Abstract
Background. Motor practice is an important component of neurorehabilitation. Imaging studies in healthy individuals show that dynamic brain activation changes with practice. Defining patterns of functional brain plasticity associated with motor practice following stroke could guide rehabilitation. Objective. The authors aimed to test whether practice-related changes in brain activity differ after stroke and to explore spatial relationships between activity changes and patterns of structural degeneration. Methods. They studied 10 patients at least 6 months after left-hemisphere subcortical strokes and 18 healthy controls. Diffusion-weighted magnetic resonance imaging (MRI) was acquired at baseline, and functional MRI (fMRI) was acquired during performance of a visuomotor tracking task before and after a 15-day period of practice of the same task. Results. Smaller short-term practice effects at baseline correlated with lower fractional anisotropy in the posterior limbs of the internal capsule (PLIC) bilaterally in patients (t > 3; cluster P < .05). After 15 days of motor practice a Group × Time interaction (z > 2.3; cluster P < .05) was found in the basal ganglia, thalamus, inferior frontal gyrus, superior temporal gyrus, and insula. In these regions, healthy controls showed decreases and patients showed increases in activity with practice. Some regions of interest had a loss of white matter connectivity at baseline. Conclusions. Performance gains with motor practice can be associated with increased activity in regions that have been either directly or indirectly impaired by loss of connectivity. These results suggest that neurorehabilitation interventions may be associated with compensatory adaptation of intact brain regions as well as enhanced activity in regions with impaired structural connectivity.
Introduction
Repetitive movement practice is an important component of neurorehabilitation after stroke. 1 Improvements in movement performance with practice provide a model for rehabilitation, 2 and clinical studies have documented performance gains during practice of a motor skill by patients, following stroke. 3-5
Imaging studies in healthy individuals have shown that gains in motor performance with practice are mediated by dynamic changes in brain activation in specific cortical and subcortical regions of the motor control network. 6-9 For example, practice-related improvements in visuomotor tracking are associated with activity changes in the prefrontal cortex and basal ganglia. 6 The neural correlates of motor performance gains with practice have only recently begun to be explored in patients after stroke, however. 10 Defining patterns of functional brain plasticity associated with motor practice could help stratify patients for intensive rehabilitation interventions 11 or guide targeted therapeutic approaches, such as brain stimulation, 12 that can enhance local plasticity during motor training.
Here, we test the hypothesis that practice-related changes in brain activity are different in patients after stroke compared with healthy controls. Furthermore, we explore spatial relationships between the practice-related changes in brain activity and patterns of structural degeneration following stroke.
To do this, we used serial functional magnetic resonance imaging (fMRI) to assess changes in task-related brain activation while participants performed a complex visuomotor tracking task before and after 15 consecutive days of practice. Practice-related changes in brain activity were contrasted between patients who had suffered a single subcortical left-hemisphere stroke more than 6 months previously and age-matched healthy controls. We also acquired diffusion-weighted MRI (shown previously to be sensitive not just to the stroke but also to secondary degeneration poststroke 13-15 ) at baseline only to assess white matter structural integrity. We tested for correlations between white matter integrity and task performance and explored the anatomic relationships between practice-related fMRI changes and cortical regions with impaired white matter connectivity at baseline as a consequence of the prior stroke.
Methods
Participants
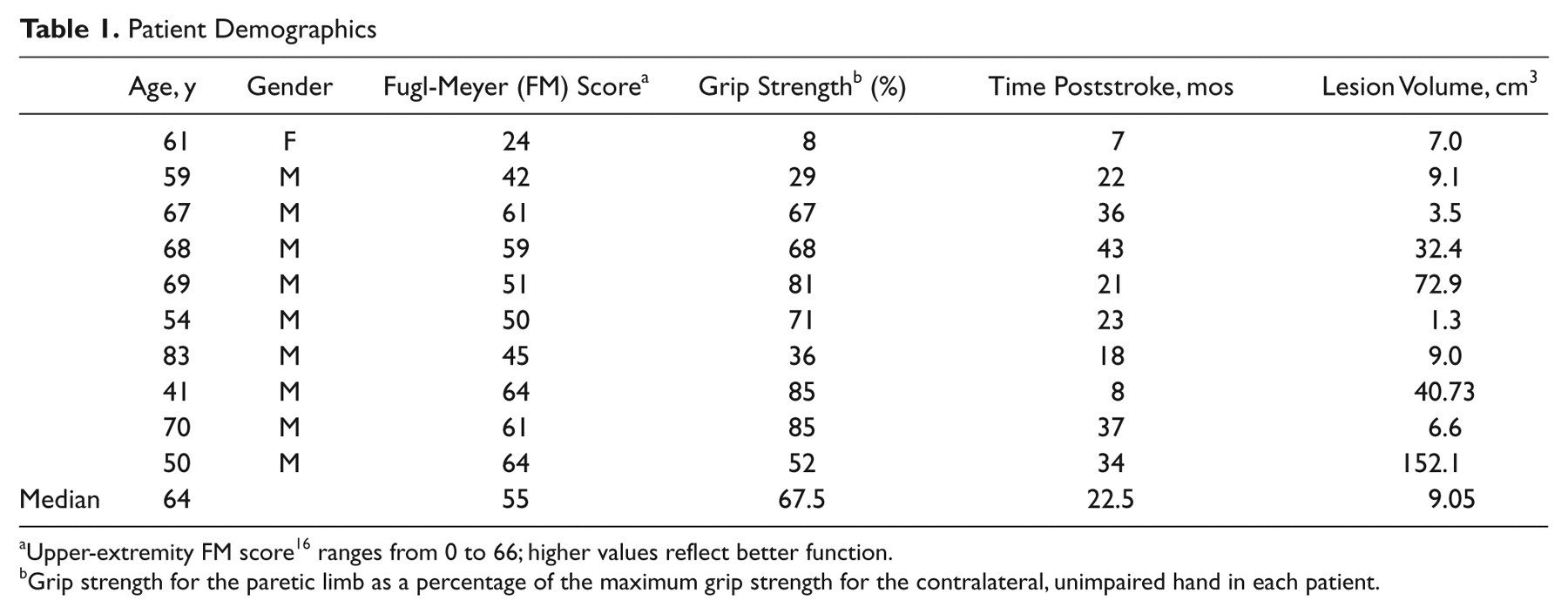
Participants were 10 right-handed patients who had previously suffered a first stroke (Table 1) and 18 age-matched right-handed controls (mean age, 58 years; range, 30-81 years; 7 women, 11 men); they gave written informed consent to participate in accordance with the Declaration of Helsinki and local Research Ethics Committee approval (05/Q1607/63). All patients were at least 6 months post a first ischemic or hemorrhagic left-hemisphere subcortical stroke affecting motor function in the right hand and were without a history or signs and symptoms of any other neurological conditions. Patients had intact sensation to light touch in affected limbs, were able to give informed consent, and did not have aphasia significantly limiting communication (National Institute of Health Stroke Scale rating >2). Patients all demonstrated visual acuity (corrected or uncorrected) that was sufficient to follow movement of the visual target on the MRI projection screen (during fMRI scanning) and on the laptop computer screen used for home training and could generate sufficient grip force for visuomotor responses to be tracked consistently by the computer.
