Abstract
Acute limb ischemia (ALI) is a large contributor to morbidity and mortality annually and can be managed either operatively or medically. ALI is most often caused by arterial embolism or in situ thrombosis and treatment is often dependent upon the severity. Anticoagulation is considered standard of care and first line therapy. However, more severe cases of ALI require surgical intervention.
Paradoxical emboli are a rare and potentially under-appreciated cause of ALI. They arise when venous emboli, from a variety of sources, traverse a patent foramen ovale (PFO) to enter the arterial system, compromising blood flow to the affected end organ. In most cases, they can only be proven if the thrombus is identified as it crosses the cardiac defect, at which point it is an indication for surgery requiring PFO closure, management for the ischemia itself, and possible intervention for the embolism.
In this report, we identify and discuss management of a series of cases where ALI was precipitated by PFOs that were discovered in the context of a pulmonary emboli that developed into paradoxical emboli. All patients had a confirmed diagnosis of COVID-19 which has been associated with a state of hypercoagulability and subsequent thrombus formation.
Keywords
Introduction
Acute limb ischemia (ALI) is a large contributor to morbidity and mortality annually and can be managed either operatively or medically. ALI is most often caused by arterial embolism or in situ thrombosis1,2 and consequences can be as severe as major limb amputation and permanent disability for the patient. 3 A rare and potentially under-appreciated cause of ALI arises from paradoxical emboli, in which venous emboli, from a variety of sources, traverse a patent foramen ovale (PFO) to enter the arterial system, compromising blood flow to the affected end organ. PFOs exist in 15-35% of the adult population 4 and most commonly manifest as cerebrovascular accidents. However, recent reports suggest that the development of ALI secondary to PFO has historically been under appreciated.5-7
Since the identification of SarsCoV2 in 2019 and the subsequent COVID-19 pandemic, researchers have discovered fascinating physiological responses to the virus. Among these findings, is increased thrombogenesis despite anticoagulant prophylaxis administration.8,9 Current data suggests that this pro-inflammatory state manifests most commonly as pulmonary embolisms (PE) and deep vein thrombosis (DVT) with high risk for venous thromboembolism in patients who have been infected with COVID-19. 10
In the five patients described here, we identify and discuss cases of ALI precipitated by PFOs that were discovered in the context of a PE within a 6 month time frame. All patients had a confirmed diagnosis of COVID-19 with an associated state of hypercoagulability. Informed consent was obtained from all patients.
Cases
Case 1
The first case is a 51-year-old male, recently diagnosed with COVID, but without other significant past medical history. At the time of initial presentation to the emergency room at a regional system hospital, he was asymptomatic from his COVID infection but reported 8/10 right leg pain with associated numbness. Upon presentation to the emergency room, pulses from the dorsal pedis and posterior tibial arteries were palpable on the left lower extremity (LLE), while the dorsal pedis and posterior tibial arteries of his right lower extremity (RLE) demonstrated monophasic signals on doppler venous ultrasound. He then represented two days later with further discoloration of the limb and having lost the ability to move the right foot. During this second emergency department visit, there was found to be an absence of pedal signals on doppler exam from the RLE with marked fixed mottling of the toes. The LLE was affected with a mild sensory deficit, but intact motor function with preserved pulsatile physical exam findings that remained unchanged from his previous visit. The patient was started on a heparin infusion and CT angiogram was obtained. The imaging revealed complete thrombosis of the right common femoral artery including the proximal segment of the right superficial femoral artery with reconstitutions of the superficial femoral artery at the middle third segment. The right profunda femoris was also occluded with distal reconstitution. Further thromboses were noted in the distal popliteal artery, tibioperoneal trunk, anterior tibial artery, posterior tibial artery, and peroneal trunks without evidence of distal reconstitution. Additional thromboses were noted throughout the left leg as well.
At this time, the patient underwent thromboembolectomy and fasciotomy of the right limb. On fasciotomy, the muscle was found to be nonreactive and edematous. Open iliofemoral thromboembolectomy was performed and successful. Distal thromboembolectomy was also performed, but flow was only restored to the tibioperoneal trunk and not the foot. At this point, an open transtibial amputation and muscle resection was performed.
