Abstract
Peripheral artery disease (PAD) is a severe manifestation of atherosclerosis. Patients with PAD are at heightened risk for atherothrombotic complications, including myocardial infarction and stroke (MACE); however, there is also an equal or greater risk of major adverse limb events (MALE), such as acute limb ischemia (ALI) and major amputation. Therefore, there is a need for effective medical therapies to reduce the risk of both MACE and MALE. Recent trials have demonstrated the role of thrombin inhibition in reducing the risk of MACE and MALE in PAD patients. One such medical therapy, vorapaxar, is a potent inhibitor of protease activated receptor-1 which mediates the cellular effects of thrombin. Vorapaxar, used in addition to aspirin, has demonstrated robust reductions in MACE and MALE in PAD patients. In this article, we provide a contemporary review of the current state of PAD and the role of antithrombotic medications in the treatment of PAD, as well as the current clinical data on vorapaxar and strategies to integrate vorapaxar into contemporary medical management of peripheral artery disease.
Peripheral Artery Disease State
Peripheral artery disease (PAD) is one of the most severe manifestations of atherosclerosis. It is estimated that in 2015 approximately 238 million people had PAD globally with an annual increase in cases since 1990. 1 In an insured cohort with an age greater than 40 years, the annual incidence of PAD was 2.4%. 2 This striking increase is due to an increased prevalence of risk factors including an aging population, increased obesity, and particularly, more diabetes, which is one of the most potent risk factors for PAD. In contrast to the incidence of myocardial infarction (MI) and stroke, which are falling, there has been a rise in acute and severe presentations of PAD, including acute limb ischemia (ALI), major amputation, and critical limb threatening ischemia (CLTI), resulting in the need for costly hospitalizations and revascularization procedures. 3,4
By nature of the underlying atherosclerosis, patients with PAD are at heightened risk of atherothrombotic complications such as MI and stroke (MACE). 3 In fact, their risk is as high or higher than patients with coronary disease. 5 The dominant driver of morbidity, loss of function, and cost in the PAD population, however, is adverse limb events. Over the course of 3 years, approximately 25% of symptomatic PAD patients will require at least one procedure to restore perfusion to their legs either to prevent amputation or to alleviate severe symptoms. 6,7 In addition, the risk of major adverse limb events (MALE), such as ALI and major amputation, is equal to or greater than the risk of MI or stroke in this population. 6,7 In spite of this risk, there are few medical therapies available that reduce the risk of MALE in patients with PAD. 8
ALI, a frequent and potentially catastrophic complication of PAD, results from sudden thrombotic occlusion of a large limb artery either due to in situ thrombosis or embolism. 9 Rates of ALI are similar to those of MI or stroke in patients with chronic PAD. 6,10 Patients with ALI are in the hospital for a median of 8 days, almost 20% require amputation, most require surgery, and at the end of hospitalization nearly 15% have died or are unable to return home due to disability. 6,10 CLTI represents about 10% of PAD patients with an overall prevalence of about 1%, although it is difficult to capture epidemiologic data in CLTI patients. 11 The amputation rates in CLTI have varied in the literature, with one systematic review finding a rate of 3.6 to 68.4 per 100,000 with rates widely varying across the globe. 12
One challenge in the PAD population has been recognition of heterogeneity of risk and an absence of mechanisms to identify subgroups at higher risk of MACE or MALE to enable personalization of therapy. Recently, several publications have demonstrated that patients with PAD and concomitant symptomatic coronary disease (CAD), or polyvascular disease, are at an extremely high risk for MACE. 5,13 In addition, several datasets have demonstrated that patients with PAD who have undergone lower extremity revascularization are at extremely high risk (∼4-fold increase) for MALE, even long-term postprocedure. 6,10,13 The recognition of these high risk profiles in patients with PAD may enable clinicians to personalize more intensive medical therapies to high risk patients rather than consider treating all patients.