Patient Demographics
Upper-extremity FM score 16 ranges from 0 to 66; higher values reflect better function.
Grip strength for the paretic limb as a percentage of the maximum grip strength for the contralateral, unimpaired hand in each patient.
Visuomotor Tracking Task
Participants viewed a computer screen displaying 2 moving bars: a computer-controlled target bar indicating the required relative force (in green) and an adjacent response bar showing the instantaneous force generated by the participant (in blue if the correct force was generated or in red if the force deviated from the target force by more than 40 pixels in the visual display). The task for participants was to match the height of the response bar to that of the target bar by altering the grip force applied to an isometric pressure-sensing device held in their right hand. 17 The force required to move the bar to its maximum amplitude was calibrated to 80% of the participant’s maximum grip strength.
During fMRI sessions, the task included alternating 38-s blocks of rest and task conditions. In the task condition of interest, blocks consisted of 2 repeats of a specific sequence of sinusoidally varying target bar movements. Participants were informed that there was a repeating sequence within the block, so this would be learned explicitly. A second task condition required them to track random bar movements, but activation during this task is not further considered here. During rest blocks, participants did not move and passively viewed 2 bars, the relative movements of which were intended to mimic those during the tracking periods (a “target” bar moved to trace a sinusoidal trajectory slightly ahead of the second, “response” bar). Participants practiced the visuomotor tracking task for 4 blocks outside the scanner before their first recorded trial.
Performance gains with practice of the visuomotor tracking task were assessed using 15 days of daily home practice (10 blocks, each containing 2 repeats of the same sequence used during fMRI, with alternating 5-s rest periods) using a laptop and identical grip device.
Behavioral Analysis of Practice Effects
For task performance during fMRI, the mean error (calculated as the absolute difference in height between the target and response bars) per block was assessed using a repeated-measures ANOVA with within-subject factors of block (1-10) and condition (random, sequence) and the between-subject factor of group (patients, controls). A short-term practice score for each participant was calculated as (ErrorBLOCK1/ErrorBLOCKS7-10). The mean error per day from the home practice sessions was assessed similarly using a repeated-measures analysis of variance (ANOVA) with within-subject factor of day (1-15) and between-subject factor of group (patients, controls). A long-term practice score for each participant was calculated as (ErrorDAY1/ErrorDAYS10-15).
MRI Data Acquisition
Functional magnetic resonance imaging
FMRI data were acquired at baseline and after 15 days of task practice on a 3-Tesla Varian MRI scanner using a multislice gradient echo planar imaging sequence (echo time = 60 ms, repetition time = 3000 ms, field of view 240 × 240 mm2, matrix 64 × 64; 21 contiguous axial 6mm thick slices). T1-weighted high-resolution MRI scans (1 × 1 × 1 mm3) were acquired for anatomic localization and lesion volume calculation.
Diffusion-weighted magnetic resonance imaging
High angular resolution diffusion-weighted imaging data were acquired at baseline on a 1.5-Tesla Siemens Sonata MR scanner with maximum gradient strength of 40 mT m−1 using echo planar imaging (TR = 8500 ms; TE = 80 ms; 53 × 2.5 mm2 thick axial slices; voxel size = 2.5 × 2.5 × 2.5 mm3; 60 isotropically distributed diffusion directions; b-value = 1000 sm m−2) and 5 volumes with no diffusion weighting.
MR Image Analysis
Analysis of MRI data was carried out using tools from the FMRIB Software Library (www.fmrib.ox.ac.uk/fsl). 18 The stroke lesion volume was segmented for each patient by manually delineating the region of abnormally hypointense signal on the patient’s T1-weighted scan using FSLview and calculating the volume of this region using fslstats.
Diffusion MRI analysis
FMRIB’s Diffusion Toolbox was used to correct for head motion and eddy currents and to fit a diffusion tensor model and calculate fractional anisotropy (FA) at each brain voxel. Voxelwise statistical analysis of FA on a white matter “skeleton” was conducted using TBSS (tract-based spatial statistics), as described elsewhere. 19 “Randomize” was used for permutation-based testing (5000 permutations) and significant clusters defined using a cluster-forming threshold of t = 3 and a corrected cluster size threshold of p < 0.05.
A general linear model was used to test for voxelwise relationships with FA. Age was included as a covariate of no interest. We constructed within-group (patient or healthy control) design matrices to test for correlations between FA and short-term practice scores for performance of the visuomotor task. We included average tracking error and (for the patient group only) Fugl-Meyer score as covariates of no interest, so results reflect the practice-related component of the task across the multiple blocks rather than simply overall performance differences.
Probabilistic tractography, based on a multifiber probabilistic diffusion model 20,21 fit to each voxel, was run to estimate intrahemispheric connectivity between 56 brain regions (48 cortical and 8 subcortical), defined using the Harvard-Oxford atlas (available within FSLview), and linearly aligned with each subject’s diffusion data using FLIRT. From every seed voxel, 5000 samples were initiated, and the number reaching every other brain region was recorded, giving an estimate of connectivity probability between pairs of brain regions for each participant. For each brain region, the average connectivity probability to all other brain regions was calculated, averaged across participants to calculate the percentage reduction in connectivity probability in patients compared with controls.
fMRI analysis
Prestatistical processing of fMRI data included motion correction, spatial smoothing using a Gaussian kernel of full-width half-maximum 8 mm, and nonlinear high-pass temporal filtering (Gaussian-weighted Least squares fitting [LSF] straight line fitting, with σ = 80.0). De-noising was performed using independent component analysis in MELODIC, 22 with the number of output components limited to 50. Artifact components resulting from ghosting, slice drop out, or head motion, for example, were defined using conservative criteria (http://www.fmrib.ox.ac.uk/analysis/research/melodic/) and removed from the data. Registration of functional images to high resolution and standard (Montreal Neurological Institute [MNI]) space was carried out using nonlinear registration (FNIRT) with manually defined lesion masks to downweight the influence of the lesion.