Angiogram of the left limb found occlusion of the below knee popliteal artery (Figure 1(A)). Suction thrombectomy was performed for this embolism and inline flow was restored to the foot with triphasic Doppler signals in the posterior tibial artery and a palpable pulse (Figure 1(B)). Three days following his initial procedures, the patient returned to the operating room for formalization of his below-knee amputation. The patient’s PFO closure was completed at a later date. (A) Pre-thrombectomy angiogram shows an occluded left popliteal artery (white arrow). (B) Post-thrombectomy angiogram shows a revascularized left popliteal artery (white arrow).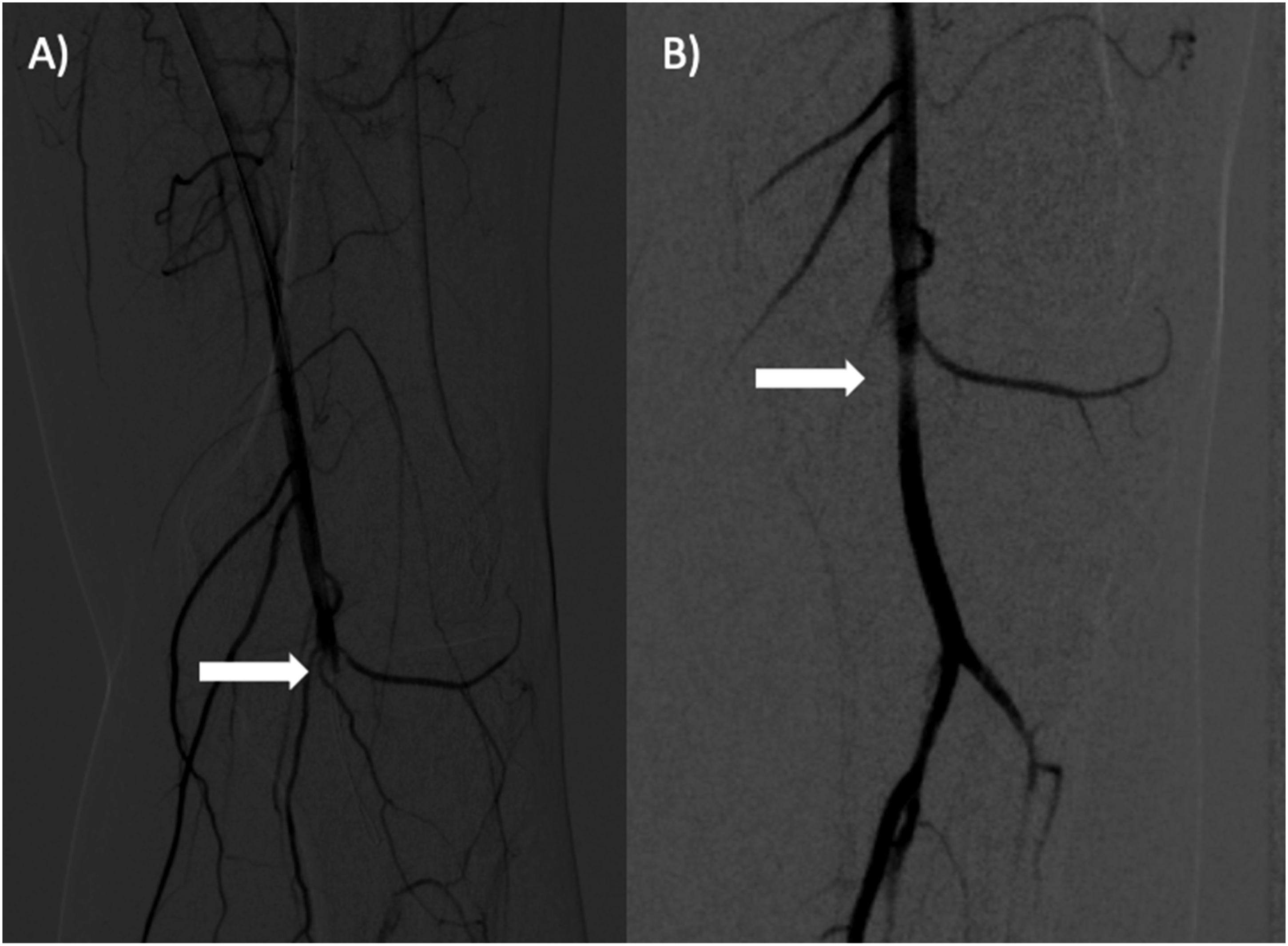
Case 2
The second case is a 57-year-old male with a confirmed history of COVID-19 infection who presented to the emergency room with severe chest pain and LLE numbness and tingling. A CT angiogram revealed a DVT in the left lower extremity, bilateral PE in the lobar and segmental branches, and thrombus within the left