Abstract
Background. Poststroke impairments of the ipsilesional arm are often discussed, but rarely receive focused rehabilitation. Ipsilesional deficits may affect daily function and although many studies have investigated them in chronic stroke, few characterizations have been made in the subacute phase. Furthermore, most studies have quantified ipsilesional deficits using clinical measures that can fail to detect subtle, but important deficits in motor function. Objective. We aimed to quantify reaching deficits of the contra- and ipsilesional limbs in the subacute phase poststroke. Methods. A total of 227 subjects with first-time, unilateral stroke completed a unilateral assessment of motor function (visually guided reaching) using a KINARM robot. Subjects completed the task with both the ipsi- and contralesional arms. Subjects were assessed on a variety of traditional clinical measures (Functional Independence Measure, Chedoke-McMaster Stroke Assessment, Purdue Pegboard, Behavioral Inattention Test) to compare with robotic measures of motor function. Results. Ipsilesional deficits were common and occurred in 37% (n = 84) of subjects. Impairments of the ipsilesional and contralesional arm were weakly to moderately correlated on robotic measures. Magnitude of impairment of the contralesional arm was similar for subjects with and without ipsilesional deficits. Furthermore, we found that a higher percentage of subjects with right-hemisphere stroke had ipsilesional deficits and more subjects with left-hemisphere subcortical strokes did not have ipsilesional deficits. Conclusions. Magnitude of contralesional impairment and lesion location may be poor predictors of individuals with ipsilesional impairments after stroke. Careful characterization of ipsilesional deficits could identify individuals who may benefit from rehabilitation of the less affected arm.
Introduction
Impairment of the ipsilesional arm after unilateral stroke is an acknowledged, but typically untreated aspect of stroke. Often, ipsilesional deficits are described as “subtle,” 1 and functional recovery after stroke is thought to be highly dependent on motor recovery of the contralesional arm.2-4 While not as overt as contralesional deficits, ipsilesional deficits can significantly affect functional recovery after stroke.5,6 In daily life, many tasks require use of both the contralesional and ipsilesional limbs (eg, eating with a knife and fork), and the presence of ipsilesional deficits may further affect a persons’ ability to perform such tasks throughout recovery after stroke. In some cases, the contralesional arm is so significantly impaired that individuals are taught to adopt compensatory strategies that rely on the ipsilesional arm, or that may require minimal assistance from the contralesional arm. In individuals who rely primarily on their ipsilesional arm, the presence of ipsilesional deficits may significantly impact daily function, and have the potential to negatively affect long-term recovery, independence, and quality of life.
To date, several studies have described impairments of the ipsilesional limb, including slower hand movements, 7 impairments in hand dexterity, 1 and coordination.8,9 The majority of these studies were conducted in individuals in the chronic phase poststroke (>1 year), suggesting that ipsilesional deficits persist long-term following stroke. Two recent studies were conducted in small groups of subjects in the subacute phase (<6 months) and examined clinical and kinematic measures of the ipsilesional arm.10,11 They found that the ipsilesional arm was impaired in reach-to-grasp type movements in the subacute phase and that these deficits persisted at the studies’ conclusion (3 months).
While the above studies suggest that ipsilesional deficits can be seen in both the subacute and chronic phase of stroke, many were conducted in small groups of subjects with highly selected lesions. Some authors have suggested that the nature of ipsilesional deficits depends on hemisphere of stroke lesion, with right-hemisphere lesions affecting ipsilesional control of movement endpoint 12 and left-hemisphere lesions affecting ipsilesional movement coordination and trajectory.13,14 Our experience suggests these findings do not allow for full appreciation of the heterogeneity of altered behavior that can occur in the poststroke ipsilesional limb nor completely describe its relation to potential lesion locations. To date, the largest study (n = 72) we are aware of investigated ipsilesional motor function after stroke and found that recovery of ipsilesional impairment was highly variable from subject to subject. 15 While this study examined the characteristics of ipsilesional deficits over time, the authors did not examine the relationship between contralesional and ipsilesional impairment.
Recently, we have developed robotic technology to assess unilateral motor behavior in individuals with stroke using an exoskeleton.16,17 Our main goal in this study was to better characterize motor function of the contralesional and ipsilesional arms in the subacute phase poststroke. Second, we aimed to make objective comparisons in motor function between the contralesional and ipsilesional limbs using robotic assessment. Last, we aimed to fully characterize the clinical and neuroanatomical characteristics of individuals with and without ipsilesional deficits.
Methods
Two hundred twenty-seven subjects with first-time, unilaterally affected stroke were recruited from 2 locations in Calgary, Alberta (Foothills Hospital and the Dr. Vernon Fanning Centre). Subjects were tested approximately 1-week post-troke (Table 1). Subjects were excluded if they could not understand 3-step instructions, had evidence of apraxia as evaluated by an experienced physiotherapist,18,19 and had significant musculoskeletal injuries, neurologic disease, or injury unrelated to stroke. Furthermore, subjects were excluded from the present study if they had bilateral strokes, brainstem, or cerebellar involvement, as confirmed on computed tomography (CT) or magnetic resonance imaging (MRI). The study was approved by the University of Calgary Research Ethics Board, and all subjects provided informed consent.
Subject Demographics.