Statistical analysis was carried out using the general linear model with local autocorrelation correction. 23 A first level analysis modeled individual participant data from each session and included 4 regressors in total: 2 regressors modeling sequence and random visuomotor tracking blocks using fixed height box-cars and 2 confound regressors that modeled these box-cars convolved with a linear trend, to covary out time-varying effects in task-related activity. Sequence box-car versus rest was the contrast of interest used for all subsequent analyses.
Higher level random effects analyses with outlier detection 24 combined data across participants and/or sessions in a series of general linear models. An analysis of baseline data in all participants included group mean regressors (patients and controls) and specified a contrast to test for differences in task-related (sequence vs rest) activity between groups. Fugl-Meyer scores of motor impairment were included in the model for the patients to covary out effects of impairment on baseline activity. An analysis of both sessions across all participants included 2 session regressors to differentiate between baseline and follow-up sessions for each group separately, a regressor for each participant to identify his/her baseline and follow-up sessions, and a (demeaned) age regressor. A contrast was specified to test for differences in the effects of session between groups (ie, a Group × Session interaction). Z-statistic images were cluster thresholded using an initial cluster-forming threshold of z > 2.3, and a corrected cluster extent threshold of P < .05. Signal change from peak voxels within clusters showing significant interactions was plotted to illustrate patterns of activation differences that were driving significant effects.
Results
Patients had a broad range (Table 1) of disability (median Fugl-Meyer score, 55; range, 24-64) and stroke lesion volumes (median, 9 cm3; range, 1-73 cm3).
Both the patients and the healthy controls performed a simple visuomotor tracking task with their right hands during the baseline fMRI examination. Patients showed a trend to greater performance error than the healthy controls (main effect of group: F(1, 24) = 3.98; P < .058), but both groups showed significant improvements in performance with practice over the multiple task blocks within the session (main effect of block: F(9, 216) = 6.90; P < .001). There was a positive correlation between this short-term improvement in performance with practice for patients and FA in clusters localized to the posterior limb of the internal capsule bilaterally for the patients (Figure 1, t > 3, corrected P < .05; r 2 = 0.58). Controls showed no significant correlation between white matter FA and improvement in performance.
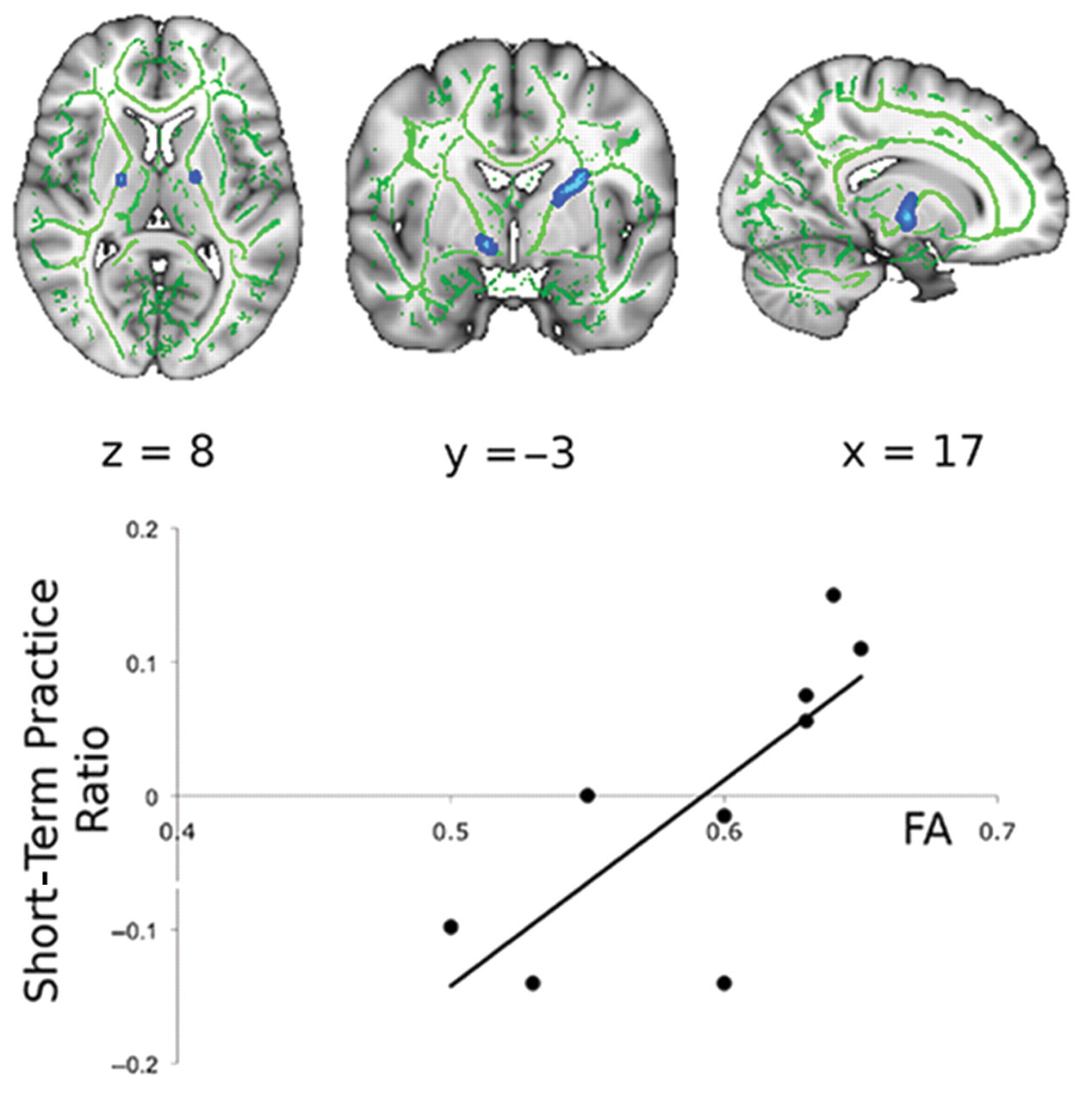
Diffusion MRI fractional anisotropy at baseline correlated strongly with improvements in visuomotor tracking performance during task practice. To the left, bilateral clusters within the posterior limb of the internal capsule showing a positive correlation of short-term practice scores and FA are illustrated (blue, thickened; FA skeleton in green; t > 3, corrected P < .05). A scatter plot defining the relationship between FA at the peak voxel within the clusters and short-term practice score is shown on the right. FA explained a majority of the variance in performance scores between patients (r 2 = 0.58). (Note that practice ratios are positive, but here, we plot the demeaned and orthogonalized values entered into the GLM). Abbreviations: MRI, magnetic resonance imaging; FA, fractional anisotropy; GLM, general linear model.
