Abstract
Background and Objectives. A tight link between linguistic functions and activation of motor areas has been consistently reported, indicating that the 2 systems share functional neural resources. Few efforts have been made to explore whether this knowledge could aid the rehabilitation of aphasia. Methods. The authors assessed whether preactivation of the leg motor cortex during standing, compared with sitting, can facilitate language production in patients with chronic aphasia. In a cross-over within-subject design, the authors assessed performance on a picture naming task and controlled for effects on processing speed and simple verbal reaction time. Results. They found that standing compared with sitting had a beneficial effect on the number of semantic self-corrections that resulted in correct naming. In the absence of effects on motor or general processing speed, this points to a specific effect on lexical retrieval and selection. This was further corroborated by an error pattern analysis. Successful semantic self-corrections during standing were only found when there was already partial activation of the target semantic network—that is, when self-corrections were preceded by an incorrect but semantically associated naming response. Discussion. These findings show that preactivation of the motor system, which extends beyond the intrinsic link between manual gestures and language, can facilitate lexical access in chronic aphasia and may open new directions in aphasia rehabilitation.
Introduction
Approximately 30% of stroke patients suffer from aphasia, which affects language production and comprehension. 1 The most frequent aphasia symptom is impaired word retrieval (anomia). 2 Despite the availability of several standardized intervention strategies, chronic anomia is relatively resistant to intervention. 3 Thus, novel training adjuvant therapies need to be devised.
One exciting new direction in aphasia rehabilitation research is exploitation of the tight link between the language and the action (motor) systems that has been discovered in recent years. 4,5 It has been suggested that close interactions between these 2 systems are a relic of the evolution of spoken language from facial and manual hand gestures. 6,7 Recently, studies in healthy individuals have assessed whether activity in one domain (eg, the language system) can facilitate activity in the other domain (eg, the motor system), or vice versa. Indeed, the excitability of the hand and mouth motor cortex is enhanced during speech comprehension and production tasks but not during nonlinguistic control tasks. This effect has been shown for action- and non-action-related language materials. 8-14 Moreover, Liuzzi et al 15 recently provided evidence for a preexcitation of the leg motor cortex during non-action-specific language processing, indicating that language–motor system interactions are more extensive than previously thought (ie, extending beyond the hand and the mouth) and might not be limited to action-specific language materials. It has also been shown that motor activity can affect language processing. Executing or observing manual gestures facilitates lexical retrieval and semantic processing, 16-18 and preventing manual gestures can slow down speech production. 19,20
This language–motor interaction system could represent an intriguing new “backdoor” approach to accessing the language system for rehabilitation. 21 Unlike direct language therapies, this system, in principle, is independent of remaining linguistic functions but is not specifically connected to the language system. Activating the motor system in aphasia may support intensive speech therapy by providing better “working conditions” to activate the remaining parts of the language network. In particular, it has been shown that even though two thirds of the neural activity during word processing is category specific (eg, hand-related word elicit activity in the somatotopic representation of the hand area of the motor cortex), the remaining activity pattern is shared across semantic categories. 22 Preactivation of these common areas of the network (eg, the inferior frontal cortex) or those that are tightly connected (eg, the ventral premotor cortex [vPMC]) 23,24 might have a beneficial effect on language functions across semantic categories.
Several previous studies demonstrated that naming impairments in aphasia can be reduced by speech accompanying symbolic or iconic gestures, 25 and even simple pointing gestures that are unrelated to the target words can have beneficial effects on naming performance in nonfluent aphasia. 26-28 Still, preactivation of the motor system as such was confounded by possible preactivation of the gestural system by the pointing movement, and the precise mechanisms by which meaningful and nonmeaningful hand gestures improve language functions in aphasia remain unclear. Some explanations include facilitation of lexical retrieval through increased net activity in a common semantic network, 25 a spillover of activity from the hand motor cortex to the face area, 26 or facilitation of compensatory right frontal activity. 27,29
From a theoretical perspective, studies in healthy people have demonstrated that language motor facilitation is not restricted to the upper extremities or the face area but also comprises other motor circuits, including the leg motor system. 4,9,13-15,30,31 However, none of the available studies that examined facilitation of language via the motor system in aphasia had tried to activate the motor system without making use of hand gestures. This might also be of clinical relevance because a number of aphasia patients have a comorbid upper-extremity paresis, which may prevent unimanual or bimanual gestures. In addition, there might be impaired ability to produce co-speech gestures in aphasia patients per se. 25
Thus, based on previous work that demonstrated increased excitability of the leg motor cortex during language tasks, 15 we assessed whether picture naming in aphasia can be improved by preactivation of the leg motor cortex. For the purpose of the present exploratory proof-of-principle study, we chose to use a simple task manipulation: In a cross-over within-subject design, 20 patients with chronic nonfluent aphasia and anomia named visually presented pictures while either sitting or standing. It has been shown that standing (vs sitting) results in relatively increased activity in the leg motor cortex. 32 To explore effects on performance, we assessed naming accuracy and latency during the 2 conditions. In addition, we assessed latency and accuracy during 2 tasks that aimed at controlling for effects of standing on motor or general processing speed.