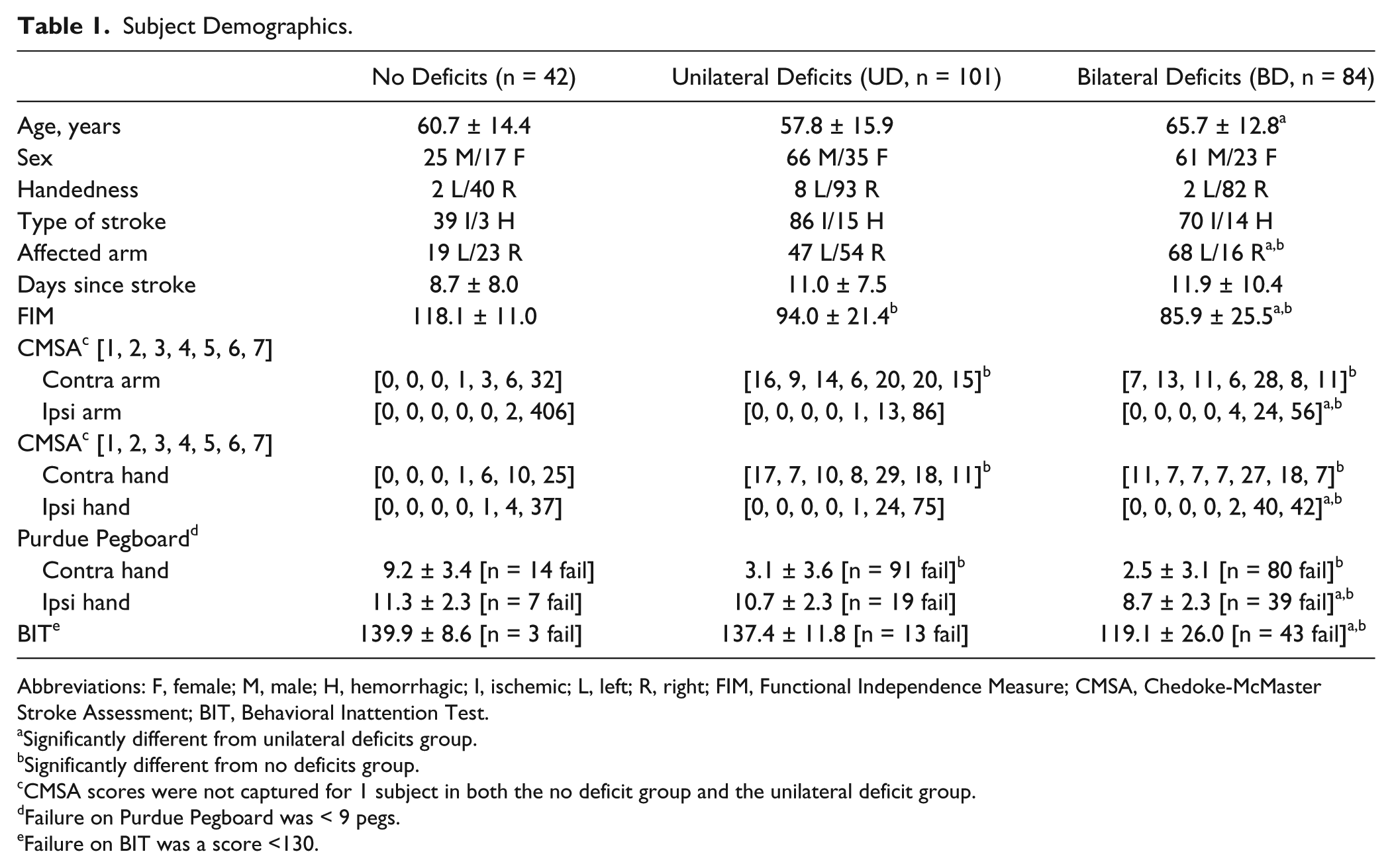
Abbreviations: F, female; M, male; H, hemorrhagic; I, ischemic; L, left; R, right; FIM, Functional Independence Measure; CMSA, Chedoke-McMaster Stroke Assessment; BIT, Behavioral Inattention Test.
Significantly different from unilateral deficits group.
Significantly different from no deficits group.
CMSA scores were not captured for 1 subject in both the no deficit group and the unilateral deficit group.
Failure on Purdue Pegboard was < 9 pegs.
Failure on BIT was a score <130.
Robotic Measures of Motor Control
To evaluate unilateral motor function, subjects performed a visually guided reaching (VGR, Figure 1) task using the KINARM robotic exoskeleton (BKIN Technologies Ltd, Kingston, Ontario, Canada, Figure 1A). The task and subsequent measurement parameters have been previously described.16,20,21 Subjects sat in the wheelchair base of the robotic exoskeleton with their arms supported against gravity by the arm troughs of the exoskeleton. With the subject aligned in the middle of the seat, the robotic exoskeleton was custom-fit to each individual’s limb geometry. Subjects without proper trunk control were fit with a restraint harness affixed to the robotic exoskeleton (<3% of subjects).
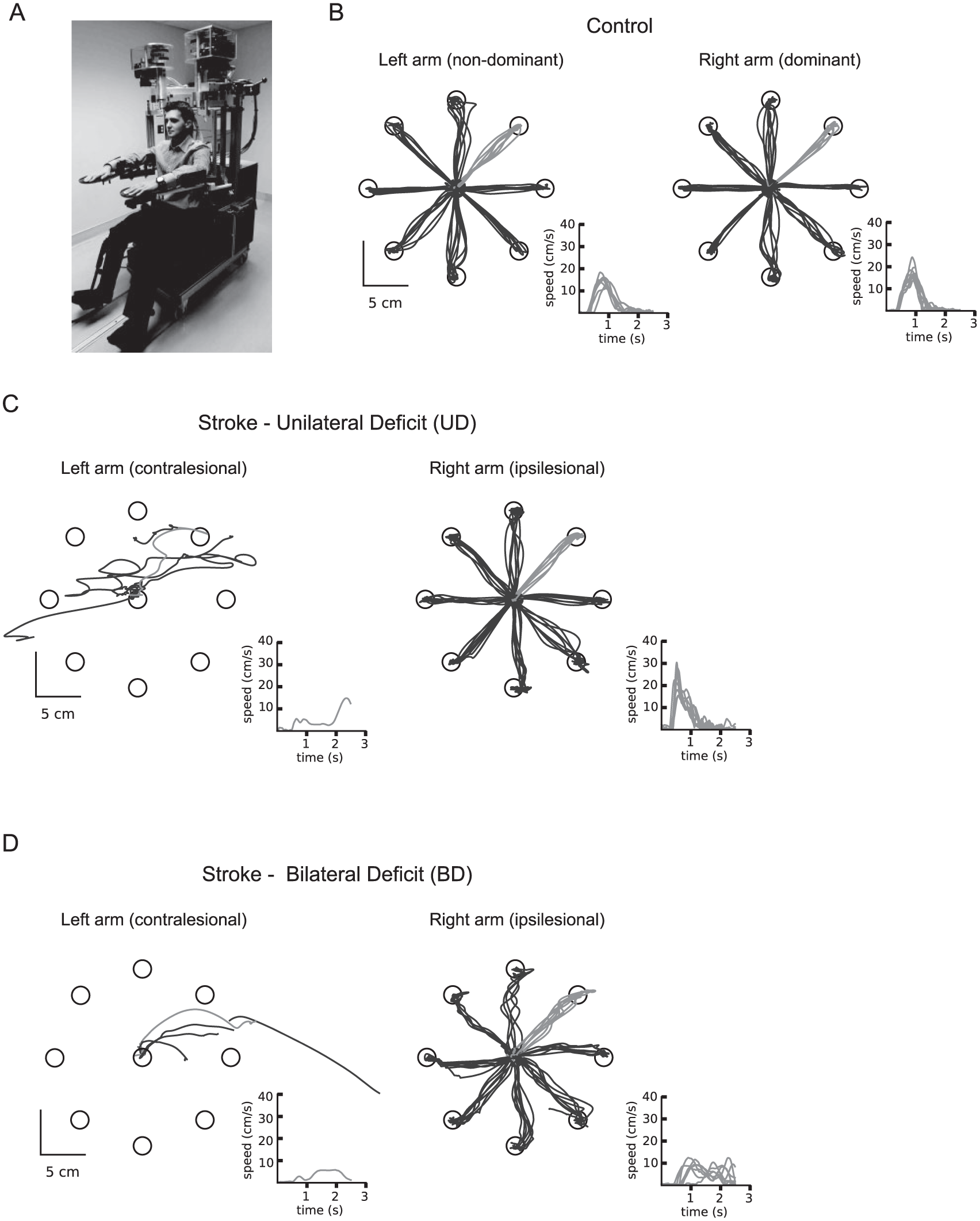
Exemplars of reaching in control, stroke with and without ipsilesional deficits. (A) Picture of the KINARM robotic exoskeleton. (B-D) Exemplar positional and hand speed data from the visually guided reaching (VGR) task for a control subject (B), a subject with stroke in the unilateral deficit (UD) group, a subject with stroke in the bilateral deficit (BD) group. Note the difference in hand path and speed traces for subjects displayed in C and D.
