Abstract
The optimal management strategy for acute limb ischemia (ALI) in patients with a concomitant malignancy is not well established. A very high mortality rate (83–100%) at 1 year has been reported in those who are treated surgically. Accordingly, a conservative management approach has been suggested as the main therapeutic modality. Our aim was to evaluate the survival outcomes of cancer patients treated for ALI at our cancer center. Cancer patients treated for ALI at the MD Anderson Cancer Center from 2001 to 2011 were included in this study. Overall survival and amputation-free survival rates were calculated. A total of 74 cancer patients with concomitant ALI were included in the study. Surgery was the most common therapy (36 patients; 49%). Percutaneous catheter-based interventions were used in 21 patients (28%). Eighteen patients (24%) received anticoagulation therapy only, and six patients (8%) received no therapy. The 30-day, 6-month, and 1-year overall survival rates were 80% (95% confidence interval [CI], 69% to 87%), 59% (95% CI, 47% to 69%), and 48% (95% CI, 36% to 59%), respectively. Eight patients (11%) underwent amputation. The 1-year amputation-free survival rate was 47% (95% CI, 35% to 58%). In conclusion, we did not find an invasive approach for the treatment of ALI in cancer patients to be associated with the very high mortality rates previously reported. In our opinion, the indications for surgery or catheter-based intervention in these patients should not differ from patients without cancer.
Introduction
Acute limb ischemia (ALI) is a vascular emergency and is associated with significant morbidity and mortality. Clinical trials in the general population have established the benefit of catheter-directed thrombolysis and surgery in treating patients who present with this condition. 1 Selection of the initial treatment modality varies and depends on several factors including etiology, duration of symptoms, and the severity of ischemia on initial presentation. In general, catheter-directed thrombolysis has been shown to improve limb salvage and to decrease early mortality in selected patients. 1 Urgent surgery is appropriate if embolic etiology is suspected or if the limb is immediately threatened, or when thrombolytic therapy is contraindicated. There is no evidence of a difference in the amputation and mortality rates at 1 year between patients initially treated with surgery and those initially treated with thrombolysis. 1 Mild ischemic events can sometimes be managed by watchful observation or by anticoagulation therapy alone. Amputation is indicated if irreversible ischemia is established on presentation. Despite the remarkable progress in treating this condition over the past 50 years, data from recent large clinical studies still show a 30-day mortality rate of up to 15% and amputation rates between 15% and 30%. 2 The 1-year survival rate is approximately 80%. 2
Much worse outcomes have been reported in patients with certain comorbidities, including cancer. In the Thrombolysis or Peripheral Arterial Surgery (TOPAS) trial, cancer was present in 12% of patients with ALI (12% in the thrombolysis arm, 11% in the surgical arm) and patients with cancer had a higher rate of morbidity and mortality.3,4 Outcomes in cancer patients whose limb ischemia was treated with surgery were extremely poor in two other separate studies that found the associated mortality to be as high as 100% at 2 months.5,6 Based on these findings, conservative management and palliative therapy have been advocated as the most appropriate treatment modality for cancer patients with limb ischemia, 7 whereas surgical intervention has been strongly discouraged.5–7 Such an approach, however, is not supported by other researchers: In their recent retrospective analysis of outcomes in 16 cancer patients with ALI, Tsang and colleagues 8 reported a 44% survival rate at 1 year following surgical revascularization. Similar findings were reported by Ouriel et al. 3 in their subgroup analysis from the TOPAS trial, with an observed 50% amputation-free survival rate at 1 year. To the best of our knowledge, outcomes of other therapeutic interventions (medical therapy, catheter-based intervention, or observation) have not been reported in cancer patients with concomitant ALI. Our objective was to describe and evaluate the survival outcomes of cancer patients presenting with and treated for ALI at our cancer center.