Methods
Patients were recruited through a stroke database (Department of Neurology of the University of Münster) and referrals by local neurologists and speech language pathologists. Inclusion criteria comprised chronic nonfluent aphasia caused by cerebrovascular stroke (>12 months poststroke) and right-handedness prior to stroke (Edinburgh Inventory Laterality Index > +70). 33 Patients with other aphasia types were excluded because previous studies have only shown effects of motor facilitation in patients with nonfluent aphasia. 26,27,29 Patients were screened for paresis using the National Institutes of Health Stroke Scale (NIHSS) (www.ninds.nih.gov/doctors/NIH_Stroke_Scale.pdf), and patients with severe paresis of the leg that prevented unassisted standing for more than 20 minutes were excluded from the study. Other exclusion criteria comprised severe psychiatric comorbidities (eg, depression or psychosis as determined by the patients’ medical records and the Beck’s Depression Inventory [BDI]), 34 dementia (Mini Mental State Examination [MMSE]), 35 and severe apraxia of speech.
A total of 20 patients with aphasia and mild to moderate anomia were recruited (7 women, 13 men; age [mean] = 55.3 years, range = 39-67 years; time since stroke [mean] = 98.5 months, range = 41-258 months; years of education [mean] = 12.8 years, range = 9-19 years; BDI [mean] = 6.6, range = 1-15; MMSE [mean] = 27.1, range = 22-29; for details, see Table 1). Aphasia was caused by a lesion within the territory of the left middle cerebral artery in 19 patients. One patient had aphasia caused by occlusion of the right middle cerebral artery (crossed aphasia; patient #10). Data were analyzed with and without this participant, and removal of this patient did not affect the outcome in the variables of interest. In all, 12 of the patients suffered from Broca’s aphasia, and 8 had amnestic aphasia as determined by the Aachen Aphasia Test (AAT). 36 See Table 1 for details of the AAT-naming subtest and the profile score, a measure of overall aphasia severity. None of the patients was above the threshold for clinical depression (BDI < 17) or dementia (MMSE < 25; in 1 patient [number 04] the lower score was explained by the primary language disorder; Table 1).
Clinical and Demographic Information
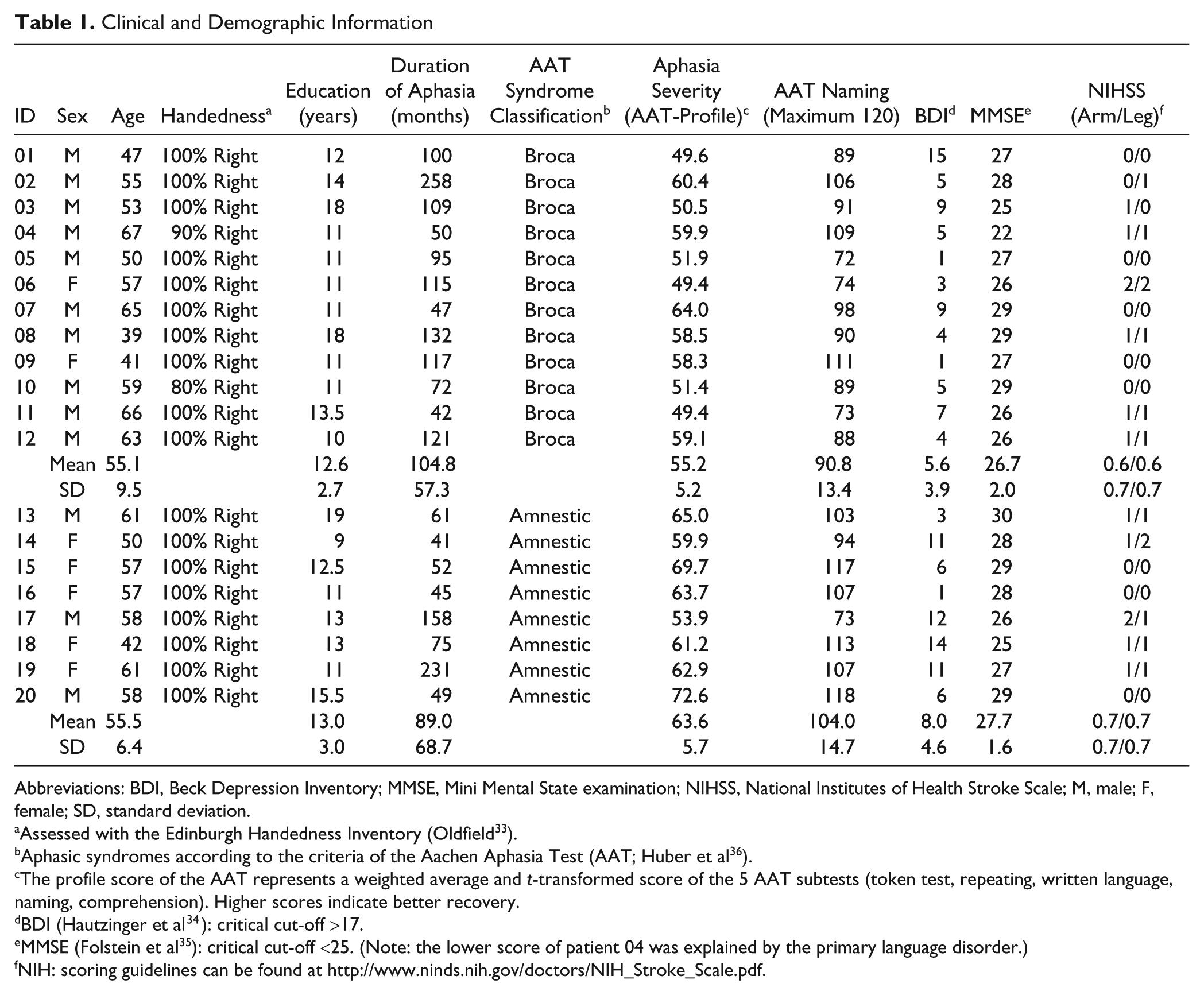
Abbreviations: BDI, Beck Depression Inventory; MMSE, Mini Mental State examination; NIHSS, National Institutes of Health Stroke Scale; M, male; F, female; SD, standard deviation.
