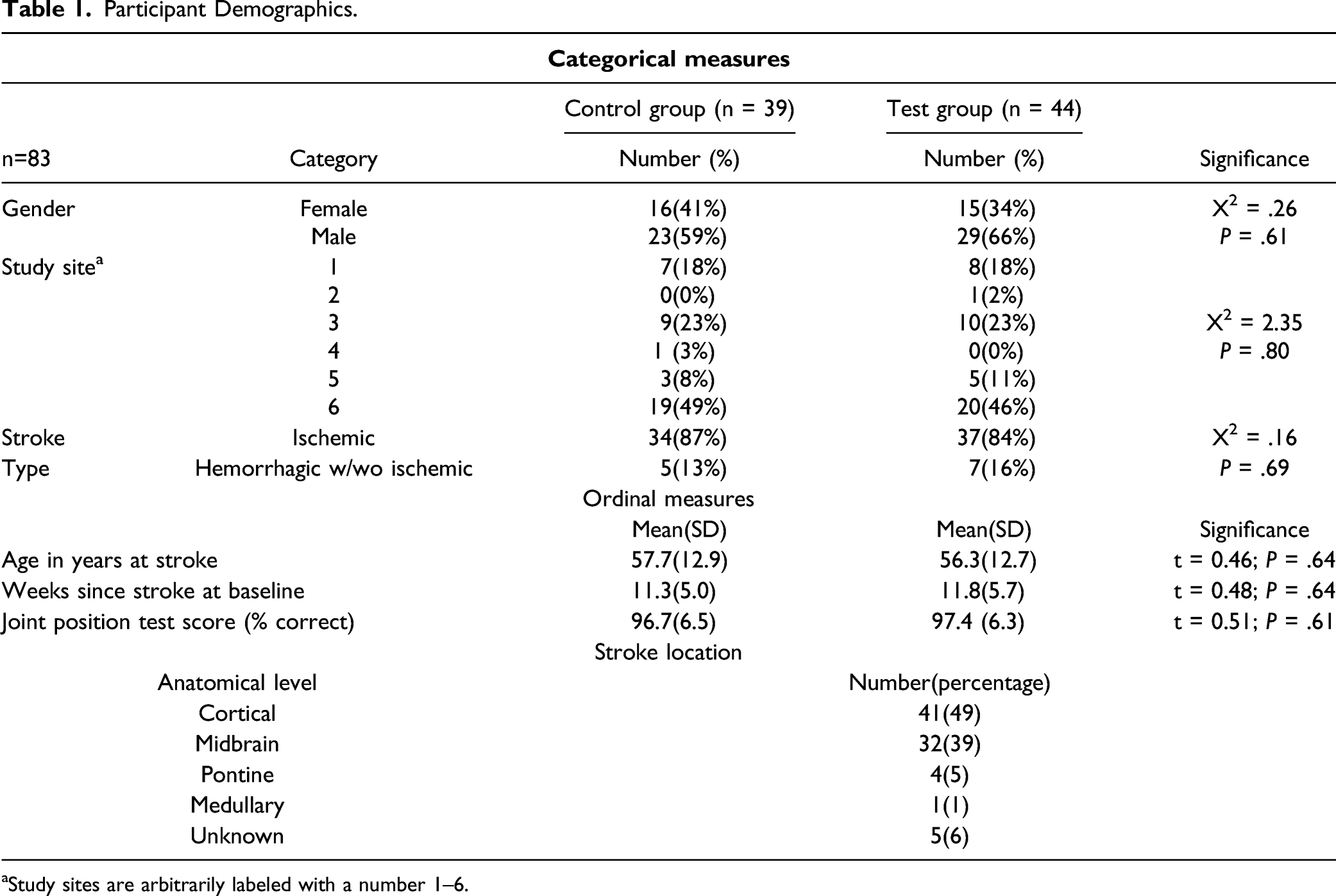
aStudy sites are arbitrarily labeled with a number 1–6.
Blind Verification and Compliance.
