Abstract
Background. Physical therapy doses may need to be higher than provided in current clinical practice, especially for patients with severe paresis. The authors aimed to find the most effective and feasible dose of Mobilisation and Tactile Stimulation (MTS), which includes joint and soft-tissue mobilization and passive or active-assisted movement to enhance voluntary muscle contraction. Methods. This 2-center, randomized, controlled, observer-blinded feasibility trial compared conventional rehabilitation but no extra therapy (group 1) with conventional therapy plus 1 of 3 daily doses of MTS, up to 30 (group 2), 60 (group 3), or 120 (group 4) minutes for 14 days. The 76 participants had substantial paresis (Motricity Index [MI] < 61) a mean of 30 days (standard deviation [SD] = 20 days) after anterior circulation stroke. MTS was delivered using a standardized schedule of techniques (eg, sensory input, active-assisted movement). The primary outcome was the Motricity Index (MI) and secondary outcome was the Action Research Arm Test (ARAT) tested on day 16. Adverse events were monitored daily. Results. No difference was found in the change in control group MI compared with each of the 3 intervention groups (P = .593) or in the ARAT. Mean actual daily treatment time for all MTS groups was less than expected. The attrition rate was 1.3%. No adverse events related to overuse occurred. Conclusion. The authors were not able to deliver a maximum dose of 120 minutes of daily therapy each day. The mean daily dose of MTS feasible for subsequent evaluation is between 37 and 66 minutes.
Background
Substantial neuromuscular weakness is prognostic of poor recovery after stroke. 1 Although recovery may be enhanced by task-specific training such as constraint-induced therapy, 2 people who cannot produce sufficient voluntary contraction of the paretic muscle to extend the wrist and fingers are unlikely to benefit. For these people, sensorimotor stimulation, such as that in Mobilisation and Tactile Stimulation (MTS), 3 might prime or augment activity in the motor execution system to enhance their ability to voluntarily contract the paretic muscle. 4,5
MTS is a module of routine therapy used in current clinical practice to treat the upper limb of people with substantial neuromuscular weakness. 3 MTS involves “hands-on” sensorimotor stimulation to the hand and forearm. The specific techniques have been described comprehensively by physiotherapists experienced in stroke rehabilitation, and generalizability of the standardized schedule of MTS has been established (unpublished data). In summary, MTS involves joint and soft-tissue mobilization techniques and passive or active-assisted movement with the aim of priming and/or augmenting activity in the motor execution system to enhance the ability to voluntarily contract paretic muscle.
The hypothesis is therefore that MTS might enhance upper-limb motor recovery of people with substantial upper-limb paresis early after stroke. 6 Before testing this hypothesis in a clinical efficacy trial, it is important to identify the most appropriate dose for evaluation within the setting of an acute stroke unit.
Dose finding has not featured prominently as a precursor to stroke rehabilitation trials, 7,8 although there is evidence from meta-analysis that intensity of therapy may be important for motor recovery. 9 However, these meta-analyses are confounded by inclusion of primary trials comparing different types as well as intensities of therapies. When meta-analysis controls for different types of therapy, the findings are less compelling. 10 Dose, however, remains an important consideration. Animal model studies have found that 400 repetitions may be required to produce motor learning and brain reorganization. 11
It is unknown, however, whether this level of dose is appropriate after human stroke to produce the same beneficial effect, not least because of the fatigue experienced by people after stroke. 12 Indeed, a more intensive dose of constraint-induced therapy delivered early after stroke might not be beneficial. 13 At present, clinical guidelines recommend the provision of at least 45 minutes a day, 14 but the research evidence to support this is sparse. 10 A search of electronic databases (Box 1) in September 2009 found no published evaluations of 3 or more prospectively decided different doses of the same nonpharmaceutical stroke rehabilitation therapy. The aim of this trial was to determine the most appropriate dose of MTS, balancing clinical efficacy, adverse effects, and feasibility of delivery early after stroke.