Assessed with the Edinburgh Handedness Inventory (Oldfield 33 ).
Aphasic syndromes according to the criteria of the Aachen Aphasia Test (AAT; Huber et al 36 ).
The profile score of the AAT represents a weighted average and t-transformed score of the 5 AAT subtests (token test, repeating, written language, naming, comprehension). Higher scores indicate better recovery.
BDI (Hautzinger et al 34 ): critical cut-off >17.
MMSE (Folstein et al 35 ): critical cut-off <25. (Note: the lower score of patient 04 was explained by the primary language disorder.)
NIH: scoring guidelines can be found at http://www.ninds.nih.gov/doctors/NIH_Stroke_Scale.pdf.
Written informed consent was obtained from all participants. The study was approved by the institutional review board of the University of Münster and was conducted in accordance with the Declaration of Helsinki.
Experimental Design and Tasks
Data were collected for 3 tasks. The primary outcome measure was a picture naming task. Control tasks were a simple verbal reaction time task (saying the word “yes” in response to a visual cue) and a visual discrimination task (deciding whether 2 geometrical objects were identical or different by pressing a button). The 2 control tasks were included to assess whether potential latency differences between the 2 conditions during picture naming could simply be explained by a more general motor facilitation (verbal reaction time) or increased processing speed (visual discrimination). We used a cross-over, within-subject design in which each patient had to execute each task while sitting (condition 1) and while standing (condition 2). In condition 1, the participants were instructed to sit in a comfortable and relaxed position in a medical armchair with back support. Upper and lower extremities were positioned on arm and leg rests. During condition 2, the patients were instructed to stand on the floor with their weight on both feet in an upright position (Figures 1A and 1B illustrate the experimental setup during the naming task for the 2 conditions).
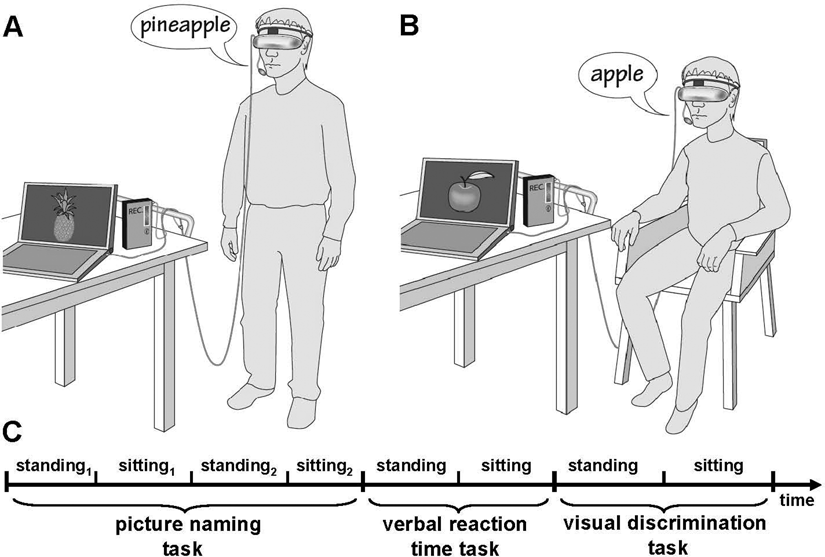
The experimental setup for the picture naming task during (A) standing and (B) sitting; the time line of an exemplary session is shown below (C)
Presentation of the experimental stimuli was accomplished by a head-mounted pair of liquid crystal display goggles (Sony Glasstron, PLM-S700E) to ensure that visual presentation (eg, visual angle, viewing distance, brightness) was comparable between conditions and tasks. Stimuli were presented using presentation software (Neurobehavioral Systems, Inc, Albany, CA).
All 3 experiments and both conditions were administered on the same day with short breaks in between the tasks. All participants started with the naming tasks; afterward, the 2 control tasks were administered. The experimental session began with condition 1 for half of the participants, and with condition 2 for the other half. All 3 tasks for each condition were administered consecutively within 1 session. For the naming task, data from 2 sessions were obtained, with the order randomized across participants (Figure 1C shows an exemplary session).