fMRI analysis showed that, at baseline, performance of the sequence-tracking task versus rest was associated with activation of contralateral sensorimotor and bilateral premotor and parietal cortical areas, basal ganglia, thalamus, and cerebellum across all participants (data not shown). A mask derived from this suprathreshold task-related activity across all participants defined a region of interest for the voxelwise contrasts between groups. Patients showed significantly reduced activation relative to controls at baseline in the left precentral gyrus (including the primary sensorimotor cortex), left superior frontal gyrus (including the dorsal premotor cortex) and in the left supplementary and presupplementary motor areas, as well as in lobules V and VI of the right cerebellum and bilaterally in the thalamus (Figure 2, Table 2).
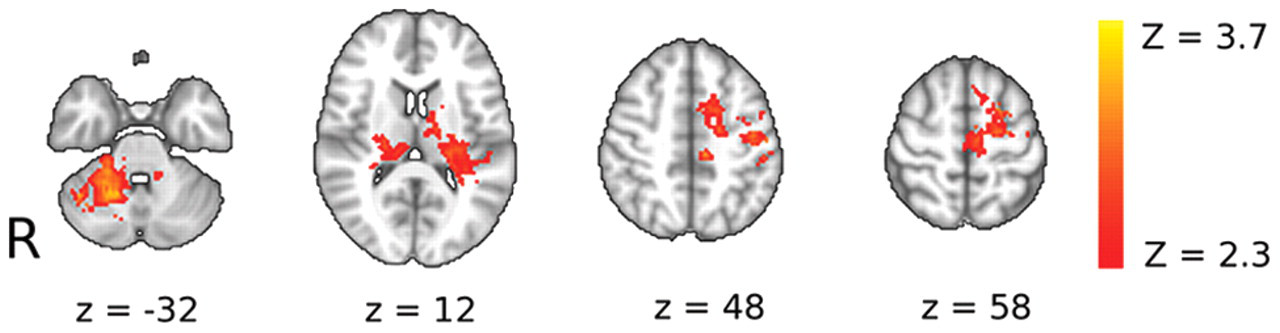
Patients showed decreased brain activation relative to healthy controls for the main effect of the visuomotor tracking task at baseline. Brain regions in which controls have greater activation than patients during performance of the visuomotor tracking task at baseline are shown. Axial slices are shown for Montreal Neurological Institute (MNI) Z-coordinates provided in the figure. The activation changes are illustrated with a cluster-forming threshold of Z > 2.3 and a corrected cluster extent threshold of P < .05 and are superimposed on a background MNI template brain image with the left-hand side of the brain (ipsilesional in patients) displayed on the right-hand side of each brain slice. See Table 2 for details of local maxima.
Local Maxima a for Significant Task-Related Activation Clusters That Differ Between Patients and Controls at Baseline
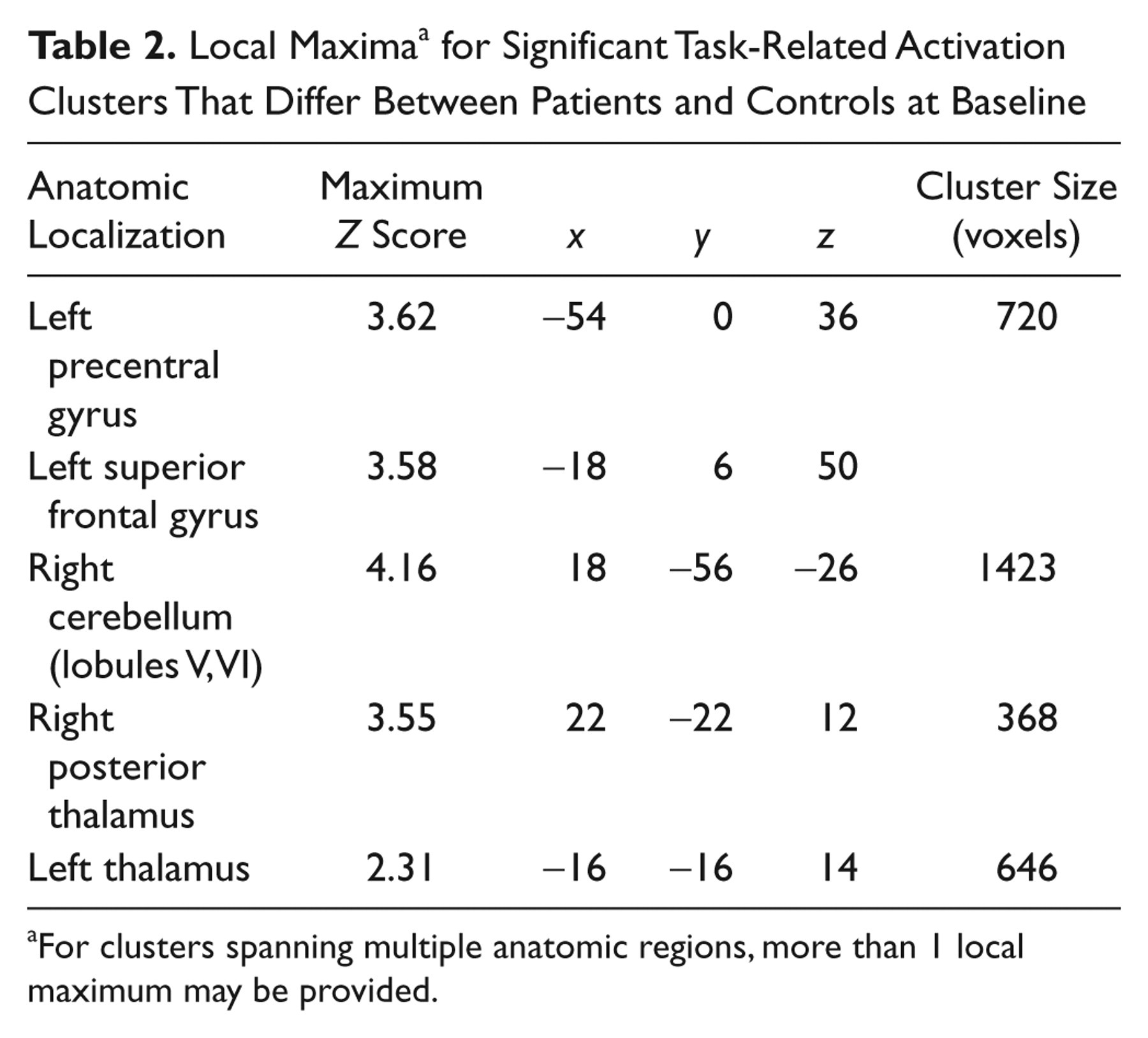
For clusters spanning multiple anatomic regions, more than 1 local maximum may be provided.