common femoral artery (CFA) extending to the deep and superficial femoral arteries (SFA) (Figure 2). The patient was started on a heparin drip and transferred for further care due to elevated pulmonary artery pressures. Axial CT-angiogram shows an occluded left common femoral artery (white arrow).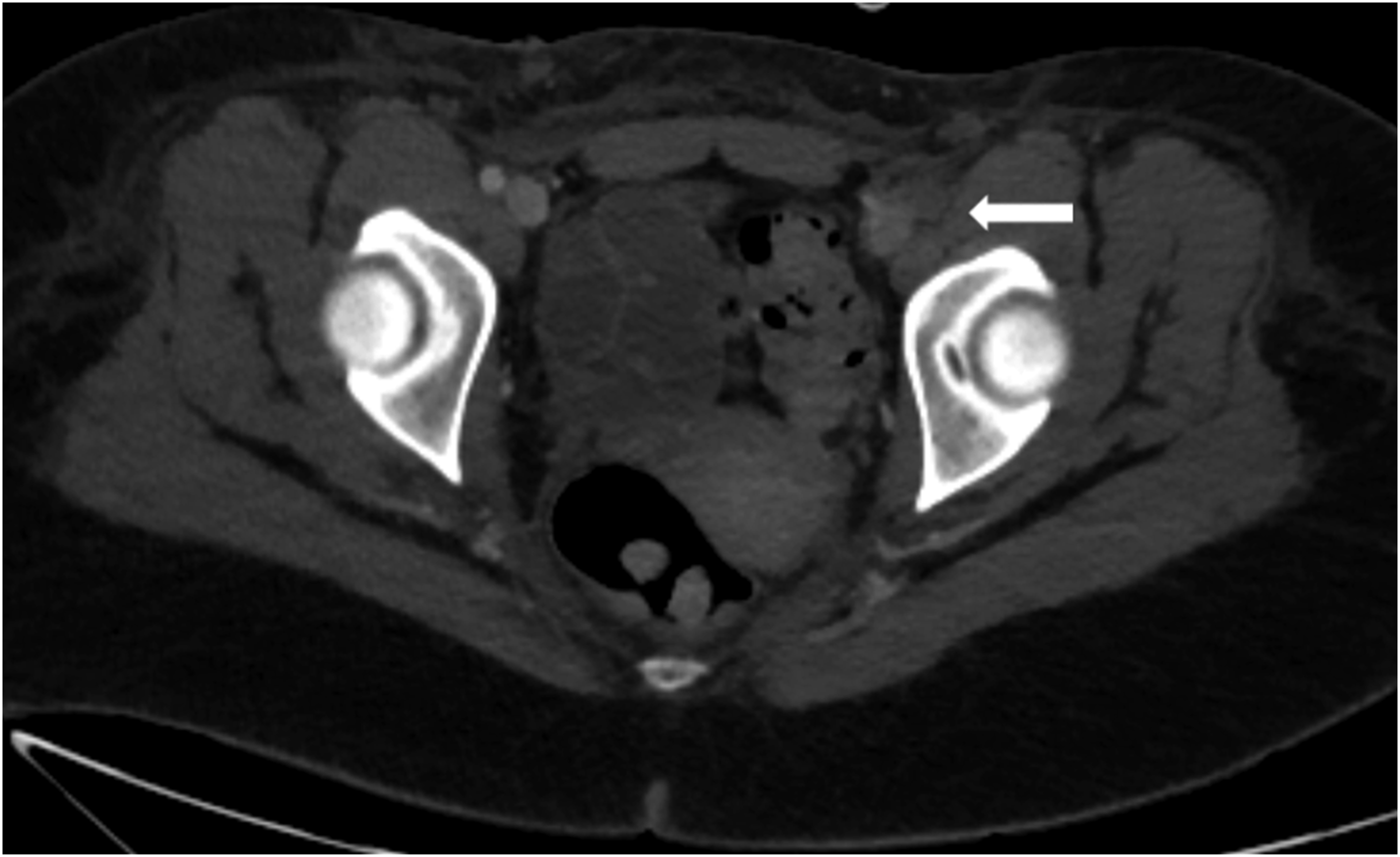
Transthroacic (TTE) and transesophageal (TEE) echocardiogram, both showed a PFO and evidence of mild right heart strain. The patient was emergently taken into the operating room for immediate LLE angiogram and suction embolectomy (Figure 3(A)). Given that the patient had a large abdominal pannus, it was felt that he was at high risk for developing groin wound complications, so an endovascular approach was chosen. During the procedure, thrombus was successfully extracted from the profunda femoris, but the device was unable to remove the embolism from the SFA, thus it remained occluded. At this time, the procedure was converted to an open thrombectomy. Significant thrombus