Search Strategy to Identify Trials of Different Doses of the Same Nonpharmaceutical Stroke Rehabilitation Therapy

Methods
Design, Randomization, and Ethical Considerations
A 2-center, randomized, controlled, observer-blinded feasibility trial with 4 groups (Figure 1) was carried out:
Group 1: control—no extra therapy
Group 2: up to 30 minutes per day MTS therapy for 14 consecutive working days
Group 3: up to 60 minutes per day MTS therapy for 14 consecutive working days
Group 4: up to 120 minutes per day MTS therapy for 14 consecutive working days
The rationale for the doses was that much physiotherapy is delivered clinically in 30-minute sessions.
Randomization order to group was generated before the trial began by an independent statistician and stratified by clinical center, severity of paresis (Motricity Index [MI] 15 score ≤ 19 and from 20 to 61), and absence or presence of unilateral spatial neglect (Catherine Bergego Scale score = 0 and >0, 16 respectively). Sequentially numbered opaque sealed envelopes for each stratification category concealed the group allocation. The blinded assessor completed baseline measures for each participant prior to the research physiotherapist telephoning the stratification categories to an independent administrator. The administrator then opened the next envelope in the appropriate numbered sequence and revealed the participant’s group allocation to the research physiotherapist. The research physiotherapists, one providing treatment in each center, remained blinded to baseline and outcome measures for each participant until the trial was completed.
Research ethics approval was obtained for this trial (Ref 05/Q0702/176), and site-specific approvals were obtained to conduct the trial in the stroke units. All participants provided written, informed consent. This trial was registered on ClinicalTrials.gov (NCT00360997).
Participants
All participants had an infarct or hemorrhage in the anterior cerebral circulation 8 to 84 days before recruitment; paralyzed or substantially paretic upper limb (<61/100 on the MI Arm Section 15 ); no clinically important upper-limb pain or visible upper-limb movement deficits attributable to pathology other than stroke; and ability to follow a single-stage command using their nonparetic upper limb.
Sample Size
The power of a comparison of the control group with a combination of the 3 experimental groups was considered. The ratio of the number of control participants to experimental participants was set to 1:3, and power calculations were done to compare the 2 independent groups. The expectation was that 20% of the controls would experience improvement in paretic upper-limb function (as assessed by an increase of one level on the MI Arm Section 15 ). A sample size of 20 in each of the 4 groups, therefore, provided 80% power at 0.05% significance to detect clinical improvement in at least 51% of people in the combined experimental group. 17
Interventions
All participants received their routine conventional physical therapy (CPT) from the clinical physiotherapists. The amount and content of upper-limb CPT given to participants during the intervention phase of this trial was documented using a treatment schedule with acceptable reliability that has been used to record CPT in a trial of functional strength training. 18,19 The clinical physiotherapists at each center were trained to use the treatment schedule and asked to record treatment given on each of the 14 intervention days for each participant (no upper-limb treatment was recorded as 0 minutes). The research physiotherapists collected the completed forms regularly. CPT was recorded to inform interpretation of the degree to which therapists’ knowledge of group allocation might influence the treatment they provide and to enhance interpretation of generalizability of results because processes of routine care vary with geographical location. 20
Participants randomized to the control condition (group 1) received the CPT provided by the clinical physiotherapists and no experimental therapy. Participants randomized to experimental groups 2, 3, and 4 received up to 30, 60, or 120 minutes of MTS, respectively, every working day for 14 days from the research physiotherapist in addition to CPT. MTS was given either on the ward or in the participants’ own homes if they had been discharged. Experimental interventions had to fit around routine care and not interfere with therapy sessions, ward rounds, meal times, or medical investigations. In addition, the delivery of MTS was done taking into account participants’ experience of fatigue, their preferences for timing of the intervention, and the visits of their family and friends. All these factors could potentially reduce the planned amount of MTS. Consequently, the research therapists paid particular attention to communication with clinical staff and participants regarding the flexible timing of intervention.
MTS, defined in a standardized schedule (Box 2), consists of the provision of tactile and proprioceptive stimulation through actions such as guided sensory exploration, massage, passive joint/soft-tissue mobilization techniques, active-assisted movements, and active movements where possible. 3 Details of the aims, content, and duration of each treatment session were documented by the research physiotherapists who had received training in the provision and recording of MTS.