Subjects viewed visual feedback of their hand as a 0.4-cm white dot aligned with their index fingertip through a horizontally mounted virtual reality display. Subjects were required to align the dot with a center target (1-cm radius), and make a 10-cm reaching movement when 1 of the 8 peripheral targets appeared. Subjects were instructed to move as quickly and accurately as possible. Target presentation was pseudorandomized within a block and subjects made 8 movements to each of 8 targets (64 total movements). Subjects performed the task with the contralesional and ipsilesional arms.
Motor performance was evaluated using 9 parameters16,20,21: posture speed (PS)—a measure of hand speed when maintaining their hand at the central target, reaction time (RT)—the time it takes a subject to initiate a movement in response to target appearing, initial direction error (IDE)—the amount of angular error between the subject movement and an ideal straight-line movement, initial distance ratio (IDR)—the ratio of initial movement length to full movement length, speed maxima count (SMC)—number of speed peaks in the movement, minimum/maximum speed difference (MMSD)—mean difference between pairs of local speed maxima and minima, movement time (MT)—time to complete movement between movement onset and offset, path length ratio (PLR)—ratio of the total distance of the subject movement length to the total distance of the ideal movement length, and maximum speed (MS)—peak hand speed of the movement.
For all subjects with stroke, parameter scores were normalized to standardized scores based on performance data that was compared with normative ranges (95% confidence interval) derived from neurologically intact control subjects (n = 157) that included the influence of age on performance. Nonnormal distributions for healthy control performance were converted to normal distributions based on Box-Cox transforms. 22 A subject with stroke was considered significantly impaired on a parameter if their score fell outside 95% performance of healthy controls (z > 1.65). Additionally, we computed an overall, global task score derived from the means of all calculated parameters (Task Score). The Task Score is calculated as the root-mean-square distance from mean performance of all parameters of neurologically intact control subjects and compared to the range of scores measured for a population of healthy controls. All parameter scores were normalized to standardized scores (z-scores). We then classified subjects with a Task Score z > 1.65, as significantly impaired and failing the task.
Some subjects had plegia of the contralesional upper limb (ie, unable to move the limb at all), and were only tested on the ipsilesional limb (n = 9). A small number of subjects (n = 7) attempted the kinematic testing, but were so significantly impaired with their contralesional arm that they did not complete enough trials for our algorithm to appropriately parse their data. Given previous suggestions of the relationship between the severity of impairments in the contralesional limb and the ipsilesional limb,6,23 it was important to present these data. Thus, these subjects (n = 16) were given the most impaired z-scores for all parameters, and were eliminated from correlation analyses (described below) to minimize skewing the data distribution.
For each subject with stroke, pass/fail task performance was determined by individual subject performance on the Task Score for the contralesional and ipsilesional arms. Those subjects that performed abnormally on the contralesional arm (Task Score contralesional arm, z > 1.65), but normally on the ipsilesional arm (Task Score ipsilesional arm, z < 1.65) were classified as having unilateral deficits (unilateral deficit [UD] group). Subjects who performed abnormally on both the contralesional and ipsilesional arms (z > 1.65 both arms) were classified as having bilateral deficits (bilateral deficit (BD) group).
Clinical Measures
Subjects were assessed on measures of functional ability (Functional Independence Measure [FIM] 24 ), motor function of the arm and hand (Chedoke-McMaster Stroke Assessment [CMSA], 25 Purdue Pegboard [PPB] 26 ), visual fields, visuospatial function (conventional subset of the Behavioral Inattention Test [BIT] 27 ) and handedness. 28
We compared performance on the PPB between neurologically intact subjects (n = 314) and those with stroke (described above). Ninety-five percent confidence intervals were calculated from neurologically intact control subjects for PPB performance of the dominant and nondominant limbs. Subjects with stroke were considered abnormal if they fell outside the 95% normative range, which was less than 9 pegs for the dominant or nondominant limb in neurologically intact control subjects.
Imaging
MRI (T2 FLAIR [fluid-attenuated inversion recovery] and DWI [diffusion-weighted imaging]) and CT scans were acquired at the time of stroke (3.1 ± 5.8 days poststroke), according to acute stroke protocols at the Foothills Medical Centre. Imaging was collected on 1 of 2 CT scanners, a General Electric 1.5-T or 3-T MRI scanner, with slice thickness varying from 3.0 to 5.0 mm depending on the scanner type. All scans had at least a 1 × 1m in-plane resolution. These scans were taken for clinical diagnostic purposes and obtained for use in this study. Stroke lesions were manually marked on each axial slice of T2 FLAIR or CT using a digitizing tablet and MRIcron software 29 to create a volume of interest (VOI). Lesion markings were confirmed by a stroke neurologist who was blinded to the nature of subjects’ behavioral impairments. DWI scans were taken as authoritative to indicate the areas of acute ischemia. Each subject’s FLAIR and VOI was then normalized to the Montreal Neurological Institute (MNI) template using the clinical toolbox in SPM 8.29,30 FLAIR and CT-specific template images were used for the normalization process and cost function masks were used to prevent distortion of damaged brain tissue. 31 Overlap maps and lesion volumes were then calculated using MRIcron software.
Lesion locations of subjects were identified from MRI scans or CT scans and those that were classified as cortical, subcortical or cortical + subcortical were included in the study. Cortical lesions were those only involving cortical gray matter areas. Subcortical lesions were those involving white matter or white matter tracts. Cortical + subcortical were a combination of cortical and subcortical lesions.