was extracted from the SFA and perfusion was eventually restored to the profunda, SFA, and left CFA respectively. The patient then developed Doppler signals in the foot, but completion angiogram revealed distal embolization in the popliteal artery below the knee that was missed on initial imaging. Suction thrombectomy successfully removed this embolism and flow was restored through the popliteal artery into all three tibial vessels. The patient developed palpable pulses in the foot at this time and completion angiogram showed resolution of arterial occlusion with complete restoration of flow (Figure 3(B)). (A) Pre-thrombectomy angiogram shows an occluded left common femoral artery (white arrow). (B) Post-thrombectomy angiogram shows a revascularized left common femoral artery (white arrow).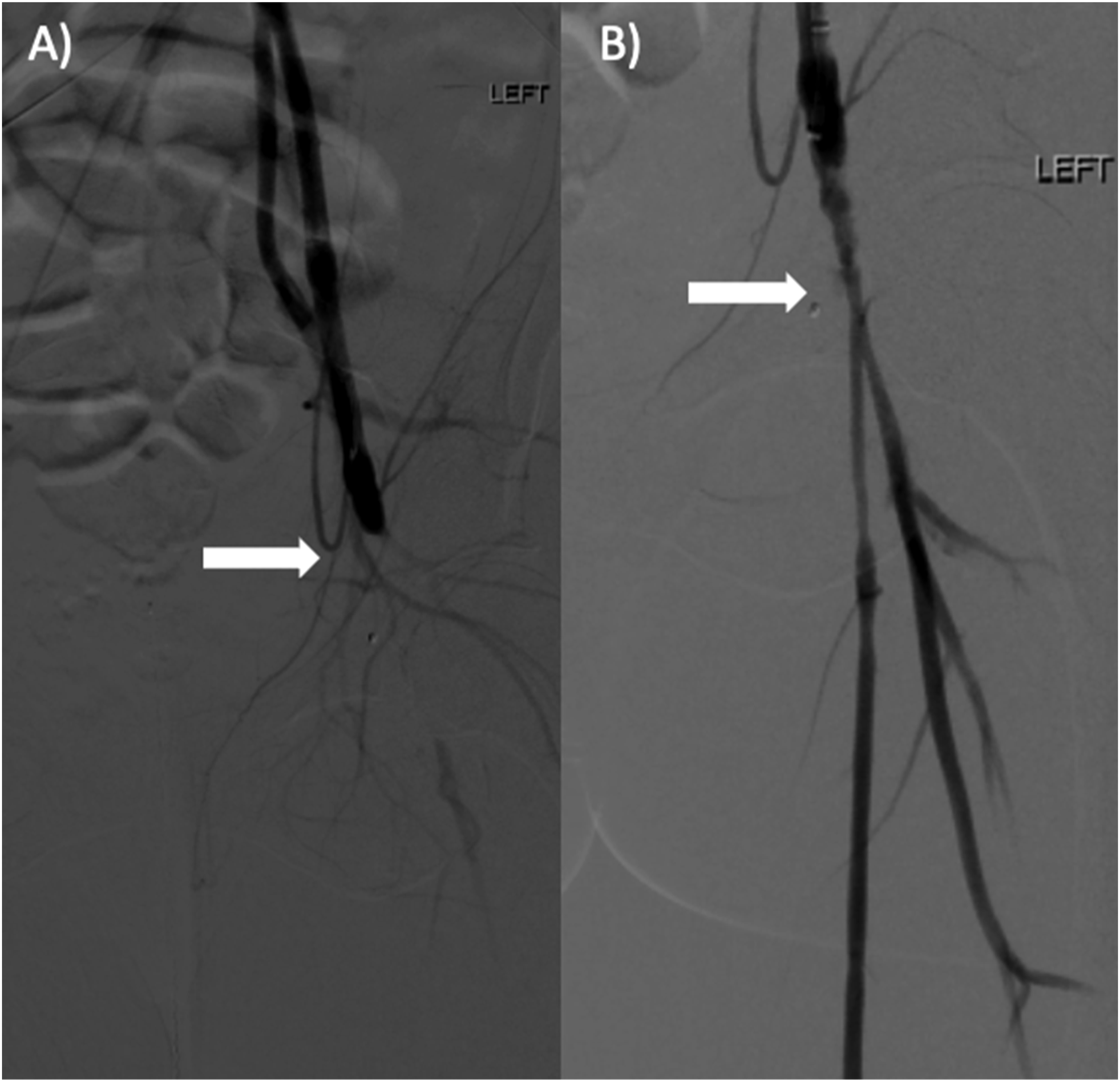
The patient underwent PFO closure during this admission and was discharged on postoperative day 6. He remains on oral anticoagulation for treatment of his pulmonary embolism.