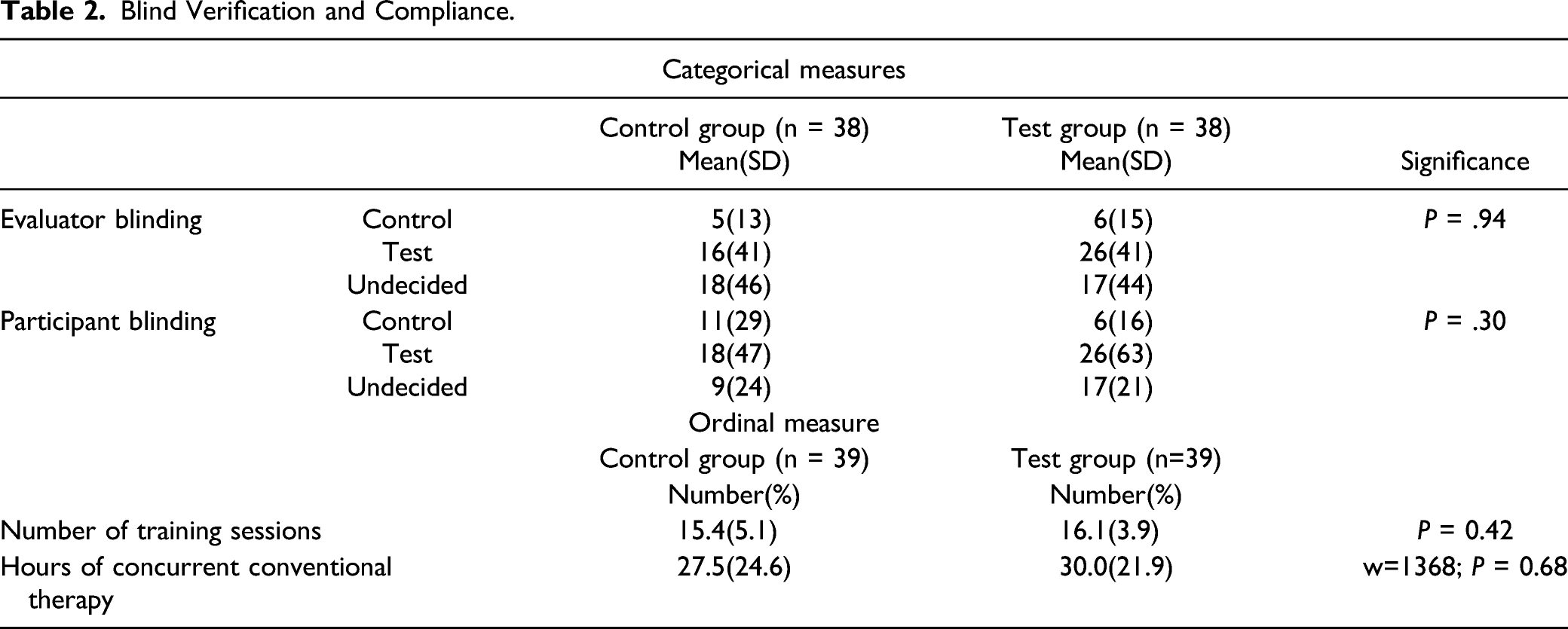
Phase 1 Results.
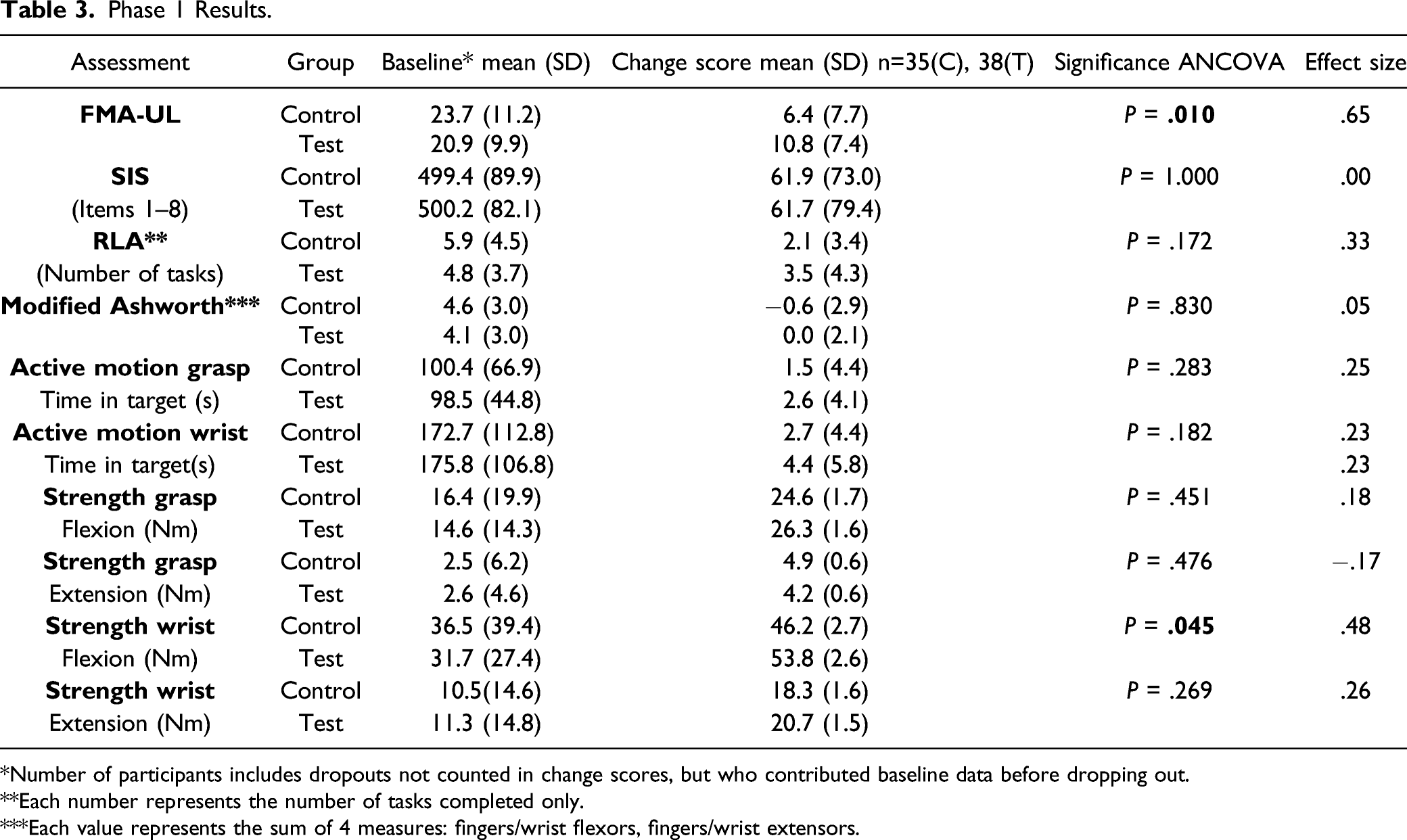
*Number of participants includes dropouts not counted in change scores, but who contributed baseline data before dropping out.
**Each number represents the number of tasks completed only.
***Each value represents the sum of 4 measures: fingers/wrist flexors, fingers/wrist extensors.
The primary outcome variable chosen for this study was the FMA-UL score as this assessment provided a comprehensive picture of a participant’s level of impairment at baseline and at post-training. Additional performance data were derived from the SIS, MAS, RLA, and the Strength and Active Motion tests. A probability of .025 was used as the criterion for statistical significance for the FMA-UL, and .05 was used for all secondary measures. All tests were two-tailed.
In the paired t-test analysis of crossover data, only those data from the 22 participants completing at least 15 training sessions were analyzed. The Phase 2 effect size was computed for each assessment as the mean of the differences divided by the standard deviation of the differences. As in Phase 1, the baseline and post-treatment Strength and Active Motion Test scores were obtained from the first 3 and last 3 training sessions.
The study was terminated after 48 months due to the elapsed time and limited funding.
Results
Screen Failures, Drop-Outs, Events, and Baseline Demographics
A total of 51 candidates failed the screening for participation: age >80 (n = 13), FMA-UL score too high (n = 7), insufficient time to complete 18 training sessions by 6 months post-stroke (n = 6), cardiovascular issues (n = 4), inability to communicate in English (n = 3), travel distance too far (n = 3), concurrent participation in a different study (n = 2), hand plegia (n = 2), not the first stroke (n = 2), alcohol abuse (n = 2), hemianopsia (n = 1), speech dyspraxia (n = 1), limb apraxia (n = 1), uncontrolled seizures (n = 1), severe cognitive deficit (n = 1), Rankin score <3 (n = 1), or loss of interest after screening (n = 1).
Eight Test Group participants and 5 Control Group participants dropped out, and 10 of the 13 chose not to participate in a second (i.e., post-training) evaluation. Reasons for dropping out were unexplained no-shows for training sessions and post-training evaluation (n = 7), travel distance too far (n = 2), initiated antispastic medication during enrollment (n = 2), UL pain (n = 1), and participant-perceived increase in muscle tone (n = 1).