Data and Statistical Analyses
Two-sample Kolmogorov-Smirnov tests were used to test for distribution differences in demographic, clinical and robotic measures. One-way analyses of variance were used to make between-group comparisons for subject characteristics and clinical measures. Tukey and Bonferroni post hoc tests were used to correct for multiple comparisons when appropriate. Spearman correlations were used to determine the relationship between contralesional and ipsilesional motor performance. Unpaired t tests were used to determine statistical differences between lesion volume across groups and across hemispheres. Fisher’s exact test was used to determine categorical group differences for stroke groupings, lesion location and/or hemisphere of lesion. Fisher’s exact test with Freeman-Halton extension was used for 2 × 3 contingency comparisons. Cohen’s kappa was calculated to test level of agreement between clinical and robotic measures.
Results
We assessed contralesional and ipsilesional reaching behavior in 227 subjects (152 male/75 female) in the subacute phase poststroke (10.9 ± 8.8 days). One hundred ninety-five subjects had ischemic strokes and 32 had hemorrhagic strokes, with 135 subjects with right-hemisphere strokes (predominantly affected on the left-side of the body) and 92 with left-hemisphere strokes (predominantly affected on the right-side of the body). Fifty-nine subjects (26%) were found to have visuospatial neglect, as measured by the BIT.
Figure 1 displays data for a typical neurologically intact control subject (Figure 1B), a subject with stroke with only contralesional (unilateral) deficits (Figure 1C), and a subject with stroke with contralesional and ipsilesional (bilateral) deficits (Figure 1D). From visual inspection, the subject with bilateral deficits (BD) makes larger directional errors and slow, longer movements with their ipsilesional arm compared to the neurologically intact control and the ipsilesional arm of the subject with unilateral deficits (UD). Notably, performance with the contralesional arm for the UD and BD subjects look similar.
For all subjects, we compared performance of the contralesional arm to the ipsilesional arm (Figure 2) to determine if level of impairment was correlated. Figure 2A displays overall task performance and shows that subjects with ischemic and hemorrhagic strokes display similar variabilities in the relationship of impairment between contralesional and ipsilesional arms. We found moderate correlations between impairment of the contralesional and ipsilesional arms for all nine individual kinematic parameters (Figure 2, Rs: PS = 0.50, RT = 0.59, IDE = 0.30, IDR = 0.40, SMC = 0.42, MMSD = 0.38, MT = 0.41, PLR = 0.34, MS = 0.59). While we found that many subjects had similar impairment on the ipsilesional and contralesional arms (Figure 2), many subjects did not. The open triangle shapes in each of the subpanels of Figure 2B-D highlight the exemplar subjects in Figure 1 who presented with similar magnitude of impairment on several parameters of the contralesional arm, but very different performance with the ipsilesional arm.
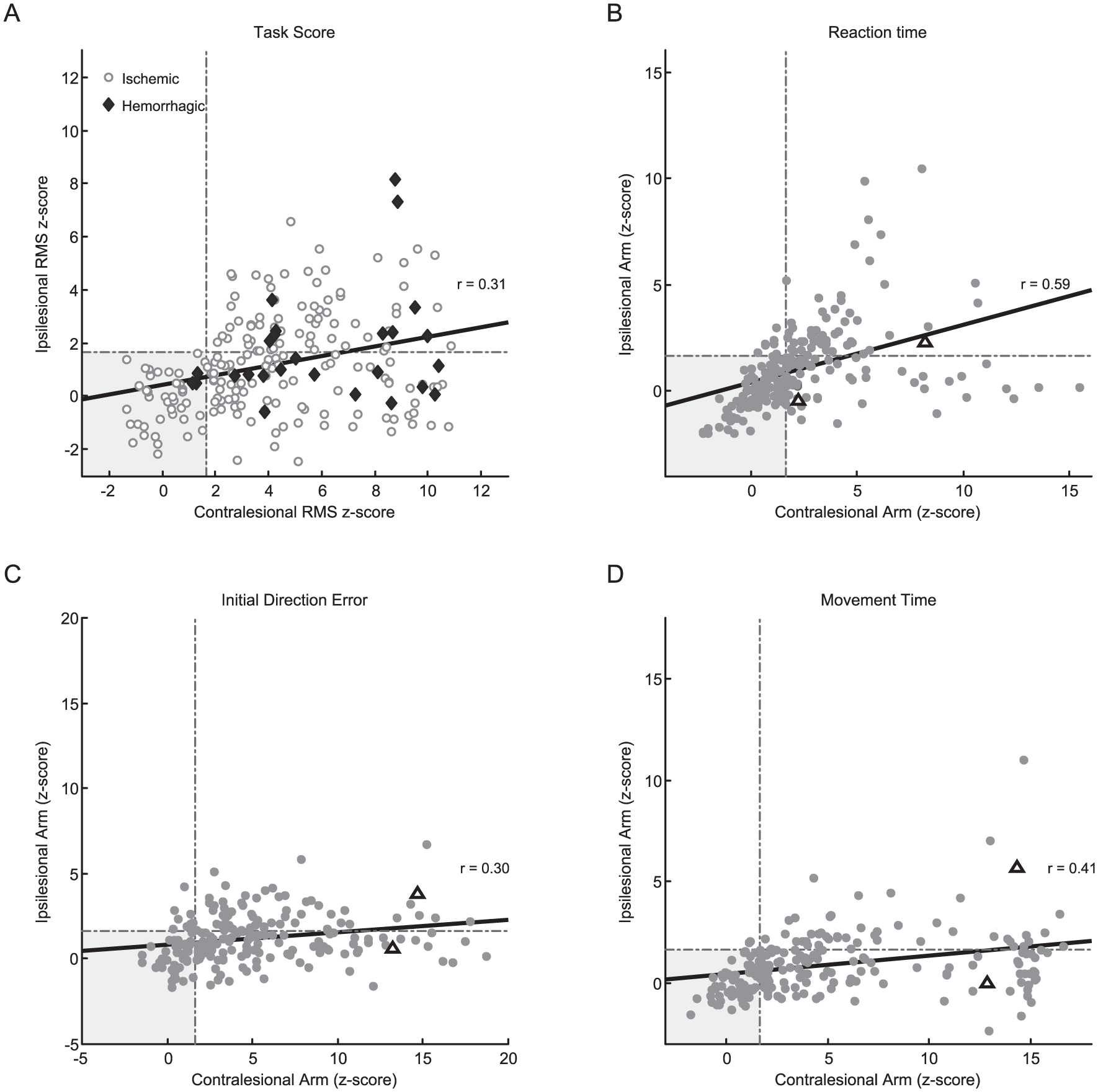
Individual parameter correlations of the affected arm to unaffected arm. Scatterplots of the contralesional arm versus the ipsilesional arm across all subjects for overall task performance (A) and 3 parameters of the reaching task (B-D). The gray box signifies the boundary of normal performance of both the contra- and ipsilesional arms, with the dotted gray lines denoting the normative control range. In A, those with ischemic (circles) and hemorrhagic stroke (diamonds) are plotted with different symbols to highlight the variability in performance between the 2 groups. In B-D, within each plot, the data of exemplar subjects in Figure 1 (C and D) are plotted as open triangles, to emphasize that impairment in the contralesional arm could be quite similar, while performance with the ipsilesional arm was quite different.