Picture Naming Task
Prior to the experiment, 4 sets of 30 line drawings from an online database 37 depicting common objects and animals were matched for word frequency (CELEX), 38 length (number of letters), name agreement, and visual complexity (frequency: mean = 1.9-2.0; F(3, 116) = 0.1; P = .9; length: mean = 6.2-6.6 letters; F = 0.3; P = .9; name agreement (maximum 1): mean = 0.89-0.91; F = 0.1; P = .9; visual complexity: mean = 18 898-20 062 pixels; F = 0.08; P = .97). Two of these matched sets were used during each of the experimental conditions (sitting1,2 and standing1,2—that is, all 4 sets were used for each patient), and the respective sets were counterbalanced between participants.
The patients were told that they would see object pictures on a screen for 5 s. Their task was to name the pictures as quickly and accurately as possible during this time interval. Each picture presentation was preceded by a blank screen (2.8 s). The entire session was digitally recorded via a microphone attached to the goggle system and transferred to a PC for offline analysis.
The number of correct responses during each experimental condition was assessed separately for the following:
Correctly produced responses: these responses included immediate correct responses (including minor phonemic errors, that is, >two thirds of the letters were correctly produced), hesitant correct responses (eg, mhm, ehm, . . . “correct response”), or corrections for minor phonemic errors (mirrot → mirror); see Tables 2 and 3 for details of the 2 response types.
Semantic paraphasias that were successfully corrected (eg, dog → cat].
Performance During the 3 Experiments for Different Types of Aphasic Syndromes, All Responses Are Included (Average Latency Sitting and Standing; Sum of Responses Sitting and Standing)
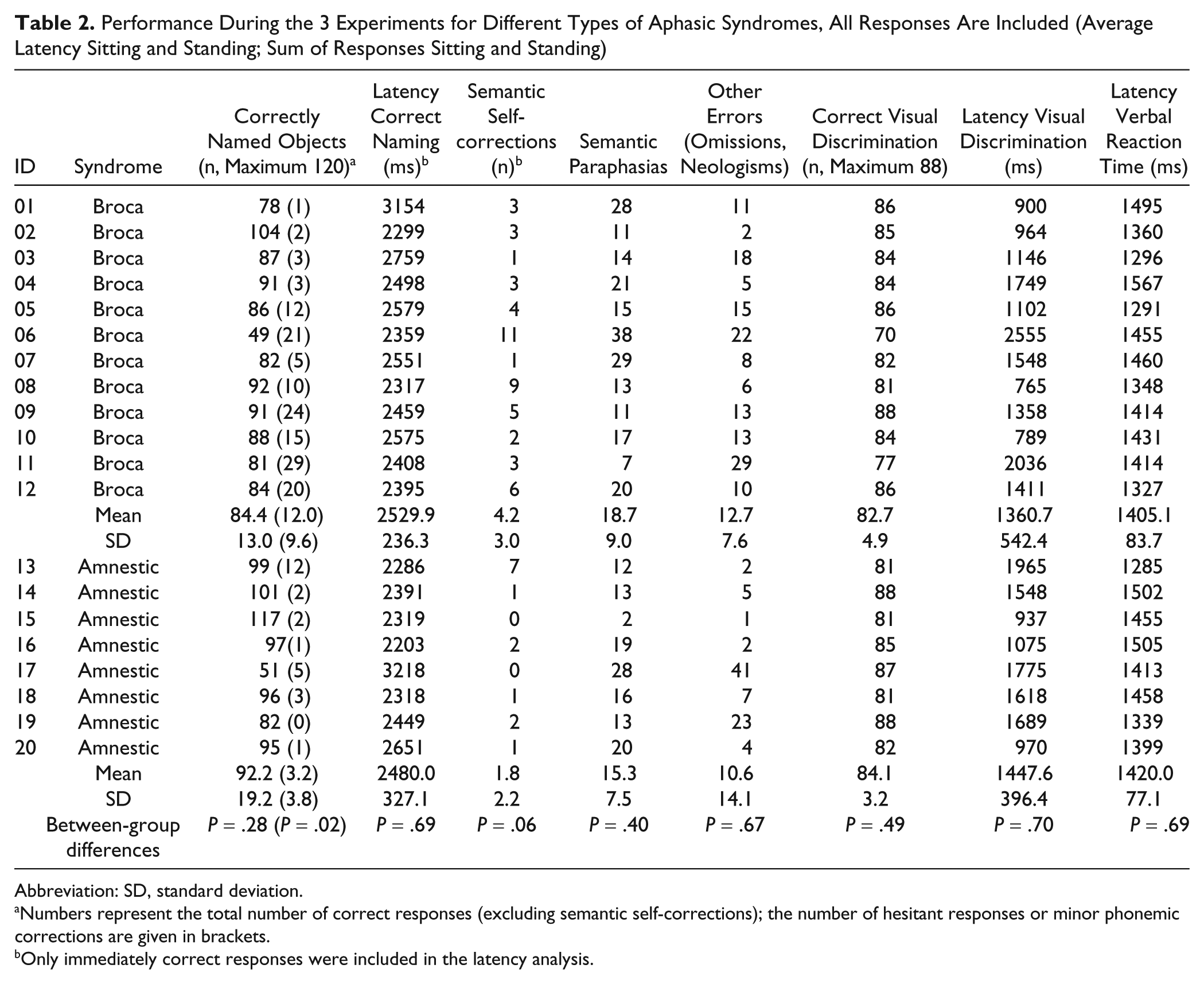
Abbreviation: SD, standard deviation.