Medical Landscape
Antithrombotic therapies play a key role in the prevention of atherothrombotic events in atherosclerosis. 14 The data supporting their use in PAD, however, are mixed. Meta-analyses show that aspirin reduces MACE and increases bleeding in symptomatic PAD; however, there is no benefit for limb outcomes. 14 Clopidogrel has been shown to be modestly superior to aspirin as monotherapy in PAD but has demonstrated no benefit for limb outcomes. 14 More potent P2Y12 inhibition with ticagrelor versus clopidogrel did not reduce MACE or MALE in PAD. 15 The combination of aspirin and clopidogrel (dual antiplatelet therapy or DAPT) does not reduce MACE or MALE in PAD and increases bleeding. 16 Anticoagulation with warfarin does not reduce MACE or MALE and is associated with an increase in severe bleeding events in PAD. 17,18 The lack of effective therapies is represented in the 2016 ACC/AHA PAD guidelines which only recommend aspirin or clopidogrel monotherapy for MACE reduction and provide no Class I recommendations for antithrombotic therapies to reduce MALE. 8 Recently, the Cardiovascular Outcomes for People Using Anticoagulation Strategies (COMPASS) and Vascular Outcomes Study of Acetylsalicylic Acid Along with Rivaroxaban in Endovascular or Surgical Limb Revascularization for Peripheral Artery Disease (VOYAGER PAD) trials have demonstrated that the combination of aspirin and rivaroxaban 2.5 mg twice daily, a low dose factor Xa inhibitor that inhibits thrombin generation, reduces MACE and MALE in PAD and increases bleeding. 19,20 Low-dose rivaroxaban in combination with aspirin is now approved for use in PAD. Uptake has been relatively low as clinicians have struggled to understand which patients are at highest risk enabling personalization of therapy. Nonetheless, the COMPASS and VOYAGER PAD trials definitively demonstrate the importance of thrombin inhibition in reducing MACE and MALE in PAD.
Vorapaxar
Vorapaxar is a first in class potent and reversible antagonist for protease activated receptor-1 (PAR-1). 21 Thrombin not only plays a role in the coagulation cascade but has many cellular effects including direct effects on inflammatory cells, vascular smooth muscle cells, and platelets. In fact, thrombin is the most potent activator of platelets. 18 These cellular effects are mediated through PARs, and PAR-1 is the most important receptor on platelets in humans. Therefore, vorapaxar enables the potent, selective, and reversible inhibition of thrombin-mediated activation of cells and platelets through activation of PAR-1. 21
Although vorapaxar potently inhibits thrombin-mediated activation of platelets, it does not decrease thrombin generation or thrombin levels. This in contrast to Factor Xa antagonists which directly decrease thrombin levels (Table 1, Figure 1). The intricate interplay between thrombin generation and platelet activation underlies the pathophysiology of atherothromobtic events in atherosclerosis. 22 This is especially important in polyvascular disease which is seen in nearly 20% of all patients with atherosclerosis. 23 A recent analysis showed benefit in more intense antithrombotic regimens in patients with polyvascular disease, which included therapeutics that both inhibited platelet activation and thrombin formation. 24
Antithrombotic Therapy.


Thrombin pathway inhibition by vorapaxar and rivaroxaban. Shown are the mechanisms of how vorapaxar and rivaroxaban modulate the thrombin pathway as well as potential adverse outcomes. DVT indicates deep vein thrombosis; MI, myocardial infarction; PE, pulmonary embolism.
Vorapaxar exhibits several favorable characteristics in that it is almost 100% bioavailable, is quickly active within 1-2 hours of ingestion, is dosed daily due to its long half-life, and is hepatically cleared. Thus, there is not a need to renally dose or exclude patients with renal dysfunction. It does not change thrombin time, activated partial thromboplastin time, or bleeding time. 21 It has been studied in more than 40,000 patients with symptomatic atherosclerosis in long-term outcome trials and has been well-tolerated with no severe adverse effects beyond the bleeding that is expected with an antithrombotic therapy. 21 Although it targets thrombin, a potent activator of platelets, clinical efficacy and magnitude of benefit must be determined in the completed outcomes trials as are described in the following section.