Components of MTS

Abbreviations: MTS, Mobilisation and Tactile Stimulation; 1, thumb; 2-5, index, middle, ring, and little fingers; DIPJ, distal interphalangeal joint; IPJ, interphalangeal joint; MCPJ, metacarpophalangeal joint; PIPJ, proximal interphalangeal joint.
Adapted from Hunter et al. 3
Measurement Battery
Measurement points were at baseline (the working day ± 2) before randomization and at outcome (the working day ± 2) after the intervention phase ended. In addition, assessment for adverse events was conducted on each working day of the intervention phase. An assessor, blinded to treatment allocation, in each center obtained all measures in accordance with standardized protocols.
The 2 outcome measures were selected to reflect the hypothesized effect of MTS—namely, that the sensorimotor stimulation would increase the ability to produce voluntary contraction of muscle (primary effect) and thus enhance ability to use the paretic upper limb in functional tasks (secondary effect). Previous findings of an improvement in MI scores (voluntary contraction of paretic muscle) followed by an improvement in Action Research Arm Scores (functional ability) support this effect. 6 The arm section of the MI 15 was the primary outcome 21,22 and correlates highly with dynamometry. 23 The MI is also sensitive to change. 24 The arm section consists of 3 subsections: pinch grip, elbow flexion, and shoulder abduction. The scores for each subsection range from 0 (no flicker of voluntary muscle activity) to 33 (normal power). The scores for each section are combined, and then, a score of 1 is added thus giving a total possible score of 100 (33 + 33 + 33 + 1). The Action Research Arm Test (ARAT 25 ) also shows good reliability 25,26 and validity. 27 The ARAT consists of 4 subsections (grasp, grip, pinch, and gross movements), each of which involves 3 to 6 items, which are scored from 0 (unable) to 3 (normal performance). Scores for each subsection are combined, and the total possible score is 57. We used a standardized protocol for conduction and scoring. 28 Specifically, “partially completing a task” (score = 1) was interpreted as the voluntary placing of the paretic hand adjacent to the object to be grasped. More recently, standardized guidelines for using the ARAT have been published. 29 These require that an object is held and lifted to justify a score = 1. This difference needs to be appreciated when comparing the results of this trial with other trials.
Adverse Events
It was considered that there was a small risk of occurrence of an overuse syndrome as expressed by
a decrease in ability to produce voluntary contraction of the upper-limb paretic muscle and
the presence of pain in the paretic upper limb (participant report, either verbal or behavioral.
If either of these adverse events occurred on 3 consecutive days and could not be explained by a clinical reason other than participation in the trial, then treatment was discontinued.
Analysis
Participants were analyzed according to their group allocation regardless of the amount of therapy they actually received. The control group was compared with the combined treatment groups using a Mann-Whitney test.
To assess within-group differences, we used descriptive statistics and the Wilcoxon test of the change from baseline. The efficacy of MTS was assessed by determining the number of participants for whom there was a minimal, clinically important difference when the control group and the intervention group were compared, using a χ2 test. The minimally important difference was set as 5.7 points for the ARAT 30 and 1 level (at least 3 points) for the MI (no published data were identified).
To identify the most appropriate dose of MTS that could be evaluated in a subsequent trial that is feasible to deliver in an acute stroke clinical setting, we considered the combined data of efficacy, frequency of adverse events, and amount of MTS actually delivered.
Results
Recruitment and Attrition
From 912 stroke patients screened, 146 were excluded for reasons unrelated to the study criteria. Of the remaining 766 patients, 675 did not meet the study criteria. Consequently, 91 people (12%) were eligible for recruitment, of whom 15 refused informed consent. Therefore, 76 people (10%) were recruited and randomized (Figure 1).
Three participants, all from group 3 (up to 60 minutes MTS), were unable to complete all the planned intervention. Outcome measures were completed for the 2 people who transferred out of the geographical area. The attrition rate at outcome was 1.3% (Figure 1).