During the study, tendon vibration caused skin abrasions in 3 participants, 1 at each of 3 study sites. In each case, the site Study Physicians judged the event not to be serious but related to participation in the study. After the abrasions healed over 1–2 weeks, the 3 participants wore a nylon sleeve on the forearm during further training and completed the training protocol without any additional problems.
At baseline, no significant differences were found between the 2 treatment groups with respect to study site, gender, proprioception, type of stroke, stroke location, age at stroke, or time since stroke (Table 1). On average, the time of enrollment was 11.5 (5.4 SD) weeks post-stroke. The anatomical levels of each stroke event in the participants are summarized in Table 1.
The number of training sessions and hours of other, outpatient therapy received by participants were not significantly different between groups (Table 2). Table 2 also shows that neither the evaluators nor the participants could consistently identify the participants’ treatment group assignment. An analysis of the effect of age and time since stroke showed that neither of these 2 factors significantly affected FMA-UL scores in either treatment group.
Phase 1 - Controlled Study
The participants’ scores on the FMA-UL (Table 3, Figure 3A) showed a significant, positive difference between the Test Group and Control Group participants (P = .010) indicating that the eighteen, 30-min training sessions significantly reduced motor impairment for Test Group participants beyond the effects of prescribed outpatient therapy and spontaneous recovery (i.e., Control Group participants). The average FMA-UL change score for Control Group participants was 6.4 and 10.8 