Abstract
Strategies to enhance the relaxometric properties of gadolinium (Gd)-based contrast agents (CAs) for magnetic resonance imaging (MRI), without the chemical modification of chelates, have recently had a strong impact on the diagnostic field. We have taken advantage of the interaction between Gadolinium diethylenetriamine penta-acetic acid (Gd-DTPA) and the hydrogel structure of hyaluronic acid to design cross-linked hyaluronic acid nanoparticles down to 35 nm for use in MRI applications. The proposed bioformulations enable the control of the relaxometric properties of CAs, thus boosting the relaxation rate of T1. Our results led us to identify this approach as an adjustable scenario to design intravascularly injectable hydrogel nanoparticles entrapping Gd-DTPA. This approach overcomes the general drawbacks of clinically approved CAs having poor relaxivity and toxic effects.
Commentary to: Russo M, Bevilacqua P, Netti PA. A microfluidic platform to design crosslinked hyaluronic acid nanoparticles (cHANPs) for enhanced MRI. Sci Rep. 2016;6:37906. doi:10.1038/srep37906
Our work 1 entitled “A Microfluidic Platform to Design Crosslinked Hyaluronic Acid Nanoparticles (cHANPs) for Enhanced MRI,” recently published by Scientific Reports, exploits a microfluidic flow-focusing approach to synthesize hydrogel nanoparticles down to 35 nm. This method provides a rational entrapment of the gadolinium-DTPA (Gd-DTPA) for magnetic resonance imaging (MRI) boosting its relaxometric properties. Furthermore, systems with monodisperse size under 100 nm are likely to improve delivery functions to the tissues, stability of the metal chelates, and also provide enhanced relaxometric properties of the Gd-based contrast agents (GBCAs; Figure 1). Gadolinium-DTPA has been selected because of its daily use in the clinical practice.
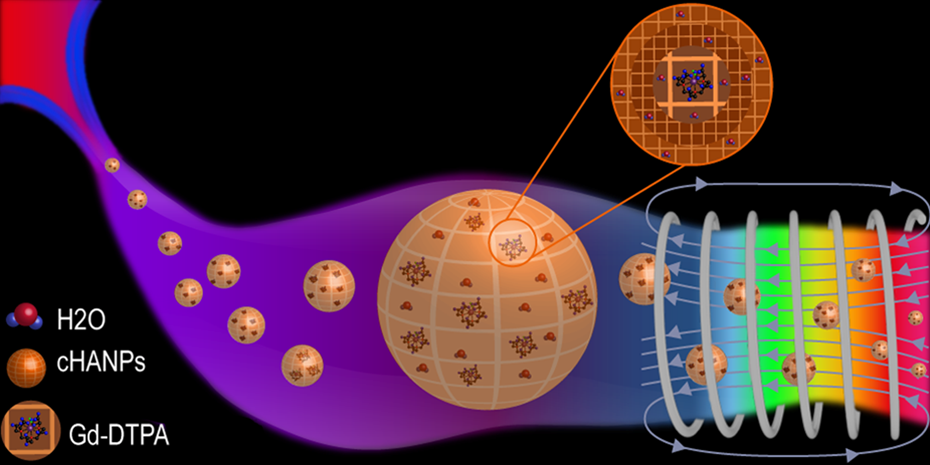
A proposed fashionable way to entrap gadolinium-DTPA (Gd-DTPA) in cross-linked hyaluronic acid nanoparticles (cHANPs) for application in diagnostics and therapy. The microfluidic flow-focusing approach used to control the interference of gadolinium in the nanoprecipitation phenomena and to design the nanoparticles is reported. Beyond the significant improvements generated by the utilization of the proposed microfluidic platform, the minimalist illustration clearly presents the idea to set up a reduced time-consuming process that is able to produce ready to use and completely biocompatible nanoparticles to impact immediately on the clinical magnetic resonance imaging (MRI) applications.
Contrast agents (CAs) are paramagnetic metal ions able to enhance the contrast in MR images by positively influencing the relaxation rates of water protons in the immediate surroundings of the tissue in which they localize. Among different CAs, GBCAs are routinely used in the clinical MRI practice. Magnetic resonance imaging represents the first-line diagnostic imaging modality for numerous indications. Gadolinium-based CAs are injected before the examination. This permits the detection of various pathologic processes otherwise not appreciable without an enhanced MR or other imaging modalities. The widespread use of GBCAs still has several limitations, such as low efficacy, toxic effects, and lack of tissue specificity. Indeed, their relaxivity is still far away from the theoretical limits, and they still present nephrotoxicity and heavy allergic effects. Furthermore, recent studies conducted by Kanda et al 2 and McDonald et al 3 -5 reported the risk of intracranial deposition of elemental Gd in neuronal tissues after GBCAs administration, even after only 1 MRI scan. Patients with normal renal and hepatobiliary function were also at risk. The authors pointed out that the clinical significance of these findings is not completely understood at this time. In the meantime, the Food and Drug Administration alarmed the medical community about the risk of deposition, recommending health-care professionals to limit the use of GBCAs unless necessary. This warning could have a potential negative impact on the early diagnosis of several pathologies (www.FDA.GOV.com/safetyannouncement7-27-2015). With this in mind, the ability to provide a formulation able to overcome several of these limitations is of great interest to the scientific and the medical community. It is essential to highlight the need to not only build a new system but also build a biocompatible probe with higher relaxivity. This probe should be able to protect the chelate around Gd ions from transmetallation phenomena and control the accumulation and the clearance of the GBCAs in a specific organ.