Following baseline testing, participants practiced the visuomotor tracking sequence at home for 15 days. Both patients and controls showed a decrease in mean tracking error over this practice period (Figure 3; main effect of day: F(13, 299) = 11.17; P ≤ .001) with a main effect of group—F(1, 23) = 5.26; P = .03—reflecting a consistently greater relative performance error in the patients. Separate ANOVA tests for patients and controls confirmed a significant decrease in error over time for both groups (main effect of day; patients, F(13, 104) = 6.11, P < .001; controls, F(13, 195) = 4.34, P < .001). There was a significant interaction between day and group—F(13, 299) = 6.28; P < .005—because of a greater practice effect for the patients (practice score = 2.02 ± 0.95) relative to the controls (1.27 ± 0.33, t = 2.3, df = 9.1, P = .048, corrected for unequal variances).
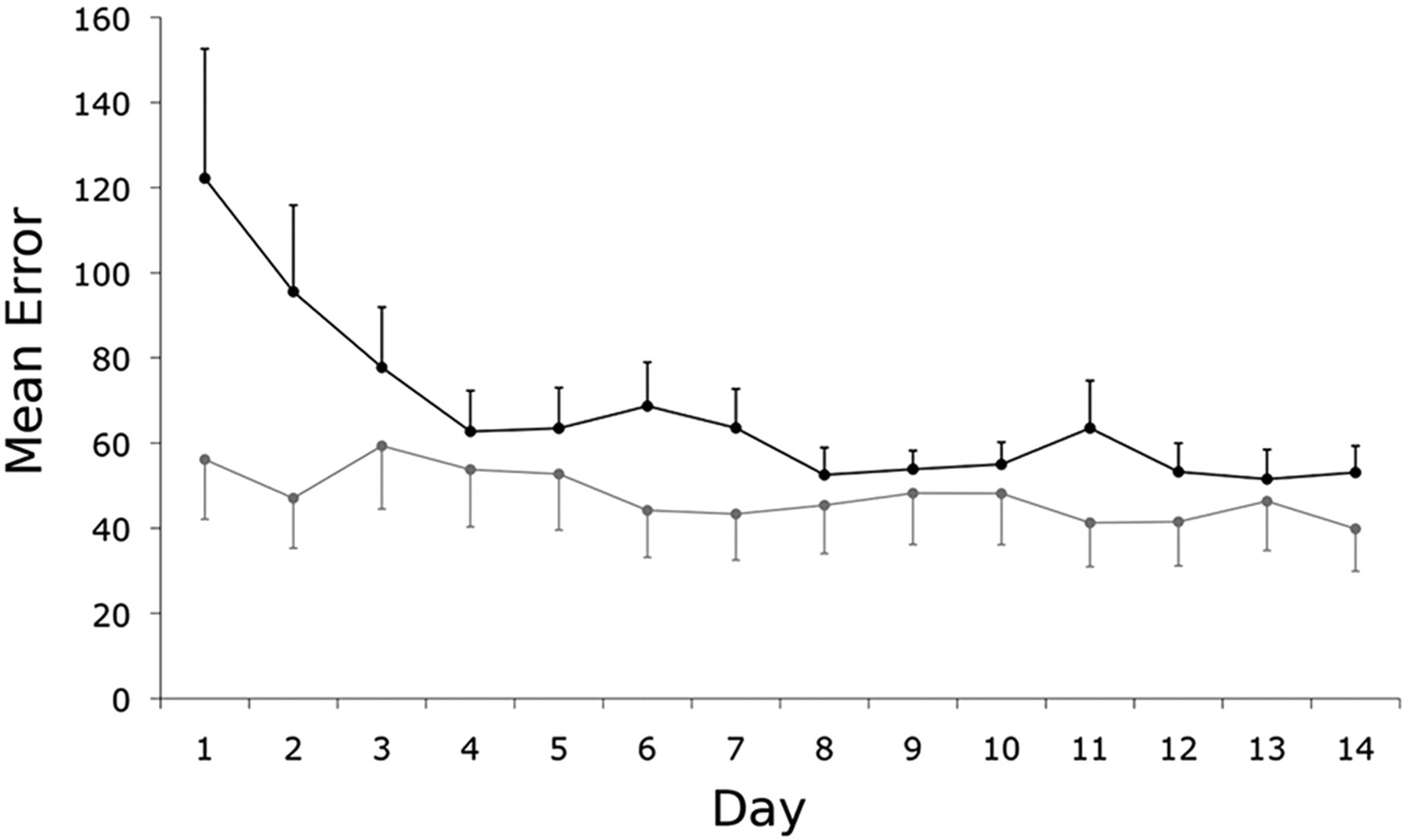
Home practice was associated with improved performance on the visuomotor tracking task for the patients. The mean error per block or per day for patients (black) and healthy controls (gray) during home practice sessions decreased monotonically over the practice period. The bars reflect standard errors of the mean.