Methods
Patient selection
This is a retrospective chart review of cancer patients treated for ALI at The University of Texas MD Anderson Cancer Center from 2001 to 2011. The study protocol was reviewed and approved by the institutional review board and a waiver of informed consent was obtained. Patients were identified by their discharge diagnostic codes, and were included if they met the criteria of concomitant malignancy and ALI with symptoms occurring within 15 days prior to current admission. We excluded patients with benign tumors or without active malignancy, and also patients with iatrogenic limb ischemia related to a tumor chemoembolization procedure. Patients’ clinical characteristics, including age, sex, type and stage of malignancy, type of chemotherapy, cardiovascular risk factors and medications at the time of the ischemic event, were extracted from the medical record and tabulated. Advanced malignancy was defined as metastatic disease in patients with solid tumors and as disease relapse or need for stem cell transplantation in patients with hematologic malignancies. The Rutherford staging criteria 9 were used to classify the severity of limb ischemia based on the reported initial clinical evaluation by the treating physician. Catheter-based interventions included catheter-directed thrombolysis, arterial angioplasty and/or arterial stenting. Surgical procedures included thrombectomy, embolectomy and/or bypass surgery. The medical therapy category included patients receiving anticoagulation therapy only (heparin or low molecular weight heparin with or without antiplatelets). Pathology reports summarizing histological findings of the retrieved specimen during surgery were reviewed and divided into four separate categories: (1) predominately atherosclerosis, (2) pure thrombus, (3) predominately malignant cells/tumor, and (4) tumor invasion of vessel wall.