for Test Group participants. The average subscore for each item of the FMA-UL is compared for the 2 treatment groups in Figure 3B. The subscores are also grouped by reflex, proximal UL, and distal UL. Only the Mixing Synergies and Coordination/Speed individual items showed a significant difference between groups. The 5 items collectively related to the proximal UL also showed a significant difference between groups (ANCOVA); however, the differences recorded for the distal UL did not reach significance (Figure 3B). Of the other more specific impairment-oriented tests (i.e., MAS, Active Motion, and Strength tests), the performances of the Test and Control Group participants only showed a significant difference on the Wrist Flexion Strength test (Table 3). FMA-UL change scores from Phases 1 and 2 of the study. In A, total FMA-UL change scores are compared for the Phase 1 Test Group participants (white bar), Phase 1 Control Group participants (gray bar), and Crossover Group participants (black bar). In B, FMA-UL individual subscores and subscores grouped by type (i.e., reflex, proximal UL, and distal UL). Asterisks indicate a significant difference in change scores between the Phase 1 Test Group (white) and Control Groups (gray) only.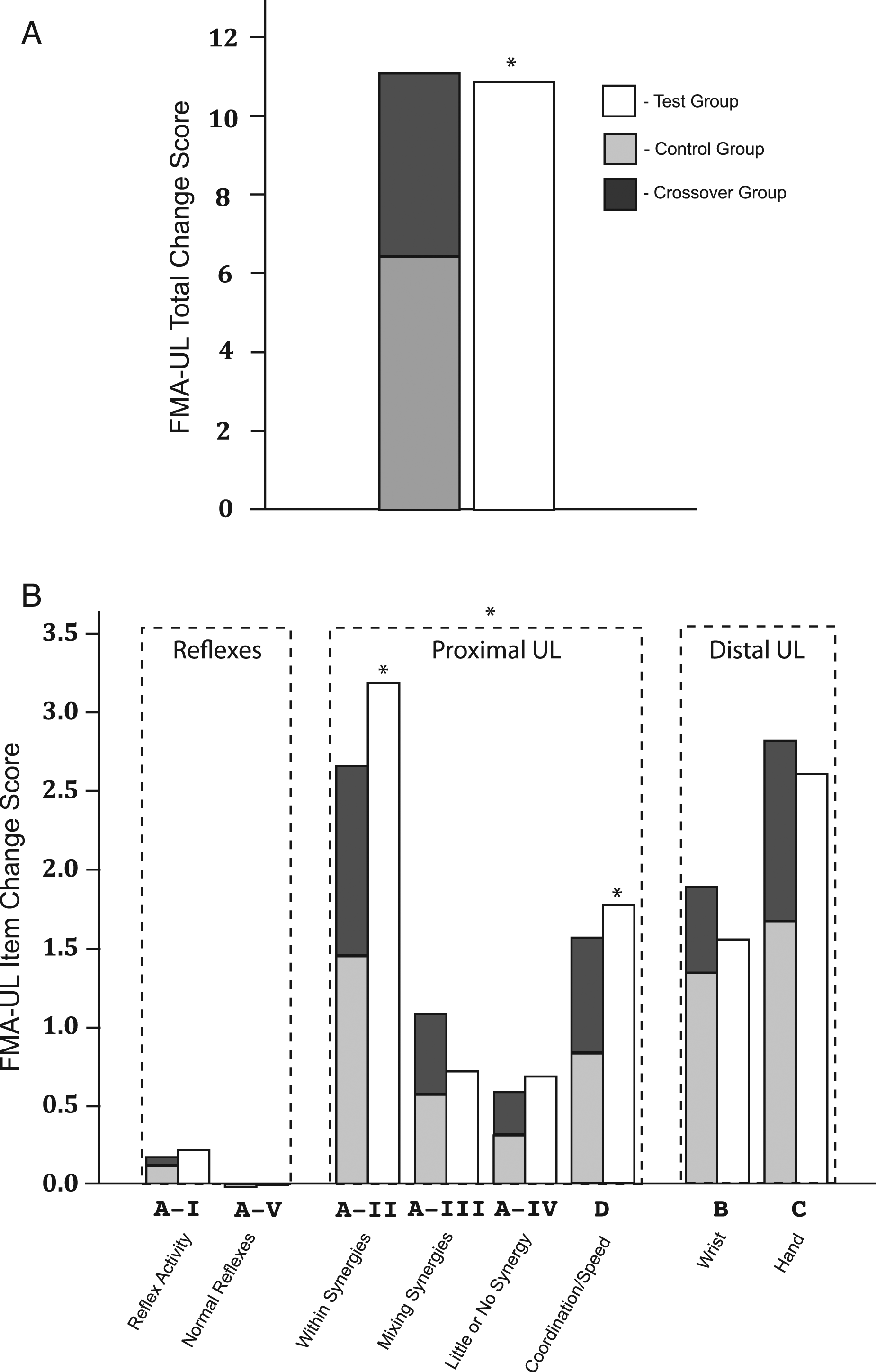
Phase 2 - Crossover Group Results
Results Phase 2.
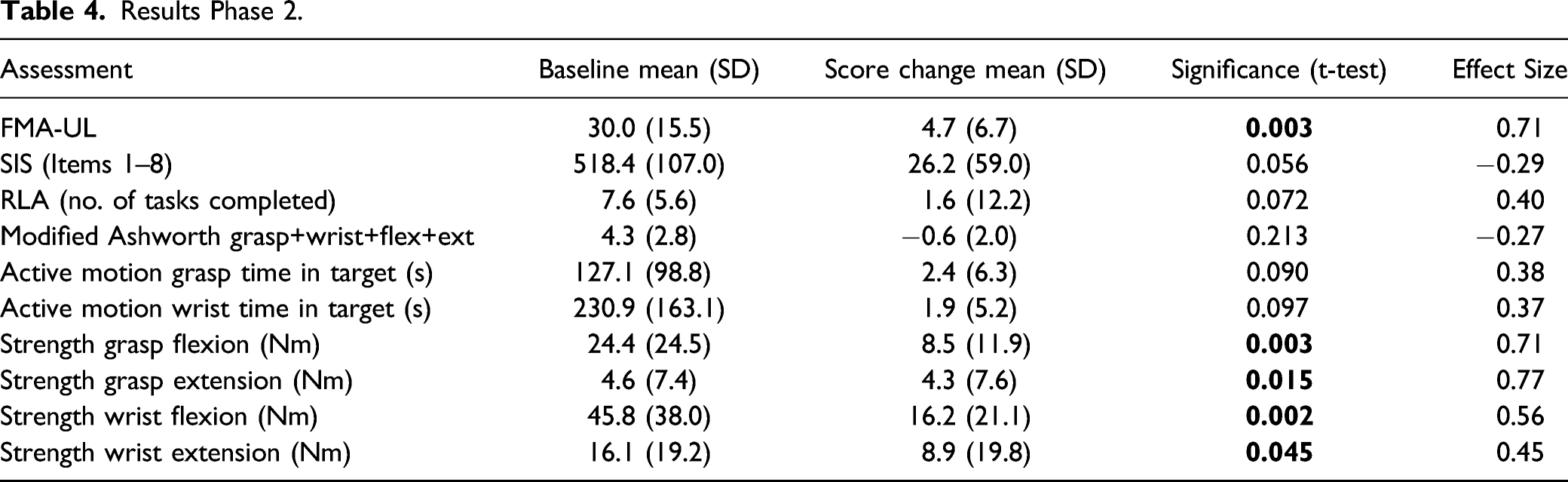
Figure 3A provides the sum of the Phase 1 and 2 FMA-UL change scores for the Crossover Group participants (6.4+4.7=11.1; that is, stacked gray and black bars). These scores were nearly the same as the Test Group participants’ average change score from Phase 1 alone (10.8). To a lesser extent, the FMA-UL subscores also exhibited this additive characteristic (Figure 3B).