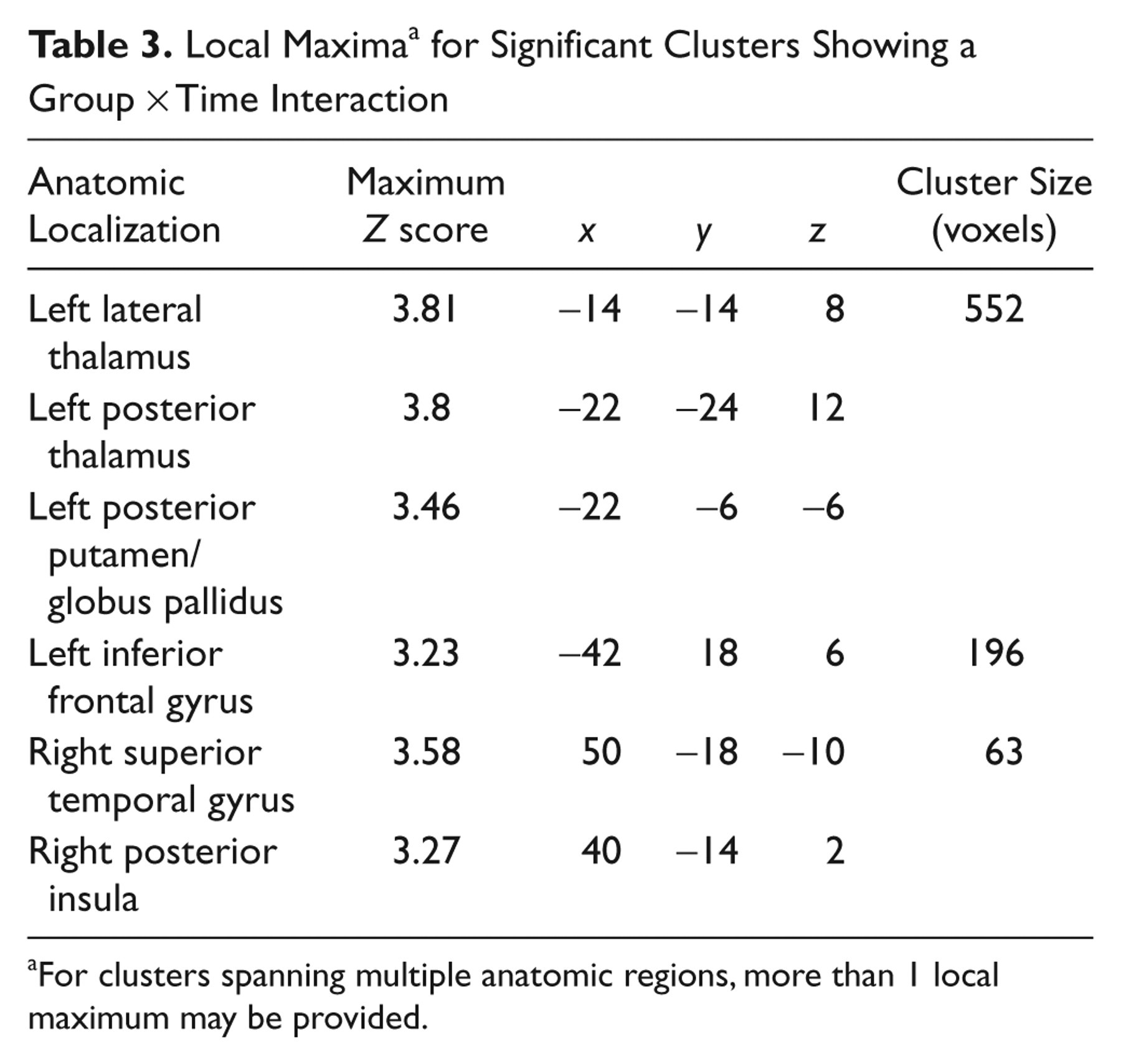
fMRI was repeated during performance of the visuomotor task after the home practice period. The main effects of task were similar to those at baseline (data not shown). We tested for between-group differences in activation changes between the baseline and postpractice follow-up examinations; significant Group × Time interactions were found in the left inferior frontal gyrus, bilateral insula, and right superior temporal gyrus. Subcortically, interactions were found in the left ventrolateral and lateral posterior thalamic nuclei, left globus pallidus, and left posterior putamen (Figure 4A; Table 3). Post hoc analyses were performed to characterize these regional activation differences (Figures 4B and 4C). We found that the group differences were a consequence of decreases in activation for the healthy controls after practice and either no change or increases in activation for the patients. A double dissociation in the direction of activation changes with practice for patients relative to controls was found in the thalamus and insula.