Benefits and Risks of Vorapaxar in Peripheral Artery Disease
Through extensive clinical investigation, including the pivotal Trial to Assess the Effects of Vorapaxar in Preventing Heart Attack and Stroke in Patients with Atherosclerosis (TRA2P-TIMI 50) trial, vorapaxar has been approved for use in secondary prevention in patients with prior MI and/or PAD in addition to aspirin and/or clopidogrel. 25 The addition of vorapaxar has been shown to reduce the risk of MACE by 20% with a number needed to treat (NNT) of 63 at 3 years. There is an associated increase in Global Use of Strategies to Open Occluded Arteries (GUSTO) moderate (requiring blood transfusion) or severe (intracerebral or hemodynamically compromising) bleeding with a number needed to harm (NNH) of 77 but no increase in severe bleeding events such as intracranial hemorrhage or fatal bleeding. When considering MACE and GUSTO severe bleeding, treating 1000 patients for 3 years with vorapaxar would prevent 16 MACE events at the cost of 3 GUSTO severe bleeds. Results are consistent in both the MI and PAD populations. 25
Recent data, however, have shown that the benefit/risk profile is more favorable in the PAD subgroup of patients with polyvascular disease. 26 As noted above, this has been recognized as a particularly vulnerable population for MACE. In a recent analysis of over 6,000 patients with PAD, 2.9% with history of amputation and 5% with history of Fontaine III/IV disease, in TRA2P-TIMI 50, it was shown that in patients with both PAD and CAD (polyvascular disease), vorapaxar reduces MACE with an absolute risk reduction of 2.2% at 3 years or a NNT of just 45. This translated into an overall net outcome difference of 2.5% or NNT of just 40. For comparison, the NNT to prevent any cardiovascular event after a myocardial infarction was 23 with the use of atorvastatin 80 mg over simvastatin 20 mg; however, this was over a median of ∼5 years which is double that for the observations from TRA2P-TIMI 50 where median follow up was 30 months (2.5 years). 27 These data highlight that patients with PAD and CAD derive a robust benefit and net benefit from the addition of vorapaxar to aspirin and/or clopidogrel. 26 It is important to note the individual variability of clopidopgrel metabolism and in those patients with loss of function genetic variants, the benefit of clopidogrel-containing antithrombotic regimens may be attenuated.
In addition to the benefits of vorapaxar for MACE, there are also robust benefits for MALE, including ALI. TRA2P-TIMI 50 prospectively captured and adjudicated ALI and reported, as part of a key secondary outcome of urgent hospitalization of a thrombotic nature, that ALI was significantly reduced with vorapaxar. The reduction in ALI was 42% in patients with PAD and statistically significant. 6,7 In addition, the need for repeat peripheral revascularizations, which are a large driver of cost, was significantly reduced. Overall, in patients with PAD, in addition to the 16 MACE events prevented at 3 years for 1000 patients treated for 3 years, vorapaxar would also prevent 13 ALI events and 39 peripheral revascularizations. These benefits for adverse limb events are robust and comparable in magnitude to those seen with rivaroxaban.