Numbers represent the total number of correct responses (excluding semantic self-corrections); the number of hesitant responses or minor phonemic corrections are given in brackets.
Only immediately correct responses were included in the latency analysis.
Performance and Response Latency During the Naming Task for the 2 Aphasic Syndromes, Separated by Condition (Sitting Versus Standing)
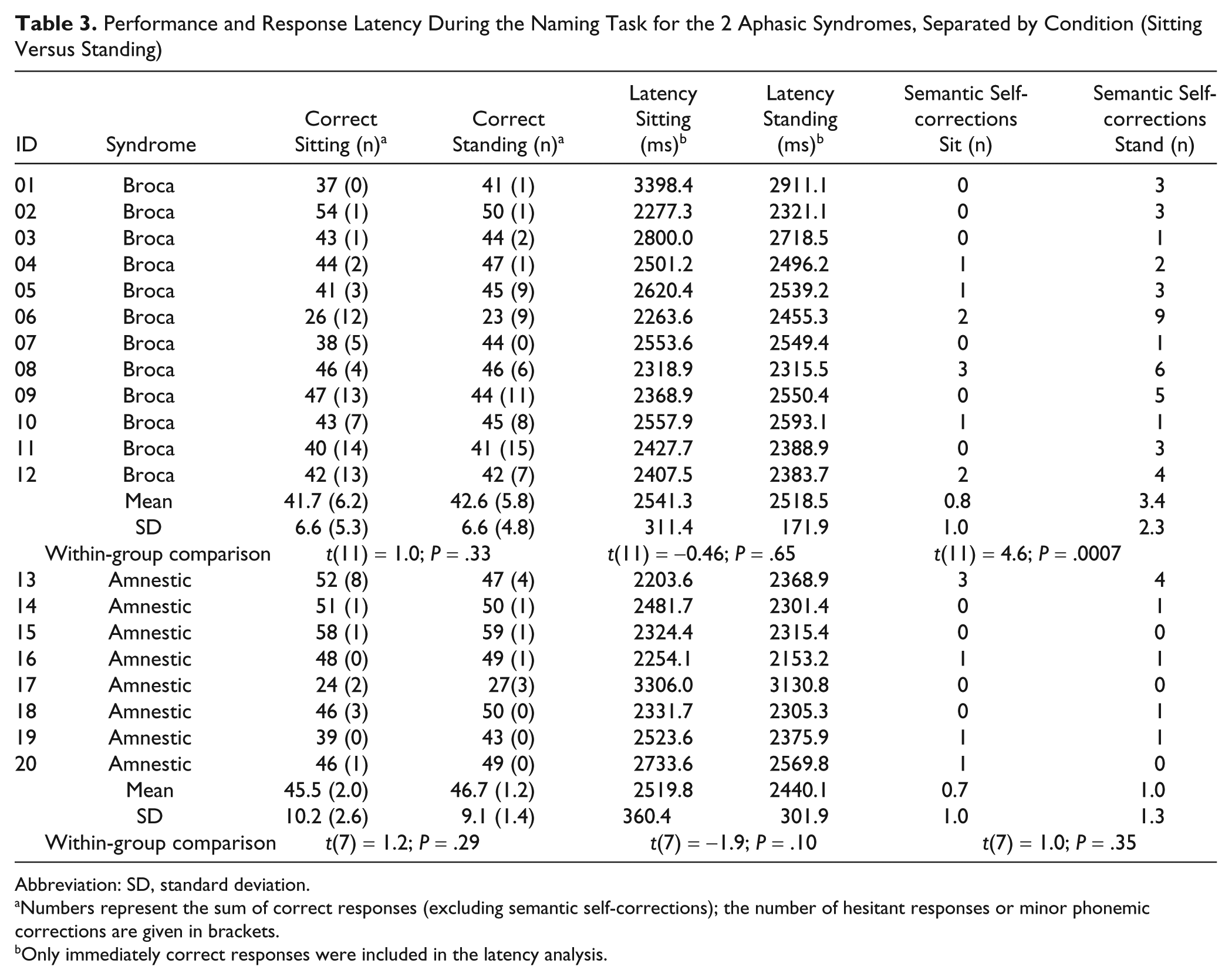
Abbreviation: SD, standard deviation.
Numbers represent the sum of correct responses (excluding semantic self-corrections); the number of hesitant responses or minor phonemic corrections are given in brackets.
Only immediately correct responses were included in the latency analysis.
Errors were grouped into 2 categories and analyzed separately: (1) omissions or other types of responses (eg, neologisms, where two thirds or more of the word was produced incorrectly, or other unrelated utterances) and (2) semantic paraphasias that were not corrected.