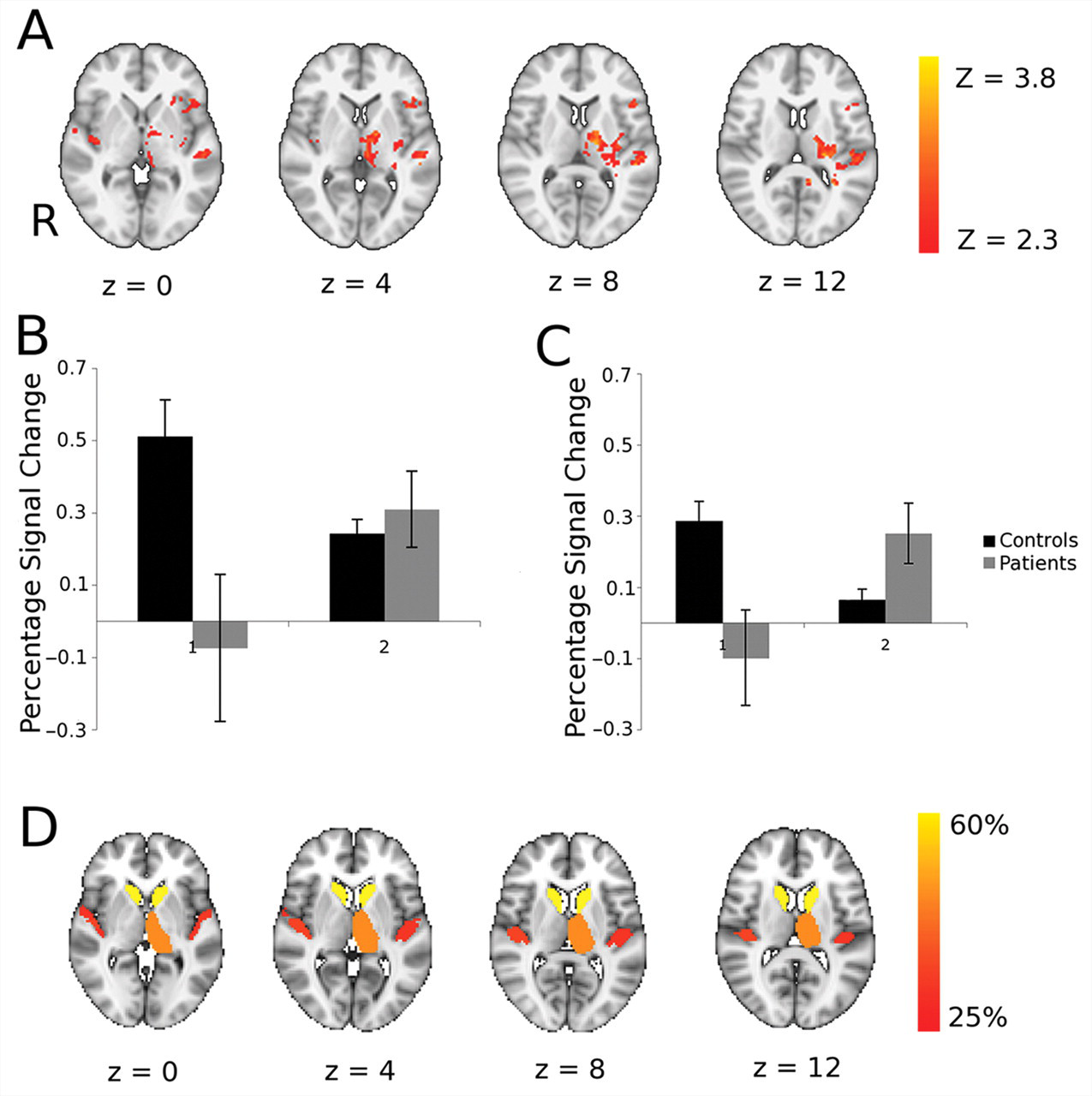
Patients showed increased brain activation with motor practice relative to controls, and many of these brain regions correspond well with those that had impaired white matter connectivity at baseline. A. Brain regions showing significant differences between patients and controls for task performance at baseline versus performance following 3 weeks of task practice. Axial slices at MNI levels indicated were thresholded at an initial cluster-forming threshold of Z > 2.3 and a corrected cluster extent threshold of P < .05. The left-hand side of the brain (ipsilesional in patients) is displayed on the right-hand side of each brain slice. See Table 3 for details of local maxima. Region-of-interest analyses to characterize direction of activation change in clusters showing Group × Time interaction in the thalamus (B) and insula (C). D. Brain atlas regions with reduced white matter connectivity at baseline in patients relative to healthy controls. To identify these, probabilistic diffusion MRI tractography was performed, and mean probabilities of connectivity to other brain regions were measured for each atlas region. The color scheme indicates the percentage reduction in connectivity probability in patients compared with controls, and all regions showing 25% or more reduction are colored.
Local Maxima a for Significant Clusters Showing a Group × Time Interaction
For clusters spanning multiple anatomic regions, more than 1 local maximum may be provided.
We tested whether the differential activation changes with practice for the patients relative to controls could be found in regions in which connectivity was impaired at baseline by white matter damage from subcortical stroke. To do this, we used probabilistic white matter tractography to estimate the mean connection probability between an atlas set of cortical and subcortical brain regions across the whole brain for the patients and for the healthy controls. Many of the regions that showed increased activation with performance gains after practice in the patients (Figure 4A) also had reduced connectivity at baseline in the patients relative to the healthy controls (Figure 4D).
Discussion
As expected, we found differences in brain activation during visuomotor tracking between patients and age-matched healthy controls. At baseline, patients had decreased task-associated activation relative to the healthy controls. After 15 days of motor practice, strikingly different patterns of practice-related brain activation changes were found between the 2 groups, notably including those regions in which white matter connectivity was reduced. These results highlight a novel anatomic overlap between brain regions showing practice-mediated increases in activation after stroke and those in which direct or indirect injury led to impaired function at baseline with reduced white matter connectivity. They suggest a specific role for motor practice in mediating functional recovery of the injured brain after stroke.
Baseline Differences Between Brain Activity in Patients and Controls While Performing the Visuomotor Tracking Task
At baseline, patients had reduced activation relative to the healthy controls in cortical regions (primary sensorimotor, premotor, and supplementary motor cortices of the lesioned hemisphere) involved in motor control 25,26 and anatomically interconnected 27,28 subcortical areas (ventrolateral and anterior thalamus bilaterally and contralesional cerebellum, lobules V and VI). This finding is consistent with the patterns of brain injury, characterized by varying degrees of interruption of corticothalamic-cerebellar motor pathways.
The relative reduction in motor cortical activity in patients relative to healthy controls that we observed should be contrasted with previous reports that have emphasized increased movement-associated activation in patients after a stroke. 29,30 The differences may arise from task differences because the majority of previous studies have used simpler movement tasks than the one used here; a limited adaptive capacity or “reserve” for compensatory activation in the injured brain may be made apparent only with more difficult or complex tasks. 31 In addition, with a learning task of the sort used here, there may be interindividual variation in the effects of a fixed amount of short-term task practice on brain functional responses. Whereas some participants may have reached a performance plateau, others could continue to improve if allowed to practice for longer (as shown in a previous study of long-term practice effects on brain responses) 32 , and so any associated gains in brain functional responses may not be maximal. Future studies with varied practice schedules, larger groups, and a range of motor tasks could test these hypotheses directly.