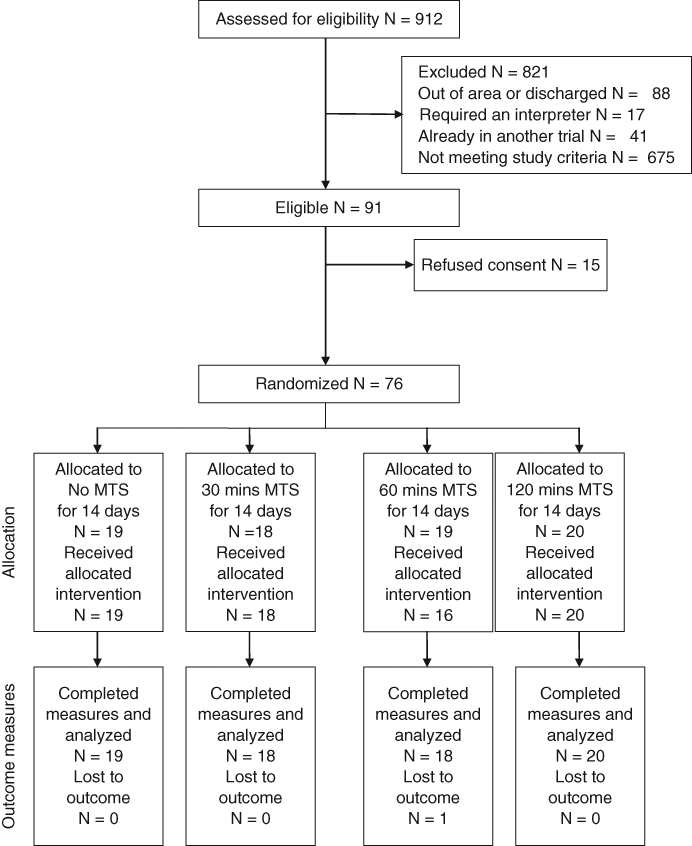
Participant recruitment and attrition flowchart.
Participants
Table 1 gives participants’ baseline characteristics. Their mean age was 72.5 years (standard deviation [SD] = 11.6 years), and the mean time after stroke was 29.6 days (SD = 18.9 days). A left-sided hemiplegia was exhibited by 53 participants (70%), and 23 participants (30%) had unilateral spatial neglect. The median (interquartile range [IQR]) score for MI was 11.0 (1-40), and for the ARAT it was 0 (0-2).
Baseline Characteristics of Participants
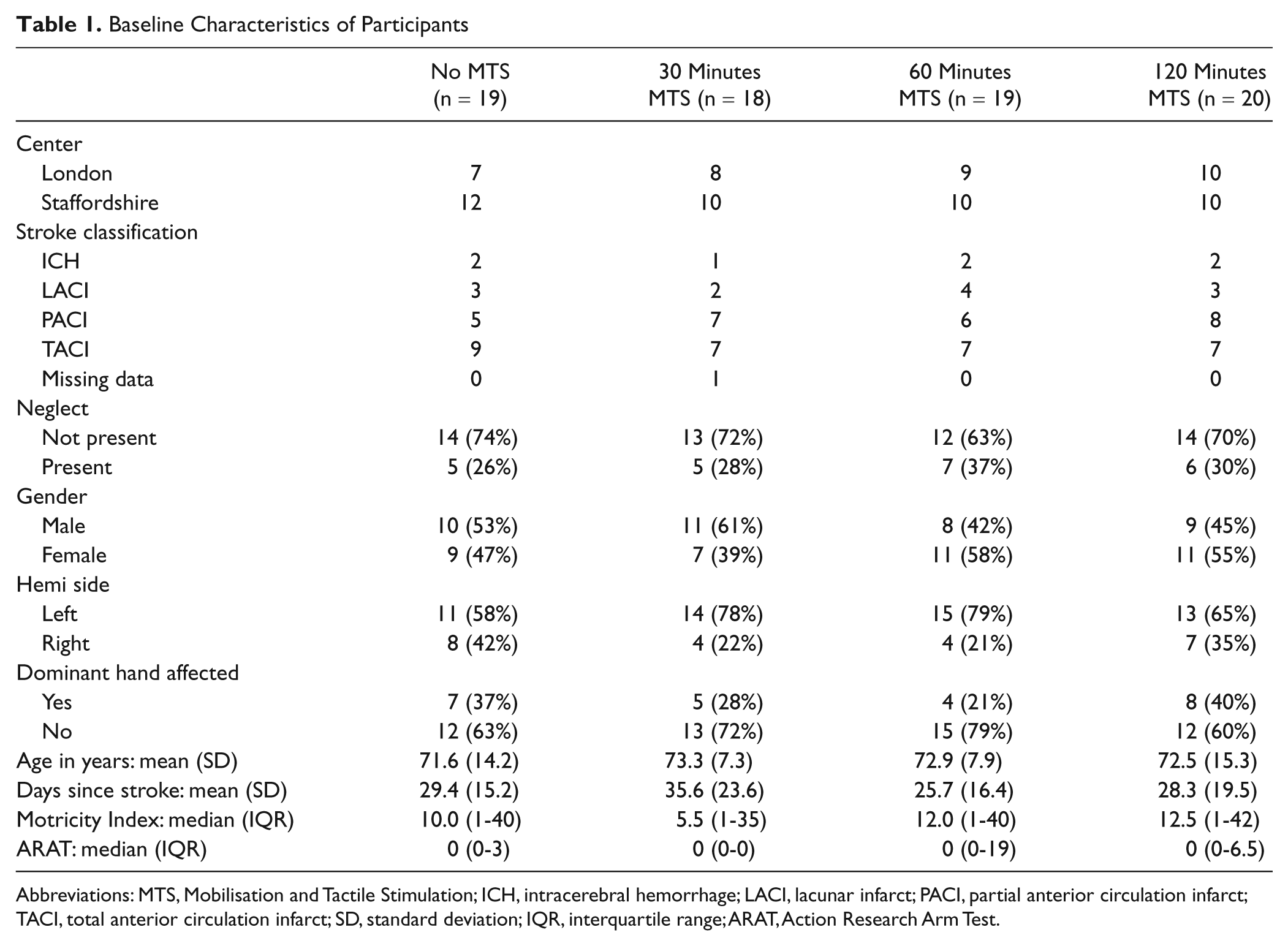
Abbreviations: MTS, Mobilisation and Tactile Stimulation; ICH, intracerebral hemorrhage; LACI, lacunar infarct; PACI, partial anterior circulation infarct; TACI, total anterior circulation infarct; SD, standard deviation; IQR, interquartile range; ARAT, Action Research Arm Test.
Amount of MTS and CPT Delivered
Table 2 shows the amount (minutes) of MTS that could have been delivered and that was actually delivered for each group. The mean daily treatment for the experimental groups was 23 minutes for group 2, 37 minutes for group 3, and 66 minutes for group 4. Field notes and informal reports from the research therapists suggest that the reason for the shortfall in dose could be a result of poststroke fatigue and ward activities/routines.
Amount (Time) of MTS That Could Have Been Delivered and That Was Received, by Allocated Group

Abbreviations: MTS, Mobilisation and Tactile Stimulation; SD, standard deviation.