The benefits of intensive therapies for MALE in absolute terms depend on the risk of the patient. Not all patients with PAD are at high risk for MALE with a clear difference in risk between intermittent claudication and CLTI. Recent data have shown that patients with prior limb revascularization are at particularly high risk for MALE both early after intervention and long-term. Therefore, the population of patients with PAD and prior revascularization may derive greater benefits from MALE reduction (Figure 2). In fact, in a recent paper by Qamar et al patients with PAD and prior revascularization were shown to derive a robust reduction in MALE with vorapaxar with an absolute reduction of 2.5% at 3 years translating to a NNT of just 40. 26
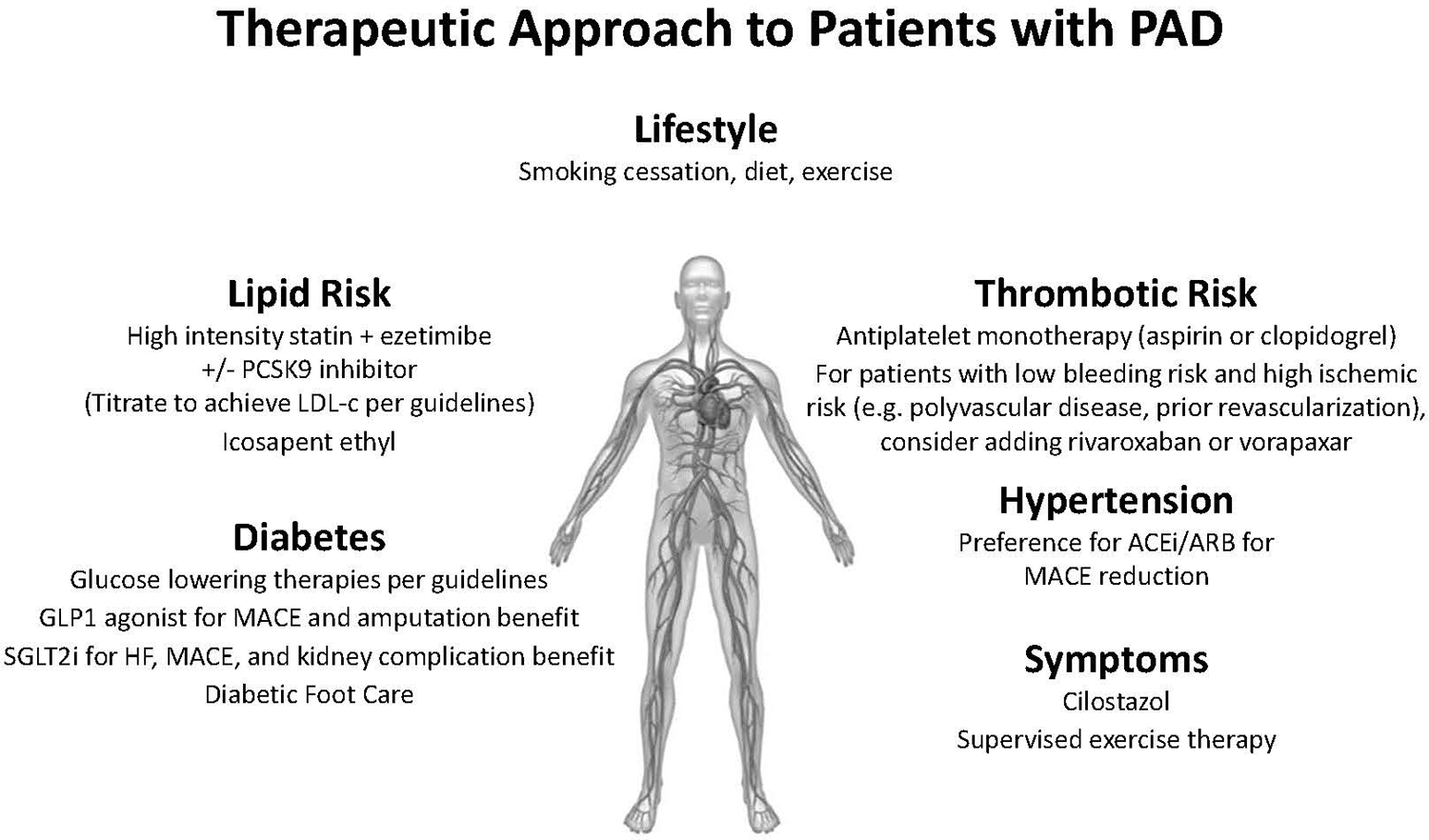
Approach to risk factor modification in PAD. Shown are recommended strategies for risk reduction in patients with PAD, including the role of vorapaxar in high-risk patients. ACEi indicates angiotensin-converting enzyme inhibitor; GLP1, glucagon-like peptide-1 receptor; LDL, low-density lipoprotein; PAD, peripheral artery disease; PCSK9, proprotein convertase subtilisin/kexin type 9 inhibitor; SGLT2i, sodium-glucose transport protein 2 inhibitor.
Integrating Vorapaxar Into PAD Treatment
Vorapaxar and rivaroxaban are the only antithrombotic therapies that have been approved for use in PAD by the Food and Drug Administration (FDA) as an addition to antiplatelet monotherapy. Both therapies show similar benefits for MACE and MALE and similar safety profiles and both work by interrupting the role of thrombin in patients with PAD. As better understanding of the risk profile of PAD and high-risk patient profiles that warrant more intensive therapies is gained, having these therapeutic options available will be important for clinicians and patients to personalize treatment strategies. Factors such as risk profile, half-life, dosing, and pharmacologic characteristics may influence choices for an individual patient. Biomarkers are another potential data point that could assist in identifying optimal therapy for those patients at increased risk of ischemic outcomes (e.g. MACE or MALE) or bleeding. Although several biomarkers including troponin, brain natriuretic peptide, or high-sensitivity C-reactive protein have been associated with increased of MACE, fewer have been demonstrated to be predictive of MALE. 28,29 Similarly, optimal risk prediction for bleeding in patients with PAD remains an area of ongoing research. Ongoing research holds promise for better risk stratification for patients with PAD for limb outcomes and for bleeding.
Because these therapies increase bleeding, broad adoption in all patients may not be suitable (Figure 2). Patients with PAD at low bleeding risk should be identified. Of these, those with polyvascular disease (PAD and CAD) and those with PAD and prior revascularization have been shown to derive robust benefits for MACE and MALE with favorable NNTs and net benefits. These patients should be considered for more intensive therapies. The choice of which agent may depend on several factors, but the once daily formulation of vorapaxar coupled with the absence of any renal restrictions or dose adjustments, as well as the ability to give in addition to clopidogrel may be attractive to some providers.
Footnotes
Authors’ Note
Institution of Reported Work: University of Colorado Anschutz Medical Campus.
Author Contributions
All authors have contributed equally to drafting and revising this manuscript and agree the content is ready for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: None of the authors disclose grant support for this review article. However, we would like to disclose that CPC Clinical Research does receive grant support from Amgen, AstraZeneca, Bayer, JanOne, Janssen, Merck, NovoNordisk, Sanafit, and Wraser.