Data analysis
Overall survival (OS) was defined as the time from the date of hospitalization with the ischemic event to the date of death or last follow-up. Amputation-free survival was defined as the time from the date of hospitalization for the ischemic event to the date of amputation or death or last follow-up. The Kaplan-Meier method was used to estimate overall survival outcomes. Cox proportional hazards models were fitted to determine the association of patient and clinical characteristics with survival outcomes. Factors that were significantly associated with survival outcomes in the univariate log-rank test were considered in the multivariate Cox models. The final multivariate model only contained factors that had statistical significance. Results were expressed in hazard ratios (HR) and 95% confidence intervals (CIs). A p-value less than 0.05 was considered statistically significant; all tests were two-sided. Statistical analyses were carried out using SAS 9.1 (SAS Institute Inc., Cary, NC, USA) and S-Plus 7.0 (Insightful Corporation, Seattle, WA, USA).
Results
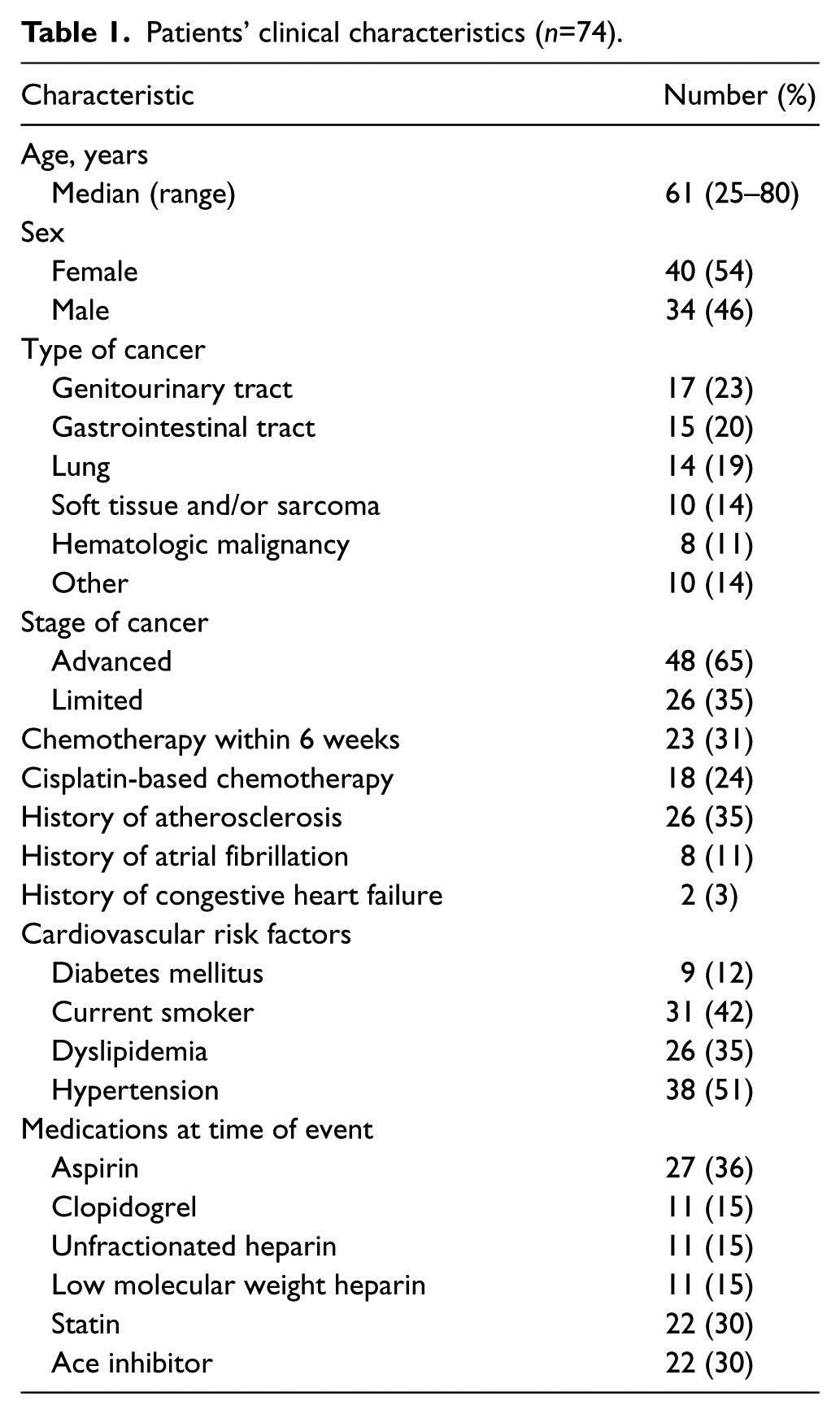
Between January 2001 and December 2011, we identified 74 cancer patients with concomitant ALI that met our inclusion criteria. Patients’ clinical characteristics are summarized in Table 1. The median age was 61 years (range 25–80 years) and 46% were men. The most common malignancies were of the genitourinary system, followed by the gastrointestinal system.
Patients’ clinical characteristics (n=74).
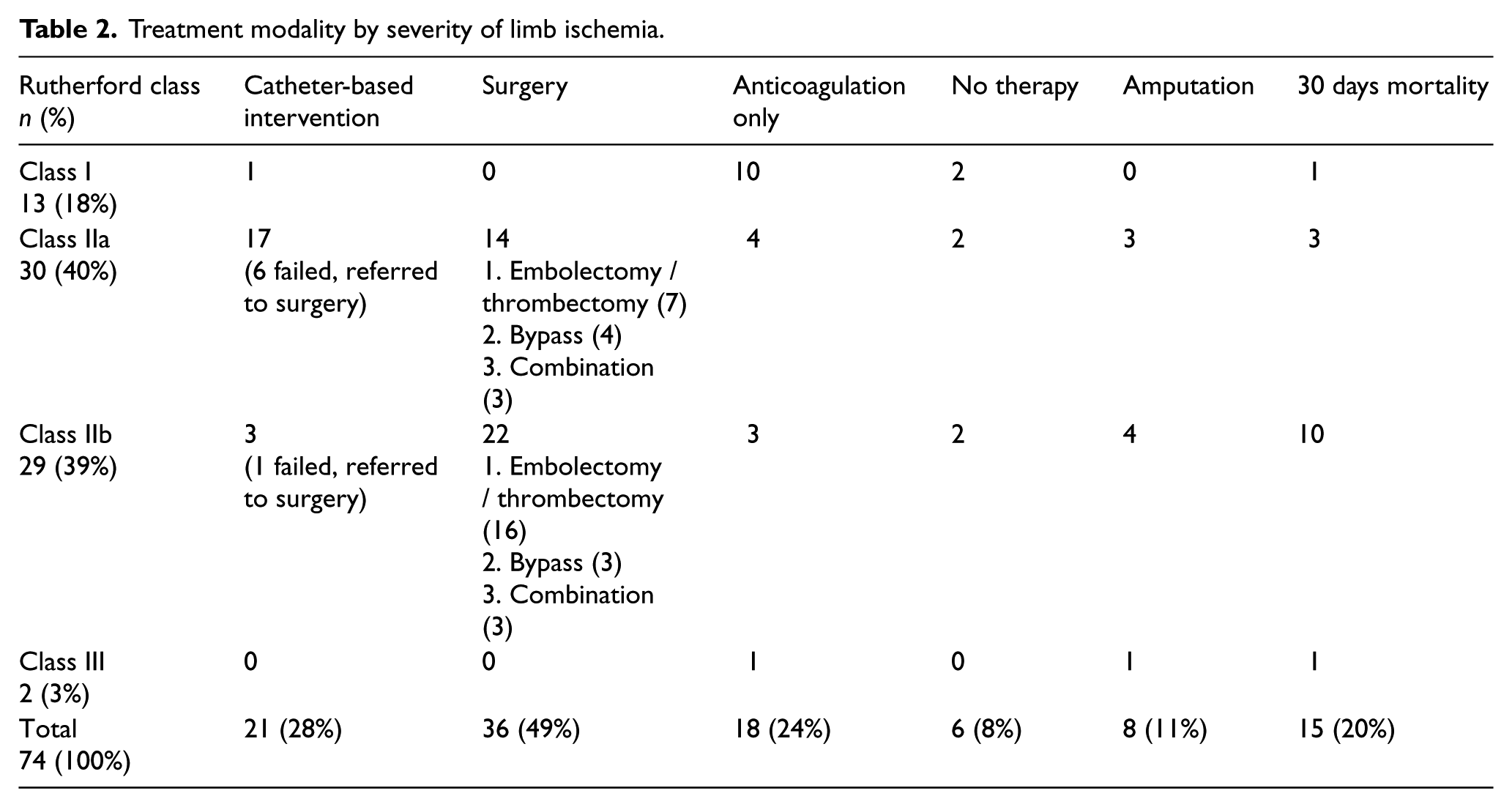