Case 3
A 23-year-old female with a history of viral myocarditis and a familial history of “enlarged heart” presented in cardiogenic shock and was found to be COVID 19 positive. The patient required extracorporeal membrane oxygen (ECMO) support for her COVID-19 respiratory illness and cardiac support. Thus, the right CFA was chosen as the access site for the arterial ECMO cannula, and the left common femoral vein (CFV) was chosen for venous ECMO cannula access. Following decannulation from ECMO, the patient developed acute pain and weakness with notable coolness in her LLE. Arterial duplex ultrasound and further workup revealed an occlusive thrombus at the left CFA. In addition, post-op echocardiogram showed thrombus located within the left atrium as well as a clot in-transit through a PFO.
The patient was placed on cardiopulmonary bypass and underwent an emergent open closure of her PFO and suction embolectomy of her clot in transit and atrial thrombus. Following rewarming, RLE angiogram and suction embolectomy was performed to restore arterial flow into the right lower extremity. The patient remained in the hospital for 4 more days before being discharged home.
Case 4
A 51-year-old male, with confirmed COVID-19 infection and past medical history of hypertension, atrial fibrillation, and a repaired mitral valve prolapse, initially presented to the emergency room with shortness of breath, tingling in his feet bilaterally extending to his right calf, and swelling in his bilateral extremities. The patient was started on a heparin infusion, and he reported improvement in the numbness/tingling in his bilateral lower extremities. Further work up showed an elevated troponin level. elevated brain naturitic peptide (BNP), and non-palpable pulses in the bilateral distal lower extremities. He underwent a CT angiogram showing acute PE in the left and right main pulmonary artery extending into multiple lobar and segmental branches with evidence of right heart strain. Lower extremity imaging