In 1 center, the clinical physiotherapists providing CPT reported that if they did not provide any treatment for the upper limb then they did not complete a recording form. Although lack of a form could indicate that no CPT was provided on that day, its absence could also indicate that CPT was provided but a form was not completed. Because of the uncertainty, a detailed analysis of CPT was not undertaken. With caution, therefore, we report that the mean time recorded as being spent treating the upper limb in CPT was only 4.4 minutes a day (range, 0 to 23.2 minutes). If the missing forms are treated as zero returns, then the mean daily amount of treatment for the upper limb is reduced to 2.4 minutes. None of the participants experienced an adverse event.
Clinical Efficacy
There was no difference when the control group was compared with the 3 intervention groups (P = .593) using a Mann-Whitney test. Similarly, with the relatively large IQRs, there is little evidence of a statistical trend across the 4 groups (Table 3). The within-group median increase in MI score change between baseline and outcome was statistically significant for all groups (P = .010 to P < .001; Table 3). The greatest median increases were 16 points in group 3 and 10.5 points in group 4. All intervention groups contained more participants with a clinically important change of 1 level or more (at least 3 points) when compared with participants in the control group, and the greatest number was in group 4 (Table 3). However, MTS did not result in a significantly higher number of clinically important changes in the combined treatment groups as compared with the control group (P = .722).
Change, Baseline to Outcome for All Groups

Abbreviations: MTS, Mobilisation and Tactile Stimulation; IQR, interquartile range; CI, confidence interval; MI, Motricity Index; ARAT, Action Research Arm Test.