The femoropopliteal or tibial segments were the most commonly affected arteries (48 patients, 65%), followed by the aorto-iliac segments (15 patients, 20%). Brachial or subclavian artery acute thrombosis was found in four patients (three and one, respectively). The majority of patients were categorized as having Rutherford class IIa or IIb (79%) ALI, and only two patients (3%) presented with class III ischemia. Table 2 summarizes the treatment modalities by severity of limb ischemia on initial presentation. The majority of patients with class I ischemia were treated with anticoagulation only with no limb loss and only one died within 30 days. Of the patients with class IIa ischemia, three patients underwent limb amputation (two after failed thrombolysis and one after failed surgery) and three died within 30 days from the ischemic event. Most patients with class IIb ischemia (22 out of 29 patients) underwent surgical revascularization with four failures requiring limb amputation. A total of 10 patients in this group died within 30 days, including two patients who received no therapy, three patients who received anticoagulation therapy only, one patient who received thrombolysis and four surgical patients.
Treatment modality by severity of limb ischemia.
Severe co-morbidities that were prohibitive to any invasive or medical intervention were encountered with six patients (8%): one recovering from brain surgery for a metastatic lesion, three with severe thrombocytopenia (<50,000/microliter) and two with active gastrointestinal bleeding. Two of these patients presented with mild class I limb ischemia and were observed. The other four presented with class IIa (two patients) and IIb (two patients) ischemia; three of these were the thrombocytopenic patients and died within the first 30 days of the ischemic event. Severe thrombocytopenia, prohibiting thrombolysis or anticoagulation, was observed in another two patients who underwent open surgical revascularization and survived the 30-day period (one failed and required amputation).