In the last few years, many efforts have been made to develop better CAs based on nanostructures with higher relaxivity and lower toxicity. Compared to other strategies based on bioconjugation or encapsulation 6 , models made on the optimization of “rigidification” 7 and “confinement” 8 of metal chelates seem to be the most effective at enhancing the effect of GBCAs without the chemical modification of the chelate 9 . Recently, we have also explored the impact in the bulk of cross-linked and uncross-linked biopolymer matrices on relaxometric properties of CAs. We found the contribution to the enhancement of CAs is highlighted and attributed to the reduced mobility of water within the hydrogel. 10
Although there has been a growth of interest in the imaging field and significant efforts have been made to apply microfluidic technologies to the production of nanoparticles, 11 the exploitation of microfluidic approaches to support the design of hydrogel nanostructures for MRI is still lacking. Microfluidics is an interdisciplinary field of research combining soft matter physics, biochemistry, and microsystems engineering. It enables the accurate control of fluids and constant manipulation of individual process parameters and phenomena, such as mixing, flow rate, chemical reaction, and phase separation behavior. 12 However, impacting the relaxometric properties of CAs without chemically modifying chelates has yet to be studied by modulating the properties of nanoparticles with controlled nanoprecipitation in a microfluidic flow-focusing system. 6 This method could be used to obtain a new class of MRI approaches based on rational polymer nanostructures.
Our study has reported, for the first time, interferences of the Gd chelate in the flow-focusing approach that could significantly advance the core knowledge of the interaction between Gd chelates and hydrogel matrix to be applied to MRI. The use of microfluidics allowed us to observe this phenomenon and allowed for the setting up of a flow-focusing strategy to modulate the nanoprecipitation playing on the hydrophilic–lipophilic properties of the surfactants and modulate the pH levels to optimize relaxometric performances of the MRI CAs.
The simultaneous addition to the middle channel of Gd together with HA solution causes significant interference on the flow-focusing pattern and consequently affects nanoparticle shape, size, and encapsulation. Process parameters were controlled to obtain monodisperse nanoparticles and improved properties of the Gd-DTPA entrapped within our nanoparticles. Cross-linked hyaluronic acid nanoparticles are produced using divinyl sulfone (DVS) as a cross-linking agent. The HA-DVS biocompatibility has been already confirmed by histological analysis, as previously reported by Oh et al. 13 The cross-linking reaction occurs simultaneously with the nanoprecipitation. This increases cHANPs’ stability in water and avoids swelling. Above all, it is essential in controlling degradation, release properties, and allows retainment of Gd-DTPA.
Given the versatility of our cHANPs, it was particularly interesting to investigate how the selection of different reaction strategies and formulations could result in advanced and tailorable CA-loading functionalities. Gd-DTPA entrapped cHANPs have the capability to retain GBCAs to boost the MR signal without the chemical modification of the chelates and at the same time could potentially provide tissue specificity and reduce nephrotoxicity and intracranial Gd deposition. In vitro relaxivity has been studied for loaded and unloaded cHANPs at 37°C and 1.5 T by a relaxometric instruments. These results were compared to the commercial CA: Magnevist. In our platform, by fine-tuning the mixing of all species, nanoprecipitation behavior and cross-linking reaction are adjusted very precisely and simultaneously, leading to relaxivity values about 12 times larger than that of free CAs clinically available.
We propose a microfluidic platform along with our efficient, straightforward, and scalable strategies to aid in the development of intravascularly injectable and completely biocompatible hydrogel nanoparticles, entrapping clinically approved CAs for application in diagnostics and therapy. The strength of our scientific work is that we have used and combined clinically approved materials, such as HA and Gd-DTPA.
Our method allowed for strict control of nanoparticle characteristics and the entrapment of the Gd-DTPA, resulting in a low polydispersity of nanoparticles and a high encapsulation efficiency of the metal in the nanoparticles, as well as easy recovery of the obtained particles without long and expensive purification steps. Additionally, this microfluidic approach enabling the tuning of the physicochemical properties of the obtained nanoparticulate architectures that have been proved to boost the MRI signals.
The aim of this work was to contribute to the solution of the physical and biological limits regarding the clinical use of CAs. We have proved that it is possible to increase relaxivity by tuning the structural parameters of hydrogel nanoparticles. At the same time, we were also able to improve tissue specificity, the stability of the chelates, imaging diagnostic acquisition, and potentially reduce the administration dosage. The developed microfluidic strategies allow for a fine-tuning of the formulations that can easily be applied to different polymers and materials, or a combination of materials, involved in the nanomedicine field. These developments will allow us to make a library of customized nanostructures.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