Discussion
This study investigated the efficacy of a novel robotic assisted motion device and impairment-oriented intervention during the subacute phase of stroke recovery in persons with moderate-to-severe upper limb impairment. Unlike other UL robotic devices, the one used here included muscle vibration to augment proprioceptive input. Muscle vibration has been shown to enhance rapidly and substantially the sensation of movement via cortical plasticity in healthy adults36,37 as well as in people recovering from stroke. 38 Vibration is also known as an effective means of relaxing spastic muscles, 39 at least temporarily, and like stretching, is sometimes used in the clinic to relax the upper limb prior to carrying out other clinical interventions with it. Based on these previous observations, pairing muscle vibration with the participant’s volitional contractions of the weakened muscles in the current intervention was expected to induce cortical plasticity that increased the participant’s strength and control of the paretic muscle(s). While we observed reduced overall impairment based on the FMA-UL in Phase 1 and, more specifically, strength gains in Phase 2, the study design did not allow for a determination of the role of muscle vibration on these gains in the current study.
Also, unlike other robotic assisted motion devices for the UL, the investigational device used in this study involved the participant assisting the device in performing a simple flexion-extension joint rotation instead of the device assisting the participant in performing a functional task, such as directing the arm towards a target in extrapersonal space.18,40,41 The participant assisting the device, as in the present study, allowed for real-time measurement and feedback of the participants’ volitional torque contribution—information closely related to the physiological impairments (e.g., neuromuscular weakness, spasticity, and desensitization) that interfere with functional movement.
After completing eighteen, 30-min training sessions in Phase 1 of this study, Test Group participants showed an overall decrease in impairment based on significantly increased FMA-UL scores relative to Control Groups participants. The 4.4-point difference between their scores (i.e., Test Group–10.8 and Control Group–6.4) presumably represents the effect of the robotic intervention beyond the participants’ spontaneous recovery and the effects of any prescribed outpatient therapy received concurrently with the study. In support of this conclusion, the Crossover Group FMA-UL change score in Phase 2 of the study was nearly the same as that for the difference between the Test and Control Groups’ scores in Phase 1. The overall increase in FMA-UL scores seen in Test Group participants (i.e., 10.8) exceeded the threshold for the MCID,26,27 whereas the increase occurring from just spontaneous recovery, prescribed outpatient therapy, and placebo intervention (i.e., Control Group score) did not. In Phase 1 of this study, despite training being delivered exclusively to the distal UL of the Test Group participants, the only item subscores on the FMA-UL that changed significantly were those most closely related to the proximal UL. A likely explanation for this outcome is that, with stroke, the distal UL is often more impaired than the proximal UL, and therefore, requires more training than the proximal limb. This result also demonstrates distal-to-proximal transference of distal limb training,42,43 whereas the converse (i.e., proximal to distal) has not been observed during proximal UL training. 42
Interpretation of Results
While previous research44–46 with the investigational device used in this study showed promise for the eventual and lasting recovery of UL function in stroke survivors with moderate-to-severe, chronic (≥1 yr post-stroke) UL impairment, use during the subacute phase was not investigated. A goal of the present study was to determine whether the investigational device used here would be efficacious ≤6 months post-injury. Studying severely impaired individuals during the subacute phase of stroke recovery is complicated due to ongoing spontaneous recovery and by any prescribed outpatient therapy received during the post-injury period. Therefore, the purpose of the Control Group during Phase 2 of the study was to distinguish the effects of the test intervention from those arising from spontaneous recovery and prescribed outpatient therapy, as well as to control for a placebo effect.