For the precise assessment of verbal response times (response latency), we used a digital stereo recorder with 2 channels. On the first channel, we recorded a computer-triggered signal that was sent 1 s prior to the picture onset and that was not audible to the patients. On the second channel, the verbal response of the subject was recorded. The 2 channels were then combined to a stereo signal with an analog audio mixer. For the analysis of the recorded data, we used a self-written software program (http://www.staff.uni-marburg.de/~sommerje/) that allowed us to manually determine the onset of the verbal response (ie, onset of correct name) relative to the trigger signal. For this purpose, the software played a short sequence of the second channel in a continuous loop. For the latency analysis, only immediately correct responses were included; that is, self-corrections with longer and more variable latencies were excluded. Correctness and latency of the respective responses were assessed by one of the authors (UW) who was blinded to the respective experimental condition (sit/stand).
Control Conditions: Verbal Reaction Time and Visual Discrimination Tasks
For the verbal reaction time task, the participants were told that they would see a black dot in the middle of a white screen. Their task was to respond to the dot by saying the word “yes” aloud as quickly as possible. A total of 32 dots were displayed at randomized intervals of 2 to 5 s during each of the conditions (sit/stand). To assess the latency of the verbal responses, we used the same dual channel methodology described above. All patients performed at ceiling level during this task; thus, all responses were included in the analysis.
For the visual discrimination task, we created a set of simple geometric shapes (Supplementary Figure 1). Two of these shapes were presented simultaneously on the screen, and the patients had to indicate whether they were identical (eg, the same stimulus, with the same orientation and size) or different. They responded manually with right index and middle fingers on a response box device. For each condition, 44 pairs of shapes were presented (50% identical and nonidentical shapes). The order was randomized across and within subjects. The response box was either placed comfortably in the lap of the patient (sitting) or on a height-adjustable table so that the patient could assume a relaxed arm position (standing). Response latencies were calculated for correct responses only.
Statistical Analyses
Statistical analyses were conducted with JMP 4.0 software (SAS Institute Inc, Cary, NC). Initially, the effects of the 2 experimental conditions were assessed for the entire patient sample and all dependent variables by 2-tailed paired t tests. To qualify whether there were differential effects of the task manipulation (sit/stand) for the 2 groups of patients (amnestic/Broca’s aphasia), we first compared patients with Broca’s aphasia and amnestic aphasia with regard to (1) the clinical and demographic variables and (2) the dependent variables (ie, latencies and correct responses for the 3 experimental tasks). For the latter analyses, the 2 experimental conditions were pooled (ie, average number of responses or latencies for sitting and standing). Then, the effect of the posture manipulation for the 2 groups of patients was further qualified using repeated-measures analyses of variance (ANOVAs) and post hoc t tests. Associations between naming ability and aphasia severity (AAT naming subtests and profile score) were assessed with Pearson correlation coefficients.
Results
All patients tolerated the experimental tasks well and were able to complete the study.
Results for the Entire Patient Sample
Effects of sitting and standing on picture naming tasks
For the total number of correct naming responses (including hesitant responses) and response latencies, no differences between sitting and standing were observed (number of correct responses: t(19) = 1.55; P = .14; latency: t(19) = −1.34; P = .19). However, a significantly larger number of semantic self-corrections were observed during standing as compared with sitting [t(19) = 3.89; P = .001].
Effects of sitting and standing on control tasks
No differences were observed between sitting and standing during the control tasks [latency verbal reaction time: t(19) = 0.63; P = .53; number of correct responses visual discrimination task: t(19) = 1.12; P = .27; latency visual discrimination: t(19) = −.79; P = .43].
Post hoc Analysis for the 2 Groups of Aphasia Patients (Amnestic/Broca’s Aphasia)
Differences between aphasic syndromes
The 2 patient groups were comparable with respect to age [F(1, 18) = 0.00; P = .93], education [F(1, 18) = 0.08; P = .77], duration of aphasia [F(1, 18) = 0.31; P = .58], and BDI and MMSE scores [BDI, F(1, 18) = 1.4; P = .24; MMSE, F(1, 18) = 1.3; P = .25]. As expected, aphasia was more severe in patients with Broca’s aphasia as indicated by a lower AAT profile score [F(1, 18) = 11.3; P = .003], and there was a trend for lower naming scores in the AAT [F(1, 18) = 4.2; P = .053] (see Table 1).
Similarly, the total number of correct responses (pooled data for sitting and standing) during the experimental naming task (including minor phonemic errors or delayed responses) was higher in amnestic patients compared with patients with Broca’s aphasia (mean/SD = 92.2 ± 19.2 vs 84.4 ± 13.0); however, this difference was not statistically significant [F(1, 18) = 1.2; P = .28]. In line with the more severe aphasia in patients with Broca’s aphasia, these patients produced more delayed or hesitant correct responses [F(1, 18) = 5.9; P = .02], and there was a trend for more self-corrections of semantic paraphasias [F(1, 18) = 3.8; P = .06]; please note that this latter comparison was also significant when we removed patient 13, who produced a disproportionally large number of semantic self-corrections relative to the other patients with amnestic aphasia; Table 2). Even though patients with Broca’s aphasia on average produced more semantic paraphasias, omissions, and neologisms, the distributions of error types was statistically not significantly different between the groups (all P > .40; Table 2).