showed occlusion of flow in the right and left distal popliteal arteries with some reconstitution bilaterally. An echocardiogram also revealed that he had a PFO. Following initial improvement in lower extremity symptoms with anticoagulation, the patient began to lose motor function and it was felt the patient would benefit from popliteal embolectomy as well as primary PFO closure.
Given the worsening ischemia, angiogram of the lower extremity with suction embolectomy was chosen for this patient. During the procedure, the patient developed acute hypoxia and tachycardia. Suction thrombectomy was stopped and the patient was placed on venous-arterial (VA) ECMO due to his extensive pulmonary emboli and hemodynamic instability. Once he stabilized on VA ECMO, bilateral popliteal and tibial artery embolectomy was performed. Intra-operative transesophageal echocardiogram (TEE) demonstrated no evidence of atrial appendate thrombus. Inflow and outflow was ultimately successfully restored, and the patient developed audible biphasic Doppler signals in the pedal vessels. The patient’s PFO closure was completed at a later date (Figure 4). Coronal CT-angiogram of the chest shows PFO closure plug in place following repair (white arrow).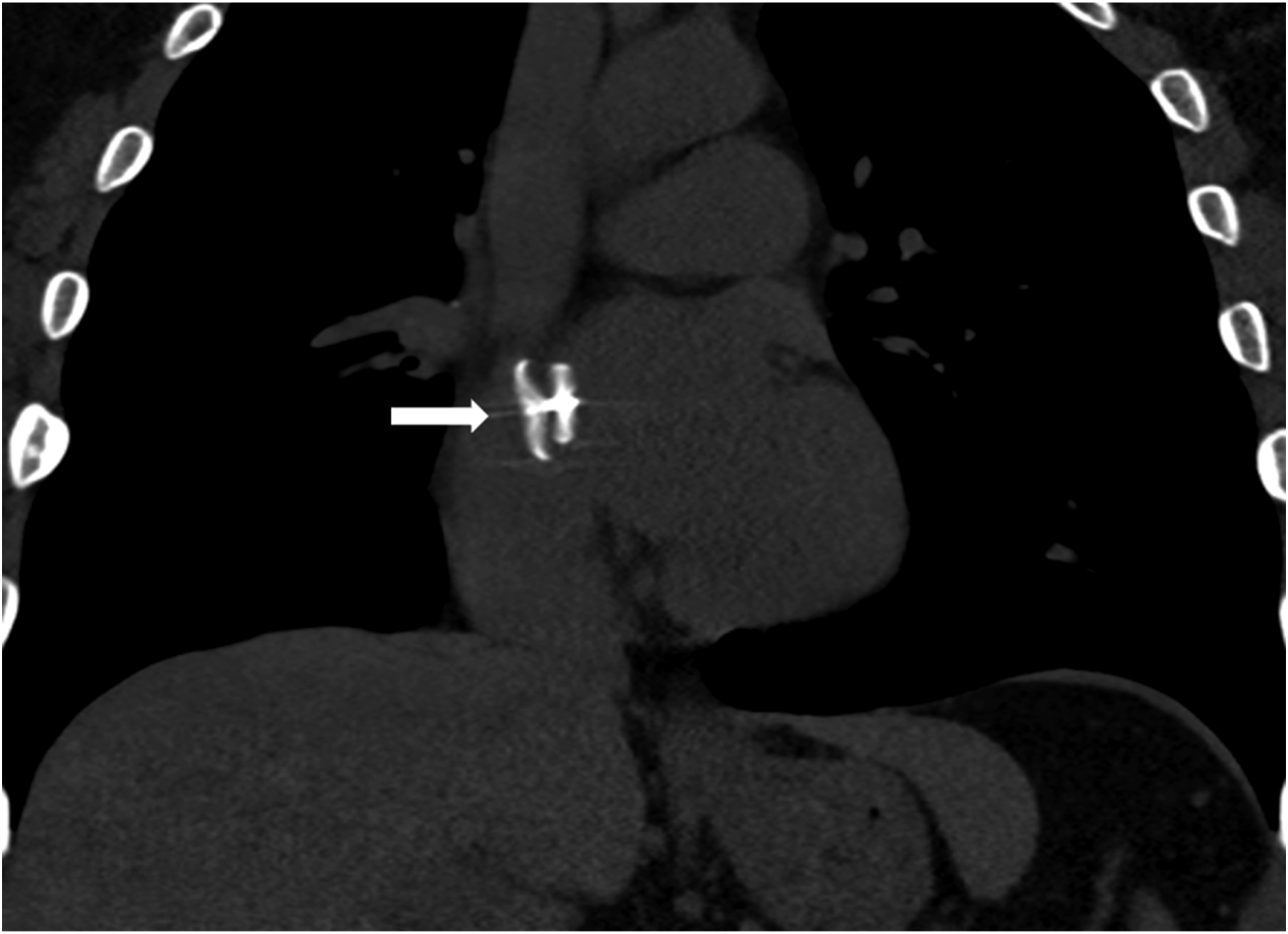
Case 5
A 43-year-old male, recently diagnosed with COVID-19, with no significant past medical history presented with numbness, weakness, and pain in his right arm. A CT angiogram revealed a right subclavian artery occlusion as well as a saddle PE. This saddle PE opened a previously closed PFO and led to subsequent acute limb ischemia. He was placed on a heparin infusion and was taken to the OR for a right brachial artery cutdown and thromboembolectomy.
After thromboembolectomy the patient developed a palpable pulse in the wrist and triphasic doppler signals. The patient then underwent PFO closure later during the hospitalization. At 4 month follow up the patient had improved sensation and function in his upper extremity.