From a procedural technical standpoint, thrombolytic infusion was given over 1–2 days with a second-look angiogram at 48 hours to guide further therapy. An adjuvant procedure was needed in 13 (17%) of the 21 patients who underwent catheter-based interventions. This included open surgery in seven (9%) patients and a percutaneous endovascular procedure in six (8%). Open surgery was the most commonly used ALI therapy (49%) including embolectomy, thrombectomy with or without patch angioplasty or insertion of a bypass graft conduit. Seven (9%) patients had previously undergone catheter-based intervention prior to surgery (three angioplasty, three thrombolytic therapy, and one a combination of both). Seven of the eight patients who underwent limb amputation had prior attempts at revascularization (three embolectomies, three catheter-directed thrombolysis, and one bypass surgery). All amputations occurred within 30 days of the presenting event.
Pathology results were available for 24 surgical patients. Thrombus-only was identified in 16 (67%) of the samples. Atherosclerosis with associated thrombus was noted in another five (20%) patients. Direct tumor invasion of the arterial wall was documented in two (8%) cases. One patient with underlying leukemia had a pathologic specimen showing clot made of a large aggregate of leukemic cells.
The median follow-up was 8 months (range 0–108 months). Forty-seven patients died, and 37 of those patients died within 1 year. The 30-day, 6-month, and 1-year OS rates were 80% (95% CI, 69% to 87%), 59% (95% CI, 47% to 69%), and 48% (95% CI, 36% to 59%), respectively. The 1-month and 1-year amputation-free survival rates were 80% (95% CI, 69% to 87%) and 47% (95% CI, 35% to 58%), respectively. Univariate analysis of baseline patients’ clinical characteristics showed that male sex (p = 0.021) had a negative impact on OS. After multivariable adjustment, men had a double increase in risk of death (HR = 1.97; 95% CI, 1.09 to 3.57; p = 0.024) compared with women. Figure 1 shows the Kaplan-Meier estimates of OS with 95% CI.
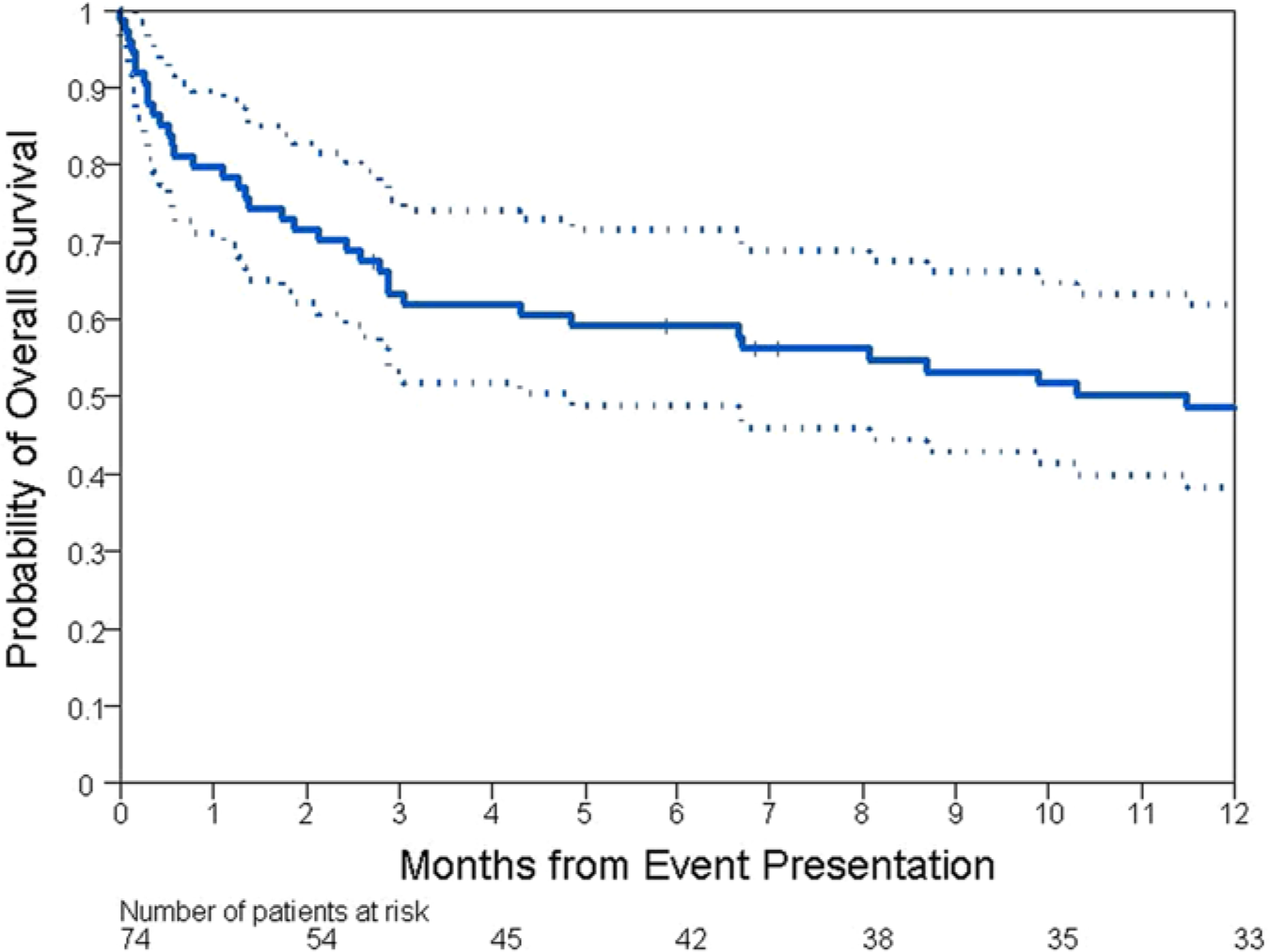
Kaplan-Meier estimates of overall survival with 95% confidence intervals.
Discussion
We did not find that an invasive approach for the management of ALI in cancer patients, including surgical procedures, was associated with high mortality rates as has been previously reported.
ALI and cancer patients are independently diverse and challenging. Thus, patients suffering simultaneously from both conditions are particularly difficult to manage. In fact, multiple factors including symptoms at presentation, severity of ischemia, cancer type, bleeding risks and life expectancy are of relevance and should be factored in to the management plan. There is a relative paucity of literature with few reports focusing on the pathogenesis, management and outcome of ALI in cancer patients.