Response latencies between the 2 groups were comparable for the 3 tasks (naming: F(1, 18) = 0.15, P = .69; verbal reaction time: F(1, 18) = 0.16, P = .69; visual discrimination: F(1, 18) = 0.15, P = .70). No differences were found for the number of correct responses during the visual discrimination task for the 2 groups (F(1, 18) = 0.47; P = .49; Table 2).
Post hoc comparison between aphasia subtypes
A subsequent repeated-measures ANOVA revealed that the overall significant beneficial effect of standing (vs sitting) on the number of semantic self-corrections was driven solely by the subgroup of patients with Broca’s aphasia who had more severe aphasia. These patients produced more semantic self-corrections during standing versus sitting compared with the amnestic patients [Interaction (Aphasia Type × Posture: F(1, 18) = 10.6, P = .004; amnestic aphasia: t(7) = 1.0, P = .35; Broca’s aphasia: t(11) = 4.63, P = .0007] (see Table 3; Figure 2). This is also corroborated by the fact that aphasia severity (AAT profile score) was negatively correlated with the relatively larger number of self-corrections during standing compared with sitting (r = −0.52; P = .01); that is, patients with more severe aphasia produced more semantic self-corrections during standing rather than when sitting.
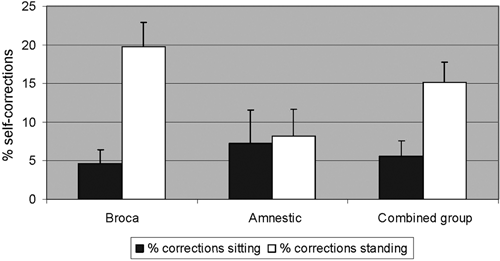
The effects of the 2 experimental conditions, expressed as percentage of semantic self-corrections relative to the individual patient’s number of incorrect responses; that is, the number of possible self-corrections. Formula: [(semantic self-correctionsstand or sit/ 60 − correctstand or sit) × 100], where 60 is the maximum number of correct responses possible. Combined refers to the combined group of patients
The same analysis was conducted for all the other dependent variables, even though no significant effects of the posture manipulation were found when we included the entire patient sample. No significant interactions were found for these variables (all F = 0.05-0.67; all P > .42). However, in the less severely affected amnestic subgroup, naming latency was shorter during standing compared with sitting in 7of the 8 patients (Table 3). However, the direct comparison between the 2 conditions was not significant [t(7) = −1.9; P = .1].
Error patterns and semantic self-corrections
We further analyzed the patterns of semantic errors and classified them as related (ie, whether they belonged to the same broad semantic category or shared features, such as cat vs dog) or unrelated errors (eg, dog vs moon). Additionally, the semantic self-corrections were classified by whether or not they belonged to the same broad category as the initial (incorrect) response. As illustrated in Supplementary Table 1A, both patient groups produced more semantically related than unrelated errors during both conditions [total number of related/unrelated errors = 288/57; related errors; Broca: related > unrelated errors during sitting/standing; F(1, 22) = 27.2/28.7, both P < .0001; amnestic: F(1, 14) = 19.2/8.0, both P < .02; please note that the larger number of semantic paraphasias during sitting in patients with Broca’s aphasia is explained by the fact that a larger number of self-corrections occurred during standing; see Supplementary Table 1B]. Thus, in about 80% of the trials in which the patients produced a semantic paraphasia, the response shared semantic features of the target picture. Moreover, when we analyzed the types of semantic self-corrections, in 58 of 65 instances (89%), the semantic self-correction was preceded by a semantically-related response (Supplementary Table 1B)—that is, the patients activated a common semantic network but initially failed to activate the target lexical form.
Discussion
The main finding of the present study was that a simple task manipulation, aimed at increasing activity in the leg motor cortex, can improve word retrieval in patients with chronic aphasia. Specifically, we found a greater number of semantic self-corrections that resulted in correct naming of objects during standing as compared with sitting. On the other hand, the total number of immediately correct responses (excluding semantic self-corrections) and response latencies were comparable between the 2 conditions, the number of phonemic error corrections was comparable, and no facilitatory effects were found on the control tasks. Taken together, these findings point to a specific impact of the task manipulation on lexical retrieval and/or selection rather than effects on processing speed or a simple spillover of activity from the motor cortex. This is also supported by the fact that successful semantic self-corrections were only found when there was partial activation of the target semantic network—that is, when self-corrections were preceded by an incorrect but semantically associated naming response.
A tight link between linguistic functions and activation of motor areas has been consistently reported by many researchers, indicating that motor and the language systems share functional neural activation patterns. 5 Owing to the action recognition system, language simultaneously activates the motor system to increase access to lexical information and to facilitate meaning construction. 5 Moreover, the involvement of the motor system in language functions expands far beyond its role in speech production. Studies have demonstrated that language processing has effects on both the mouth and facial motor systems, 10 the hand motor system, 11,12 and the leg motor system. 15 Thus, action–perception networks constitute a core mechanism of language production and comprehension, and activation of the language system leads to automatic and significant activation of the motor system.