Characteristics and Magnitude of Ipsilesional Impairment
On kinematic testing, we found that 19% of subjects (n = 42) had no deficits in either arm, 45% of subjects (n = 101) had deficits in only the contralesional arm (UD group) and 37% of subjects (n = 84) had deficits in both the contralesional and ipsilesional arms (BD group, Table 1). We found no significant differences in measures of sex or handedness across the 3 groups, but found that the BD group was significantly older than the UD group, F(2, 226) = 5.97, P = .003, Tukey, P < .05. Further, a larger proportion of subjects in the BD group had right-hemisphere lesions compared with both groups, F(2, 226) = 16.0, P < .001, Tukey, P < .05.
Many UD and BD subjects had large impairments of the contralesional arm on kinematic testing. Notably, the distribution of impairments of the contralesional arm was similar for subjects in the UD and BD groups (Figure 3). Within the contralesional arm, between group (UD and BD) comparisons revealed that only 1 parameter (SMC) was significantly different (Supplemental Table 1). This suggests similar motor impairment of the contralesional arm for UD and BD subjects. Not surprisingly, the frequency of ipsilesional impairment in individual parameters was much higher in BD subjects (eg, MT, 12% fail [UD group], 52% [BD group]), with 7 out of 9 parameters demonstrating a significant difference for the ipsilesional arm between UD and BD subjects (Supplemental Tables 1 and 2). Furthermore, we compared the impact of left- and right-hemisphere damage on ipsilesional and contralesional limb performance. We found no differences in the prevalence of impairment in any of our robotic parameters based on the hemisphere that was damaged within either the UD or BD group (Supplemental Table 2).
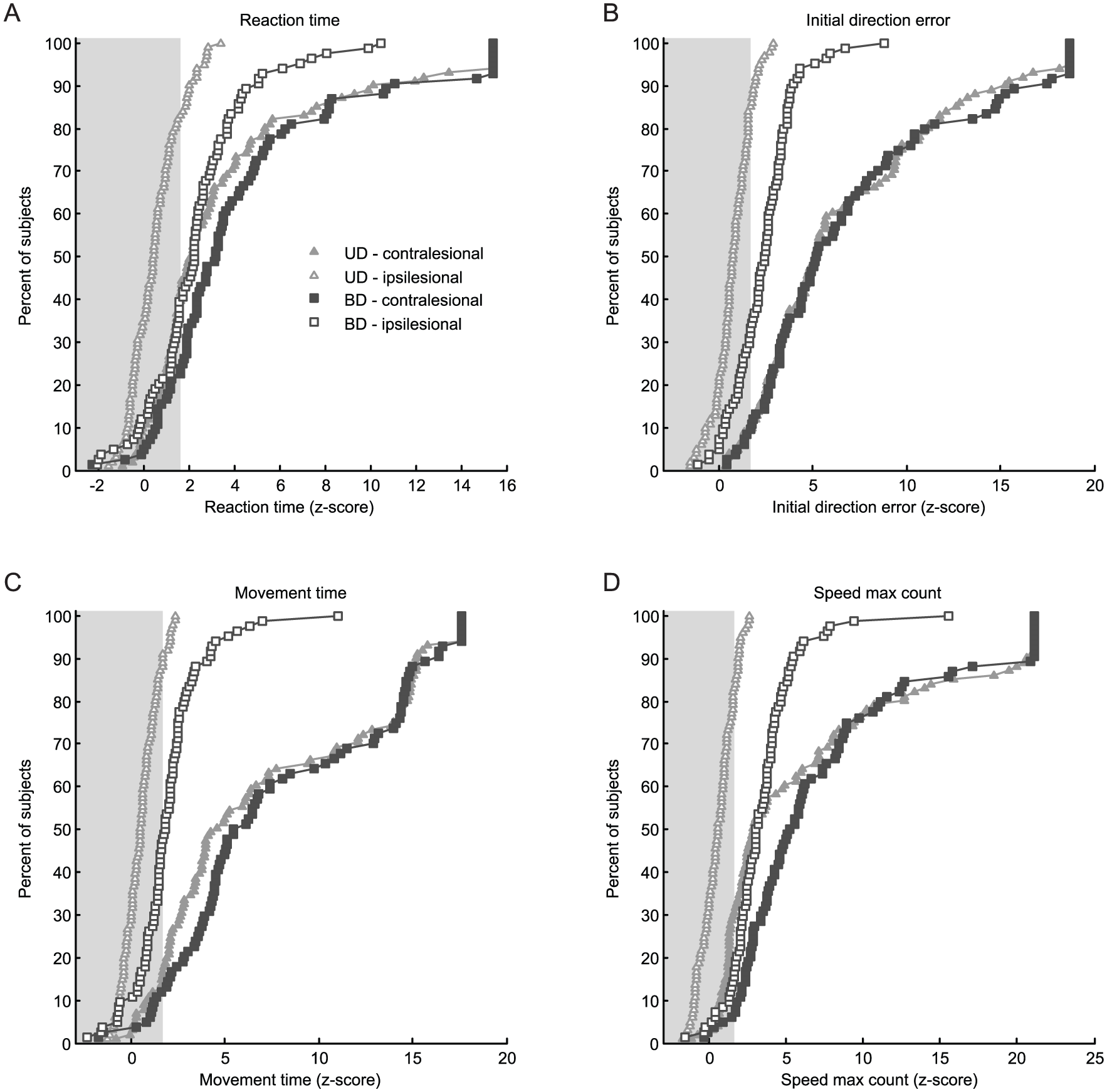
Cumulative sum histograms of the ipsi- and contralesional arms for subjects with and without ipsilesional deficits for several parameters of the visually guided reaching (VGR) task. (A) Reaction time (RT), (B) initial direction error (IDE), (C) movement time (MT), and (D) speed maximum count (SMC). The gray box indicates the 95% normative control range, any value outside this range is significantly different from controls. We found that the percent of subjects with impairments for the contralesional arms in the unilateral deficit (UD) and bilateral deficit (BD) groups within parameters were highly similar (solid shapes). In contrast, the number of subjects with impairment in the ipsilesional arm (open shapes), was highly different, with many more subjects in the BD group failing individual reaching parameters.