The association between malignancy and peripheral artery ischemic disease has long been recognized. In 1987, Naschitz et al. reported the first series of patients with known stable peripheral artery disease who experienced worsening of their claudication symptoms when they developed cancer, followed by a significant improvement when their malignancy responded to therapy. 10 In a series of 62 patients who presented with critical limb ischemia, El Sakka et al. diagnosed occult malignancy in 16% of these patients. 7 This is likely an underestimation of the true co-prevalence of these two conditions since only patients with strong suggestive clinical features were screened for cancer. In the TOPAS trial, cancer was present in 12% of patients with ALI, and those with cancer had a higher rate of morbidity and mortality.3,4 In fact, it is also well established that the second most common cause of death in patients with peripheral artery disease is malignancy. 11 On the other hand, the prevalence of acute arterial ischemia in cancer patients was studied and reported by Khorana et al. and found to be in the range of 1.5–3.1%.12–14 Thromboembolism was found by the same researchers to be a leading cause of death in this patient population (9.2%). 14 A substantial proportion of these deadly ischemic events are related to arterial thromboembolism, including peripheral artery events. A much higher rate of arterial ischemic events has been reported in patients with specific malignancies15–18 or in patients who are treated with certain chemotherapy agents (cisplatin and angiogenesis inhibitors).18–22
Several mechanisms can lead to arterial ischemic events in patients with cancer. In addition to the typical causes of arterial ischemia related to traditional cardiovascular conditions, patients with an underlying malignancy or a hematologic disorder have added risks for in situ thrombosis related to acquired thrombophilia associated with cancer and its therapy. Other potential mechanisms for ischemia include tumor embolization, mechanical compression of blood vessels by the neoplastic process, endothelial damage caused by blood vessel catheterization, surgery, and chemotherapeutic agents. 18
Very few studies have looked at outcomes in patients presenting with this syndrome. Javid et al. reviewed prospectively the outcomes of 20 cancer patients who presented with critical limb ischemia (95% of them had ALI), eight of whom underwent surgery. 5 The mortality rate was 50% at 6 months and the 1-year survival rate was only 17%. 5 In another surgical study, Morris-Stiff and Lewis compared the outcomes of surgery in a cohort of 14 patients with cancer and ALI to surgical outcomes in a group of 102 non-cancer patients. 6 Mortality at 2 months was significantly higher in the cancer group (100% vs 35%; p<0.001), despite similarities in management. 6 Based on these two observational studies, ALI in cancer patients has been regarded as an agonal and terminal event with a bleak prognosis. Surgical intervention in managing these patients has been previously discouraged by many, and palliative care has been advocated as the main therapeutic modality.