Dependence of Baseline Task Performance on White Matter Microstructure
We found that the loss of white matter integrity in the PLIC explained a substantial proportion of the variance in performance error reductions with baseline short-term task practice for the patients. The PLIC region implicated includes fiber tracts projecting from frontal cortical regions, including the premotor cortex. 33 Previous work has identified premotor regions as important for hand motor control following damage 30,34,35 and has emphasized the importance of descending motor outputs in both the lesioned and the contralesional hemisphere. 36
We believe that the association between the behavioral effects of motor practice and white matter microstructure in the contralesional PLIC most likely reflects transsynaptic changes occurring as a consequence of the strokes. 37,38 However, it also could reflect differences in the microstructure of white matter prior to the stroke; individual differences in performance are associated with variation in FA in task-relevant pathways, even in healthy individuals. 39 Although regional correlations between FA and performance were not found for the healthy controls in the current study, the dependence of performance on the microstructure of undamaged white matter regions may be greater in patients as a consequence of their brain injury. This is consistent with clinical observations of worsening motor impairment in a stroke-affected paretic limb following the development of a new lesion in the ipsilateral cortex. 40
Effects of Motor Practice on Task-Related Brain Activity
Control of grip force demands integration of sensory input and motor output in brain regions, including the motor, somatosensory, premotor, and parietal cortices. 41 Implicit learning of a sequence of grip forces is associated with a shift of activation from cortical to subcortical activation in a thalamic–basal-ganglia–premotor network. 6 Whereas previous studies of the effects of motor practice following stroke have provided useful insights into changes specifically in motor cortical areas, 42 we assessed changes across the brain more widely and were therefore able to provide novel evidence on changes in polymodal areas and subcortical nuclei. With long-term task practice, we found a double dissociation between patterns of practice-related changes in brain activity in patients and healthy controls in cortical regions, including the inferior frontal gyrus, insular and superior temporal regions, and in the basal ganglia and thalamus subcortically. In healthy controls, task performance at follow-up was associated with significantly lower activity in these brain regions than performance of the same task at baseline, consistent with the notion of increasing efficiency of motor activity for performance of highly practiced or overlearned movements. 43 However, opposite trends were seen in patients, who showed an increase in task-related activity in these regions following practice, analogous to observations of increased recruitment of task-relevant areas with short-term motor learning for healthy participants. 6,44-46 The observation of increased brain activation with practice for patients is consistent with that in previous studies of rehabilitation interventions, which have shown that good treatment outcomes are associated with increased activation in brain regions relevant to the task. 32,47,48
Earlier studies of brain activation with simple movement have emphasized adaptive changes in secondary motor control regions such as the premotor cortex. 29,34,35,49 A recent, well-designed study of implicit visuomotor learning with a joystick highlighted increasing prefrontal activation in patients poststroke with learning, in contrast to decreasing activity in healthy controls. 10 This difference was interpreted in terms of adaptive increases in attentional control with learning for the stroke patients.
Increased Activation of Brain Regions With Impaired Connectivity in Patients After Motor Practice
We found evidence for reduced structural connectivity not only local to the stroke but also in more distant regions, including the contralesional hemisphere. Whereas diffusion studies of FA have revealed patterns of anterograde (Wallerian) and retrograde white matter tract degeneration following focal damage, 13-15,50 such effects are rarely reported in the contralesional hemisphere. However, observations of transhemispheric diaschisis 37 support the concept that widespread interconnected regions, even in the contra lesional hemisphere, can be functionally and structurally altered after a focal lesion. Elsewhere, we have used a subset of the data reported here to characterize a novel, multivariate approach to the analysis of white matter connectivity that suggests a potential for explicitly network-based approaches for detecting this kind of distributed neuropathology. 38
However, in the current report, we have assessed connectivity simply using averaged pairwise connection probabilities generated from a tractography approach and have compared the results of this analysis with fMRI findings. The regions with reduced anatomic connectivity at baseline showed a striking overlap with regions in which functional activity increased (despite general decreases in activation in healthy controls) with task practice in the stroke patients. The between-group differences in longitudinal change of brain activity suggest that motor practice plays distinct roles in patients as compared with healthy individuals. In the healthy controls, practice is associated with regionally increased functional efficiency, 43 but in patients, practice enables increased functional recruitment of at least some of the regions that are structurally compromised. Although we presume that both rely on mechanisms of practice-related brain plasticity, we infer from the different directions of longitudinal activation change that the local circuit adaptations are distinct.
There is considerable overlap between the structurally impaired thalamic, basal ganglia, and superior temporal regions showing practice-related increases in patients (Figure 4) and areas showing reduced activity at baseline (Figure 3). Taken together, these findings further suggest that chronic, degenerative transsynaptic changes after a stroke contribute to impaired performance but that a behavioral intervention (eg, motor practice) can increase functional recruitment of affected areas, presumably reflecting local, activity-dependent plasticity.
We recognize clear limitations to our study. First, the number of patients tested was relatively small. Although observation of significant effects despite modest patient numbers emphasizes the magnitude of the detected relationships, other more subtle associations may have been missed. By selecting a relatively homogeneous patient population, we attempted to improve our sensitivity to detect effects, but this may limit generalization to a wider stroke population. More complete evaluation with a much larger patient group could test the relationship between differences in the anatomic distribution of impaired connectivity with different sizes or localization of stroke and individual differences in practice-related brain activation. Furthermore, we used only a single visuomotor paradigm, although we took care to ensure that this provided objective and quantitative performance measures. Future work should contrast outcomes with practice of a broader range of activities and compare their effects with appropriate control activities and explore the influence of differences in practice duration and schedules. Although longitudinal changes with practice of a single motor task provides a simple model for recovery, 2 it cannot capture the range of cognitive processes that contribute to outcomes from a clinical rehabilitation intervention.
Conclusions
We found evidence for differences in the dynamics of motor practice–related brain functional plasticity in patients following stroke relative to healthy controls and related these to patterns of structural degeneration. Our observations provide novel, direct evidence that motor practice—a central component of most approaches to neurorehabilitation—promotes functional recovery of brain regions in which structural integrity is directly or indirectly impaired by stroke.
Footnotes
Acknowledgements
We are grateful for technical assistance from Iain Wilson.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PMM became a full-time employee of GlaxoSmithKline after the design and initial implementation of this experiment.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Stroke Association (Grant Number