Representation of Clinical Scores
Subjects that were identified with ipsilesional deficits (BD group) during reaching were the most impaired on many clinical measures. Compared with subjects with no deficits and those in the UD group, BD subjects had significantly lower functional (FIM), motor (CMSA, PPB), and visuospatial (BIT) scores (Table 1). We found that 27% of subjects (n = 59) had visuospatial neglect, with the BD group having the highest percentage of neglect subjects (ND: 7% [n = 3], UD: 13% [n = 13], BD: 51% [n = 43]). We found that those subjects with neglect did not necessarily have the highest magnitude of motor impairment of the contralesional or ipsilesional arms (Supplemental Fig. 1).
While robotic measures identified 37% (n = 84) of the 227 subjects with ipsilesional deficits, we found that the CMSA arm identified 19% (n = 44), CMSA hand identified 32% (n = 72) and PPB identified 29% (n = 65) of subjects with ipsilesional impairment (Table 1, Supplemental Figures 2 and 3). Furthermore, these clinical measures had fair agreement with the robotic measures according to Cohen’s kappa for the ipsilesional arm (CMSA arm: κ = 0.24, CMSA hand: κ = 0.30, PPB: κ = 0.30), and moderate to substantial agreement for the contralesional arm (CMSA arm: κ = 0.54, CMSA hand: κ = 0.49, PPB: κ = 0.62). Despite these levels of agreement, we found that our kinematic measures identified many subjects that scored a 7 (normal) on the CMSA as having significant impairment in the contra- or ipsilesional arms (Supplemental Figure 3).
Characteristics of Lesion Location Compared With Robotic Performance
In order to better delineate the lesion locations of our sample, we created maps (Figure 4, Supplemental Figure 4) to display the areas of lesion overlap for subjects in the entire sample, the ND, UD, and BD groups. In the BD group, we observed fewer lesions in the left than right hemisphere, but observed the most anatomic overlap in the thalamus (n = 8) within the left hemisphere, and the anatomic areas of most overlap in the right hemisphere were the insula, external capsule, putamen, internal capsule, and superior corona radiata (n = 25). Lesion volume in the BD group was, on average, 14.7 ± 19.3 cm3 in the left hemisphere, and 44.0 ± 53.5 cm3 in the right hemisphere. In the UD group, the anatomic areas of most overlap in the left hemisphere were the putamen and internal capsule (n = 22), and in the right hemisphere were the putamen, internal capsule, external capsule, and insula (n = 22). Lesion volume was, on average, 24.7 ± 39.6 cm3 in the left hemisphere and 32.9 ± 48.3 cm3 in the right hemisphere. In the ND group, the anatomic area of most overlap in the left hemisphere was the external capsule (n = 5), and in the right hemisphere was the putamen, external capsule, superior temporal gyrus, and superior parietal cortex (n = 5). We found that lesion size was only significantly different between the ND and UD groups, F(2, 226) = 3.52, P = .03, Tukey, P < .05, and observed no hemispheric differences between groups for left hemisphere, F(2, 226) = 1.3, P > .05, or right-hemisphere lesion size, F(2, 226) = 1.6, P > .05 (Figure 5).
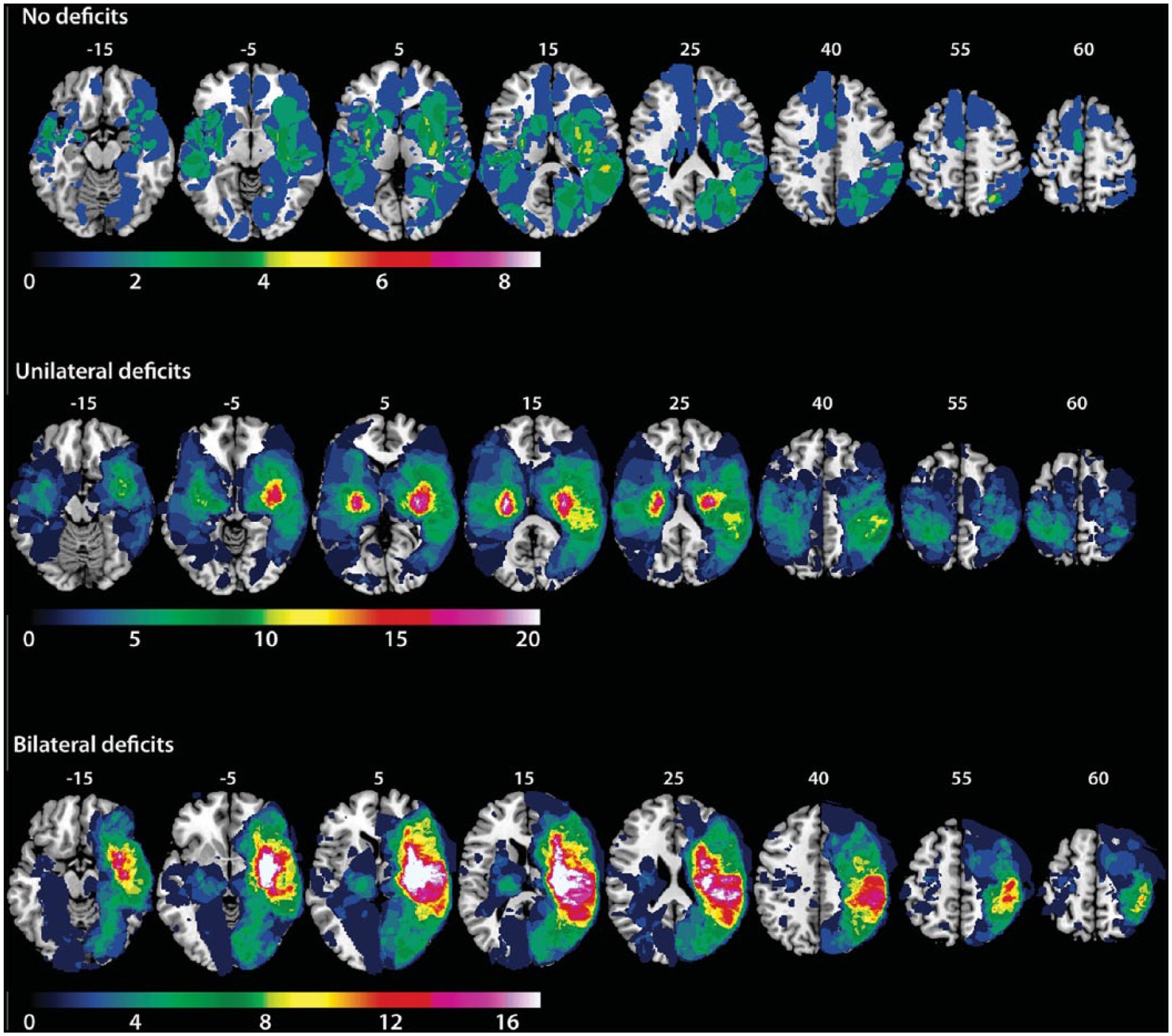
Lesion overlap maps for subjects in the no deficit (ND) (n = 42), unilateral deficit (UD) (n = 101), and bilateral deficits (BD) (n = 84) groups. Gradient color bar indicates the number (n) of subjects with overlap. Maximum overlap was set at 20% of the total n for each of the subject groups sample. Montreal Neurologic Institute (MNI) z-coordinates are labeled on top of each brain section.