In contrast with these two surgical studies, the main finding in our patient population was higher survival rates at 30 days, 6 months, and 1 year. Similar 1-year survival rates were reported previously by Ouriel and Veith 4 and more recently by Tsang et al. 8 In a multivariate analysis of 544 patients from the TOPAS trial, Ouriel and Veith 4 found that a history of malignancy was one of four variables associated with worse outcomes with a risk ratio of 1.6 (95% CI, 1.07 to 2.44; p = 0.024). Twelve percent of their patient population had cancer. They were divided equally between two treatment groups: the thrombolytic group (32/272) and the surgical treatment group (33/272). Their 1-year amputation-free survival rate was 50%. It is not clear if there was a difference in outcomes between the two groups since outcome by treatment modality was not reported. More recently, a 44% survival rate at 1 year was observed by Tsang and colleagues 8 in a retrospective analysis of 16 cancer patients with ALI treated with surgery.
To our knowledge, our study is the largest cohort of patients with concomitant malignancy and ALI and, contrary to previous studies, our patients underwent treatment modalities covering the entire spectrum of available interventions. There was no difference in outcomes between patients who had advanced cancer and patients with limited disease. Multivariate analysis showed male sex was the only clinical parameter associated with statistically significant worse outcomes. A total of 23 (31%) patients had undergone chemotherapy within 6 weeks preceding their clinical event. The majority of these patients (78%) had cisplatin as part of their chemotherapy regimen, a drug well known to be associated with arterial thrombotic complications. 19 The pathology findings in the 24 surgically treated patients suggest that limb ischemia in this group of patients is much more likely to be thromboembolic and atherosclerotic in etiology than tumor invasion or tumor embolization.
The 1-month survival rate of 80% (95% CI, 69% to 87%) in our patients was similar to that of non-cancer patients with ALI. At 1 year, the overall survival rate in our patients was 48% (95% CI, 36% to 59%). The amputation rate in our series was 11%, and the 1-month and 1-year amputation-free survival rates were 80% (95% CI, 69% to 87%) and 47% (95% CI, 35% to 58%), respectively.
This study has several limitations including the retrospective review of a clinical database with no comparable controls. The retrospective data collection also did not allow us to factor in other potential benefits of invasive treatment for ALI, such as pain relief and quality of life implications. Another shortcoming of this study was our inability to accurately determine the exact cause of death (despite very serious and rigorous efforts). Historically, however, it is known that beyond the acute event, the main cause of death in patients with peripheral artery disease is coronary artery disease (40–60%), followed by malignancy (7–23%) and cerebrovascular disease (2–15%). 23 In our patient population, it is possible that a significant percentage of patients who survived the acute event beyond the first 30 days subsequently died of their malignancy. This would explain the lower 1-year survival rate compared with that of non-cancer patients (48% vs 80%).
In conclusion, we found that an invasive approach for the treatment of ALI in cancer patients does not appear to be associated with a prohibitively high mortality rate, as has been previously reported. We speculate that the low 1-year survival rate that we observed in our patient cohort compared with the published data in non-cancer patients is likely related to the cancer prognosis than to acute limb ischemia or its treatment modality. It is our belief that conservative management as the main therapeutic modality would only be justified in cases of terminal cancer where palliative care supersedes. Otherwise, cancer patients with ALI should be managed in line with the established standard of care for ALI in the regular population
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