Conversely, it should be possible to enhance activity in language areas by preactivation of different parts of the motor action system. In other words, motor stimulation could be used to activate the language network and to increase cortical activation in regions within the network, such as Broca’s area. 39 Rapid functional interaction and information exchange between these 2 brain systems during language comprehension was found to contribute to semantic processing and to facilitate responses to linguistic stimuli. 14,16,40,41 In fact, this mechanism and implications for aphasia rehabilitation have already been suggested by findings from a few previous studies: Hanlon et al 26 demonstrated that a pointing gesture with the (affected) right arm has beneficial effects on naming performance in nonfluent aphasic patients. Crosson et al 28 found that a 2-week anomia treatment, where naming trials were preceded by a complex left hand/arm movement, leads to improved lexical retrieval. However, the potentially beneficial impact of motor system preactivation on language processing has not been studied extensively in aphasia patients. Moreover, none of the previous studies has assessed the impact of parts other than the hand motor cortex on language functions in aphasia, and such studies are confounded by the fact that manual gestures are critically involved in language comprehension and production per se 25 and do not allow us to assess the effects of more general motor cortex activation. Given this theoretical background, our findings now provide, for the first time, experimental evidence that there is indeed a general preactivation of the language system by the motor system that extends beyond the hand motor cortex, and this can be used to facilitate language functions in aphasic patients.
According to the neuronal language model based on Hebbian learning mechanisms, 5,42 language acquisition at the neuronal level leads to the formation of semantic networks. These networks are distributed across cortical areas, mainly in the left frontotemporal language regions and prefrontal motor regions. 43 Moreover, within these distributed networks, common lexical and semantic processes that overlap with the inferior frontal action recognition system can be distinguished from processes that code for distinct physical and psychological features of the stimuli (eg, category-specific somatotopic activations related to arm vs. mouth related words). 22 Two critical features of these distributed representations are that: (1) even partial stimulation can activate these networks, and (2) feedforward and feedbackward connections between different cortical areas amplify cortical activation within the networks. Thus, inducing unspecific activity in the motor system may increase net activity in areas responsible for lexical retrieval across different categories (the above-mentioned common parts of the network), which may have facilitated lexical access in our patient sample. Indeed, it has been demonstrated that unspecific preactivation of the language network with transcranial magnetic stimulation facilitated picture–word verification in healthy participants. 44 In the present study, this was expressed as a larger number of semantic self-corrections from semantically related associates, suggesting that motor system preactivation was only sufficient to enhance naming performance under the condition of partial access to an object name (ie, category-specific preactivation). Indeed, as shown in Supplementary Table 1, approximately 90% of semantic self-corrections that resulted in correct naming of the pictures were preceded by an incorrect but related response. This might also explain why the patients with amnestic aphasia, who in general had more correct responses and less semantic errors, on average did not benefit from the standing intervention.
Also, Pulvermüller et al 22 demonstrated that about two thirds of the neurometabolic activity during word processing was accounted for by category-specific activation, indicating that unspecific input in more general nodes of the network might enhance net activity and facilitate selection processes; on the other hand, this may not be sufficient to improve word retrieval without category-specific preactivation. This remains to be explored in future studies, using a larger number of stimuli belonging to different semantic categories (eg, leg- vs hand-related pictures).
Based on the present data and the fact that lesion information was not available for all the patients, we can only speculate about the neural mechanisms responsible for improved naming performance during leg motor cortex preactivation in our study. It is known that the left inferior frontal gyrus (Broca’s area) and the vPMC, areas which are tightly linked anatomically, show a common activation when healthy people observe gestures 45 and during gait control (eg, standing). 46 Moreover, anatomical studies show that part of Broca’s area (BA44) and the vPMC (BA6) share specific cytoarchitectonic properties. 23,24 Thus, because of shared cytoarchitectonic features, tight anatomical links, functional connectivity, and topographic proximity, the vPMC may be a good candidate for the remapping of linguistic functions in aphasia patients. This hypothesis needs to be scrutinized in future studies, which should combine structural and functional imaging data with brain-stimulation targeted specifically at the vPMC.
Clinical Implications
In summary, we demonstrated that inducing activity in the leg motor cortex selectively facilitates naming in aphasia patients. This facilitation was limited to patients with more severe (Broca’s) aphasia; however, for patients with amnestic aphasia, there was a trend toward reduced naming latency. Thus, the positive findings in this proof-of-principle study provide a rationale for assessing the effectiveness of motor cortex stimulation by more sophisticated and clinically applicable techniques such as anodal transcranial direct current stimulation 47-49 or high-frequency repetitive transcranial magnetic stimulation. 50 Future studies should explore whether the combination of speech and language therapy and motor cortex stimulation can enhance treatment outcome.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This work was supported by the Bundesministerium für Bildung und Forschung (to AF: FKZ 0315673A; and by 01EO0801); the Deutsche Forschungsgemeinschaft (ME 3161/2-1; Fl 379-4/2 379-8/1; DFG-Exc 257); the Volkswagen Foundation (I/80 708) to CB and SK; the Stiftung Neuromedizin Münster and the Interdisciplinary Center for Clinical Research (Floe 3-004-008) to AF; and the National Institute of Health (T32DC008768) to SH.