Discussion
COVID-19 is widely understood to be capable of inducing a hypercoagulable state in certain patient populations. Clinicians speculate that this could be due to COVID-19 induced downregulation of ACE2 receptors that ultimately dysregulate the renin-angiotensin-aldosterone system (RAAS) and lead to immunothrombosis via activation of the complement system. 8 While the exact mechanism is unclear, COVID-19 induced coagulopathy can still be diagnosed through lab testing. Clinically, it has been shown that patients infected with SARS-CoV2 exhibit elevated IL-6 and IL-8 cytokines and D-dimer levels along with fibrinogen degradation products. This hypercoagulability ultimately leads to an increased risk for thrombotic events and subsequent mortality.11,12 A recent autopsy analysis has reported that the incidence of pulmonary embolism and DVT in deceased COVID-19 patients was ∼42% and ∼58% respectively. 9 Thus, patients with a confirmed diagnosis of COVID-19 are susceptible to a variety of vascular symptoms and clinical sequelae secondary to thrombus formation, including increased risk for developing ALI through the forced opening of a previously closed foramen ovale due to increased right sided intracardiac pressure.12,13
Acute limb ischemia is mainly caused by a decrease in limb perfusion due to arterial blockage via an embolism or thrombus. 14 While the hypercoagulable state of COVID-19 may serve as a catalyst for such events, unique pathways for thrombus migration may certainly arise. A rare yet concerning example of such thrombotic events, is transformation of the thrombus into a paradoxical embolism after traversing a PFO. Paradoxical emboli are more difficult to diagnose and treat than typical causes of ALI. In most cases, they can only be proven if the thrombus is identified as it crosses the cardiac defect, at which point it is an indication for surgical intervention. 15 The pathophysiology involves increased right sided heart pressures from a pulmonary embolus impeding forward flow through the lungs, and thus forcing open a previously undetected PFO. Specifically, the increased right ventricular pressure will shunt the suspected thrombus from the right atrium to the left atrium before occluding peripheral vasculature. 16 However, it is also possible that the suspected thrombus will become entrapped due to forceful opening or complex morphology of the PFO. 17 Despite the pathophysiology being well established, longitudinal data on clinical presentation related to limb ischemia in such special cases is still lacking. Treatment, nevertheless, is still often dependent upon the severity of disease. In typical ALI cases, anticoagulation is considered standard of care and first line therapy. However, more severe cases of ALI require surgical intervention involving endovascular or open surgical revascularization. 14 In alternate settings, such as in the treatment of patients with PFO induced ALI, extra considerations are required such as the need for PFO closure in addition to the management of the ischemia itself, and possible intervention for the pulmonary embolism.18,19
Although paradoxical embolism represents less than 2% of all arterial emboli,16,20 a handful of case studies have still been reported in the context of limb ischemia.16,21,22 Treatment for such cases usually involved a combination of embolism mobilization, PFO closure, and post-op anticoagulation medication. Greenberg et al 21 reports successfully performing an urgent embolectomy and PFO closure with prophylactic fasciotomy for a hypercoagulable patient with lower limb ischemia secondary to a thromboembolism that traversed through a previously silent PFO. Takafuji et al 22 further reports successfully performing a thrombectomy and percutaneous PFO closure for a patient with paradoxical embolism and left leg ischemia. Both patients were administered post-operative anticoagulation medication and developed no complications. In terms of general management for paradoxical embolism, treatment options are similar. Usually, patients are observed, prescribed anticoagulation medication, and/or undergo percutaneous or open closure of the PFO to prevent recurrence of paradoxical embolism. 23 However, recent studies have suggested that surgical closure of the PFO exhibits better long-term outcomes compared to isolated medical therapy. Specifically, percutaneous closure of PFO seems to demonstrate lower rates of recurrent thromboembolic events and secondary cerebrovascular events long-term.24-28 Certain studies have even reported success rates as high as 90% in preventing patients from developing recurrent embolism after performing percutaneous PFO closure. 29
ALI is an emergent concern in patients with confirmed cases of COVID-19. Although treatment protocols for such cases are well established, early suspicion and diagnosis are difficult but key factors for positive prognosis. While the hypercoagulable state of COVID-19 and the presence of PFOs should be considered in pertinent patients with ALI, imaging modalities should be used to confirm etiology. Clinicians treating patients who have previously tested positive for COVID-19 and present with arteriovenous embolism should be strongly suspicious of PFO mediated paradoxical embolism as an etiology for ALI.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
IRB Approval
The University of Maryland, Baltimore (UMB) Institutional Review Board (IRB) has approved this study under IRB #HP00099092. As this was a de-identified retrospective case review, this study was exempt from requiring informed consent under 45 CFR 46.104(d).