For ARAT, there was no difference when the control group was compared with the 3 intervention groups (P = .5011) using a Mann-Whitney test. Similarly, there was no statistical trend across the 4 groups (Table 3). The within-group median (IQR) change in score was similar across all 4 groups and statistically significant within each group (P = .024 to P = .003; Table 3). Groups 3 and 4 contained more participants with a clinically important change of 5.7 points or more than did the control group (Table 3). However, MTS did not result in higher clinically important changes in the combined treatment group compared with the control group (P = .850).
Most Appropriate Dose for Further Evaluation
No adverse events occurred in the present trial; therefore, all the doses evaluated might be appropriate. However, the least efficacy, considering the MI and ARAT results together, was found in group 2, with no difference between groups 3 and 4 (Table 2). The findings (Tables 2 and 3) suggest that benefit might occur from a mean dose ranging from 37 (SD = 15) to 66 minutes (SD = 19), and this range may be feasible for subsequent evaluation.
Discussion
This phase I trial found that between 37 and 66 minutes of daily MTS might be the most appropriate dose for subsequent evaluation of clinical efficacy. The findings indicate that it may be feasible to deliver doses in this range in rehabilitation units early after stroke. The highest dose (group 4) did not result in any adverse events, suggesting that 66 minutes of MTS each day may be delivered without causing harm.
There was a difference between the maximum amount of MTS that could have been delivered in each experimental group and that actually delivered. Differences between planned and delivered amounts of extra therapy are also evident in earlier trials that evaluated 2 doses of the same stroke therapy: for example, 44%, 19 65%, 31 62%, 32 and 96% 33 of the planned amounts were delivered. In the study reported here, the shortfall increased as the potential for higher doses increased, suggesting that the highest possible dose of up to 120 minutes a day in group 4 was neither practicable nor feasible in real-life scenarios. Although the maximum doses were not delivered and the reasons for this were not recorded prospectively, field notes suggest that the amount of MTS might have been shaped by poststroke fatigue experienced by participants and also by ward activities/routines. Whatever the reason, the present study does indicate that a daily dose of between 37 and 66 minutes of MTS is a feasible one for subsequent evaluation of clinical efficacy.
As in other rehabilitation studies, it was not possible to ensure that clinical colleagues were blinded to group allocation of participants. For example, if a clinical therapist knew that an individual was receiving up to 60 minutes a day of MTS from a research therapist, a reduction in routine clinical therapy could have occurred. This potential for bias was recognized in the design, in that clinical physiotherapists were required to record the amount and content of upper-limb CPT. However, potential inaccuracy was encountered in documenting the absolute dose of CPT because the center did not always fully comply with guidelines. Subsequent trials will need additional procedures. Recording upper-limb therapy delivered by occupational therapists will also need to be considered in a subsequent trial.
Interpretation of the results of the present trial needs to consider the possibility that it was underpowered. The power calculation to estimate the sample size for the present trial was based on the assumption that 20% of the participants in the control group would improve between baseline and outcome measures. This turned out to be an underestimate, with 11 of the 19 control participants actually improving (58%). A possible factor contributing to the difference between the expected and actual change in the control group could have been the introduction of thrombolysis into the United Kingdom during the course of this trial. 34 Another factor is a potential learning effect from the repeated use of the MI in adverse event monitoring. It is also possible that the control group received additional attention from the clinical therapists. Using our data, estimation of the sample size for subsequent trials should be less prone to clinical assumption.
To the best of our knowledge, the present trial is the first prospective randomized controlled trial of 3 or more amounts of the same nonpharmaceutical stroke rehabilitation therapy. Although the results do not provide a definitive dose of MTS for further evaluation, the successful completion of the trial addresses the need to evaluate progressively stroke rehabilitation therapies to enhance the design of phase III trials. 6 Successful completion of a prospective dose-finding trial in stroke rehabilitation has been demonstrated, and further exploration of the effectiveness of MTS as a module of routine therapy is justified.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: We are grateful to The Stroke Association for the provision of funding for this study. The Stroke Association had no role in the design, conduction, or interpretation of the results of this study.