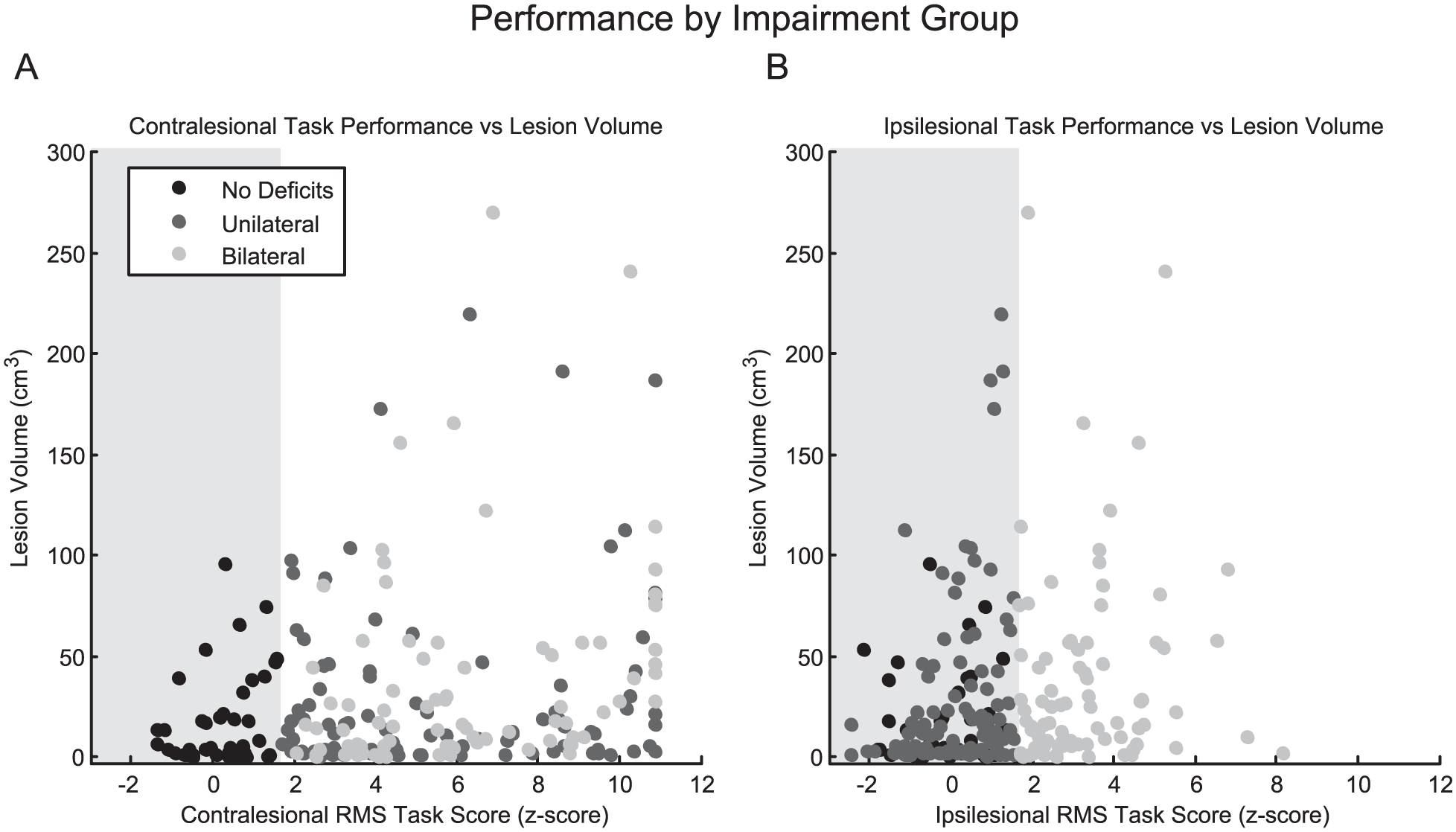
Comparison of lesion size to impairment group. Scatterplots of overall performance on the robotic reaching task versus lesion volume as a function of impairment group (no deficit [ND], unilateral deficits [UD], or bilateral deficits [BD]).
We examined general lesion location of all subjects and categorized stroke lesions into 3 groups: cortical (C), subcortical (SC), or cortical + subcortical (C + SC). We aimed to determine if lesion location categorization (C, SC, C + SC) or hemisphere of lesion (right or left) was related to robotic group classification (UD or BD). We observed a significant relationship between lesioned hemisphere and robot group classification (Fisher’s test, P < .001), where a larger number of BD subjects had right-hemisphere strokes. Furthermore, we found that significantly more subjects in the UD group had left-hemisphere SC strokes (Fisher’s test, P = .04), and that significantly more subjects in the BD group had right-hemisphere C + SC strokes (Fisher’s test, P = .0002). In our sample, 26% (n = 59) of subjects had neglect. Since individuals with neglect often have ipsilesional deficits related to visuospatial impairment, we removed these subjects and recalculated the above analyses. On removing these subjects, we found that the only statistical relationship that remained was BD subjects had a significantly larger number of right-hemisphere strokes.
Discussion
We found that over a third of the subjects tested had ipsilesional deficits. Furthermore, ipsilesional deficits were not strongly correlated to contralateral impairment, and the magnitude of contralesional impairment was not indicative of the presence or absence of ipsilesional impairment (Figure 2). While these deficits are an acknowledged aspect of stroke impairment and recovery, they are infrequently treated. We must highlight that many of the ipsilesional motor deficits we observe here are subtle and they may neither be detected using casual observation in the clinic, nor even by some clinical measures such as the CMSA.