The placebo version of the intervention was designed to have little or no therapeutic effect by omitting any volitional assistance to the applied movement and by vibrating the UL in a manner so as to provide little or no useful proprioceptive input. However, the passive motion applied to the limb during the placebo intervention could have produced a mild therapeutic effect, and vibration applied to the “wrong” (i.e., shortening) muscle(s) may have been weakly transmitted through dense tissues of the forearm to the “correct” (i.e., lengthening) muscles. Such inadvertent effects would result in underestimating the efficacy of the test intervention.
In Phase 2 of the study, the Crossover Group participants showed not only significant improvement in their FMA-UL scores, but also increased scores relating to the secondary outcomes. Strength Tests in all involved muscles increased significantly, and change scores in the Active Motion test (i.e., both hand and wrist), the RLA, and the SIS nearly reached levels of significance, possibly forecasting the return of some UL function with further robotic training. However, Phase 2 of the study often extended into the seventh or eighth month post-stroke, so the timing of receiving the intervention could potentially explain the differences in secondary outcomes between the Test and Crossover Groups.
Study Limitations
One purpose of this study was to determine to what extent the test intervention would reduce UL impairment during the subacute phase of stroke recovery. However, the study design precluded the opportunity to identify the explicit causative factors resulting in significantly improved impairment levels following the robotic training. Candidate causative factors include exercise (i.e., participant-assisted movement) and muscle vibration. The Phase 1 placebo treatment lacked any active assistance by the Control Group participants, so differing amounts of exercise between the 2 treatment groups could, by itself, explain the difference in improvement between Control and Test Group FMA-UL scores.42,43 However, strength gains in Phase 1 did not differ for Test and Control Group participants (Table 3), suggesting that strength alone is unlikely to explain the entire 4.4-point additional gain achieved by Test Group participants. Alternatively, since previous studies showed that muscle vibration decreases spasticity 39 and somatosensory loss,36–38 2 common impairments resulting from stroke that interfere with movement, possible roles for reduced spasticity and augmented sensation in these gains are suggested. Differentiating the relative contributions of these factors will require additional study.
In Phase 1 of this study, the significant increase in FMA-UL scores (a broad measure of impairment) without significant change in most of the secondary outcomes (more specific measures of impairment) suggests that the effect of the robotic training was broadly, but weakly, distributed since, among the different types of sensorimotor impairment (e.g., weakness, spasticity, and loss of sensation). Scores on the RLA and SIS improved, but not to a significant level, indicating that changes in impairment levels over 18 training sessions were insufficient to improve UL function. However, a previous study involving 6 months of (near) daily training on an early version of the investigational device showed that chronic, moderately-to-severely impaired stroke survivors’ UL impairments continued to decrease on average over ∼90, 30-min training sessions. 44 In another stroke study using this investigational device on participants with a chronically plegic or severely paretic UL, 30 training sessions were sufficient to return some hand function (i.e., non-zero scores on the Box and Block Test 45 ) to 4 of 28 participants receiving zero scores at baseline. These results suggest that increasing the dosage of the robotic training during the subacute phase of recovery might lead to further improvement in impairment and, possibly, to some recovery of hand function.
No follow-up analysis was conducted in this study to measure the extent to which impairment changed in the months following the end of the robotic training period. In several previous studies, however, employing the same intervention used in this study, follow-up analyses showed that gains were retained or continued to improve.45,46 Future studies conducted during the subacute period of recovery should include follow-up evaluations during the first 6–12 months post robotic training. Neuroimaging studies are also needed to characterize the neuroanatomical basis of neuroplasticity resulting from the intervention.
Conclusion
In individuals with moderate-to-severe UL impairment in the subacute phase of recovery from stroke, robotic impairment-oriented training was a viable intervention to reduce UL impairments beyond that achievable spontaneously and with prescribed outpatient therapy.
Footnotes
Acknowledgments
The authors wish to acknowledge the contributions to this study by Sandra Oster, technical editor of this manuscript, as well as numerous evaluators, trainers, and study coordinators at the study sites.
Declaration of Conflicting Interest
Author Cordo and Oregon Health and Science University (OHSU) have financial relationships with AMES Technology, Inc., a company that may benefit from the work presented in this manuscript; this potential conflict of interest is managed by the OHSU Research Integrity Office.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by AMES Technology, Inc. Preliminary results of this study were published as a conference abstract.