The large number of subjects we identified with ipsilesional deficits suggests that rehabilitation after stroke should consider formal assessment of the ipsilesional arm. It has been shown that motor training of the ipsilesional arm can improve motor function. 32 Some rehabilitation methods rely on eliminating contributions from the ipsilesional arm, such as constraint-induced movement therapy (CIMT). 33 In cases where there is ipsilesional arm impairment, it is possible that bimanual rehabilitation therapies may be more desirable to allow for or promote recovery of deficits in the ipsilesional arm. 10 Furthermore, it is thought that by not allowing the ipsilesional arm to participate in rehabilitation, that functional synergy between the arms may be negatively affected throughout the recovery process. 34 The frequency with which we see ipsilesional deficits in our sample suggests that to promote personalized rehabilitation after stroke, it is important to assess and consider treating the ipsilesional arm to potentially improve functional outcomes and quality of life after stroke.
Characteristics of Ipsilesional Deficits
We had expected that individuals with the most severe contralesional impairments would be more likely to have ipsilesional deficits, but this was not the case (Figure 2, Supplemental Table 1, Supplemental Figure 1). The magnitude of contralesional arm impairment was not predictive of ipsilesional impairment, and the magnitude of ipsilesional arm impairment did not scale with impairment of the contralesional arm. Our results suggest that it is difficult to predict the presence of ipsilesional deficits based on motor performance of the contralesional arm alone.
Surprisingly, we found few differences in contralesional or ipsilesional motor behavior when we compared individuals with left- and right-hemisphere strokes (Supplemental Table 2). Previous work has found differences in motor impairment of the ipsilesional limb based on lesioned hemisphere. 23 It has been reported that individuals with left-hemisphere damage have ipsilesional difficulty with kinematic elements related to movement planning, whereas individuals with right-hemisphere damage have difficulty with end of movement errors.13,14,35 A separate study found ipsilesional deficits in a pointing task only in subjects with left-hemisphere damage. 36 In this study, we used several kinematic measurements very similar to those previously reported in other studies, such as initial direction error and reaction time, 14 as well as movement time.13,14,35 Unlike previous studies, we failed to observe any differences in ipsilesional or contralesional motor behavior due to lesioned hemisphere (Supplemental Table 2). There are a few potential explanations. First, the other studies we are aware of that demonstrate hemispheric differences were conducted in chronic stroke, after the time period when most poststroke plasticity and reorganization occurs, 37 whereas we are examining subjects in the subacute phase. Second, we recruited subjects with stroke that would typically arrive in a hospital environment whereas these earlier studies specifically recruited individuals with chronic stroke lesions to examine underlying mechanisms of motor control. Third, our subject group is more than ten times larger than these studies and this can lead to heterogeneity in lesion location, which may have contributed to the differences seen. Despite the difference in sample sizes, we observed similar areas of brain damage and proportion of subjects with lesion overlaps in comparison to these previous studies (Figures 4 and 5, Supplemental Figure 4). Finally, chronic stroke subjects in these previous studies made much faster movements (nearly 3 times faster, ~0.6 m/s13,14) with the ipsilesional limb compared with those reported in this study (average of 0.2 m/s with ipsilesional limb) and the speed of the movement may have contributed to the differences seen.
However, when we examined the differences based on subject grouping (ND, UD, or BD), we found that subjects with left-hemisphere subcortical strokes were less likely to have ipsilesional deficits, while those subjects with right-hemisphere C + SC strokes were more likely to have ipsilesional deficits. We must note that the majority (63% [n = 31]) of these subjects (right hemisphere, C + SC, BD group, total n = 49) had neglect, and as a whole, the majority of subjects in the BD group had neglect (n = 43 [51%], Table 1). However, while many subjects in the BD group did indeed have neglect, these subjects with neglect did not always have the greatest degree of ipsilesional impairment (Supplemental Figure 1). This makes an important point that while ipsilesional deficits can often be due to perceptual deficits, there are many other factors that may impact ipsilesional motor function after stroke.
Overall, our results may suggest that lesions to left subcortical areas can result in relative sparing of ipsilesional motor function after stroke, contrary to several studies.13,14,36,38 However, most of these studies have been conducted in chronic stroke, whereas the participants in the current study were tested in the subacute phase. A limitation of our study is that we cannot recruit some subjects with left-hemisphere strokes that have aphasia, because they have difficulty understanding and executing the 3-step command required to participate.
Relationship to Clinical Measures
One of the challenges when validating newer methods of measuring motor impairments such as robotics, is that there are no true gold standards with which to make comparisons. Most clinical measures of motor function rely on observer-based ordinal scales. The level of agreement between kinematic testing and the CMSA or PPB for the ipsilesional limb were only fair. In the case of CMSA of the hand or the PPB (Supplemental Figures 2 and 3), this is somewhat to be expected because of the fact that the robot does not specifically test hand function. However, the robot identified 31 more subjects with ipsilesional arm impairments than the CMSA of the arm. We put forward that clinical testing with the CMSA of the arm likely misses a large number of individuals with ipsilesional deficits (Supplemental Figure 3). Our results highlight the importance of assessing kinematics of both arms in addition to clinical measures in order to truly appreciate underlying motor deficits.10,21,39
Functional Impact of Ipsilesional Deficits
It is likely that ipsilesional deficits are oftentimes not as overtly noticeable to the clinician or the patient as contralesional deficits. Previous studies have referred to ipsilesional impairments as a general clumsiness with the limb, 1 suggesting while these deficits are subtle, they have much larger functional implications.5,6 It is thought that ipsilesional deficits may lead to longer recovery times, difficulty with interlimb coordination, 8 and general functional impairment.5,6 In our study, those subjects with bilateral motor deficits show the largest functional impairments, as measured by the FIM (Supplemental Figure 2). The large number of subjects in which we observed motor deficits of the ipsilesional limb speaks to the necessity of assessing motor function of both arms after stroke.
Footnotes
Acknowledgements
We would like to thank Janice Yajure, Kim Moore, Mark Piitz, and Megan Metzler for data collection and subject recruitment; Helen Bretzke and Justin Peterson for technical expertise; and Amy Yu and Jamsheed Desai for assistance with image marking.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SHS is the co-founder and Chief Scientific Officer of BKIN Technologies, the company that manufactures the KINARM.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded through a Canadian Institutes of Health Research operating grant (MOP 106662), a Heart and Stroke Foundation of Canada Grant-in-Aid and an Ontario Research Fund grant (ORF-RE 04-47). JAS was supported by an Alberta Innovates Health Solutions postgraduate fellowship.
Supplementary material is available for this article online on the Neurorehabilitation & Neural Repair website.
